International Journal of Pediatric Research
Leptospirosis Prevalence and Associated Factors in School Children from Valle de Chalco-Solidaridad, State of Mexico
Dr. Joel Navarrete Espinosa1*, MC Beatriz Rivas Sanchez2 and Dr. Maria Luisa Soriano Rodriguez3
1Division of Epidemiology, Epidemiological Surveillance Coordination, IMSS, Mexico
2Department of Microbiology and Parasitology, Medicine Faculty, UNAM, Mexico
3Unidad de Medicina Familiar # 180, IMSS, Mexico
*Corresponding author: Joel Navarrete Espinosa, Division of Epidemiology, Epidemiological Surveillance Coordination, IMSS, Mexico, E-mail: joel.navarrete@imss.gob.mx
Int J Pediatr Res, IJPR-1-008, (Volume 1, Issue 2), Research Article; ISSN: 2469-5769
Received: September 25, 2015 | Accepted: October 09, 2015 | Published: October 12, 2015
Citation: Navarrete EJ, Rivas SB, Soriano RML (2015) Leptospirosis Prevalence and Associated Factors in School Children from Valle de Chalco-Solidaridad, State of Mexico. Int J Pediatr Res 1:008. 10.23937/2469-5769/1510008
Copyright: © 2015 Navarrete EJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Material and Methods: Cross-sectional study in school children 8-12 years of age; Leptospira identification tests (IFI and Dark Field: Microbiology and Parasitology Department, FM-UNAM) were performed and risk factors were researched. Those positive to IFI (cutoff 1: 80) and dark field in blood and urine were considered positives. Prevalence, prevalence ratios, and multivariate analysis were estimated.
Results: 220 children were studied; the prevalence of Leptospira infection was 37% (IFI) and 83% (dark field in blood). The highest prevalence was for females, children younger than 10 years old, children from the 16 de Septiembre school, those who had dogs, and those whose pet was allowed inside the house. According to the IFI results, in the final multivariate model the main associated risk factors were: parents' schooling (p < 0.05), belonging to the school 16 de Septiembre (p < 0.05), and allow the dogs inside the house (p < 0.05). Factors associated with Leptospira in blood were parents' schooling (p < 0.05), belonging to the school 16 de Septiembre (p < 0.05), allowing the dogs inside the house (p = 0.6). No association with contact with stagnant water was found.
Conclusions: Children who live in the Valley of Chalco are exposed to several factors, exposing them to the transmission chain of leptospirosis. It is important to continue this research line to determine the implications of leptospirosis in this population. To educate to avoid close contact with animals to improve health must be permanent.
Keywords
School children, Prevalence, Leptospirosis, Dark field microscopy, Immunofluorescence (IFI)
Introduction
Leptospirosis has been considered a zoonotic disease distributed worldwide and of occupational type, usually in adult population and predominant in tropical regions [1-3]; almost always associated with poverty [4]. However, this concept has changed in recent decades and it is now known that susceptibility to the disease is universal and happens more frequently as a result of exposure and contact with the urine of infected pets and vermin [5,6].
In recent years, an increase in the number of cases reported of this disease has been observed worldwide, due to the changes in the environment and the presence of strong rains and floods, which expose the population to water contaminated with urine of sick animals [4,7-10]. In Mexico different studies have been performed, with different techniques and cut values, and the prevalence of the infection may reach up to 80% of the general population [11-14].
The clinical behavior of the infection presents a large variety of signs and symptoms that are similar in many diseases [15-18]. The knowledge of the disease is almost exclusively based in the description of acute and severe presentations of the disease (Weil Syndrome). Nevertheless, leptospirosis has been currently described as a disease that may be chronically present, with multiple manifestations and complications [19-21].
In children, the infection may simulate a number of infectious diseases, such as viral meningoencephalitis, pharyngitis, urinary tract infection, mononucleosis, and other [22]. This is especially important in the case of hepatitis, for the presence of severe leptospirosis cases with a fatal ending have been documented, and which were initially treated as viral hepatitis [23].
It is currently known that children are a high risk group for the disease, for they are in contact with their pets, especially with dogs [24] and their urine. The children in Valle de Chalco, Estado de Mexico, are in addition exposed to frequent and large floods and thus to contact with stagnant waters, increasing the risk for infection.
Although studies have been performed in the general population, and being considered an occupational-type disease [25], children are almost always excluded; in the same way, most of these studies have been performed in diseased persons in acute state, and the infection prevalence in children in unknown and little is known regarding the presence in carriers and/or chronic Leptospira infection in them. In the same way there is little information about the clinical manifestations and complications that happen or the factors associated to the infection in the children group.
In this context, it is known that animals that have been infected may remain free of symptoms and excrete leptospiras for the rest of their life; however, there is not enough information regarding this in humans, concluding that when humans are infected they may excrete Leptospira through urine for several months [15].
There are few international research works exploring this fact, therefore it was necessary to perform the present study with the main objective of knowing the prevalence of Leptospira background infection in the school population (children) in the Valle de Chalco and which are the main risk factors associated with the infection in this population. Knowing the symptoms associated with the infection in acute phase, and knowing the proportion of children in whom persists the presence of Leptospira in blood and urine, and signs and symptoms associated with this condition were established as specific objectives of this study, which will allow us estimate the magnitude of the problem in a community that might be representative of many others in our country and, at the same time, provide elements for the diagnosis and treatment of leptospirosis in children.
Materials and Methods
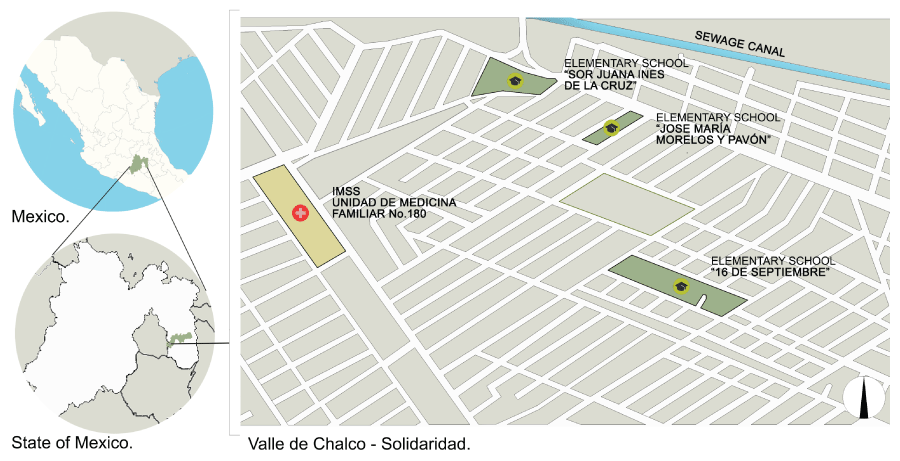
To determine the prevalence of Leptospira infection, a cross-sectional study was conducted from January to November 2014 in children aged 8-12 years in the localities of El Triunfo and San Isidro (Valle de Chalco-Solidaridad), registered in 4th to 6th grades of the following elementary schools: 16 de Septiembre, Sor Juana Ines de la Cruz, and Jose Maria Morelos y Pavon, during the 2013-2014 school year; these were randomly chosen from the list of schools in the vicinity. The selection of the children who participated in the study was also done through a table of random numbers, based in the school lists. Once the parents of the children objective of the study were informed of the importance and the procedures that would be performed, signed consent was required to participate in the study.
From each child who agreed participating in the study, blood and urine samples were required. A sample of 5 ml of blood was obtain from the vein in the left arm with the Vacutainer system; the sample was drawn by a specialist nurse. The sample was kept in the cold-chain system, in a Coleman-type thermos, at a temperature between 2-8°C; it was labelled and foliated according the corresponding individual questionnaire. Then a part of each sample was sent to the Parasitology and Microbiology Department of the Medicine Faculty from the Universidad Nacional Autonoma de Mexico (UNAM), where the detection of specific antibodies for IFI was performed [26]; additionally, dark field microscopy was performed to every sample to identify the bacteria y blood and urine [27]. In the same way, another portion of the sample was used to perform a general blood exam by conventional methods, being done at the Unidad de Medicina Familiar # 180 from the Instituto Mexicano del Seguro Social (IMSS) in Chalco, Estado de Mexico, which was used as recruitment center of the studied population.
A questionnaire was applied for the personal identification of the child, as well as to measure the factors related with the disease: time living in the area, socioeconomic stratus (AMAI index), coexistence with domestic animals, close contact with pets (hug, kiss, sleep with), presence of rodents in the household, if living near a breeding place, and exposure to water during floods. Also an interview was conducted to know the history of morbidity in the children. The answers to the questionnaire were obtained from the child by one person of the research team, previously trained for such task (epidemiology medic) in the presence of a parent or tutor, who corroborated, or in its case, corrected the answers given by the child.
Since the purpose of the study was the identification of infections that occurred in the past, it was determined to identify the presence of IgG antibodies. For the detection of IgG antibodies against Leptospira interrogans antigens, a reference pull de 14-serovars of Leptospira interrogans: was used Canicola, Icterohaemorrhagiae, Pomona, Pyrogenes, Ballum, Autumnalis, Muechen, Hardjo, Wolffi, Tarassovi, Shermani, Grippotyphosa, Bratislava, and Lai lai, as well as a pull of 9 isolates of Leptospira serovar Pomona obtained from patients with chronic leptospirosis and typified by means of monoclonal antibodies at the International Laboratory of Leptospirosis Reference (WHO/FAO/OIE) in Amsterdam, Holland.
According to the Norma Oficial Mexicana for Leptospirosis [28] (Mexican Official Normativity), it was considered as case any child who presented a ≥ 1:80 titers of antibodies, and those in whose blood and urine was observed the presence of Leptospira in the dark field test.
For data analyses, the frequencies and percentages were done after clearing the data base. In the same way, the comparison between the proportion of children with and without antibodies against Leptospira, was performed, with the socio-demographic and exposure variables by means of the X2 test and Fisher's exact test with confidence intervals of 95%, and a value of p ≤ 0.05.
Finally, a logistic regression model was performed with the variables that resulted significant in the bivariate analysis, using the statistical Stata, version 13.1 package. Under the same procedure a model was run considering the presence of Leptospira in blood and urine in the children in addition to the pathologic background from the specific interrogatory.
Results
In total, 220 school children who accepted to participate were studied. The mean age was 10.6 years (± 1.3). Regarding the gender of the children, the highest proportions were females. In the same way, the highest percentage of the sample was for children older than 10 years of age.
The proportion of children originating from Valle de Chalco was higher, and 90% referred having lived at least 7 years in the locality. The percentage of children registered in the 6th grade was higher. The frequencies of the main studied factors are shown in table 1.
Regarding personal history of important pathologies in the children, 7% expressed having some disease. According to the signs and symptoms suggestive of leptospirosis, it was found that 12% presented an allergic disease to different substances; only 2% referred having symptoms suggesting Hepatitis. 11% referred excessive fatigue and 24% referred frequent fatigue; similarly, 15% referred excessive sleepiness and 27% having regular headaches.
The presence of exanthema was documented in 13%, epistaxis in 22%, bone pain in 23%, abdominal pain in 18%, repetitive infections in superior airways in 15%, conjunctivitis in 16%, and depression in 7%.
The averages in weight and size for the children were 41.04 kilograms (± 11.9) and 1.42 meters (± 0.11) respectively. The blood studies presented the following averages for the total group of children: erythrocytes 5.2 million/ul. (± 0.35), hemoglobin 14.8 g/dl. (± 0.84), hematocrit 43.1% (± 2.7), and leukocytes 6.8 million/ul (± 1.6).
The prevalence of leptospirosis with the cutoff IgG ≥ 1:80 was 37% (0.37; CI95% 0.28-0.47), this cut was the basis for all analysis. In the same way, the prevalence of the presence of the bacteria in blood by dark field was 83% (0.83; CI95% 0.74-0.90) and in urine 40% (0.40; CI95% 0.30-0.50).
The highest prevalence was obtained with the dark field test in blood. The prevalence to each of the main studied factors are shown in table 1.
![]()
Table 1: Prevalence of antibodies against Leptospira and Leptospira in blood and urine in the studied children.
View Table 1
In total, 188 children (86%) were positive to the IFI test with cut points from 1:20 to 1:640, and 31 (14%) were negative. In the same way, the presence of the bacterium in blood was demonstrated in 48.9% of the children, in blood and in urine in 32.9%, and in urine in 7.8%. Six children (2.7%) were negative to all studies. From the 31 children in who the presence of antibodies in blood was not demonstrated, 25 had the bacteria in blood, urine, or both (Table 2).
![]()
Table 2: Infection by Leptospira cases by IFI (different cutoff values) and Dark Field in blood and urine.
View Table 2
For the bivariate analysis, the presence of antibodies (IgG 1:80) and the observing the bacteria in blood and urine were compared with the main studied variables (Table 3).
![]()
Table 3: Bivariate analysis for infection by Leptosipira: IFI and Dark Field in blood and urine.
View Table 3
The association of the following variables was not demonstrated: flooding of the street where they live, contact with flooding water, presence of rodents in the house, and walking barefoot.
For the multivariate analysis, three models were constructed considering the presence of antibodies in blood (IgG ≥ 1:80), the presence of the bacteria in blood and urine, using the strongest association variables in the bivariate analysis (Table 4).
![]()
Table 4: Multivariated model for infection by Leptospira* with IFI and Dark Field in blood and urine.
View Table 4
To measure the relation with the presence of antibodies in blood, a model adjusted by age and gender was used, using the most significant variables found in the bivariate analysis. It was demonstrated a protective effect for children with parents with low schooling (p < 0.05) in comparison to those who had higher schooling. On the contrary, when comparing the school of the child, and taking as reference the Jose Maria Morelos school, it was demonstrated a 2.2 (p = 0.1) and 5.7 (p < 0.05) higher risk for the presence of antibodies for the Sor Juana Ines de la Cruz and 16 de Septiembre schools, respectively. Regarding the place where pets live, a 4.3 higher risk (p < 0.05) was demonstrated for those who referred allowing the dogs inside the house, when compared with those who did not have a pet.
In relation to the presence of Leptospira in blood, a 3.1 higher risk (p < 0.05) was found for those with parents with low schooling when compared with children with parents with higher schooling. As to the schools where children went, a higher risk was found for the Sor Juana Ines de la Cruz school (p < 0.05) and 16 de Septiembre (p < 0.05) school in comparison to the Jose Maria Morelos school. In the same way, although it was not statistically significant, it was found a higher risk (80%) for those who allowed the dog inside the house when compared to those who did not have a dog.
Finally, although the model showed less precision, when measuring the relation of the same variables with the presence of the bacteria in urine, a higher risk (80%) was found for those whose parents had a basic education in comparison with those whose parents had a higher education. In the same way, a higher risk was demonstrated for the students from the Sor Juana Ines (p = 0.08) and 16 de Septiembre (p = 0.4) schools in comparison to the students from the Jose Maria Morelos school, although the statistical significance was marginal. On the contrary, no association significant was found with the place in which the pet lives (Table 4).
Discussion
There is a relation between the results of the present work with the information generated by different studies, both national and international, and it confirms the importance of leptospirosis as a public health problem yet to be met.
The prevalence of the history of leptospirosis in children living in Chalco, Mexico state is high and even higher than that reported in other works in which symptomatic children with a probable diagnosis of Leptospira [11] have been studied. However, according to what is stated in some studies and similarly to those studies performed in endemic areas, most of the studied children were anicteric, asymptomatic or presented undifferentiated symptoms during the infection in the acute stage [15], for they did not manifest any typical symptomatology associated with the acute stage at the time of the interview. Consistently with that reported in other works, it was demonstrated that the contact with domestic animals, especially dogs and their excrete (urine), are related with the presence of the infection in children [22,23]. This is more evident when considering that as a complementary part of the study, a group of children who referred having a dog in the house was selected to take samples of the pet, and by means of dark field analysis, find the presence of Leptospira in blood. From 26 sampled dogs, the infection was confirmed in 25; in the same way, the 26 children who owned these dogs were also positive to the test.
On the contrary, despite the fact that Chalco is a region where floods occur annually due to the overflow of sewage canals adjacent to the area, and thus the high exposure that these population suffers with stagnant and residual waters, we were unable to prove this as a risk factor, as it has been demonstrated in other researches [25,29,30]. Nevertheless, the closeness with the sewage canal (Figure1) and the background of a higher frequency in floods in Sor Juana Ines de la Cruz school, as well as a higher proportion of children with pets, and a higher number of these reported by the children in the 16 de Septiembre school, could explain the higher risks found in these schools. In general, the found prevalence place Chalco area as an endemic area for leptospirosis.
On the other hand, as it is shown on table 2, its noteworthy the high proportion of children in whom the presence of bacteria in blood or urine and positivity, in low titers (49%), to antibodies was demonstrated, and even more those in whom the presence of antibodies in blood was not detected (14%). This last point is highly relevant, for they are infection cases that might be unperceived, if we only take into account the diagnosis through serology. These facts could have many implications for the individuals, and it results fundamentally relevant determining the role that Leptospira plays in the their health, and also for the conceptualization of these subjects as asymptomatic carriers of the bacterium or as carriers of a chronic infection, especially considering that a high proportion of the children (40%) presented the elimination of the bacterium through urine, which could represent activity of the Leptospira and potential kidney damage [20,21,31-33]; and even as a possible source of infection [34]. Currently there is the hypothesis that this bacterium could remain for a long time in the individual and behave as an opportunistic germ.
If it is true that it is hard to identify Leptospira and obtain a leptospirosis diagnosis by means of dark field study, and that highly trained personnel is needed to perform the tests, it is also true that the personnel participating in the observation of the samples in this study, is personnel specifically trained in the identification of the bacterium in blood and urine, having done this professionally for many years, performing immune-dye and electronic microscopy, working specifically with leptospiras in national institutions (InDRE, UNAM), making the results totally reliable [35].
One of the limitations we faced in our study was the size of the studied simple, as a consequence of the difficulty to perform this type of research due to the operative problems to work with children (blood-sample drawing). In this sense, if it is true that the number of schools included in the study and the size of the studied sample do not allow to completely generalize the results to all the child population in Valle de Chalco, for only three schools and a small amount of school children were studied, leaving aside those who were not in school at the time of the selection which could, in turn, bias (healthy child) the study, it is also true that the selection of the schools and children were done randomly, taking into consideration the total number of schools in the area, implying that all the children in Chalco had the same probability of being included in the study. On the other hand, according to official national surveys (Censo Nacional de Poblacion y Vivienda 2010), the proportion of children between 6 and 14 years of age that go to a school is of 94%, in such a way that the studied children may very well represent the child population of this area.
Another factor that contributed to the limited size of the studied sample was the high cost of the tests for the diagnosis of the infection (IFI and dark field). These facts did not allow obtaining a larger sample, enough to identify evaluate more precisely the magnitude of association strength with some of the risk factors related with the infection. Including a higher number of schools and children would allow decreasing random errors and obtaining statistical significance and more precise trust intervals.
Similarly, the magnitude and the low risks found in this study have a direct relation with the high exposure of all the population to the studied factors of both the infected and the non-infected.
However, the relevance of the research does not reside so much in the risk factors for the infection that, although important, have been identified in multiple studies [3,4,15], but in the identification of the proportion of children with infection background, as well as those in whom the presence of the bacterium was identified in blood and urine "in a persistent way" without the presence of clinical data [33]. The need to continue this research line to corroborate if this fact determines a condition that does not represent a hazard for the children or it is a preclinical or chronic phase of leptospirosis already described in studies performed in Mexico by some of the members of our work team [19-21].
Other aspect to be considered was the cutoff value (1:80) used to classify the positive according to the official normativity [28], since when we performed the exercise with the lowest cut (1:40) the risk was increased. However, although the risks were low with the cutoff that was used, there was consistency in the findings from other studies and it fulfilled the established normativity that defines as cutoff value ≥ 1:80 titers to consider an individual as infected. Currently, there is evidence that refer infected individuals in acute of chronic stages that may react without significant increases, depending on the infected strains or the immune response of each individual as a consequence of repeated exposure [3,15,20,21].
This is the first study performed in children apparently healthy in a conurbation in our country, with environmental and cultural conditions that reflect in a representative way what might be happening in other areas; especially nowadays, when the close contact with dogs has substantially increased, corresponding to the concept of "dog humanization", and that are one of the main transmitters of the disease in urban areas [24,36]. We consider this highly important, even more if we consider the close relation between the number of infected dogs and children found in the present study.
Getting out this information is fundamental to offer doctors the tools for diagnosis and treatment of this disease in children, once the prognosis in infected individuals depends of an early diagnosis [23].
Promoting health to avoid close contact with potentially sick animals or with their excrement, as well as areas or water contaminated by these, are fundamental for the prevention and control of this zoonosis.
References
-
Roca B (2006) Leptospirosis. Rev Med Univ Navarra 50: 3-6.
-
Najera S, Alvis N, Babilonia D, Alvares L, Mattar S (2005) Leptospirosis ocupacional en una region del Caribe colombiano. Salud Publica Mex 47: 240-244.
-
(2008) Leptospirosis humana: guia para el diagnostico, vigilancia y control. Organizacion Mundial de la Salud; traduccion del Centro Panamericano de Fiebre Aftosa. - Rio de Janeiro: Centro Panamericano de Fiebre Aftosa -VP/OPS/OMS.
-
Informal Consultation on Global Burden of Leptospirosis: Methods of Assessment. WHO. Department of Food Safety, Zoonoses and Foodborne Diseases Sustainable Development and Healthy Environments. Geneva, 25-27 October 2006.
-
Heymann David L (2005) El control de las enfermedades transmisibles 18a (edn). Publicacion Cientifica y Tecnica No. 613. OPS. Washington, D.C., USA, 415-418.
-
Hartskeerl Rudy A (2005) International Leptospirosis Society: objectives and achievements. Rev Cubana Med Trop 57: 7-10.
-
Jansen A, Schoneberg I, Frank C, Alpers K, Schneider T, Stark K (2005) Leptospirosis in Germany 1962-2003. Emerg Infect Dis 11: 1048-1054.
-
Cespedes M (2005) Leptospirosis: Enfermedad Zoonotica Reemergente. Rev Peru Med Exp Salud Publica 22: 290-307.
-
Bharadwaj R (2004) Leptospirosis - a reemerging disease? Indian J Med Res 120: 136-138.
-
Sanders EJ, Rigau-Perez JG, Smits HL (1999) Increase of leptospirosis in dengue-negative patients after a Hurricane in Puerto Rico in 1966. Am J Trop Med Hyg 61: 399-404.
-
Vado-Solis IA, Cardenas-Marrufo MF, Laviada-Molina H, Vargas-Puerto F, Jimenez-Delgadillo B, et al. (2002) Estudio de casos clinicos e incidencia de leptospirosis humana en el estado de Yucatan, Mexico durante el periodo 1998 a 2000. Rev Biomed 13: 157-164.
-
Colin-Ortiz J, Perez-Serna J, Caballero-Servin A, Garcia-Romero J, Ibarra-Lopez L, et al. (2004) Seroprevalencia a leptospiras en grupos de riesgo de Guadalajara, Jalisco. Enf Inf Microbiol 24.
-
Benavides PL, Lopez HE, Torres BJ (2006) Niveles de anticuerpos antileptospira en poblacion humana aparentemente sana de la ciudad de Mexico. Revista Mexicana de Ciencias Farmaceuticas 37: 10-15.
-
Navarrete-Espinosa J, Moreno-Munoz M, Rivas-Sanchez B, Velasco-Castrejon O (2011) Leptospirosis Prevalence in a Population of Yucatan, Mexico. J Pathogens.
-
Ajay RB, Jarlath EN, Jessica NR, Michael A M, Diaz MM, et al. (2003) Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis 3: 757-771.
-
Perez SJ, Colin OJ, Caballero SA, Cuellar EA (2005) Seropositividad a Leptospiras de pacientes sospechosos de hepatitis viral negativos a marcadores serologicos. Rev Cubana Med Trop 57: 57-58.
-
Elizalde CAA, Tenorio GG, Velasco CO (2004) Identificacion de Leptospira en la patogenesis de la uveitis cronica en la ciudad de Mexico. Rev Mex Oftalmol 78: 165-70.
-
Velasco-Castrejon O, Rivas-Sanchez B, Gutierrez E, Chavez L, Duarte P, et al. (2005) Leptospira simulador o causante de leucemia? Rev Cubana Med Trop 57: 17-24.
-
Velasco-Castrejon O, Rivas-Sanchez B, Sanchez-Spindola ME, Soriano J, Rivera-Reyes HH, et al. (2009) Leptospirosis cronica en Mexico: diagnostico microscopico y evidencias que respaldan su existencia e importancia. Rev Mex Patol Clin 56: 157-167.
-
Velasco CO, Rivas SB (2006) Leptospirosis humana cronica. En: Narro RJ, coordinador, Diagnostico y tratamiento en la practica Medica. Manual Moderno Mexico 641-650.
-
Velasco-Castrejon O, Rivas-Sanchez B, Rivera-Reyes HH (2002) Transicion de la leptospirosis aguda a cronica. Rev Cubana Med Trop 54: 66-67.
-
Suarez HM, Rodriguez MG, Torres MO, Marrero AJL, Alonso SJM (2006) Leptospirosis en ninos de una provincia cubana. Rev Mex Pediatr 73: 14-17.
-
Garcia LIS, Cruz CM, Lugo IZ, Sanchez BR, Castrejon OV, et al. (2013) Fatal Leptospirosis Case in Pediatric Patient: Clinical Case. OJMM, 3: 12-17.
-
Luna AMA, Moles CLP, Gavaldon RD, Nava VC, Salazar GF (2008) La Leptospirosis canina y su problematica en Mexico. Rev. Salud Anim 30: 1-11.
-
Gamage CD, Amarasekera J, Palihawadana P, Samaraweera S, Mendis D, et al. (2012) Analysis of Hospital-Based Sentinel Surveillance Data on Leptospirosis in Sri Lanka, 2005-2008. Jpn J Infect Dis 65: 157-161.
-
Aguedo-Flores P, Restreto M, Loreto M (2006) Evaluacion de la prueba de inmunofluorescencia indirecta para el diagnostico de leptospirosis humana. Biomedica. 1019-1024.
-
Velasco-Castrejon O, Rivas-Sanchez B, Becker A (2002) Vecovision, un nuevo metodo imagenologico para el diagnostico definitivo de leptospirosis. Rev Cubana Med Trop 54: 67.
-
Norma Oficial Mexicana NOM-029-SSA2-1999, para la vigilancia epidemiologica, prevencion y control de la leptospirosis en el humano.
-
Wasinski B, Sroka J, Wojcik-Fatla A, Zajac V, Cisak E, et al. (2012) Seroprevalence of leptospirosis in rural populations inhabiting areas exposed and not exposed to floods in eastern Poland. Ann Agric Environ Med 19: 285-288.
-
Cespedes M, Tapia R, Balda L, Gonzalez D, Glenny M, et al. (2009) Brote de leptospirosis asociado a la natacion en una fuente de agua subterranea en una zona costera, Lima - Peru. Rev Peru Med Exp Salud Publica 26: 441-448.
-
Mena-Castro E, de Luna E, Gonzalez A, Ramirez AR, Jarvis V (2008) Leptospirosis e Insuficiencia Renal en Ninos. Arch Dom Ped 41: 3-6.
-
Visith S, Kearkiat P (2005) Nephropathy in Leptospirosis. J Postgrad Med 51: 184-188.
-
Ganoza Christian A, Matthias MA, Mayuko S, Manuel C, Eduardo G, et al. (2010) Asymptomatic renal colonization of humans in the Peruvian Amazon by Leptospira. PLoS Negl Trop Dis 4: 1-10.
-
Velasco Castrejon O, Rivas Sanchez B (2010) Leptospirosis transmitida del hombre al perro. Enf Inf Microbiol 30: 106-109.
-
oscar VC, Beatriz RS, Jacqueline EH, Enrique MH (2007) Diagnostico de leptospirosis cronica, comparacion entre la aglutinacion microscopica y 3 tecnicas diagnosticas confirmatorias. Rev Cubana Med Trop 59: 1-8.
-
Rivera Flores A, de la Pena Moctezuma A, Roa Riol MA, Ordonez Badillo ML (1999) Seroprevalencia del leptospirosis en perros callejeros del norte de la ciudad de Mexico. Vet Mex 30: 105-107.