International Journal of Clinical Cardiology
Early Therapy with Beta Blockers Plus Ivabradine Versus Beta Blockers Alone in Patients Hospitalised with Heart Failure and Reduced Ejection Fraction (ETHIC-AHF Study): Results at One-Year Follow-Up
Francisco J Hidalgo*, Francisco Carrasco, Juan C Castillo, Sara Rodríguez, Laura Pardo, Enrique Durán, Carlos Ferreiro, José Sánchez, Mónica Delgado, Dolores Mesa, Martín Ruiz and Manuel Anguita
Department of Cardiology, Hospital Universitario Reina Sofía, Spain
*Corresponding author: Dr. Francisco J Hidalgo Lesmes, Department of Cardiology, Hospital Universitario Reina Sofía, Avda, Menéndez Pidal 1, 14004, Córdoba, Spain, Fax: +34957760237, E-mail: fjhl.87@gmail.com
Int J Clin Cardiol, IJCC-4-093, (Volume 4, Issue 1), Original Article; ISSN: 2378-2951
Received: September 28, 2016 | Accepted: March 02, 2017 | Published: March 06, 2017
Citation: Hidalgo FJ, Carrasco F, Castillo JC, Rodríguez S, Pardo L, et al. (2017) Early Therapy with Beta Blockers Plus Ivabradine Versus Beta Blockers Alone in Patients Hospitalised with Heart Failure and Reduced Ejection Fraction (ETHIC-AHF Study): Results at One-Year Follow-Up. Int J Clin Cardiol 4:093. 10.23937/2378-2951/1410093
Copyright: © 2017 Hidalgo FJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Heart rate (Hr) has prognostic value in patients with heart failure, depressed ejection fraction (HFrEF) and sinus rhythm.
Objective: Is to analyze the effect of the early co-administration of ivabradine added to beta-blockers (intervention group) versus only beta blockers (control group) in patients hospitalized with HFrEF on Hr and clinical and functional outcomes at long term.
Methods: We have analysed the one-year follow-up results of a randomized study comparing these two treatment strategies after hospitalization for acute HFrEF, sinus rhythm and Hr > 70 bpm, following the usual titration doses of both drugs.
Results: 33 patients in the intervention group and 38 in Control group were included. Hr at 28 days was lower in the intervention group (64.3 ± 7.5 versus 70.3 ± 9.3 bpm, p = 0.01), keeping this difference at one year (61.8 ± 5.5 versus 68.4 ± 9.3 bpm, p = 0.01). The doses of beta-blockers at one year were similar in both groups. The EF at one year was significantly higher in the intervention group (48.2 ± 17 versus 41.8 ± 10%, p = 0.002). The probability of cardiovascular death was 26% lower in the ivabradine group, although without reaching statistical significance (HR 0.74; IC 95%: 0.12-4.43). No severe side effects drugs were observed.
Conclusion: Early co-administration of ivabradine and beta-blockers during hospital admission for acute HFrEF, sinus rhythm and Hr > 70 bpm is feasible and safe, and it produced a significant decrease in Hr at 28 days post-discharge and at 1 year after hospital discharge, as well as an improvement in left ventricular systolic function.
Introduction and Objectives
Heart failure (HF) is a clinical syndrome with enormous relevance, given its constantly growing prevalence and its poor prognosis [1,2]. Within this group, approximately half of the patients have reduced left ventricular ejection fraction (HFrEF) [1]. Heart rate (HR) seems to have prognostic value in patients with HFrEF and sinus rhythm. A directly proportional relationship between HR reduction and prognostic improvement has been observed [3,4]. Recent clinical practice guidelines have set HR target values (< 70 bpm) and have included a pure heart rate slowing drug in the therapeutic algorithm (ivabradine), a specific inhibitor of the If-current in the sinus node [5], for treatment of patients that already received an optimal dose of beta-blockers or the maximum-tolerated dose of these drugs [6,7] and continued having a HR > 70 bpm. Ivabradine has shown to have a prognostic benefit and to improve functional parameters, even in patients with chronic severe systolic HF [8].
Some studies have reported that there is a relationship between the reduction of HR and the use of beta-blockers, but no correlation has been found with the dose of these drugs [9-11]. Moreover, it's also known that the percentage of patients who achieve optimal beta-blockers doses and optimal HR values with beta-blocker administration is low [12]. Ivabradine not only has been recognized as having a prognostic benefit, but a prognostic significance for the time required to achieve HR control has also been found, as seen in the SHIFT study [4]. Therefore, introducing and up-titrating drugs early during the vulnerable phase of post-hospitalization can be important in order to reduce mortality and early re-hospitalizations. Current indication of ivabradine is based on the SHIFT design, but until the recent publication of ETHIC-AHF study [13], there were no data about the safety and potential benefits of its use during hospitalization, added to beta-blockers. The aim of this study is to present the results a tone year follow-up of patients included in the ETHIC-AHF study to evaluate if the positive results obtained in the short-term follow-up after discharge were maintained at long-term.
Methods
The ETHIC-AHF study, acronym of "Early Therapy with Ivabradine in patients with Congestive Acute Heart Failure", whose protocol, design and initial results have recently been published [13], it's a prospective, comparative and randomized trial with a simple randomization strategy in which we compared the current strategy recommended by the current clinical practice guidelines [6,7], i.e. to use beta-blockers in increasing doses and to add ivabradine only in those patients who after reaching the optimal dose or the maximum-tolerated dose of beta-blockers, persisted with heart rate > 70 bpm (control group), versus the strategy of simultaneous and early start, 24-48 hours after heart failure admission, of beta-blockers and ivabradine, with simultaneous and progressive up-titration of both (intervention group). The details of the study design, the inclusion and exclusion criteria, and the drug titration protocol have already been presented [13]. In summer, patients who met these criteria and granted informed consent were randomized within 24-48 hours after admission to the usual pattern of beta-blockers (control group) or to received ivabradine, at a dose of 5 mg/12 hours, plus the beta-blocker (carvedilol or bisoprolol) (intervention group). After discharge, dose titration of beta-blockers was performed in visits at 14 days, 28 days, 4 months, 8 months and 1 year in both groups, with the objective to reach a HR < 70 bpm and about 60 bpm. Ivabradine dose could be increased to 7.5 mg/12 hours in each visit in the intervention group, and could be added to beta-blocker after the 28 days follow-up visit in the control group if HR was > 70 bpm and the patient had received the optimal dose of beta-blockers (10 mg/day of bisoprolol or 25 mg/12 hours of carvedilol) or the maximum-tolerated dose of these drugs.
Heart rate at 28 days after discharge was the primary outcome [13]. Secondary outcomes consisted in the evolution of HR at 4 months and 1 year, left ventricular ejection fraction, BNP levels, NYHA functional class, drug safety (adverse effects and withdrawal from medication due to adverse effects), and major clinical cardiac events (cardiac death and readmissions for heart failure) at 1 year after discharge. Patients were subjected to follow-up evaluation at 15 days, 28 days, 4 months, 8 months and 1 year after discharge. The scope of the previously published article [13] was mainly to analyse the effect of the two strategies on the primary objective (HR at 28 days after discharge) and results at 4 months, but it seems also important to evaluate if the short-term effects of the early use of ivabradine were maintained at long-term (1 year), and this is the scope of the present article. We also want to know the effect of the early use of ivabradine in acute heart failure on mortality and readmissions at one year after discharge.
For statistical analysis, qualitative variables were expressed as absolute numbers and percentages, and the quantitative variables were expressed as mean ± standard deviation. To determine the relationship between quantitative variables, we used Student's t-test for independent data and analysis of variance (ANOVA) for paired data, as appropriate. To determine the relationship between qualitative variables, we used the chi-square test. Events during follow-up were analysed by Kaplan-Meier curves and compared using the log-rank test. In all cases, differences were considered statistically significant if p < 0.05. We used the IBM®-SPSS (version 21.0 for Macintosh, SPSS Corp., New York, Armonk). To calculate the sample size, we used the Epidat 3.1 program, with a confidence level of 80%, a 5% percentage of error, and an objective of reducing the heart rate of the control group by 15%. We obtained a sample size of 72 patients.
Results
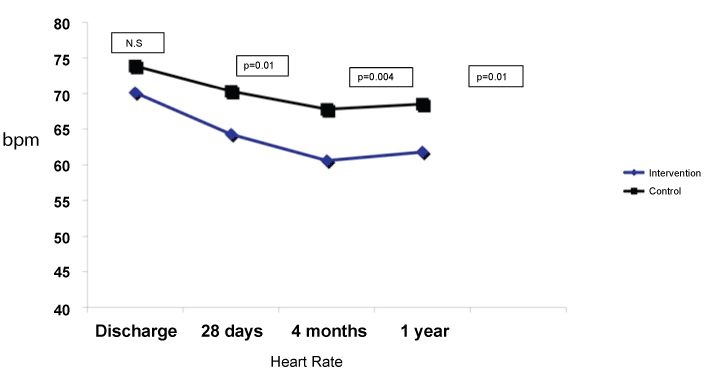
Between November 2013 and April 2015, 72 patients were included, one of which was removed early before discharge after removing his consent. Therefore, 71 patients were analysed, 33 in the intervention group and 38 in the control group. All of them could complete the planned follow-up. The flowchart of patients included and excluded and the reasons for it have been published [13], as well as baseline characteristics and treatment received during hospitalization and at discharge [13]. Table 1 summarizes the most important characteristics of both groups, showing no differences between both groups. There were no differences in diuretics use, AICEs/ARB or MRA [13]. Heart rate at 28 days after hospital discharge was significantly lower in the intervention group, staying this difference at 4 months (Figure 1).
![]()
Table 1: Summary of the main characteristics of the two groups during hospitalization.
View Table 1
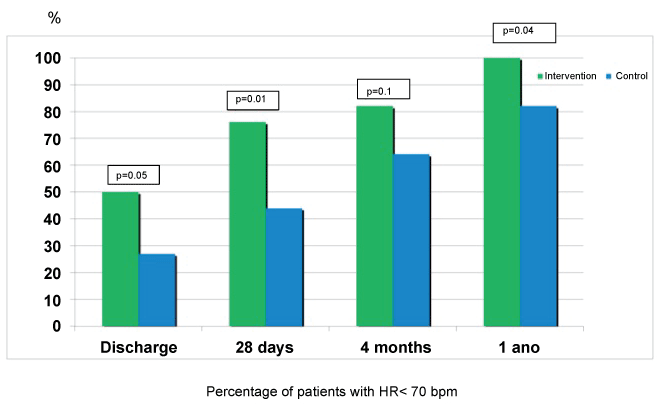
At one year, heart rate remained significantly lower in the intervention group (61.8 ± 5.5 versus 68.5 ± 9.3 bpm, p = 0.01), as it is shown in figure 1. No differences in the percentage of patients with medium/high doses of beta-blockers were found at one-year follow-up (58 versus 60%, p = 0.86). Regarding the use of ivabradine at one-year follow-up, 22 patients in the intervention group (81.5%) were receiving ivabradine (8 patients 5 mg/12 hours and 14 patients 7.5 mg/12 hours, with a mean dose of 13.2 ± 2 mg/day), versus 11 patients (33.3%) in the control group (9 patients 5 mg/12 hours and 2 patients 7.5 mg/12 hours, with a mean dose of 10.9 ± 1.7 mg/day, p < 0.001). No severe adverse effects related to the drugs used (beta-blockers or ivabradine) that forced their withdrawal were recorded, although ivabradine was discontinued in 3 patients to continue titrating beta-blockers due to heart rate less than 60 bpm (2 patients in the control group and 1 patient in the intervention group), and its dose was reduced in 1 patient of the intervention group (from 7.5 mg/12 hours to 5 mg/12 hours), to allow increased dose of beta-blocker. All patients in intervention group had a HR < 70 bpm at one year follow-up for 82% in the control group (p = 0.04) (Figure 2).
.
Figure 2: Percentage of patients in both groups with heart rate lower than 70 bpm.
View Figure 2
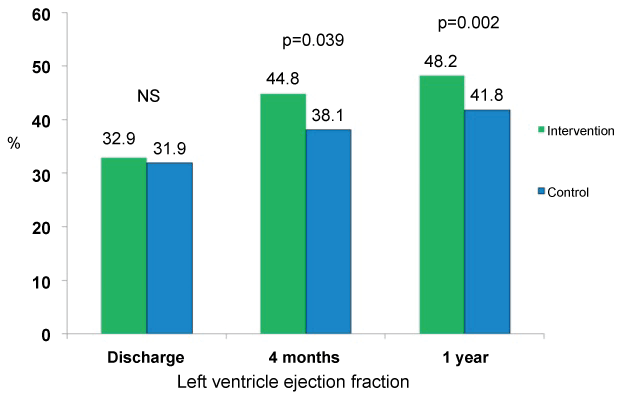
Left ventricular ejection fraction, similar in both groups at admissions and discharge [13], was significantly higher at 1-year follow-up in the intervention group (48.2 ± 17 versus 41.8 ± 10 %, p = 0.002) (Figure 3). BNP values were significantly lower in the intervention group at 4 months follow-up, but no significant differences between both groups were recorded at one year follow-up (300.9 ± 117.4 versus 484.3 ± 137 pg/ml, p = 0.3). A trend to a lower percentage of patient in NYHA class III or IV ambulatory was observed in the intervention group, although without reaching statistical significance (14.8 versus 21.2 %, p = 0.5).
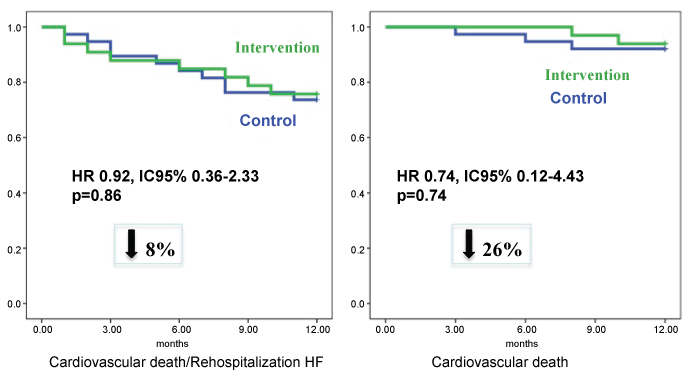
The rate of clinical events at one-year follow-up was similar in both groups. The probability of cardiovascular death was 26% lower in the ivabradine group, although without statistical significance (HR 0.74; IC 95%: 0.12-4.43) (Figure 4), and the incidence of cardiovascular death or heart failure re-hospitalizations was similar (Figure 4). Total mortality was similar in both groups too (10.5 versus 15.5%, NS). During follow-up, three patients of intervention group and three patients of control group received electric devices (Cardiac Resynchronization Therapy-CRT or Implantable Cardiac Defibrillator). One patient in the intervention group who remained in ambulatory functional class IV despite optimal medical therapy, have severe systolic biventricular dysfunction and was not candidate to CRT, was successfully transplanted. At one-year follow-up, we did not record any severe adverse effects related to the used drugs.
.
Figure 4: Probability at one year follow-up of not suffering cardiovascular death and/or hospitalization for heart failure (left), and of not suffering cardiovascular death (right) in both treatment groups.
View Figure 4
Discussion
Control of heart rate is an increasing priority in patients with HFrEF, due to improved prognosis in terms of mortality and heart failure events [3,4]. Current practice guidelines [6,7] recommend in patients with ambulatory HFrEF and sinus rhythm, Hr < 70 bpm, receiving optimal doses of beta-blockers (I A indication) the addition of ivabradine for patients who nevertheless fail this objective (indication IIa B). However, no data on the usefulness of HR control in the most vulnerable stage of the disease, i.e. in patients hospitalized for an episode of acute heart failure. The ETHIC-AHF study, recently published [13], carried out in patients with acute heart failure, has demonstrated the safety and efficacy of the early administration of ivabradine plus beta-blockers during hospitalization, resulting in a significant reduction in heart rate, increased in left ventricular ejection fraction, reduced BNP levels and a trend to a better functional class at 4 months after discharge [13].
In this article, we present the results of one-year follow-up of this study, which confirm the initial results observed in the short-term. Heart rate remained significantly lower in the intervention group treated during admission with ivabradine and beta-blockers (Figure 1), and this was despite a similar rate of use of beta-blockers and ivabradine in both groups at one year. The percentage of patients with heart rate lower than 70 bpm was 100% in the intervention group versus 82% in the control group (Figure 2). Left ventricular ejection fraction was significantly higher in patients initially randomized to ivabradine plus beta-blockers, being this difference higher than that obtained at 4 months (Figure 3). Also, the tendency to a lower symptoms severity persisted at one year. Both groups had a similar incidence of cardiovascular events, although cardiovascular death was 26% lower in the ivabradine group at one-year follow-up (not significant difference, probably related to the small sample size and the low incidence of events (Figure 4).
These results, with a 12 months follow-up, demonstrate the safety of the combined use of ivabradine plus beta-blockers early during hospitalization in patients with acute HFrEF, chronic or de novo, achieving a better control of heart rate, in similar percentages than reported in other studies with up-titration of only beta-blockers [9-11]. In addition, this combination therapy appears to improve left ventricular ejection fraction, as already reported in the SHIFT study in stable patients [14,15]. As far as we know, the ETHIC-AHF [13] study is the first analysis of the safety and benefit of using ivabradine in this clinical context. The results reported in this article reinforce the idea that it is possible to improve the treatment of patients during acute phase, the most vulnerable and which confers an increased risk of short and medium term events, as stated in the new heart failure guidelines [6]. Early and rapid up-titration of beta-blockers and concomitant addition during hospitalization of ivabradine is safe, allows to achieve a heart rate < 70 bpm early, and is associated with a significant improvement in left ventricular ejection fraction at short and long term follow-up.
Limitations
Single-centre and not blinded study, with a limited number of patients (although enough for the analysis of primary endpoint).
Conclusions
The strategy of early co-administration of ivabradine and beta-blockers during the episode of decompensated heart failure in patients with systolic left ventricular dysfunction and who are in sinus rhythm is feasible and safe, getting better and earlier control of heart rate in these patients, without entailing a reduction in the dose of beta-blockers. This is associated with a significant improvement of systolic left ventricular function and a trend to a better clinical status of patients.
Clinical Perspectives
Thesis the first randomized study that analyzed the use of ivabradine in acute heart failure, offering significant data of safety and clinical relevance, without involving a reduction in the dose of beta-blockers.
Translational Outlook
It is necessary a larger study, with a higher number of patients, to confirm if thesis findings have a favourable long-term prognostic effect.
References
-
Anguita M, Crespo MG, De Teresa E, Jiménez M, Alonso-Pulpón L, et al. (2008) Prevalence of Heart Failure in theSpanish General Population AgedOver 45 Years. The PRICE Study. Rev Esp Cardiol 61: 1041-1049.
-
Macintyre K, Capewell S, Stewart S, Chaimers JWT, Boyd J, et al. (2000) Evidence of improving prognosis in heart failure. Trends in case fatality in 66547 patients hospitalized between 1986 and 1995. Circulation 102: 1126-1131.
-
Bohm M, Swedberg K, Komajda M, Borer JS, Ford I, et al. (2010) Heart rate as a risk factor in chronicheart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet 376: 886-894.
-
Bohm M, Borer J, Ford I, González-Juanatey JR, Komajda M, et al. (2013) Heart rate at baseline influences the effect of ivabradine on cardiovascular outcomes in chronic heart failure: Analysis from the SHIFT study. Clin Res Cardiol 102: 11-22.
-
Savelieva I, Camm AJ (2008) If inhibition with ivabradine: electrophysiological effects and safety. Drug Saf 31: 95-107.
-
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, et al. (2016) 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 37: 2129-2200.
-
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, et al. (2016) 2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation 134: e282-e293.
-
Borer JS, Bohm M, Ford I, Robertson M, Komajda M, et al. (2014) Efficacy and safety of ivabradine in patients with severe chronic systolic heart failure (from the SHIFT study). Am J Cardiol 113: 497-503.
-
McAlister FA, Wiebe N, Ezekowitz JA, Leung AA, Armstrong PW (2009) Meta-analysis: B-Blocker dose, heart rate reduction, and death in patients with heart failure. Ann Intern Med 150: 784-794.
-
Swedberg K, Komajda M, Bohm M, Borer J, Robertson M, et al. (2012) Effects on outcomes of heart rate reduction by ivabradine in patients with congestive heart failure: is there an influence of beta-blocker dose? : findings from the SHIFT (Systolic Heart failure treatment with the I(f) inhibitor ivabradine Trial) study. J Am Coll Cardiol 59: 1938-1945.
-
Cullington D, Goode KM, Clark AL, Cleland JG (2012) Heart rate achieved or beta-blocker dose in patients with chronic heart failure: which is the better target? Eur J Heart Fail 14: 737-747.
-
Maggioni AP, Dahlstrom U, Filippatos G, Chioncel O, Leiro MC, et al. (2010) EUR Observational Research Programme: The heart failure pilot survey (ESC-HF Pilot). Eur J Heart Fail 12: 1076-1084.
-
Hidalgo FJ, Anguita M, Castillo JC, Rodriguez S, Pardo L, et al. (2016) Effect of early treatment with ivabradine combined with beta-blockers versus beta-blockers alone in patients hospitalized with heart failure and reduced left ventricular ejection fraction (ETHIC-AHF): A randomized study. Int J Cardiol 217: 7-11.
-
Swedberg K, Komajda M, Bohm M, Borer JS, Ford I, et al. (2010) Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet 376: 875-885.
-
Tardif JC, O'Meara E, Komajda M, Bohm M, Borer JS, et al. (2011) Effects of selective heart rate reduction with ivabradine on left ventricular remodelling and function: results from the SHIFT echocardiography sub-study. Eur Heart J 32: 2507-2515.