International Journal of Pediatric Research
Six-Month Therapy with Metformin in Association with Nutritional and Life Style Changes in Children and Adolescents with Obesity
María Cristina Azcona-Sanjulián*, Ana Catalán Lambán and Borja Laña Ruiz
Pediatric Endocrinology Unit, Department of Pediatrics, Clínica Universidad de Navarra, Faculty of Medicine, University of Navarra, Pamplona, Spain
*Corresponding author: Cristina Azcona. Department of Pediatrics, Clinica Universidad de Navarra, Avda Pio XII s/n 31080 Pamplona, Spain, Tel: 948-296236, Fax: 948 296500. E-mail: cazcona@unav.es
Int J Pediatr Res, IJPR-1-002, (Volume 1, Issue 1), Original Article; ISSN: 2469-5769
Received: February 18, 2015 | Accepted: March 11, 2015 | Published: March 13, 2015
Citation: Azcona-Sanjulián MC, Lambán AC, Ruiz BL (2015) Six-Month Therapy with Metformin in Association with Nutritional and Life Style Changes in Children and Adolescents with Obesity. Int J Pediatr Res 1:002. 10.23937/2469-5769/1510002
Copyright: ©2015 Azcona-Sanjulián MC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To assess the effect of metformin on weight, BMI, body fat, and insulin sensitivity in obese children and adolescents.
Methodology: The study was prospective, and included 21 patients with obesity (8 male, 13 female) with a mean age of 12.31 (3.87) years. Inclusion criteria were a fat mass percentage (FM%) of over 25% in males and over 30% in females, a BMI of greater than the 95th percentile, and a lack of response to nutritional and lifestyle changes over three months. Informed consent was obtained from children, parents and the treatment was approved by the hospital's Ethics Committee, and the Spanish health ministry. All subjects received treatment with metformin for six months and each month was instructed to follow nutritional and lifestyle changes. The oral glucose tolerance test (OGTT) was done for 95% of the patients.
Results: A decrease in weight-SDS (p< 0.001), BMI-SDS (p< 0.001), FM% (p = 0.002), waist/hip ratio (p = 0.141), HOMA (p = 0.198 and plasma glucose level at 120 min. (p = 0.008) was observed after six months of metformin therapy in association with nutritional and lifestyle changes. At the onset of the study, plasma glucose level at 120 min. was above 140 mg/dL in 38% (n = 8) of patients, and the HOMA index was above 3.8 in 42% (n = 9). Altogether, 11 patients had insulin resistance. Mean dose of metformin was built up to a maximum of 1275 mg/day. Twenty-four percent of the subjects complained of dyspepsia or diarrhea or both.
Conclusion: In this study, metformin, combined with diet and lifestyle changes, contributed to an improvement in BMI and to a loss of weight and body fat in children with obesity, whether or not that obesity was associated with insulin resistance.
Introduction
Obesity during childhood has became a growing public health problema throughout the world to the extent that accordint to the European Association for the study o Obesity (EASO), about 16-22% of european adolescents between 14-17 years old are overweight or obese, with an anual increase of the prevalence of around 2% in the 1990s and 2000s [1].
Conventional therapy for obesity is sometimes unsuccessful, especially in those children who develop hyperinsulinemia and insulin resistance, which often precede the development of glucose intolerance [2]. Hyperinsulinemia has a strong lipogenic effect and therefore a positive energy balance is established. Fat deposition persists and so it seems that insulin-stimulated lipogenesis is unimpaired despite the resistance to carbohydrate metabolism. Therefore, it is hypothesized that, in the obese, if the insulin level falls, lipogenesis will decrease and weight gain will diminish.
Metformin (dimethylbiguanide) is an insulin-sensitizing and antihyperglycemic agent used in the treatment of type 2 diabetes. The beneficial role of metformin in young patients with diabetes type 2 was demonstrated in a randomized trial [3]. Metformin has been recently approved by the FDA for the treatment of diabetes type 2 in children over 10 years old.
Metformin acts by stimulating intracellular glucogen synthesis, decreasing hepatic glucose production (inhibition of gluconeogenesis), decreasing intestinal absorption of glucose, and increasing insulin sensitivity [4]. It also increases muscle uptake of glucose and interfere with mitochondrial activity. The use of metformin in nondiabetic obese adults and children has been associated with reduced food intake [5-7].
The first clinical application of metformin in children with obesity was described in 1977; a beneficial effect on weight and insulin concentrations was reported [8].
Subsequent data from randomized, doubled-blind, placebo, controlled trials with children given metformin therapy for exogenous obesity with insulin resistance [9-14] liver dysfunction and obesity [15] as well as for psychotropic drug induced weight gain [16] have shown improvement in body mass index (BMI), in levels of fasting serum glucose and insulin, and in the lipid profile.
Recently, Srinivasan et al. [17] assessed the effect of metformin on body composition and insulin sensitivity in 28 pediatric patients (mean age 12.5 years) and found a significant improvement in body composition and fasting insulin levels.
In our work on pediatric patients with obesity which we report here, we seek to evaluate the effect of metformin after six months of therapy.
Subjects and Methods
Participants were 5- to 18- year-old with obesity, as defined by the International Obesity Task Force [18], referred to the pediatric endocrine clinic at the University Hospital of Navarra between January 2006 and January 2007. Prior to metformin therapy, all participants had been subjected to nutritional intervention and lifestyle changes but had not responded with a loss of weight. Exclusion criteria were known type 1 or type 2 diabetes; dysmorphic features; diabetes mellitus; renal disorders; obesity due to hormonal, chromosomal, or neurological disorders; and contraindications to metformin therapy.
Written informed consent was obtained from all parents and from all participants over 12 years old. Participants under 12 years old were also informed about the therapy and expressed their agreement to it. The therapy was approved by the Spanish health ministry and by the University Hospital of Navarra Ethics Committee.
Study design
All subjects received treatment with metformin for six months. Subjects were seen monthly by a dietitian to receive standardized instructions for healthy eating and exercising. We encourage children to do aerobic exercise such as swimming, cycling, running, dancing, 3 times per week. They also do physical exercise at school twice a week. After a two week period, metformin dose was gradually built up from 425mg once daily to a maximum final dose of 850mg twice daily. Up to 40 kg the daily dose was 425mg after breakfast and dinner, and above 40kg the dose was 850mg after breakfast and dinner. Unused tablets were counted when the patients' pill dispensers were refilled with the drug, that is, after three months and at the end of the study period. Pill counts were conducted to calculate percent adherence to therapy, based on number of tablets consumed versus anticipated tablet consumption for each three month period.
Clinical assessment and anthropometry
At baseline and six months, participants attended the University Hospital of Navarra for clinical assessment including anthropometry, body composition analysis by bioelectric impedance, oral glucose tolerance test or basal glucose and insulin tests, liver function test, and determination of folic acid and vitamin B12 levels.
The medical history of each participant was investigated in detail, and subjects were examined for clinical signs of adrenarche or gonadarche according to Tanner stage [19]. A participant was considered pubertal if he or she was in at least Tanner stage 2 with respect to breast development or testicular volume.
All participants were measured anthropometrically by the same observer. Weight was measured to the nearest 0.01 kg using the calibrated BP electronic scale (Life Measurement Instruments, Concord, CA, U.S.A.). Height was measured to the nearest 0.1cm using a Harpender stadiometer. BMI was calculated as weight/height2 (kg/m2) [20]. Waist and hip circumferences were also measured, as described in the literature [21].
Acanthosis nigricans on the neck was assessed for severity by a validated scale ranging from grade 0 (not present) to grade 4 (severe: extending anteriorly, visible when the participant is viewed from the front) [22].
We assessed degree of appetite on a categorical scale of one to five: 1 being very poor, 2 poor, 3 fair, 4 good, and 5 very good [23].
Bioimpedance
BIA was performed with a TANITA BIA body fat analyser, which measures impedance of only the lower part of the body (TBF-410, Tanita, Tokio, Japan). Participants were asked to stand barefoot on the four metal sole-plates of the machine. Gender and height details were input via a keyboard. Bioelectric resistance was measured after induction of a 50 KHz electrical signal with an interval current of 150 to 900 mA. Percentage body fat was automatically estimated by the prediction equations (for children) which had been programmed into the system. The prediction equations are not provided by the manufacturers.
Biochemical and hormonal assays
Glucose was measured in plasma by an enzymatic method (Roche diagnostics). Insulin was measured in serum by an EIA assay (DPC). Sensitivity to insulin was evaluated using the HOMA index (fasting insulin x fasting glucose mmol/L/22.5) [24]. After an overnight fast, an oral glucose bolus of 1.75 g/kg (up to a maximun of 75g) was administered. Blood for determination of glucose and insulin was obtained at 0, 30, 60, 90, and 120 minutes. The liver function test and determination of vitamin B12 and folic acid levels were carried out as standard clinical assays.
Statistical analysis
Computer analysis: The statistical analyses and data recordings were performed on a personal computer using the Statistical Package of the Social Science program (SPSS), version 20.0 (Chicago, Illinois).
Descriptive analysis: Descriptive statistics are reported as means and SD because all the variables were normally distributed. Weight, BMI, and height were converted to z-scores (standard deviation scores). To obtain the z-scores (x-mean/SD), we used Spanish growth standards [25].
Statistical analysis: Since all the variables were normally distributed, we used the paired-sample t-test to compare data before and after metformin therapy. All tests were two-tailed.
Results
Twenty-five patients were referred to the study. Two patients declined to participate, and two did not meet the inclusion criteria. Therefore, 21 patients, of which 8 were male and 13 female, participated in the study. One patient declined the OGT test or any other blood investigation at month 0. At month six, only 7 patients agreed to have an OGT test. One patient discontinued the study after one month of metformin therapy because of dyspepsia and diarrhea.
Baseline characteristics
The mean age of the participants was 12.3 ± 3.9 years, with 13 being in Tanner stage 1-2 and 8 in Tanner stage 3-5. There were not significantly more girls than boys in puberty (Tanner stage 3-5) (p=0.055). All other characteristics were similar for both males and females (Table 1). All participants but one were of Caucasian origin. A family history of metabolic syndrome in either first or second degree relatives was noted in 15 patients (71%). Thirteen patients (61.9%) had acanthosis nigricans. At onset of therapy, 38% of the participants had plasma glucose concentrations at 120 min. higher than 140 mg/dL, and 42% had HOMA higher than 3.8.
![]()
Table 1: Baselinecharacteristics of the patients
View Table 1
Metformin treatment effect on anthropometry, body composition and appetite
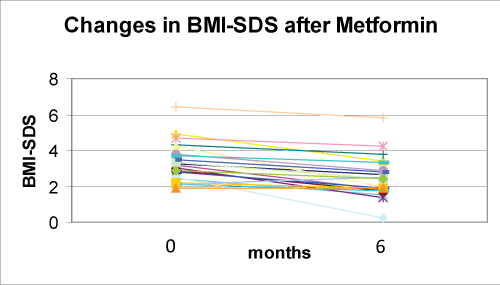
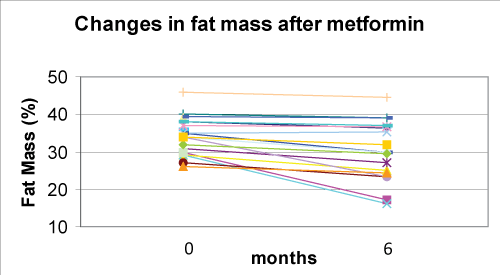
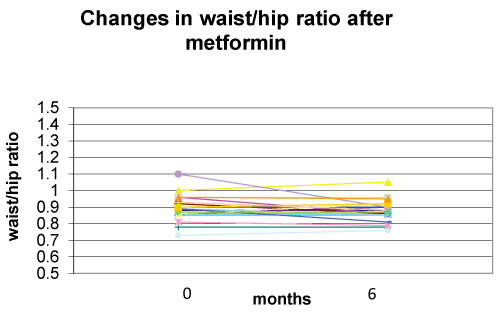
Metformin associated with nutritional and lifestyle intervention decreased weight-SDS (p< 0.0001), BMI-SDS (p< 0.0001) (Figure 1), FM% (p=0.001) (Figure 2), and waist/hip ratio (p=0.141) (Figure 3) at six months of follow-up (Table 2). Fat free mass increased significantly (p= 0.012). Appetite decreased (p< 0.001) from 5 to 4 in 80% of subjects and from 5 to 3 in 20%.
![]()
Table 2: Metformin treatment effect
View Table 2
Effect of Metformin treatment on parameters of insulin sensitivity
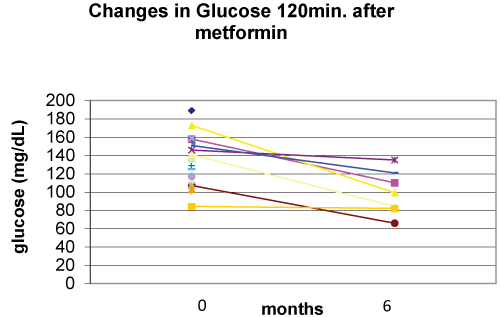
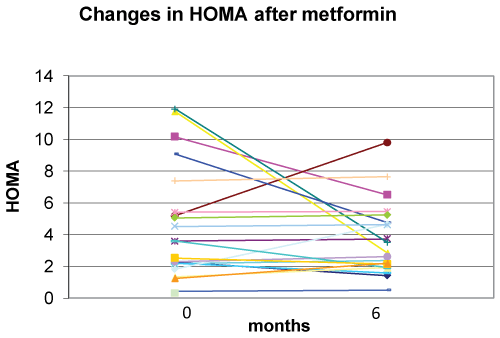
Metformin in association with nutritional and lifestyle intervention decreased plasma glucose level at 120 min. after glucose loading at six months of follow-up (p=0.008) (Figure 4). There were statistically non-significant decreases in basal insulin (p=0.142), basal glucose (p=0.183), and HOMA (p=0.198) (Figure 5) at six months of follow-up (Table 2).
Side effects, adherence to therapy, and safety profile
Metformin was well tolerated by the majority of patients. One patient had to stop therapy after a month because of dyspepsia. Only 23.8% of the participants referred gastrointestinal side effects: transient abdominal discomfort, diaorrhea, or both. After reducing the metformin dose, these gastrointestinal problems resolved within two weeks of therapy. Liver function tests, and levels of B12 and folic acid remained normal in all patients throughout the study. Mean basal vitamin B12 levels were 650pg/mL and after treatment 690pg/mL. Mean folate basal levels were 15,5ng/mL and after treatment 16,5ng/mL. Based on pill counts, adherence to therapy was 78% (range 14-98%).
Discussion
This study demonstrates that, when diet and exercise alone are not effective, metformin helps obese patients lose weight. The patients studied had been referred to the Pediatric Endocrine Unit for management of obesity and, despite being given appropiate instructions and guidance about how to change their nutrition and lifestyle, were not losing weight, some were even putting on weight. Some were developing features of insulin resistance, such as, acanthosis nigricans.
Metformin was well tolerated and had a beneficial effect on weight, BMI, waist circumference, and fat mass as has been described previously in previous other studies [9-15]. Visceral fat was not accurately measured in this study; body composition was assessed by BIA, a method which has its limitations in estimating body fat and fat-free mass. Moreover, the BIA system used in this study does not measure body fat in different body areas. Other researchers have performed whole body DEXA but did not find loss of visceral fat [17]. Loss of visceral fat might require metformin therapy for a longer period of time.
Although, based on HOMA index a number of patients had improved insulin sensitivity, this improvement was not statistically significant for the group as a whole. Similar results have been previously observed by Srinivarsan et al. [17] using more complex methods to assess insulin sensitivity.
There are several possible explanations for the lack of statistically significant improvement in insulin sensitivity. Firstly, most of the participants who had insulin resistance (52.3%, n=11) were in puberty, a period when insulin resistance due to obesity can be enhanced. This physiological insulin resistance characteristic of puberty may have masked the effect of metformin. To study the effects of metformin on insulin resistance during puberty was not the main objective of this study and would require a very different experimental approach to that adopted here. Moreover, there were not enough patients in this study to statistically assess the effect of pubertal stage on response to metformin.
Secondly, the methods we used to assess insulin sensitivity, HOMA and OGT, whilst easy to perform clinically, are not the most accurate. However, Srinivarsan et al. [17] also failed to detect a significant improvement in insulin sensitivity assessed with the frequently sampled intravenous glucose tolerance test.
Thirdly, the doses of metformin were small and the adherence to therapy was poor. We used a maximum dose of 1700 mg whereas data from adults with type 2 diabetes suggest that a total dose of 3g may be required to maximize the metabolic benefits of metformin [26]. Many patients did not adhere well to our prescribed therapy. This is not uncommon in the treatment of obese patients, especially with adolescents.
The precise way in which metformin acts is unknown. It is believed to increased insulin sensitivity and glucose uptake in subjects with type 2 diabetes mellitus [27]. It has also been suggested that metformin exerts its antyhyperglycemic effect by decreasing hepatic glucose output through inhibition of gluconeogenesis. Previous studies have demonstrated that metformin treatment reduces food intake in both humans and experimental animals [28,29]. A study has demonstrated that metformin can inhibit the complex I of the electron transport chain in mitocondria and lead to a loss of mitocondrial membrane potential and inhibition of ATP production [30]. They conclude that metformin pharmacological effects are mediated, at least in part through a time-dependent, self-limiting inhibition of the respiratory chain that restrains hepatic gluconeogénesis while increasing glucose utilization in peripheral tissues [30]. However, it is diffcult to know its precise mechanism of action in humans because most of the studies have been performed in animal cells.
In this study, we did not analyze diet in order to assess caloric intake. However, we did use a categorical scale to assess appetite and found it decreased significantly. Therefore, loss of appetite is another mechanism by which metformin can exert its anti-obesity effect.
Metformin was well tolerated. There were only minor and transient side effects, which in the majority of patients resolved spontaneously after decreasing the dose. We did not measure lactic acid, but it should be observed that an increased level of lactic acid has been reported as a complication, albeit very rare, and that it can be more frequent in patients with renal disease.
In conclusion, for certain patients with obesity who have difficulties in losing weight with conventional therapy, additional medical therapy can help. Obesity is a chronic disease with severe complications, specially insulin resistance, that become more difficult to treat the longer they persist. Metformin, by helping patients to lose weight, represents a way to try to forestall progression to type 2 diabetes in those patients predisposed to it. However, longer term, control-placebo studies are needed to fully assess the safety and behaviour of this drug.
References
-
Baker JL, Farpour-Lambert NJ, Nowicka P, Pietrobelli A, Weiss R, et al. (2010) Evaluation of the overweight/obese child-practical tips for the rpimary health care provider: recommendations from the Childhood Obesity Task forcé of the European Association for the Study of Obesity. Obes Facts 3: 131-137.
-
Le Stunff C, Bougnères P (1994) Early changes in postprandial insulin secretion, not in insulin sensitivity, characterize juvenile obesity. Diabetes 43: 696-702.
-
Jones KL, Arslanian S, Peterokova VA, Park JS, Tomlinson MJ (2002) Effect of metformin in pediatric patients with type 2 diabetes: a randomized controlled trial. Diabetes Care 25: 89-94.
-
Garber AJ (2004) Metformin and other biguanides: pharmacology and therapeutic usage. In Defronzo RA, Ferrannini E, Keen H, Zimmet P (eds): International textbook of Diabetes Mellitus, vol 1. West Sussex, UK, John Wiley and sons Ltd., pp 851-869.
-
Fontbonne A, Charles MA, Juhan-Vague I, Bard JM, André P, et al. (1996) The effect of metformin on the metabolic abnormalities associated with upper-body fat distribution. BIGPRO Study Group. Diabetes Care 19: 920-926.
-
Lee A, Morley JE (1998) Metformin decreases food consumption and induces weight loss in subjects with obesity with type II non-insulin-dependent diabetes. Obes Res 6: 47-53.
-
Adeyemo MA, McDuffie JR, Kozlosky M, Krakoff J, Calis KA, et al. (2014) Effects of metformin on energy intake and satiety in obese children. Diabetes Obes Metab .
-
Lütjens A, Smit JL (1977) Effect of biguanide treatment in obese children. Helv Paediatr Acta 31: 473-480.
-
Kay JP, Alemzadeh R, Langley G, D'Angelo L, Smith P, et al. (2001) Beneficial effects of metformin in normoglycemic morbidly obese adolescents. Metabolism 50: 1457-1461.
-
Wiegand S, l'Allemand D, Hübel H, Krude H, Bürmann M, et al. (2010) Metformin and placebo therapy both improve weight management and fasting insulin in obese insulin-resistant adolescents: a prospective, placebo-controlled, randomized study. Eur J Endocrinol 163: 585-592.
-
Wilson DM, Abrams SH, Aye T, Lee PD, Lenders C, et al. (2010) Metformin extended release treatment of adolescent obesity: a 48-week randomized, double-blind, placebo-controlled trial with 48-week follow-up. Arch Pediatr Adolesc Med 164: 116-123.
-
van der Aa MP, Elst MA, van Mil EG, Knibbe CA, van der Vorst MM (2014) Metformin: an efficacy, safety and pharmakocinetic study on the short-term and long-term use in obese children and adolescents- study protocol of a randomized controlled study. Trials 15: 207.
-
Kendall D, Vail A, Amin R, Barrett T, Dimitri P, et al. (2013) Metformin in obese children and adolescents: the MOCA trial. J Clin Endocrinol Metab 98: 322-329.
-
Freemark M, Bursey D (2001) The effects of metformin on body mass index and glucose tolerance in obese adolescents with fasting hyperinsulinemia and a family history of type 2 diabetes. Pediatrics 107: E55.
-
Freemark M (2007) Liver dysfunction in paediatric obesity: a randomized, controlled trial of metformin. Acta Paediatr 96: 1326-1332.
-
Morrison JA, Cottingham EM, Barton BA (2002) Metformin for weight loss in pediatric patients taking psychotropic drugs. Am J Psychiatry 159: 655-657.
-
Srinivasan S, Ambler GR, Baur LA, Garnett SP, Tepsa M, et al. (2006) Randomized, controlled trial of metformin for obesity and insulin resistance in children and adolescents: improvement in body composition and fasting insulin. J Clin Endocrinol Metab 91: 2074-2080.
-
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH (2000) Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 320: 1240-1243.
-
Tanner JM (1962) Growth at adolescence. 2nd ed. Oxford: Blackwell Scientific.
-
Cameron, N (1978) The methods of auxological anthropometry. In: Human growth. Eds. F. Falkner and J.M. Tanner, pp. 35-87. Plenum, New York;
-
Moreno LA, Joyanes M, Mesana MI, Gonzalez-Gross M, Gil CM (2003) Harmonization of anthropometric measurements for multicentre nutrition survey in spanish adolescents. Nutrition 19: 481-486.
-
Burke JP, Hale DE, Hazuda HP, Stern MP (1999) A quantitative scale of acanthosis nigricans. Diabetes Care 22: 1655-1659.
-
Tchekmedyian NS, Hickman M, Siau J, Greco FA, Keller J, et al. (1992) Megestrol acetate in cancer anorexia and weight loss. Cancer 69: 1268-1274.
-
Haffner SM, Kennedy E, Gonzalez C, Stern MP, Miettinen H (1996) A prospective analysis of the HOMA model. The Mexico City Diabetes Study. Diabetes Care 19: 1138-1141.
-
Ferrandez-Longas A, Mayayo E, Labarta JI, Baguer L, Puga B, et al. (2004) 1980-2002 En: Patrones de crecimiento y desarrollo en Espana. Atlas de grafica y tablas. Editores: Carrascosa A, Delgado P, Ferrandez-Longas A, Garcia-Dihinx, Hernandez-Rodriguez M, Romo A, Sobradillo B. Ed. ERGON, Madrid.
-
Garber AJ (2000) Using dose-response characteristics of therapeutics agents for treatment decisions in type 2 diabetes. Diabetes Obes Metab 2: 139-147.
-
Abbasi F, Carantoni M, Chen YD, Reaven GM (1998) Further evidence for a central role of adipose tissue in the antihyperglycemic effect of metformin. Diabetes Care 21: 1301-1305.
-
Stumvoll M, Nurjhan N, Perriello G, Dailey G, Gerich JE (1995) Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N Engl J Med 333: 550-554.
-
Rouru J, Huupponen R, Pesonen U, Koulu M (1992) Subchronic treatment with metformin produces anorectic effect and reduces hyperinsulinemia in genetically obese Zucker rats. Life Sci 50: 1813-1820.
-
Owen MR, Doran E, Halestrap AP (2000) Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J 348 Pt 3: 607-614.