International Journal of Clinical Cardiology
Coronary-Cameral Fistula Presenting as Worsening Refractory Heart Failure
Dhruti Mankodi1*, Ashish Sharma1, Fulvia Banu1, Sameer Shaharyar1, Pedro Martinez-Clark2 and Ali Bazzi2
1Department of Graduate Medical Education, Aventura Hospital and Medical Center, USA
2Cardiology Department, Aventura Hospital and Medical Center, USA
*Corresponding author: Dhruti Mankodi, MD, Department of Graduate Medical Education, Aventura Hospital and Medical Center, 20800 Biscayne Blvd, Aventura, FL 33180, USA, Tel: 306 319 9043, E-mail: dmnkodi@gmail.com
Int J Clin Cardiol, IJCC-3-086, (Volume 3, Issue 2), Case Report; ISSN: 2378-2951
Received: July 12, 2016 | Accepted: August 09, 2016 | Published: August 12, 2016
Citation: Mankodi D, Sharma A, Banu F, Shaharyar S, Martinez-Clark P, et al. (2016) Coronary-Cameral Fistula Presenting as Worsening Refractory Heart Failure. Int J Clin Cardiol 3:086. 10.23937/2378-2951/1410086
Copyright: © 2016 Mankodi D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Coronary-cameral fistula (CCF) is a rare abnormalitythatcan cause significant cardiac insufficiency. It is found in only 0.002% of the general population. Most of the abnormal communications originate from the right coronary artery, of which approximately 2% drain into left ventricle [1]. We present a case of such communication causing severe mitral regurgitation and refractory heart failure in a 49 year old female. The symptoms resolved with successful percutaneous closure of the fistula.
Introduction
Coronary-cameral fistula (CCF) is a rare condition most commonly caused by an embryologic developmental anomaly [1]. Fifty-five percent of CCFs originate from the right coronary artery, 35% from the left coronary artery, and 5% from both. More than 90% of them drain into the venous system and low pressure sites such as the right ventricle (41%), right atrium (26%), pulmonary artery (17%) and only 3% drain into the left ventricle or atrium [2,3]. We present a case of refractory systolic heart failure with severe mitral regurgitation, and atrial fibrillation with ventricular response as a result of a high flow CCF originating from the right posterolateral coronary artery terminating into the left ventricle.
Case Report
A forty-nine year old African American woman presented with orthopnea and dyspnea on exertion typical for NYHA class IV heart failure symptoms. She had an extensive history of end-stage renal disease (ESRD), and was on hemodialysis for the past seven years, had multiple hospital admissions for refractory systolic and diastolic heart failure over the past two years, chronic atrial fibrillation (A.fib), hypertension, and emphysema. On admission, the vital signs showed a BMI of 32.9, pulse of 140 bpm, blood pressure 138/98 mmHg and oxygen saturation at 92% on room air. Physical exam was pertinent for obesity, positive JVD, tachycardia with irregularly irregular rhythm, grade 2/6 systolic murmur at the apex, bibasilar crackles on lung auscultation and trace bilateral lower extremity pitting edema. There was a functional arterio-venous fistula on the left arm for hemodialysis. Electrocardiogram revealed A.fib with rapid ventricular response (RVR) of 136 bpm. Labs revealed B-natiuretic peptide (BNP) of 28,000 and there was mild pulmonary edema visible on chest X-ray. The patient reported compliance with her hemodialysis schedule, fluid, and diet intake. The patient was treated with diuretics and started on a Diltiazemdrip without any resolution. She also received hemodialysis during her inpatient stay, as per her routine schedule. A chart review indicated that she had been on optimal therapy for CHF and A.fib for over 2 years with constant decline in left ventricular (LV) function despite being on hemodialysis. Transthoracic echocardiography (TTE) revealed an ejection fraction of 10-20% which was 20% less than previous TTE done a year ago. She also had moderate to severe mitral regurgitation (MR) and left atrial dilatation. A cardiac catheterization was performed to further evaluate the worsening of LV function. It revealed a coronary congenital abnormality with fistulous formation from the right coronary posterolateral branch to the LV cavity near the mitral valve annulus which was contributing to worsening of mitral regurgitation. The fistulous formation was consistent with Sakakibara Type B however no coronary dilatation was noted on the angiography. A transcatheter closure of the fistula was recommended. However, before we could proceed, the patient developed bacteremia and further decompensation of CHF due to uncontrolled A.fib with RVR. A transesophageal echocardiography (TEE) was done which did not reveal a thrombus or any evidence of endocarditis; thus, a cardioversion was performed. The patient was converted to normal sinus rhythm but continued to be volume overloaded. Once the bacteremia was resolved, the patient underwent another cardiac catheterization with JR4 guide. A SL10 neural interventional catheter and 300 cm Pilot 50 wire was run over to the fistulous communication between the right coronary artery and the left ventricular cavity size. This was done under coronary angiography using regular contrast. Subsequently, the catheter was placed in the most proximal portion of the fistulous connection in a 2-0 mm Stryker neuro coil was delivered successfully with complete obliteration and closure of the fistulous communication. There were no peri-procedure complications.
One week after the procedure, the patient's symptoms of volume overload resolved and she remained in normal sinus rhythm. She was discharged with medical therapy which included Losartan, Hydralazine, Warfarin, Clopidogrel and Carvedilol. A transthoracic echo performed six months after the procedure revealed an improved left ventricular ejection fraction of 25% and reduction in mitral valve regurgitation to mild. Over the past year and a half since the procedure, our patient has remained to be at NYHA class II and has had significantly less number of hospitalizations for heart failure exacerbation (Figure 1).
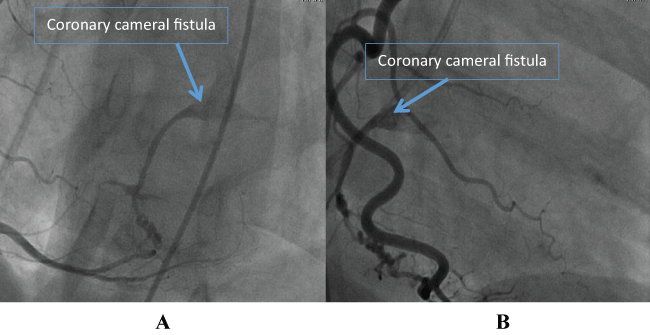
.
Figure 1A and B: Two different views of the posterolateral branch of right coronary artery forming a fistulous connection (blue arrow) that drains into the left ventricular cavity.
View Figure 1
Discussion
We present a rare case of right postero-lateral coronary artery to LV fistula causing worsening refractory heart failure. Coronary cameral fistulae most commonly arise from RCA but rarely drain into the left ventricle. Such a communication has only been reported a few times in the literature and most such cases are suspected to be acquired [4]. Hence as per our review, thisis thefirst case that reports a symptomatic congenital RCA to LV fistula. The fistula in our patient, terminated below the mitral valve annulus which was suspected to contribute to a rise in LV filling pressures and severe MR leading to atrial tachycardia. Most coronary cameral fistulae are asymptomatic but from the ones that are symptomatic, most commonly present with either chest pain or heart failure. However tachycardia or arrhythmia has been reported in one of the cases of CCF originating from RCA and draining in LV [4]. CCF most commonly causes a myocardial steal phenomenon, which becomes symptomatic late in adulthood [5]. Normally, blood flow follows the path of least resistance, but when the drainage site of the fistula is in a left heart chamber it creates a left-to-left shunt, mimicking aortic valve regurgitation that overloads the left heart [6]. Increase in LV end diastolic pressure, LV hypertrophy, congestive heart failure, and arrhythmias are the most common complications that can develop from a CCF [7,8]. A continuous murmur at the left lower sternal border and/or a collapsing pulse may be appreciated on physical exam for fistulas to left heart chambers [9]. However, coronary angiography remains the definitive diagnostic tool. Computed tomography (CT) and magnetic resonance imaging (MRI) can also be used to detect CCFs and are increasingly being applied [5].
Treatment options for such a condition include surgical ligation with or without coronary artery bypass grafting, or percutaneous trans-catheter closure. Although surgical obliteration of the fistula is the most effective treatment, both techniques result in good prognosis [5,10]. Some of the contraindications for the transcatheter technique include large or wide size of the fistula, multiple communications, distal location of fistula, adjacent vessels at risk, and it is not recommend for use in young patients. Also, if the patient requires surgical repair for any other concomitant anomaly, anopen procedure should be performed [11]. Major complications associated with such techniques include perforation, ventricular arrhythmias, and coronary artery spasm [5].
Conclusion
Clinicians should maintain high suspicion for CCF in patients with refractory heart failure. Presence of continuous murmur at left sternal border is the most suggesting sign for CCF in adults however it may not be present in all cases. The present case describes an extremely rare coronary cameral fistula, and provides a brief overview of the subject. Diagnosis is usually made by imaging, and treatment involves mechanical closure. Coronary cameral fistulas should be considered in the differential diagnosis of refractory heart failure, in order to aid early diagnosis and avoid life threatening complications.
References
-
Dodge-Khatami A, Mavroudis C, and Backer CL (2000) Congenital Heart Surgery Nomenclature and Database Project: anomalies of the coronary arteries. Ann Thorac Surg 69: 270-297.
-
Basit A, Nazir R, Agarwal A (2013) Unusual case of surgically failed coronary-cameral fistula: successful outcome with transradial coil embolization. J Invasive Cardiol 25: 56-57.
-
Hauser M (2005) Congenital anomalies of the coronary arteries. Heart 91: 1240-1245.
-
Said SA, el Gamal MI, van der Werf T (1997) Coronary arteriovenous fistulas: collective review and management of six new cases-changing etiology, presentation, and treatment strategy. Clinic Cardiol 20: 748-752.
-
Mangukia CV (2012) Coronary artery fistula. Ann Thorac Surg 93: 2084-2092.
-
Sommer RJ, Hijazi ZM, Rhodes JF Jr (2008) Pathophysiology of congenital heart disease in the adult: part I: Shunt lesions. Circulation 117: 1090-1099.
-
Kugelmass, Manning WJ, Piana RN, Weintraub RM, Baim DS, et al. (1992) Coronary arteriovenous fistula presenting as congestive heart failure. Cathet Cardiovasc Diagn 26: 19-25.
-
Liberthson, Sagar K, Berkoben JP, Weintraub RM, Levine FH (1979) Congenital coronary arteriovenous fistula. Report of 13 patients, review of the literature and delineation of management. Circulation 59: 849-854.
-
Ogden JA, Stansel HC Jr (1972) Coronary arterial fistulas terminating in the coronary venous system. J Thorac Cardiovasc Surg 63: 172-182.
-
Dimitrakakis, Von Oppell U, Luckraz H, Groves P (2008) Surgical repair of triple coronary-pulmonary artery fistulae with associated atrial septal defect and aortic valve regurgitation. Interact Cardiovasc Thoracic Surg 7: 933-934.
-
Mavroudis C, Backer CL, Rocchini AP, Muster AJ, Gevitz M (1997) Coronary artery fistulas in infants and children: a surgical review and discussion of coil embolization. Ann Thorac Surg 63: 1235-1242.





