International Journal of Clinical Cardiology
Radial-Linear Ablation in Peri-pulmonary Vein Area Prevents Inducible Atrial Fibrillation in Dogs
Xue Zhao1*, Yinxiang Zhu1#, Xiang Mei1#, Jiayou Zhang1, Jianqiang Hu2*, Wenliang Lu3, Xingui Guo3, Li Dai3, Kailei Shi3 and Zhenguo Liu4
1Division of Cardiac Electrophysiology, Translational Medicine Center, Changzheng Hospital, Second Military Medical University, Shanghai, China
2Department of Cardiology, Changhai Hospital, Second Military Medical University, Shanghai, China
3Department of Cardiology, Huadong Hospital, Fudan University, Shanghai, China
4Davis Heart & Lung Research Institute and Division of Cardiovascular Medicine, The Ohio State University Medical Center, Columbus, Ohio, USA
#Equally contribution as first author
*Corresponding authors: Xue Zhao, MD, PhD, Department of Cardiology, Third Affiliated Hospital, Second Military Medical University, 225 Changhai Road 200433, China, Fax: 021-635-20020, Tel: 021-818-85294, E-mail: xuezhao_cn@163.com
Jianqiang Hu, MD, Department of Cardiology, Changhai Hospital, Second Military Medical University, 168 Changhai Road, Shanghai 200433, China, Tel: 021-311-61263, E-mail: hujianqiang@medmail.com.cn
Int J Clin Cardiol, IJCC-3-073, (Volume 3, Issue 1), Research Article; ISSN: 2378-2951
Received: November 22, 2015 | Accepted: March 12, 2016 | Published: March 16, 2016
Citation: Zhao X, Zhu Y, Mei X, Zhang J, Hu J, et al. (2016) Radial-Linear Ablation in Peripulmonary Vein Area Prevents Inducible Atrial Fibrillation in Dogs. Int J Clin Cardiol 3:073. 10.23937/2378-2951/1410073
Copyright: © 2016 Zhao X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aims: Substrate abnormality in pulmonary vein (PV) antrum plays a critical role in the initiation and maintenance of atrial fibrillation (AF). The present study aimed at examining the efficacy of PV antrum radial-linear ablation for the prevention of inducible AF.
Methods and Results: A total of 22 dogs of both sexes (5-7 years old) were divided into two groups: 10 in control and 12 in ablation group. Multiple radial-linear lesions were created from PV orifice to peri-PV area in the dogs with CARTO guidance. The control animals went through the same procedure except ablation. AF inducibility was evaluated before and immediately after the procedure. All animals had inducible AF (100%) by atrial pacing combined with or without isoproterenol at baseline. After ablation, AF was induced in 5/12 dogs (41.7%) in the ablation group, and in 10/10 dogs (100%) for the control (p < 0.01). The frequency and duration of inducible AF were significantly decreased, and AF cycle length increased significantly in the animals with inducible AF after ablation over the baseline, while no changes were observed in the control. The ablation lesion scars in peri-PV area were confirmed pathologically with no PV stenosis.
Conclusion: This study suggested that the efficacy of radial-linear ablation in peri-PV area of dogs was consistent with clinical outcomes in paroxysmal AF ablation, and provided the basis for clinical studies in patients who needed invasive cardiac procedures or had significant risk for AF to verify the efficacy of this ablative technique on preventing AF.
Keywords
Atrial fibrillation, Catheter ablation, Dog, Pulmonary vein, Left atrium
Introduction
Pulmonary vein (PV) plays a critical role in the initiation and maintenance of atrial fibrillation (AF) [1]. Multiple reentrant circuits in the PV antrum area have been proposed to be the substrate for AF [1-3]. In a computer-assisted canine model, the reentrant circuits have been demonstrated to be dependent on heterogeneous and anisotropic conduction [4].
Despite the considerable body of clinical evidence which strongly support the importance of PV isolation (PVI), there is also a growing body of evidence which challenges the well-established concept of PVI as the cornerstone of AF ablation [5]. Narayan et al. have developed an ablation strategy which maps and targets localized sources (electric rotors and focal impulses) [6]. The use of this ablation strategy in conjunction with PVI resulted in improved efficacy as compared with PVI alone [6]. Recently, we demonstrated that radial-linear ablation in the PV antrial area in patients with paroxysmal AF was an effective strategy to treating the patient with paroxysmal AF with significant decrease in the recurrence of AF and left atrial remodeling [7], suggesting that the linear ablation effectively modified the substrate for AF in PV antria. This study aimed to explore if the radial-linear ablation in the PV antrum area could be also effective on primary prevention of AF.
Methods
Animal
Twenty two adult mongrel dogs of either sex, weighing 20 to 34 kg, at the age of 5 to 7 years old were purchased from the Laboratory Animal Center of Second Military Medical University. The animals were hospitalized in the experimental Clinic for at least one day before the procedure. Exclusion criteria were presence or history of any respiratory disease and thoracic surgery. All the animal experiments were performed in accordance with the Guidelines of the Animal Care Committee of the Second Military Medical University in Shanghai. The experimental protocols for the present study were reviewed and approved by the Animal Care Committee of the Second Military Medical University in Shanghai. Each animal was anesthetized with intramuscular injection of 3% sodium pentobarbital (40 mg/kg) at first followed by peritoneal injection at the same dose every two hours during the procedure. The animal was intubated with a cuffed endotracheal tube, and ventilated with a pressure-controlled ventilator. Heart rate, respiratory rate, pulse oximetry, temperature, and limb-lead ECG were continually monitored. The femoral artery was accessed for continuous pressure monitoring.
Study protocol
A 6F quadripolar catheter (CRD-2, St Jude Medical, St Paul, MN, USA) was positioned at the high right atrium via right femoral vein, and a 6F decapolar deflectable-tip catheter (Curve D, Biosense Webster) in the coronary sinus via right external jugular vein. The dogs included in the present study had to have at least 5 episodes of non-sustained AF or one episode of sustained AF after 10 attempts of burst atrial pacing with or without isoproterenol from CS or high RA catheter. The episode rate of inducible non-sustained AF was calculated using the formula of [(total numbers of AF episodes/10 attempts) × 100 %]. A total of 22 dogs in the present study were randomly divided into two groups: ablation group (12 dogs) and control group (10 dogs) (Figure 1). The animals in ablation group underwent radial-linear ablation with CARTO guidance, while the dogs in control group experienced the same procedure without ablation.
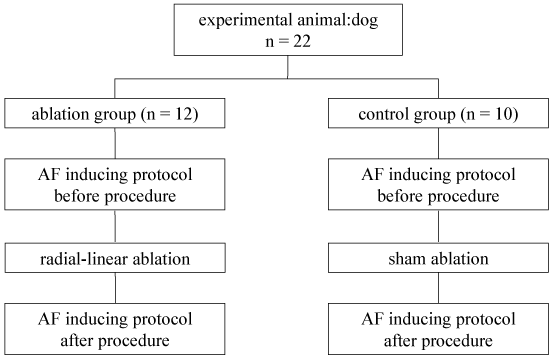
.
Figure 1: Study design and experimental protocol in the dogs. AF: atril fibrillation.
View Figure 1
![]()
Table 1: AF inducing protocol before surgery.
View Table 1
Lesion design for radial-linear ablation in peri-PV area
Radial-linear ablation lines were designed in the peri-PV area in the animals based on the morphology of the PVs defined by PV angiography. The local electrogram in the ablation target areas was the combination of potentials from PV and LA which was characterized by a wide wave of electrical activities rather than a single PV potential or LA potential. Radial-linear lesions were created with radiofrequency ablation, starting from PV orifice towards PV-LA junction guided by local electrogram, and ending at the site with single LA potential. Six to seven linear lesions were produced in both left and right peri-PV areas.
Induction of AF
AF was induced in the animals using atrial burst pacing protocol as described previously [8-10]. The minimum 1:1 atrial capture cycle length was identified and used to determine the pacing frequency. Train of burst pacing with 2 × threshold, pulse width of 2 ms, and duration of 30 seconds was delivered for the induction of AF. A total of 10 attempts of burst pacing were introduced with 5 attempts from coronary sinus and 5 attempts from peri-PV area. Irregular atrial tachyarrhythmia of ≥ 5 seconds but < 30 min was defined as induced non-sustained AF, and an induced AF episode of ≥ 30 min as sustained AF. While there had been different criteria to define non-sustained AF in the literature with the shortest duration from 2 seconds to 10 seconds, rapid and irregular atrial activity with duration of ≥ 5 seconds but < 30 min was defined as induced non-sustained AF in the present study that was consistent with published methods [11,12]. Atrial tachycardia (AT) was diagnosed when there was an abrupt increase by more than 50 bpm in the atrial rate to over 200 bpm that persisted for at least 10 seconds [13].
Ablation procedure
After a single transseptal puncture with a Brockenbrough needle, the transseptal sheath was placed into the LA, followed by PV angiography. A 3D LA geometry was reconstructed with CARTO system (Carto XP Navigation System, Biosense-Webster, Diamond Bar, CA). A mapping catheter in coronary sinus was used to record LA electrical activities and to pace. After transseptal puncture, intravenous heparin was infused to maintain an activated clotting time (ACT) between 300 and 400 seconds. Radiofrequency energy was delivered to the peri-vein area at a maximum temperature of 43°C, a maximum power of 35 W and 17 ml/min saline flow for 30 seconds at each site with a 3.5 mm irrigated-tip ablation catheter (ThermoCool, Biosense-Webster). Occasionally, the power was increased to 40 W to achieve the desired voltage reduction in some areas especially near the left atrial appendage. If for some reason the voltage reduction criteria were not met with increased power of 40 W, the saline flow could be increased up to 25 ml/min. A lesion creation at each point was considered to be successfully completed when the local bipolar voltage was decreased by 90% or to less than 0.05 mV. The linear lesions started at the PV orifice and ended at the PV-LA junction. The continuity of the linear lesions was confirmed by moving the ablation catheter back and forth along the ablation lines to check the local voltage for two times, and then pacing capture was not produced along the ablation line [14]. The procedural end point was the completion of the designed radial-linear lines of lesions. If AF was present or initiated during ablation, ablation would continue until the designed radial-linear lines were completed. Before and after ablation, the PV electrograms were recorded with a Lasso catheter (Biosense-Webster), attempts of burst pacing were made to determine the inducibility of AF using the same pacing protocol for AF induction. After the procedures were finished and the data obtained as mentioned above, the animals were humanely euthanized immediately with overdose of anaesthetic. The ablation lesions in left atrium (LA) and PV were examined histologically.
Statistics
The measurement data were expressed as mean ± SD and analyzed by t test. The measurement data was analyzed with Fisher's exact test. The data for the duration of inducible AF was expressed as median and analyzed by non-parametric test due to its non-normal distribution. The difference was considered statistically significant when a p value was < 0.05.
Results
All 22 dogs had inducible AF (100%) at baseline with non-sustained AF in 19/22 (86.4%) dogs and sustained AF in 3/22 (13.6%) dogs (Table 1). A total of 12 dogs in the ablation group underwent radial-linear ablation with CARTO guidance. Representative CARTO images of radial-linear ablation were shown in Figure 2. After ablation, AF in the ablation group was inducible in 5/12 (41.7%) dogs, while all the 10 animals in the control group (10/10 or 100%) had inducible AF (p < 0.01). One run of AT was induced in each of 4 dogs without AF in the ablation group, all of which were converted to sinus rhythm with overdrive pacing. Sustained AF occurred in 3 animals at baseline was terminated during ablation. Only 1 episode of non-sustained AF was induced in 1 of these three dogs, and 1 run of AT in another one post ablation. Although AF was still inducible in 5 dogs after ablation, the episode rate of inducible AF were significantly decreased along with increased AF cycle length in the animals after ablation compared to the baseline and to the control group (p < 0.01) (Figure 3 and Figure 4). The median (interquartile range) duration of inducible AF was 32 (1378 - 9) second at baseline and 0 (18-0) second post-procedure in ablation group (p < 0.001), and was 31 (50-11) second at baseline and 41 (68-31) second post-procedure in control group. In control group, no significant differences were observed in the episode rate and duration as well as cycle length of inducible AF (p > 0.05) after procedure over the baseline (Figure 3 and Figure 4). One dog developed cardiac tamponade requiring pericardiocentesis during the procedure, and no electric isolation occurred to all the PVs in the ablation group.
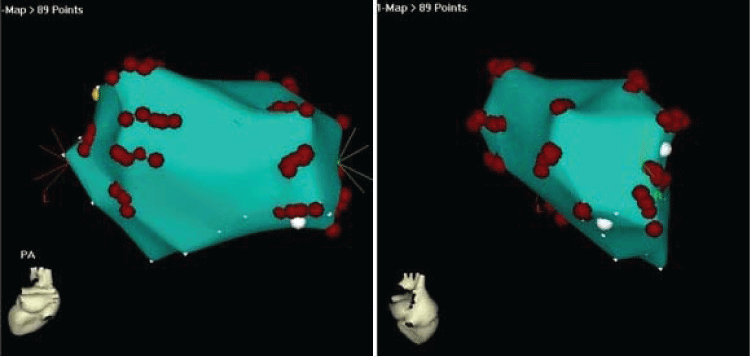
.
Figure 2: Representative CARTO images of LA with radial-linear ablations in peri-PV area. Left panel: posterior-anterior view, Right panel: right lateral view. Red dots indicated the ablation sites.
View Figure 2
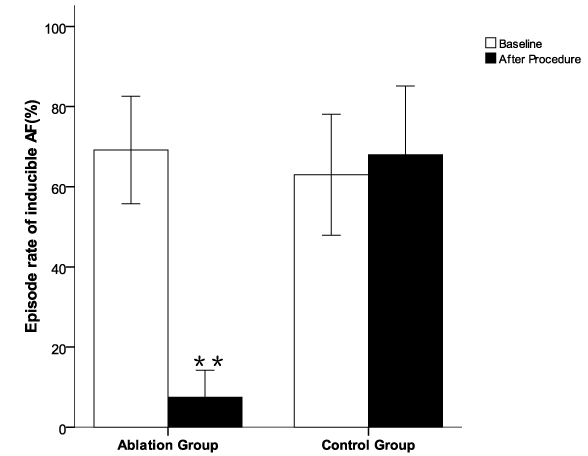
.
Figure 3: The episode rate of acutely induced atrial fibrillation by burst pacing from CS or high RA in the dogs before and post radial-linear ablation. Irregular atrial tachyarrhythmia of ≥ 5s but < 30 min was defined as induced non-sustained AF, and an induced AF episode of ≥ 30 min as sustained AF. The episode rate of inducible non-sustained AF was calculated using the formula of [(total numbers of AF episodes/10 attempts) × 100 %]. The episode rate of inducible AF was significantly decreased after ablation compared to the baseline and to the control group **p < 0.01 vs baseline in ablation group and post procedure in control group.
View Figure 3
No dogs died as a direct result of the intervention. No PV stenosis or LA thrombus was observed during pathological examination in the animals 2 ~ 4 weeks after the ablation as examined under direct vision. Chronic linear lesion scars were observed in the peri-PV areas. Microscopic examination of ablation lesion along longitudinal PV axis shows fibrotic tissue in the peri-PV area (Figure 5).
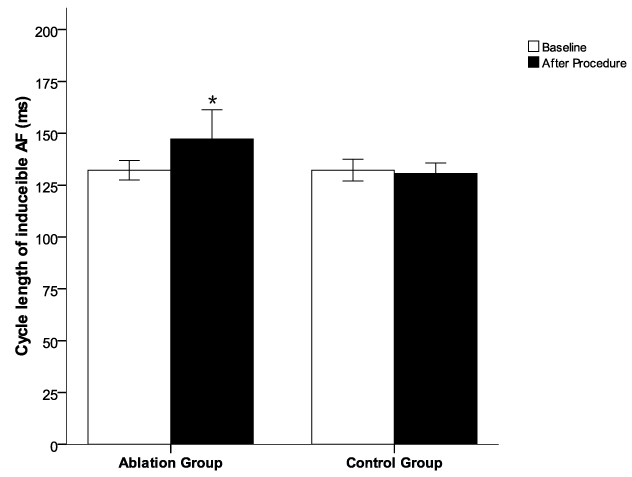
.
Figure 4: The cycle length (CL) of acutely induced atrial fibrillation by burst pacing from CS or high RA in the dogs before and post procedure. The AF cycle length was increased in the animals post procedure in ablation group compared to the baseline in ablation group and to the control group. *p < 0.05 vs baseline in ablation group and control group.
View Figure 4
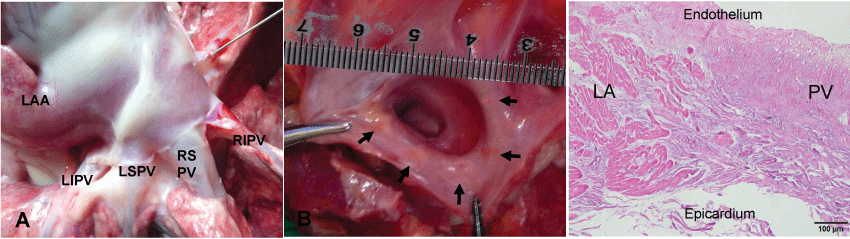
.
Figure 5: Pathological examination of LA showing linear scar tissue formation in peri-PV area after ablation. (A) Outside of left atrium in control group; (B) Inside of left atrium in ablation group. Chronic linear lesion scars were observed in the peri- PV area as indicated with the arrows, which were produced by radial-linear ablation; (C) Historical ablaton lesion. Microscopic examination of ablation lesion along longitudinal PV axis shows fibrotic tissue in the peri-PV area (HE staining).
PV: Pulmonary Vein; LA: Left Atrium; LAA: Left Atrial Appendage; LIPV: Left Inferior Pulmonary Vein; LSPV: Left Superior Pulmonary Vein; RIPV: Right Inferior Pulmonary Vein; RSPV: Right Superior Pulmonary Vein.
View Figure 5
Discussion
The main finding of the present study was that radial-linear ablation in peri-PV area significantly decreased the frequency and duration of inducible AF in dogs. These results were comparable to previous observation that incomplete PV isolation decreased AF duration and lengthened the AF cycle length in a similar canine AF model [9]. Rapid atrial pacing has been established to evaluate AF ablation outcome in dogs [9] and pigs [8]. In the present study, aging dogs with easily inducible AF using atrial pacing at baseline were enrolled to increase the susceptibility and reproducibility. Because the PV anatomy might be highly variable and the PV antrum could be difficult to identify in dogs, peri-PV area was selected as the ablation target as guided by the combination of anatomical mapping and local electrogram mapping. The morphology of the PVs was identified by PV angiography. The local electrogram was characterized by the combined potentials from both PV and LA in peri-PV area. It has been found that large atria are more likely than small atria to develop intra-atrial reentrant circuits [15], and PVs with large circumferences are correlated with increased reentrant events [4]. In an ovine model, the anatomic reentrant circuit has been identified as about 1 cm in diameter [2,16]. Based on multi-wavelets theory, perpetuation of AF requires the presence of a minimum number of coexisting wavelets, and is favored by slow conduction, shortened refractory periods, and increased atrial mass. Maintaining AF needs a certain number of active wavelets that require a critical excitable mass of atrial tissue. The surgical Maze procedure was developed based on this AF model. Based on the conduction velocity of 0.3 mm/ms and refractory period of 200 ms in atrium, the reentrant circuit is calculated to be about 6 cm in circumference and 1.5-2.0 cm in diameter in humans. When the PV antrum was divided with radial-linear lesions into several small regions, barriers were created to block the electrical conduction circumferentially in that area and could prevent the formation of reentrant circuits. In addition, the five major left atrial autonomic ganglionic plexi and ligament Marshall could be affected with the ablation lesions due to their anatomic locations [17].
This study suggested that the efficacy of radial-linear ablation in peri-PV area of dogs was consistent with clinical outcomes in paroxysmal AF ablation, and provided the basis for clinical studies in patients who needed invasive cardiac procedures or had significant risk for AF to verify the efficacy of this ablative technique on preventing AF.
Acknowledgement
This study was supported by the National Natural Science Foundation of China (NNSFC grant 30872423 and 81370442 to XZ). The authors thank Win-Kuang Shen, MD from Mayo Clinic, for his kind comments on the manuscript preparation.
References
-
Lee G, Spence S, Teh A, Goldblatt J, Kalman JM, et al. (2012) High-density epicardial mapping of the pulmonary vein-left atrial junction in humans: insights into mechanisms of pulmonary vein arrhythmogenesis. Heart Rhythm 9: 258-264.
-
Jalife J, Berenfeld O, Mansour M (2002) Mother rotors and fibrillatory conduction: a mechanism of atrial fibrillation. Cardiovasc Res 54: 204-216.
-
Dewire J, Calkins H (2010) State-of-the-art and emerging technologies for atrial fibrillation ablation. Nat Rev Cardiol 7: 129-138.
-
Cherry EM, Ehrlich JR, Nattel S, Fenton FH (2007) Pulmonary vein reentry--properties and size matter: insights from a computational analysis. Heart Rhythm 4: 1553-1562.
-
Calkins H (2013) Has the time come to abandon the concept that "pulmonary vein isolation is the cornerstone of atrial fibrillation ablation"? Circ Arrhythm Electrophysiol 6: 241-242.
-
Narayan SM, Krummen DE, Shivkumar K, Clopton P, Rappel WJ, et al. (2012) Treatment of atrial fibrillation by the ablation of localized sources: CONFIRM (Conventional Ablation for Atrial Fibrillation With or Without Focal Impulse and Rotor Modulation) trial. J Am Coll Cardiol 60: 628-636.
-
Zhao X, Zhang J, Hu J, Liao D, Zhu Y, et al. (2013) Pulmonary antrum radial-linear ablation for paroxysmal atrial fibrillation: interim analysis of a multicenter trial. Circ Arrhythm Electrophysiol 6: 310-317.
-
Ott P, Kirk MM, Koo C, He DS, Bhattacharya B, et al. (2007) Coronary sinus and fossa ovalis ablation: effect on interatrial conduction and atrial fibrillation. J Cardiovasc Electrophysiol 18: 310-317.
-
van Brakel TJ, Bolotin G, Nifong LW, Dekker AL, Allessie MA, et al. (2005) Robot-assisted epicardial ablation of the pulmonary veins: is a completed isolation necessary? Eur Heart J 26: 1321-1326.
-
Lemola K, Chartier D, Yeh YH, Dubuc M, Cartier R, et al. (2008) Pulmonary vein region ablation in experimental vagal atrial fibrillation: role of pulmonary veins versus autonomic ganglia. Circulation 117: 470-477.
-
Razavi M, Zhang S, Yang D, Sanders RA, Kar B, et al. (2005) Effects of pulmonary vein ablation on regional atrial vagal innervation and vulnerability to atrial fibrillation in dogs. J Cardiovasc Electrophysiol 16: 879-884.
-
Horikawa-Tanami T, Hirao K, Furukawa T, Isobe M (2007) Mechanism of the conversion of a pulmonary vein tachycardia to atrial fibrillation in normal canine hearts: role of autonomic nerve stimulation. J Cardiovasc Electrophysiol 18: 534-541.
-
Tan AY, Zhou S, Ogawa M, Song J, Chu M, et al. (2008) Neural mechanisms of paroxysmal atrial fibrillation and paroxysmal atrial tachycardia in ambulatory canines. Circulation 118: 916-925.
-
Steven D, Reddy VY, Inada K, Roberts-Thomson KC, Seiler J, et al. (2010) Loss of pace capture on the ablation line: a new marker for complete radiofrequency lesions to achieve pulmonary vein isolation. Heart Rhythm 7: 323-330.
-
Allessie M, Ausma J, Schotten U (2002) Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc Res 54: 230-246.
-
Fynn SP, Kalman JM (2004) Pulmonary veins: anatomy, electrophysiology, tachycardia, and fibrillation. Pacing Clin Electrophysiol 27: 1547-1559.
-
Calkins H, Kuck KH, Cappato R, Brugada J, Camm AJ, et al. (2012) 2012 HRS/EHRA/ECAS Expert Consensus Statement on Catheter and Surgical Ablation of Atrial Fibrillation: Recommendations for Patient Selection, Procedural Techniques, Patient Management and Follow-up, Definitions, Endpoints, and Research Trial Design. Heart Rhythm 9: 632-696.e21.





