International Journal of Pediatric Research
Linear Growth Predicts Acute Post-Transplant Outcomes in Paediatric Liver Transplant Patients
Jillian S Owens1, Michele Strom1,2, Farsad Farassati1,2, Krista Van Roestel1,2,3, Kathryn Chambers1,2,3, Penni Kean1,2,3, Megan Carricato1,2,3, Vicky L Ng1,2,3,4, Yaron Avitzur1,2,3,4 and Glenda Courtney Martin1,2,3,5*
1Research Institute, The Hospital for Sick Children, Toronto, Canada
2Gastroenterology, Hepatology and Nutrition, The Hospital for Sick Children, Toronto, Canada
3SickKids Transplant Centre, The Hospital for Sick Children, Toronto, Canada
4Department of Pediatrics, University of Toronto, Toronto, Canada
5Faculty of Kinesiology & Physical Education, University of Toronto, Toronto, Canada
*Corresponding author: Glenda Courtney-Martin, Department of Clinical Dietetics, The Hospital for Sick Children, 555 University Avenue, Toronto, ON, Canada M5G 1X8, Tel: 416-813-5744, E-mail: Glenda.courtney-martin@sickkids.ca
Int J Pediatr Res, IJPR-2-019, (Volume 2, Issue 1), Research Article; ISSN: 2469-5769
Received: January 04, 2016 | Accepted: May 21, 2016 | Published: May 24, 2016
Citation: Owens JS, Strom M, Farassati F, Roestel KV, Chambers K, et al. (2016) Linear Growth Predicts Acute Post-Transplant Outcomes in Paediatric Liver Transplant Patients.
Int J Pediatr Res 2:019. 10.23937/2469-5769/1510019
Copyright: © 2016 Owens JS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: Minimal research has been conducted on the effect of nutritional status on acute post-transplant outcomes in pediatric liver transplant patients. The purpose of this study was to determine the effect of pre-transplant nutritional status (height, weight, albumin, hemoglobin, Vitamin A, Vitamin E, Vitamin D, mean corpuscular volume (MCV), calcium, magnesium and phosphorus) on acute post-transplant outcomes (length of hospital stay and infection rate).
Methods: We conducted a retrospective chart review of all children who received a liver transplant between January 1999 and December 2008 at the Hospital for Sick Children in Toronto.
Results: Data were analyzed for a total of 128 children; average age at transplant was 6 years. Children with a height z-score ≤ -2.5 had a longer length of stay and greater number of infections than those who had a height z-score of > -2.5 (54.7 vs. 34.8 d). In addition, those with a height z-score of ≤ -1.5 had a longer length of stay and more infections than those with a height z-score of > -1.5 (45 vs. 35 d).
Conclusions: The acute post-transplant outcomes of pediatric patients who underwent liver Transplantation for liver failure was significantly affected by height z-scores pre-transplantation. Children with poor linear growth had a length of stay up to 20 days longer and a 30% higher infections rate than those with improved linear growth. Children with liver disease should be aggressively monitored pre-transplant to ensure adequate linear growth.
Keywords
Nutritional status, Growth, Liver transplantation, Length of stay, Infection rate
Abbreviations
ESLD: End Stage Liver Disease; MCV: Mean Corpuscular Volume; CDC: Centre for Disease Control; ICU: Intensive Care Unit; MCH: Mean Corpuscular Haemoglobin; TPN: Total Parenteral Nutrition
Introduction
Chronic liver disease is associated with poor nutritional status in children particularly if cholestasis is longstanding and the onset is in infancy [1,2]. Early satiety due to enlarged liver, spleen or ascites as well as anorexia leads to inadequate food intake and protein energy malnutrition. Changes in amino acid metabolism and increased energy requirements [3] contribute to suboptimal energy and nitrogen balance, and fat malabsorption as a result of decreased bile flow further contributes to negative energy balance as well as micronutrient deficiencies [4].
Liver transplantation is a successful treatment for both children and adults with end stage liver disease (ESLD) and provide a high quality of life post-transplant [5-8]. However, malnutrition impacts negatively on acute post-transplant outcomes [9,10]. Eighty percent of adults with ESLD requiring transplantation are considered malnourished [11,12]. This results in increased morbidity and mortality [12] longer length of stay in the intensive care unit (ICU) as well as a longer overall length of hospital stay [13]. In children, those who are malnourished pre-transplant have higher infections rates, more surgical complications, and higher mortality compared to those that are better nourished [14,15]. In addition, children with height z-scores < -1.5 stayed in hospital twice as long as children with height z-scores > -1.5 with an increased hospital cost of $40,000 average per child [10].
The Hospital for Sick Children liver transplant program has performed over 350 transplants since its inception in 1986. Although nutritional assessments are performed routinely pre-transplant, an understanding of the specific nutritional factors that predict adverse outcomes will provide a focused approach to nutritional interventions pre-transplant so that post transplant outcomes can be optimized. The goal of this study therefore was to determine the preoperative nutritional parameters that are predictive of post-operative outcomes after liver LT in pediatric patients at a large pediatric liver transplant center.
Methods
Subject selection and description
A retrospective chart review was conducted to identify patients who underwent isolated liver transplantation between January 1999 and December 2008. The following patients were excluded: 1) those who received a LT because of parenteral nutrition associated liver disease secondary to an underlying diagnosis of intestinal failure and 2) those who received multiple organ transplants (liver and small bowel or liver and kidney), and those who died before discharge. All other patients were included. Those included fell into one of the following five categories; cholestatic liver disease, metabolic disease, tumors, immune-mediated condition, and acute liver failure.
January 1999 to December 2008 was chosen to eliminate the period when both cyclosporine and tacrolimus (FK 506) were both used as immunosuppressive drug to capture a period when mainly tacrolimus was used. Our intention was to have a cohort of patients on the same immunosuppressive drug.
After patient identification, data were collected on subject characteristics: age, height, weight and diagnosis requiring transplant, markers of nutritional status: serum albumin, Vitamin A, Vitamin E, Vitamin D, hemoglobin, MCV, mean corpuscular hemoglobin (MCH), on the day of or within two weeks of transplant and outcomes: total length of in-hospital stay and number of infections. Standard practice at our hospital is to do standing height on children ≥ 2 years and recumbent length on those ≤ 2 years. Therefore, we expect that this procedure was followed when measuring height for all children included in this study. An infection was categorized as a diagnosis made in the medical record of a viral, bacterial, or fungal infection and treated with antibiotic or antifungal medication for bacterial or fungal infection and conservative management for viral infection. The acute post-transplant timeframe was defined as up to and including six months. Six months was chosen because it has previously been chosen as an acute post transplant period in the published transplant literature [15].
Prior to data collection, the data collection team (JO, MS, FF, KVR, KC, PK, MG and GCM) met on several occasion to discuss the process; including how and where the data will be collected and the relevant data to be collected. A standardized data collection form (approved by the Research Ethics Committee) was used to collect all relevant information for the study. Data collation did not begin until each one felt comfortable and followed the standard procedure of obtaining the relevant data. In addition, severalmeetings were held over the course of the data collection period to ensure the standardized process was maintained. The principal investigator GCM was responsible for the supervision of the data collection.
This study was approved by the Human Research Ethics Committee at The Hospital for Sick Children (Toronto, Canada).
Statistics
Statistical analyses were performed on primary and derived variables. Patients were stratified into five categories based on diagnosis for initial analysis. These categories were: cholestatic disease, acute liver failure, metabolic disease, tumors, and immune-mediated disease. The purpose of this stratification was to determine if there were significant differences among the outcomes of interest based on diagnoses. Analysis of variance was used to test for differences in outcome among the different diagnosis. Simple linear regression analysis was used to examine associations between each predictor variable and each outcome variable. Where no relationship existed the variables were removed from subsequent analysis. Significant associations were then assessed using multiple linear regressions, with ANCOVA used to test for confounders. Weight and height Z-scores were calculated using the Centre for Disease Control (CDC) reference data set: (http://spitfire.emmes.com/study/ped/resources/htwtcalc.htm). The CDC data set was used to calculate the weight and height z-scores because at our hospital, the CDC growth charts had been used for many years to clinically assess nutritional status in our patients. Therefore, the growth status of all patients included in this cohort would have been plotted on CDC growth charts. Since we only switched to using the World Health Organization (WHO) growth in 2013, we felt it was inappropriate to use the WHO charts to assess the height and weight z-score of the children in this study because their nutritional status and growth pre and post liver transplantation would have been monitored using the CDC growth charts.
All analysis for heights and weights were conducted using the corresponding z-scores. Z-score stratification of ≤ -2.5 and > -2.5 as well as ≤ -1.5 and > -1.5 were then used to test associations between the outcomes of interest.
Patients were subsequently stratified by cholestatic liver disease and non-cholestatic liver disease for a sub-group analysis as cholestatic liver disease is known to be associated with more significant malnutrition. Statistical significance was established at p < 0.05. Statistical analyses were performed using SAS version 9.3 for Windows.
Results
A total of 150 children received a liver transplant from January 1999 to December 2008. Of those, 22 were excluded, 3 received liver and kidney, 15 were transplanted as a result of underlying intestinal failure and parenteral nutrition related liver disease and 4 died in hospital before discharge. A total of 128 patients who received a liver transplant from January 1999-December 2008 met inclusion criteria and were included in the study. Characteristics of all 128 patients included in this study's cohort are presented in table 1. Briefly, the average age of the patients was 5.98 ± 5.6 (mean ± SD). Seventy two patients (56%) had cholestatic liver disease, 19 (15%) had acute liver failure, the other 29% were transplanted for metabolic disease, tumors or immune mediated disorders (Table 1). Height data was only available for 111 of the 128 patients whereas weight data was available for all patients. The average height and weight z-score pre-transplant were -0.60 ± 2.03 and -0.49 ± 1.63 respectively (mean ± SD). When our main outcome of interest were assessed, post liver transplantation the average length of hospital stay per patient was 38.14 ± 39.81) days (mean ± SD) and patients developed 1.13 ± 1.51 (mean ± SD) infections. The results of the ANOVA showed no differences in outcome based on diagnosis therefore, all groups were collapsed for future analysis.
![]()
Table 1: Subject characteristics of 129 children who received a liver transplant (LT) between January 1999-December 2008.
View Table 1
Associations
When the entire cohort was assessed, there was no association between age at transplant and length of hospital stay post-transplant. There was also no association between age at transplant and infection rates post-transplant. There were no associations between overall weight and length z-scores and length hospital stay or infection rates post-transplant. There were no associations between any of the markers of nutritional status and the outcomes of interest. Serum albumin was available for 118 of the 128 patients. The mean albumin concentration was 30.19 ± 6.26 (mean ± SD) with an upper and lower 95% CI of 29.05 to 31.34. There was a trend towards significance between pre-transplant serum albumin and infection rates (p = 0.085) but it was not statistically significant.
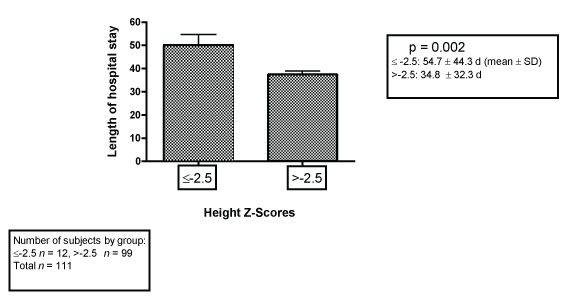
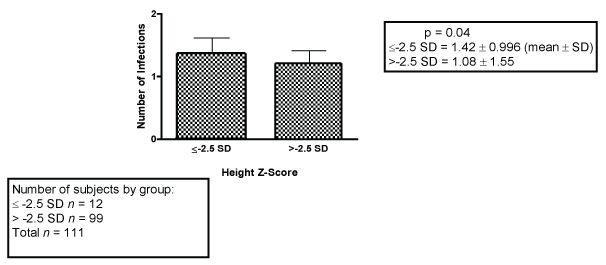
After stratification by height and weight z-scores (All 128 patients): using cut-off of -2.5 SD: After stratification by -2.5 SD, weight z-score did not affect length of hospital stay or number of infections post liver transplant in this cohort of pediatric patients who underwent liver transplantation between January 1999 and Dec 2008. Data on height was available for 111 of the 128 patients in this cohort. Children with a height z-score of ≤ -2.5 had longer hospital stay post liver transplantation than those with a height z-score > -2.5 (p = 0.002). The average length of stay for those with a height z-score of ≤-2.5 was 54.7 days whereas average length of stay for those with a height z-score of > -2.5 was 34.8 days (Figure 1). Additionally patients who had a height z-score ≤-2.5 had a significantly higher number of infections post transplantation that those with a height z-score > -2.5 (p = 0.04) (Figure 2). Those with a height z-score of ≤ -2.5 had an average of 1.42 infections compared with 1.08 in those with height z-score > -2.5 (Figure 2). Of the 12 patients with height z-score ≤ -2.5, (Figure 1 and Figure 2), 10 had cholestatic liver disease and 9 of those had Biliary Atresia.
.
Figure 1: Relation between Height Z-score (≤ -2.5 SD and > -2.5 SD) and length of hospital stay post liver transplantation in pediatric patients who underwent liver transplantation between January 1999 and December 2008.
View Figure 1
.
Figure 2: Relation between Height Z-score (≤ -2.5 SD and > -2.5 SD) and number of infections post liver transplantation in pediatric patients who underwent liver transplantation between January 1999 and December 2008.
View Figure 2
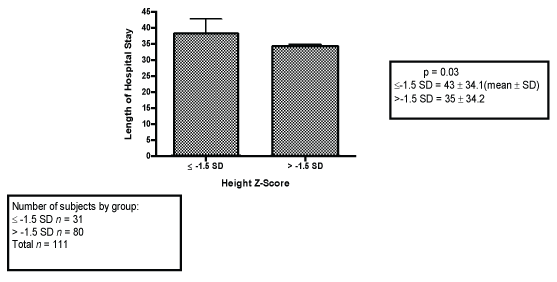
After stratification by height and weight z-scores (all 128 patients): using cut-off of -1.5 SD: Using a cut off of -1.5 SD, weight z-score was not associated with any of the outcomes of interest. Of the 128 patients data on height was available for 111 patients. There was a trend towards a higher number of infections in those patients with a height z-score ≤ -1.5 than in those with height z-score > -1.5. However, it was not statistically significant (p = 0.08). Patients with a height z-score ≤ -1.5 spent 43 ± 34.1(mean ± SD) days in hospital post liver transplantation whereas those with height z-scores > -1.5 spent 35 ± 34.2 days (p = 0.03) (Figure 3). Of the 31 patients with height z-score ≤ -1.5 , 24 had cholestatic liver disease (22 Biliary Atresia, 2 Alagille's Syndrome).
.
Figure 3: Relation between Height Z-score (≤ -1.5 and > -1.5) and number of infections post liver transplantation in pediatric patients who underwent liver transplantation between January 1999 and December 2008.
View Figure 3
Results of sub-analysis: Children with cholestatic liver disease only: Subject characteristics for this subset of children can be found in table 2. This subset of patients with cholestatic liver disease was mostly made up of children with Biliary Atresia 79% they were younger that the full cohort of patients described in table 1, with a mean age of 4.83 vs 5.98 years. However, there was no relationship between z-score for weight or length and length stay or infection rate in this subset of patients.
![]()
Table 2: Subject characteristics of a subset of 72 patients - those with cholestatic liver disease who received a liver transplant between January 1999 - December 2008.
View Table 2
Discussion
In this large cohort of 128 pediatric patients who underwent liver transplant at a single centre between January 1999 and Dec 2008, poor linear growth as characterized by height z-score ≤ -2.5 standard deviations (SD) below the mean was associated with an almost 60 percent increase in hospital stay and a 30% increase in the number of infections in patients after receiving a liver transplant. Further, when a height z-score cut off of -1.5 SD was used, those with height z-score ≤ -1.5 has a 23% longer hospital stay post liver transplantation than those with height z-score > -1.5 SD.
Many have evaluated the relationship between nutritional status pre transplant and post liver transplant outcomes in adults [9,15-17] as well as pediatric patients [10,14,18]. However, only one study looked at the relationship between height z-score in children and length of hospital stay post transplant [10]. Variation in the indices used to classify malnutrition in the adult studies have led to conflicting results because many of the indices evaluated (such as serum albumin, prothrombin time) are a reflection of the severity of the underlying liver diseases rather than malnutrition [12]. In children, growth is the best indicator of nutritional status as growth responds rapidly to changes in caloric and nutrient intake; weight first and height later (usually within three months depending on age) [19,20]. In the developing world, children with a weight-for height z-score of -3 to < -2 or < -3 are three and ten times respectively, more likely to die than those with weight-for height z-score of -1 [21].
We did not see a relationship between weight z-score in the present study and any of our outcomes of interest. Shepherd et al. [18] previously showed an increased 1 year survival of 88% in children transplanted for end stage liver failure with a weight z-score > -1.0 when compared with a 38% survival in those with a lower weight z-score. Since those outcomes are different than the acute post transplant examined in our study, it is difficult to compare. However, in clinical studies evaluating the effect of nutritional status on liver transplant outcomes, linear growth is a more reliable indicator than weight [10,18], because in liver disease weight is affected by the presence of varying degree of ascites and increasing liver and or spleen size [11].
The results for the entire cohort of patients in the current study show that poor linear growth even -1.5 SD below the population mean had a significant negative effect on length of hospital stay post liver transplantation but that the negative effects of poor linear growth on susceptibility to infections manifest itself at higher growth deficits i.e. ≤ -2.5 SD below the mean. Our results are in agreement with those of Barshes et al. [10] who showed that pediatric patients with a lower height z-score (≤ -1.5) had a longer length of stay in hospital after liver transplant than those with a higher height z-score (> -1.5). In that study, poorer linear growth resulted in an increased cost of an additional $40,000 per patient. This is important because in an era of rising healthcare costs it is important to minimize these costs while at the same time optimizing patient's outcomes and achieving high value for patients. Thus efficiency which relates outcome to cost is an important consideration [22]. The patients in our study had an overall longer hospital stay post liver transplant than those in the Barshes study [10]. Our patients with height z-score ≤ -1.5 stayed an average 43 days compared with 20.5 in the Barshes study. In addition those with a height z-score ≤ -2.5 stayed 54.7 days which is almost 30% longer than those with a height z-score ≤ -1.5. It is unclear what accounted for the higher length of stay in the current study compared with the Barshes study. It is possible that differences in clinical protocol could play a role. The results of our study suggest that assessing/evaluating nutritional factors such as pre-transplant height with an aggressive intent to minimize failure could potentially decrease morbidity, and reduce healthcare costs associated with pediatric liver transplantation.
In prospective studies in adults on the effect of nutritional status on outcome in liver transplantation [15,16], malnourished patients had up to 7 times the number of infections when compared with those who were well nourished [16]. Thus our data is in agreement with those data that malnutrition increased infection rate in liver transplantation. In the current study, patients with height z-score ≤ -2.5 had a 31% increase number of infections compared with those with a height z-score > -2.5. A total of 12 patients had height z-score ≤ -2.5 and of those 10 had cholestatic liver disease. Interestingly, when the height z-score was stratified by -1.5 SD, the number of infections was not different between those ≤ -1.5 or > -1.5. This is of interest as it suggests a threshold beyond which chronic undernutrition begins to exert a negative effect on the immune system. In the developing world, children with weight-for height z-score of -3 to < -2 are three times more likely to die from malnutrition related causes whereas those with weight-for-height z-scores < -3 are ten times, more likely to die than those with weight-for height z-score of -1.0 [21]. This phenomenon could likely be at work in liver transplantation as well based on the observation from the current study.
In a sub analysis of the 72 patients with cholestatic liver disease in this cohort, height z-score whether stratified by -2.5 or -1.5 SD, did not have a significant impact on the outcomes of interest. It is recognized that cholestatic liver disease results in significant growth retardation and malnutrition in children [23]. The malnutrition observed in cholestatic liver disease is not only characterized by decreased nutrient intake but also by significant malabsorption, abnormalities in protein metabolism and increased energy expenditure [23]. Patients with cholestatic liver disease can be characterized as having a heavier disease load than other patients with ESLD. Therefore, we expected to see a relationship between height z-score and outcome in this study. Failure to see a relationship could have been because of the reduction in the number of patients in this sub- analysis or a greater similarity among patients.
We did not find any relationship between albumin or any of the other predictor variables assessed and our outcomes of interest. This could likely be because they are not reflective of nutritional status, but rather the severity of the underlying disease. Nevertheless, it must be noted that many of the data were missing from the medical charts therefore, were available for inclusion.
The results of this study showed that pre-transplant height z-score is an important predictor of post-transplant outcomes. In the current study patients with height z-scores of ≤ -2.5 and ≤ -1.5 had a longer length of hospital stay which was 30% longer in those with a height z-score ≤ -2.5 than in those who were ≤ -1.5. Additionally, patients with a height z-scores ≤ - 2.5 had a 30% higher infection rate than those with a height z-score > -2.5.
The main limitation of our study is that it is retrospective in nature. The number of patients evaluated however makes the current study the largest to our knowledge to evaluate the relationship between nutritional status and outcomes after liver transplant in a cohort of pediatric patients. The results suggest that pediatric patients with ESLD experience growth stunting pre-transplant which has a negative impact on post transplant length of hospital stay as well as infection rate. Whether aggressive pre-transplant nutrition therapy implemented from the onset of diagnosis could alleviate these negative consequences post-transplant remains to be assessed in a prospective trial.
In summary, children with pre-transplant height-for age z-scores ≤ -2.5 had an almost 60% longer hospital stay than those with a height-for age z-score > -2.5. Those with a height-for age z-scores ≤ -1.5 had a 23% longer stay than those with a height-for age z-scores > -1.5 and those with a height-for age z-scores ≤ -2.5 had a 27% longer stay than those with a height-for age z-scores ≤ -1.5. Additionally, children with a height-for age z-scores ≤ -2.5 experienced a 30% higher infection rate than those with a height-for age z-scores > -2.5.
Given that length of stay and infection rates are increased in pediatric liver transplant recipients with compromised liner growth, the results of this study supports the application of aggressive medical nutritional management strategies by the clinical team caring for patients with chronic liver disease in order to increase value in pediatric liver transplantation. Therefore, medical nutritional managements of pediatric patients with chronic liver disease requiring transplantation should aim to achieve a height z-score > -1.5 SD in order to optimize outcome and decrease morbidity and mortality in the acute post liver transplantation period.
Authors Contributions
G.C-M, Y.A and V.N designed the study, J. O, M. S, F. F, K. V R, K.C, M.C, P.K, and G.C-M, collected the data, J.O and G.C-M analyzed the data, J.O and G. C-M wrote the manuscript. All authors have read and contributed to the final version of the manuscript. G. C-M is responsible for the final contents of the manuscript.
Conflict of Interest
Authors J Owens, M Strom, F Farassati, K Van Roestel, Y. Avitzur, K Chambers, Vicky Ng, P Kean, M Carricato and G Courtney-Martin have no conflict of interest.
Source of Funding
Canadian Society of Transplantation
References
-
Sokol RJ (1987) Medical management of the infant or child with chronic liver disease. Semin Liver Dis 7: 155-167.
-
Kaufman SS, Murray ND, Wood RP, Shaw BW Jr, Vanderhoof JA (1987) Nutritional support for the infant with extrahepatic biliary atresia. J Pediatr 110: 679-686.
-
Pierro A, Koletzko B, Carnielli V, Superina RA, Roberts EA, et al. (1989) Resting energy expenditure is increased in infants and children with extrahepatic biliary atresia. J Pediatr Surg 24: 534-538.
-
Chin SE, Shepherd RW, Thomas BJ, Cleghorn GJ, Patrick MK, et al. (1992) The nature of malnutrition in children with end-stage liver disease awaiting orthotopic liver transplantation. Am J Clin Nutr 56: 164-168.
-
Chin SE, Shepherd RW, Cleghorn GJ, Patrick MK, Javorsky G, et al. (1991) Survival, growth and quality of life in children after orthotopic liver transplantation: a 5 year experience. J Paediatr Child Health 27: 380-385.
-
Avitzur Y, De Luca E, Cantos M, Jimenez-Rivera C, Jones N, et al. (2004) Health status ten years after pediatric liver transplantation--looking beyond the graft. Transplantation 78: 566-573.
-
Spada M, Riva S, Maggiore G, Cintorino D, Gridelli B (2009) Pediatric liver transplantation. World J Gastroenterol 15: 648-674.
-
Ng VL, Alonso EM, Bucuvalas JC, Cohen G, Limbers CA, et al. (2012) Health status of children alive 10 years after pediatric liver transplantation performed in the US and Canada: report of the studies of pediatric liver transplantation experience. J Pediatr 160: 820-826 e3.
-
Figueiredo F, Dickson ER, Pasha T, Kasparova P, Therneau T, et al. (2000) Impact of nutritional status on outcomes after liver transplantation. Transplantation 70: 1347-1352.
-
Barshes NR, Chang IF, Karpen SJ, Carter BA, Goss JA (2006) Impact of pretransplant growth retardation in pediatric liver transplantation. J Pediatr Gastroenterol Nutr 43: 89-94.
-
Lautz HU, Selberg O, Korber J, Burger M, Muller MJ (1992) Protein-calorie malnutrition in liver cirrhosis. Clin Investig 70: 478-486.
-
Ferreira LG, Anastacio LR, Lima AS, Correia MI (2011) Assessment of nutritional status of patients waiting for liver transplantation. Clin Transplant 25: 248-254.
-
Stephenson GR, Moretti EW, El-Moalem H, Clavien PA, Tuttle-Newhall JE (2001) Malnutrition in liver transplant patients: preoperative subjective global assessment is predictive of outcome after liver transplantation. Transplantation 72: 666-670.
-
Moukarzel AA, Najm I, Vargas J, McDiarmid SV, Busuttil RW, et al. (1990) Effect of nutritional status on outcome of orthotopic liver transplantation in pediatric patients. Transplant Proc 22: 1560-1563.
-
Harrison J, McKiernan J, Neuberger JM (1997) A prospective study on the effect of recipient nutritional status on outcome in liver transplantation. Transpl Int 10: 369-374.
-
Merli M, Giusto M, Gentili F, Novelli G, Ferretti G (2009) Nutritional status: its influence on the outcome of patients undergoing liver transplantation. Liver Int 30:208-214.
-
Selberg O, Bottcher J, Tusch G, Pichlmayr R, Henkel E, et al. (1997) Identification of high- and low-risk patients before liver transplantation: a prospective cohort study of nutritional and metabolic parameters in 150 patients. Hepatology 25: 652-657.
-
Shepherd RW, Chin SE, Cleghorn GJ, Patrick M, Ong TH, et al. (1991) Malnutrition in children with chronic liver disease accepted for liver transplantation: clinical profile and effect on outcome. J Paediatr Child Health 27: 295-299.
-
UNICEF Wa (2009) WHO Child growth standards and the identification of severe acute malnutrition in infants and children. Genever, Switzerland 2009.
-
Mehta NM, Corkins MR, Lyman B, Malone A, Goday PS, et al. (2013) Defining pediatric malnutrition: a paradigm shift toward etiology-related definitions. JPEN J Parenter Enteral Nutr 37: 460-481.
-
Black RE, Allen LH, Bhutta ZA, Caulfield LE, Onis M de, et al. (2008) Maternal and child under nutrition: global and regional exposures and health consequences. Lancet 371: 243-260.
-
Porter ME (2010) What is value in health care? N Engl J Med 363: 2477-2481.
-
Pawlowska J, Socha P, Jankowska I (2010) Factors affecting catch-up growth after liver transplantation in children with cholestatic liver diseases. Ann Transplant 15: 72-76.





