International Journal of Pediatric Research
Infantile Blount's Disease: Histopathological Changes in the Proximal Tibial Metaphysis. Comparison between Medial and Lateral Specimens
Freek Hollman1, Pascal Vroemen1, Lodewijk Van Rhijn1, Dam Surtel1, A Cremers1, Paul Rompa2, Prosper Moh3 and Heleen Staal1*
1Department of Orthopaedic Surgery, Maastricht University Medical Centre, The Netherlands
2St. Anthony's Hospital, Ghana, West Africa
3St. John of God Hospital, Ghana, West Africa
*Corresponding author: Heleen Staal, MD, Department of Orthopedic Surgery, Research School Caphri, Maastricht University Medical Centre, P.O. box 5800, 6202 AZ, Maastricht, The Netherlands, Tel: 00-31-0-43-3875038, E-mail: h.staal@mumc.nl
Int J Pediatr Res, IJPR-2-025, (Volume 2, Issue 2), Original Article; ISSN: 2469-5769
Received: April 05, 2016 | Accepted: November 03, 2016 | Published: November 05, 2016
Citation: Hollman F, Vroemen P, Rhijn LV, Surtel D, Cremers A, et al. (2016) Infantile Blount's Disease: Histopathological Changes in the Proximal Tibial Metaphysis. Comparison between Medial and Lateral Specimens. Int J Pediatr Res 2:025. 10.23937/2469-5769/1510025
Copyright: © 2016 Hollman F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: This study will present the histological findings of biopsies acquired from the medial and lateral part of the proximal tibia during the W/M serrated osteotomy in patients with infantile Blount's disease. It is hypothesized that the medial metaphyseal area of the proximal tibia will present a different ossification pattern compared with the lateral area.
Methods: Patients (n = 4) were included prospectively. During two unilateral and two bilateral W/M serrated osteotomies, biopsies of the medial and lateral cortex of the proximal part of the serrations were taken. The specimens were fixated in formalin, decalcified, embedded in paraffin, sections (5 μm) were acquired and stained with hematoxylin/eosin or Safranin-O to determine the presence of glycosaminoglycans. All specimens were evaluated by light microscopy.
Results: From the medially obtained specimens, all showed presence of endochondral ossification. Intra-cortical chondrocyte-rich areas as well as presence of multiple irregular growth arrest lines were identified. Normal cortical and trabecular bone histology was observed in all lateral specimens.
Conclusions: In conclusion, in patients with infantile Blount's disease intra-cortical endochondral ossification was only found in the medial part of the metaphysis. This may reflect the clinical tibial vara deformity in Blount's disease in which increased mechanical load on the medial metaphysis causes failure of normal ossification.
Keywords
Blount's disease, Tibia vara, Osteotomy, Infantile, Histology, Aetiology
Introduction
Blount's disease (also known as tibia vara or osteochondrosis deformans tibiae) is characterized by tibial bowing, torsion and beaking of the posteromedial proximal tibia and involves the epiphysis, physis and metaphysis [1-3]. Infantile Blount's disease manifests between the age of four and ten years with progressive pain and laxity for which early surgical correction is recommended when conservative treatment with plaster correction has failed. The aetiology of infantile Blount's disease is probably multi-factorial. Associations are found between Blount's disease and the Afro-Caribbean race, early walking age and obesity [3-5]. Genetic predisposition is suggested and mechanical loading on the physis is often implicated [4,6,7]. Radiologically, morphologic changes are classified by Langenskiold and colleagues, which is based on age and severity of the down sloping of the medial epiphysis [8].
Macroscopic findings in the tibia on Magnetic Resonance Imaging (MRI) in cases of Blount's disease show a thicker layer of epiphyseal cartilage and increased height of the medial meniscus in the medial compartment [9,10]. These macroscopic findings may reflect the compensation of the diminished height of the ossified part of the medial epiphysis, amongst others caused by increased biomechanical stress [9,10]. Histopathological abnormalities of the proximal medial high tibial condyle are described in infantile and late-onset Blount's disease and consist of densely packed islands of hypertrophic chondrocytes, areas of acellular cartilage matrix and abnormal clusters of capillaries [11-13]. The disorganization and misalignment of the medial physeal zones might indicate asymmetric compressive and shear forces causing suppression and deviation of normal endochondral ossification in both the medial area of the metaphysis and corresponding epiphysis [1]. These alterations cause progression of the varus deformity for as long as blockage of ossification is present and the bone continues to grow in the lateral part of the growth zone [14].
Most histopathological studies are on late onset cases of Blount's disease and do not compare the medial with the lateral aspect of the proximal tibia. It is hypothesized that the medial metaphyseal area of the proximal tibia will present a different ossification pattern compared with the lateral area. In this study the histopathological findings in the medial metaphysis in cases of infantile Blount's disease are described and compared with the lateral side of the metaphysis.
Methods
Between November and October 2013, specimens (n = 6) were taken from the proximal metaphysis of four patients (2 unilateral, 2 bilateral) with infantile tibia vara in the St. Anthony's Hospital in Dzodze (Volta region, Ghana). All patients underwent uni- or bilateral W/M serrated osteotomy. Patients and their parents were informed about the study protocol and informed consent was given orally at the outpatient department. Mean age at surgery was 7.8 years (range: 6-10) and all were female. All patients were of Western African origin. Age of onset was in all patients before the age of 6 years. Based on clinical characteristics, as tibia vara combined with tibial torsion and radiological findings as described by Langenskiöld, patients were diagnosed with infantile Blount's disease. There was no history of trauma or infection in the affected legs. Patients were included prospectively. The tibias were radiologically classified according to the Langenskiöld classification (Table 1) [8]. The femorotibial angle was measured to point out the severity of the disease (Table 1). Two orthopaedic surgeons performed all osteotomies. An anterior vertical incision was used for subperiosteal exposure of the proximal shaft of the tibia just below the tibial tuberosity. Along the lateral compartment the proximal fibula was exposed and an oblique fibular osteotomy was performed with an osteotome about 6-8 cm distal to the fibular head. The 'M' shape was marked on the proximal tibia just distal to the physis of the tibial tuberosity. The width of the proximal tibia determines the length and width of the dentations of the 'M'. The distal parts of the serrations were disengaged anteriorly and replaced with a lateral shift of one tooth. During this stage biopsies of the medial and lateral cortex of the proximal part of the serrations, together with periosteum and trabecular bone, were taken (Figure 1). The size of the biopsies varied from 5 to 10 mm in longitudinal orientation. Finally, the tibia was derotated as desired and re-engaged in the corrected position. The specimens were fixated in 4% buffered formalin.
.
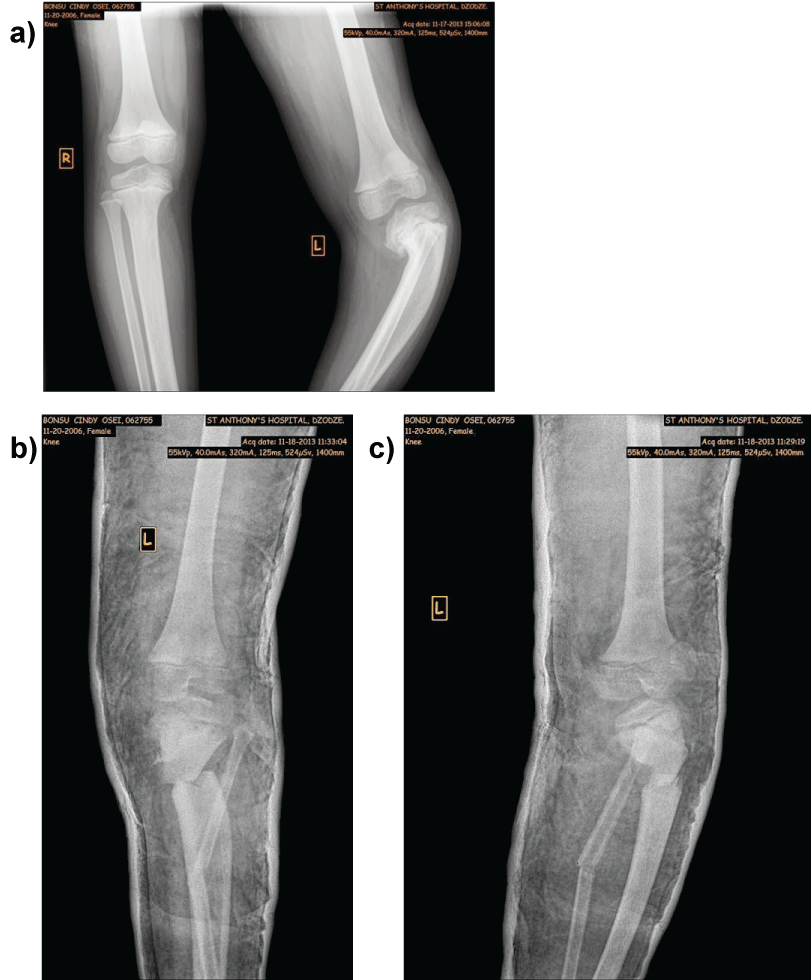
Figure 1: a,b) Clinical representation of a 7-year-old girl with Blount's disease, pre and post-operative radiographs.
a) Typical clinical manifestation of infantile Blount's disease with left sided unilateral tibia vara and tibial torsion; b) preoperative conventional radiograph with the growth plate inclined distally at the medial side and epiphysis extending down over the metaphyseal beak (Langenskiöld stage 4), the two arrows mark the location were the biopsies are obtained from the medial and lateral proximal metaphyseal tibia; c) directly postoperative conventional radiographic examination after performing the W/M serrated high tibial osteotomy.
View Figure 1
![]()
Table 1: Patient characteristics.
View Table 1
The specimens were decalcified using 0.5 M ethylenediaminetetraacetic acid pH 7.8 (EDTA (VWR)) followed by additional brief rapid decalcification in Shandon TBD-1 decalcifier (Thermo Scientific) after which they were dehydrated and embedded in paraffin in similar orientations of all specimens. Five micrometer thick contiguous sections were cut and positioned on Superfrost Plus slides. Sections were rehydrated using standard protocols and stained with Mayer's Hematoxylin (Dako). Additional, staining with Safranin-O (0.1%, Sigma-Aldrich) and Fast Green (0.1%, Sigma-Aldrich) was performed for all obtained specimens to identify the presence of glycosaminoglycans as found in cartilaginous tissue. Subsequently, sections were dehydrated and mounted in Histomount (Thermo Scientific). All specimens were evaluated by light microscopy. The medial sections were compared with the lateral sections for the presence of periosteum and its thickness, cortical and trabecular bone, cartilaginous tissue/chondrocytes, bone marrow, calcification or growth arrest lines, the presence of necrosis and other abnormalities which were not pre-determined.
.
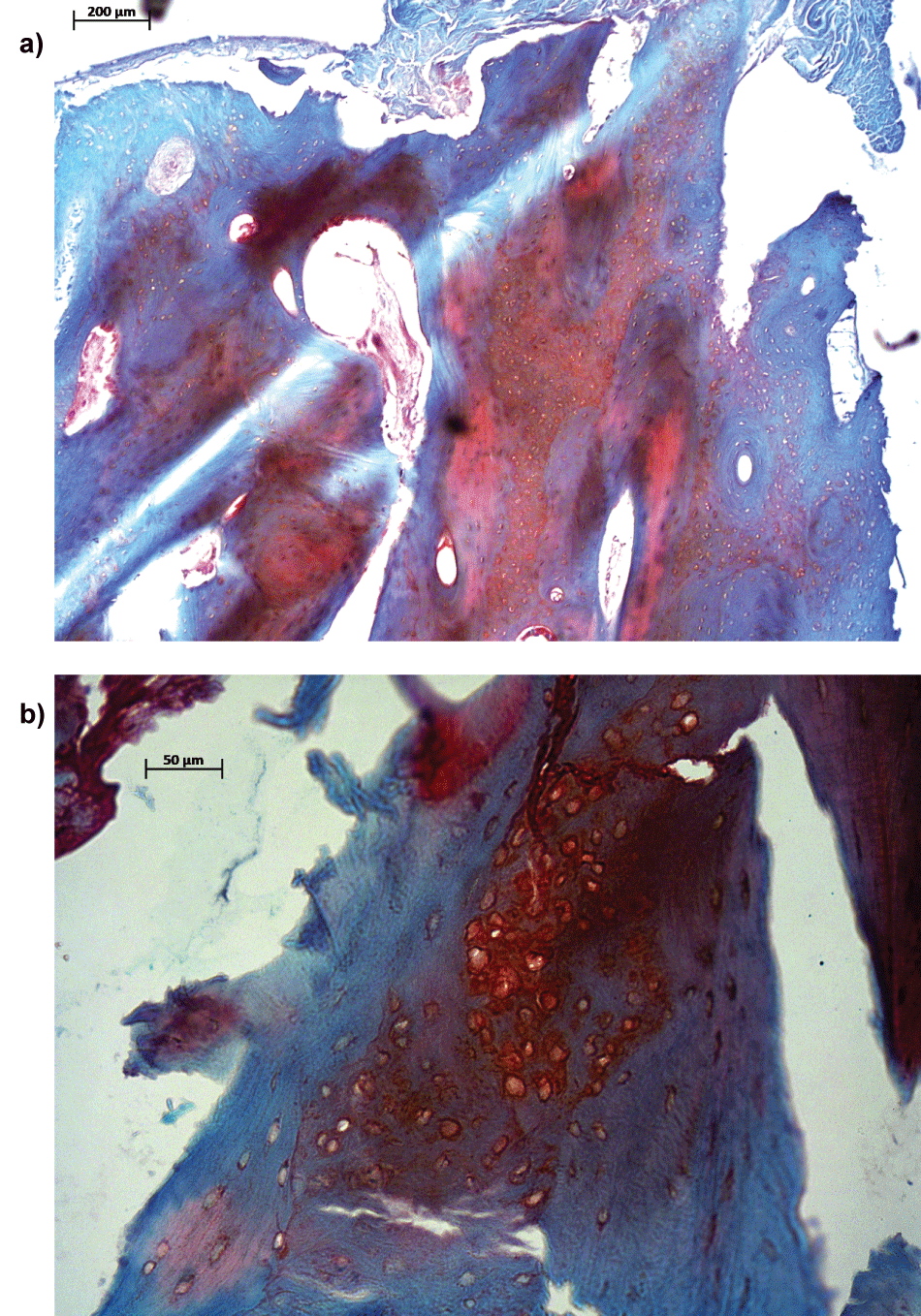
Figure 2: Endochondral ossification present in sections from medial side of the tibia from the same patient as in figure 1.
a,b) Sections from the medial side of the tibia were stained by Safranin-O/Fast Green to determine the presence of glycosaminoglycans. Bar = 200 µm for first micrograph (A) and bar = 50 µm for second micrograph (B).
a) the arrow indicates endochondral ossification which colours orange because of presence of glycosaminoglycans; b) the arrow indicates a close up view glycosaminoglycan rich area as shown in figure 2A.
View Figure 2
![]()
Table 2: Histological findings.
View Table 2
Results
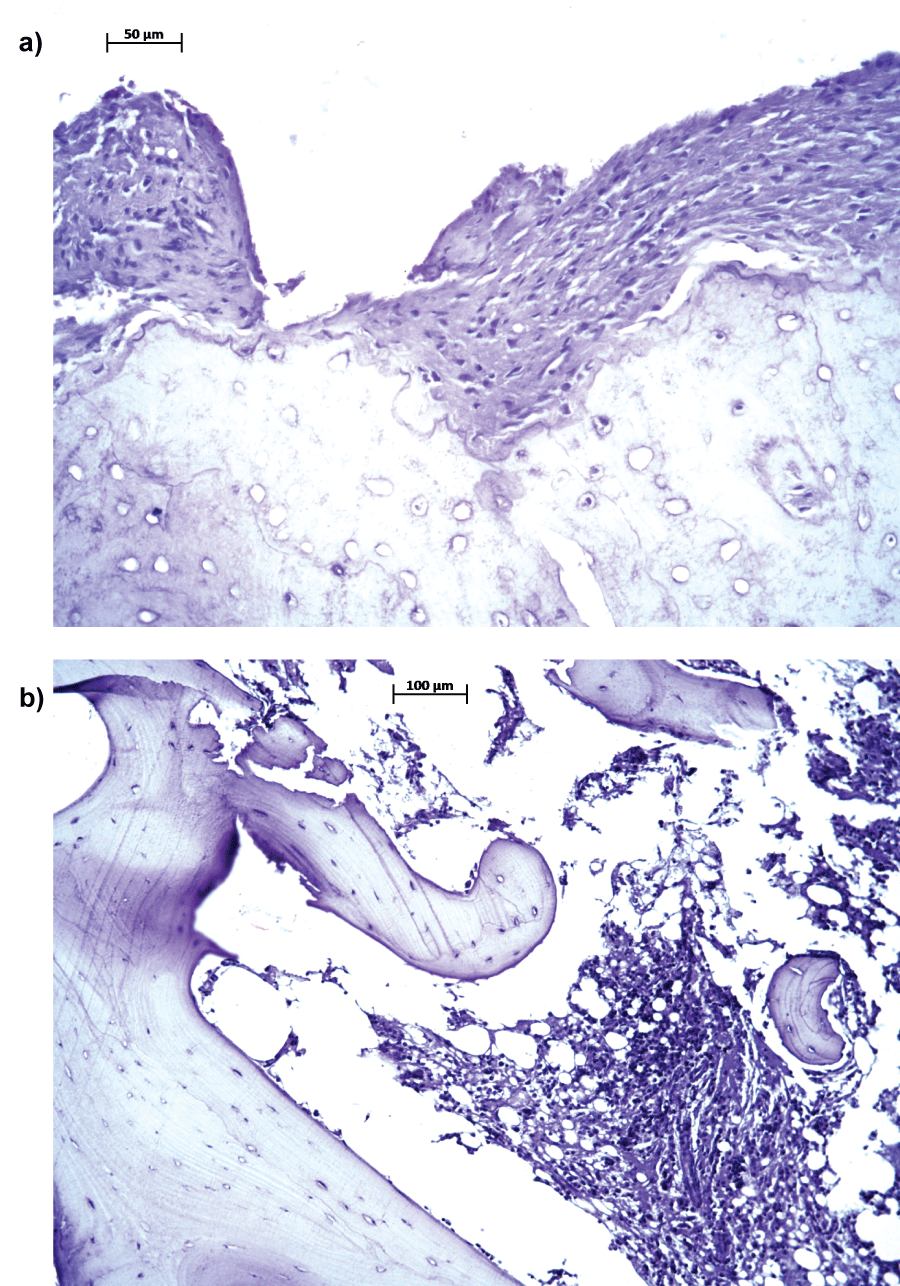
In this study we compared the histological morphology of the medial aspect of the proximal tibial metaphysis with the lateral area. Six specimens derived from 4 patients with infantile Blount's disease were analysed macroscopically and histologically. Patient's characteristics are presented in table 1. All patients were diagnosed with early age onset and Langenskiöld stage 4 infantile Blount's disease (epiphyseal secondary ossification centre passes more distally and crosses distal to the physeal level to fill the metaphyseal step) on the preoperative radiographic examinations (Figure 1). Pre- and postoperative radiographic examinations and clinical photograph were taken for each patient to evaluate the accuracy of the surgical correction (Figure 1A, Figure 1B and Figure 1C). Macroscopically, all specimens contained cortical and trabecular bone. Histological analysis consisted of systematic evaluation on abnormalities of metaphyseal cortical and trabecular bone, systematically comparing the medial with the lateral specimens (Table 2). The medially obtained specimens showed presence of intra-cortical endochondral ossification in all specimens (Figure 2A and Figure 2B). The Safranin-O staining showed subperiosteal/intra-cortical chondrocyte-rich areas in 4 out of 6 specimens (Figure 3A and Figure 3B). This was never observed in the opposite lateral specimens (Figure 4A and Figure 4B). Furthermore, in the medial specimens bone marrow was found subperiostally, which is unusual in this area of the metaphysis and indicates endochondral ossification as well (Figure 2A and Figure 3). Other findings were the presence of irregular growth lines located closely to the periosteum in the medial compartment, which was not observed in the lateral specimens (Figure 5). Two of the lateral specimens were of poor quality for proper histological analysis. The remaining lateral specimens (n = 4) showed no abnormalities compared to normal cortical and trabecular metaphyseal bone (Figure 4A and Figure 4B).
.
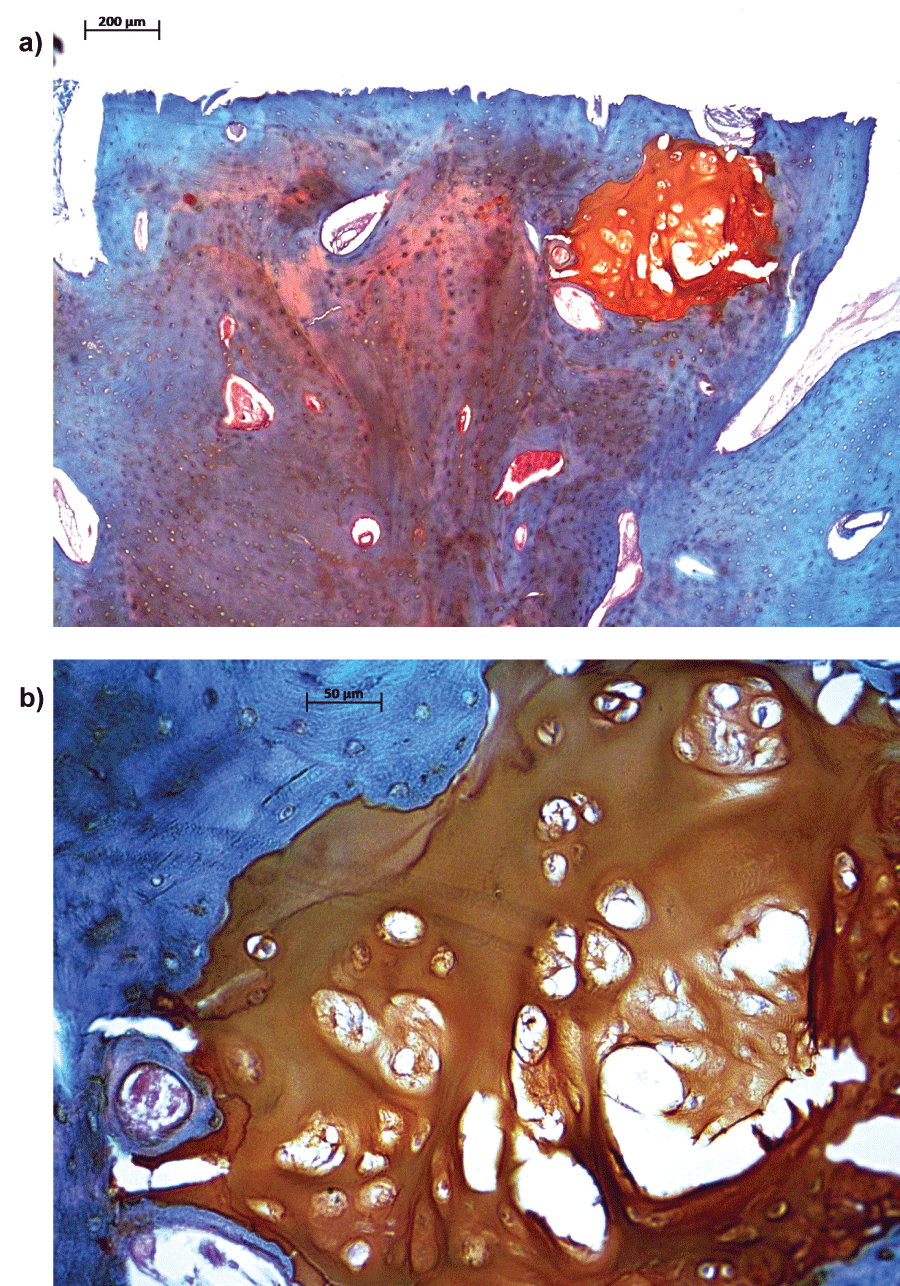
Figure 3: a,b) Islands of hypertrophic chondrocytes in sections from the medial side of the tibia.
a,b) Sections from the medial site of the tibia were stained by Safranin-O/Fast Green to determine the presence of glycosaminoglycans. Bar = 100 µm for first micrograph (A) and bar = 50 µm for second micrograph (B). The orange coloured tissue consists of islands of hypertrophied chondrocytes.
View Figure 3
.
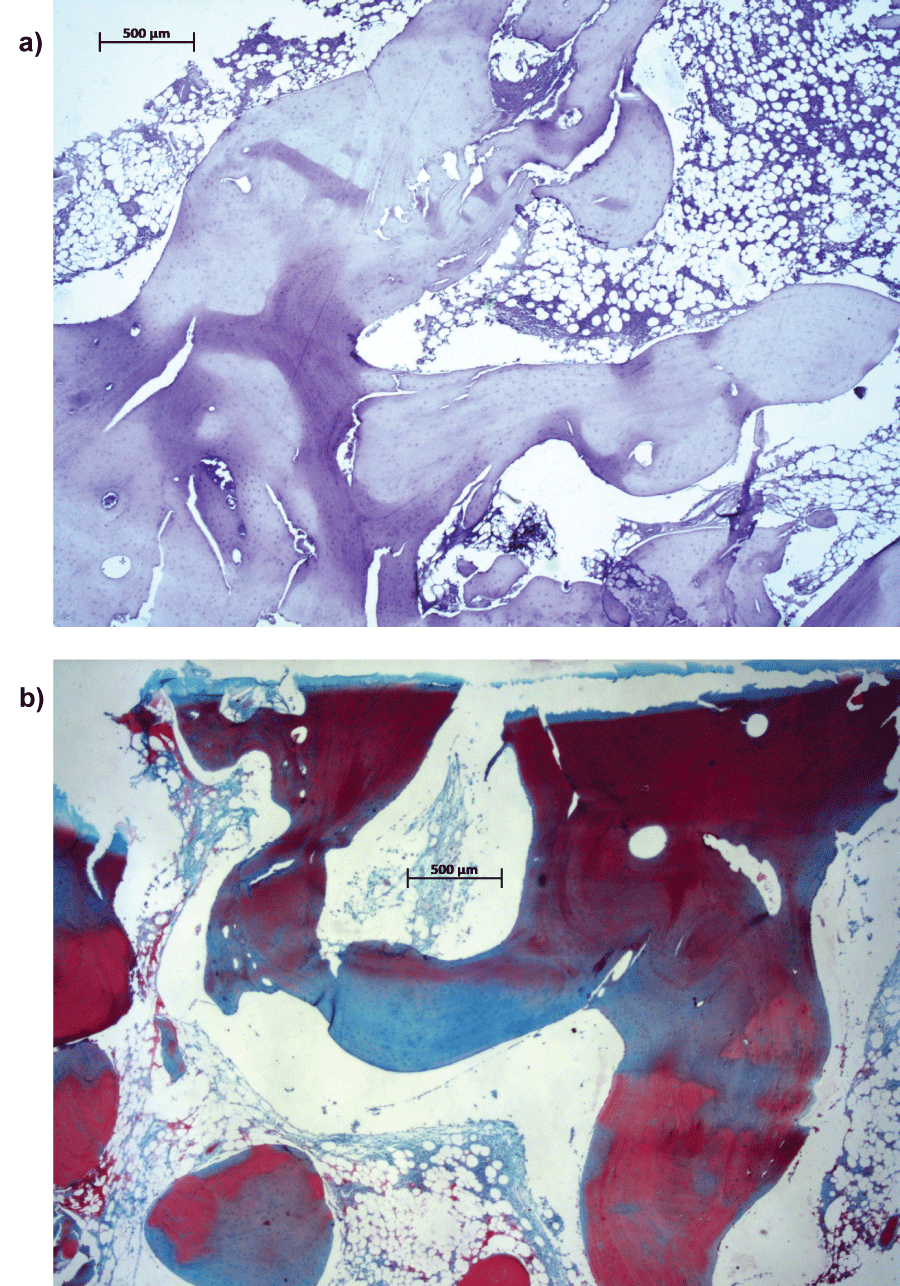
Figure 4: a,b) Specimens from the lateral side of the tibia.
a,b) Sections from the lateral site of the tibia were stained by Mayer's Hematoxylin (Dako) and Safranin-O/Fast Green to determine the presence of glycosaminoglycans as in cartilaginous tissue. Bar = 500 µm for both micrographs.
a) The micrograph shows organized woven bone; b) Safranine-O/Fast Green staining.
View Figure 4
Discussion
In this prospectively designed study, specimens from patients with infantile Blount's disease were obtained from the medial and lateral aspect of the metaphyseal proximal tibia. All medial specimens showed presence of intra-cortical endochondral ossification with the presence of intra-cortical chondrocyte-rich areas (4 out of 6 specimens), subperiosteally located bone marrow and multiple growth arrest lines. The laterally obtained specimens did not show any sign of endochondral ossification in the trabecular and cortical aspect of the proximal metaphyseal bone. The chondrocyte-like cell morphology is most likely of cartilaginous origin undergoing endochondral ossification. This finding is unusual in the metaphyseal area in the proximal tibia. The combination of growth arrest lines and cartilaginous tissue undergoing endochondral ossification may reflect repetitive trauma or failure of ossification attempts due to increased biomechanical load on the medial proximal tibia, resulting in the typical tibial vara deformity as seen in Blount's disease.
.
Figure 5: a,b) Subperiostally located growth arrest/calcification lines, medial versus lateral.
a,b) Sections from the medial site of the tibia were stained by Mayer's Hematoxylin (Dako) to determine the presence of growth arrest lines/calcification lines. Bar = 50 µm for A.
a) Medial tibial metaphyseal specimen (arrows: growth lines); b) Lateral tibial metaphyseal (arrow: growth line).
View Figure 5
The cartilaginous like tissue found in de medial area of the metaphysis also indicates the disease extends from the physeal area to the distal part of the proximal tibial metaphysis. Densely packed islands of chondrocytes from the physeal area were described previously amongst patients with infantile and late-onset Blount's disease by Langenskiold, Carter, et al. Thompson, et al. and Wenger, et al. [1,12,15,16]. These islands were located subperiosteally, outlined distinctively and contained numerous hypertrophic chondrocytes. In this study, this was typically present in one of the medial obtained specimens. The other medial specimens contained of wide spread chondrocyte rich areas (Figure 3A and Figure 3B). Increased pressure on the medial proximal tibial (meta-) physis is thought to be responsible for defective endochondral ossification, causing these histologic and morphologic changes as seen and described in fracture healing [17]. Langenskiold [12] described the presence of islands of densely packed hypertrophic chondrocytes, islands of almost acellular fibrous cartilage and abnormally large groups of capillary vessels in 9 specimens which were obtained from the medial metaphyseal beak. In contrast to Langenskiold, et al. [12], who specifically analysed the affected medial metaphysis of the proximal tibia, we compared the affected side with the lateral aspect of the proximal metaphysis. This new obtained information strongly suggests the lateral metaphysis of the proximal tibia appears to be unharmed. Another observation in this study was the presence of active endochondral ossification, which might indicate bone remodelling since the opposite lateral metaphysis showed normal woven bone. Furthermore, bone marrow was found subperiosteally, which is unusual in the proximal cortical area and might suggest active intramembranous ossification originating from the inner cambial layer of the periosteum [18]. The irregular growth arrest lines, found in the medial specimens, indicates active ossification. This might have been be caused by the increased mechanical load on the medial metaphysis. These histologic alterations described, suggest a mechanical disruption or injury and is remarkably similar to those observed in Scheuermann's disease [19]. Scoles, et al. [19] found comparable disorganization of endochondral ossification with islands of chondrocytes of the anterior area of thoracic vertebrae in patients with Scheuermann's disease [19]. This anterior defective endochondral ossification results in thoracic kyphosis, which is comparable with the tibial bowing in Blount's disease and is also seen in slipped proximal femoral epiphysis [19,20].
Comparable histological changes in Blount's disease and slipped capital femoral epiphysis, described by Lacroix, et al. [20], implies irregular vascular fibrocartilaginous tissue invasion causing progressively disorganized ossification, which finally results in early epiphysiodesis [20]. Furthermore, the presence of endochondral or metaplastic ossification of residual cartilaginous or fibrocartilaginous elements and fibrous zone invasion was observed, resulting in limited and atypical residual growth. Since both diseases show morphologic similarities, common pathophysiology could be suggested. From biomechanical perspective, the medial part of the proximal femoral and tibial physis are under increased amount of loading, which could cause these alterations. This might explain an increased incidence amongst overweighed patients. In addition, slipped capital femoral epiphysis has been described in patients with Blount's increasing the suspicion on a common etiology [21]. The initial cause however of both diseases is still unknown. An underlying genetic cause might be responsible for the expression and is in our opinion still an important, unproven aspect of the disease.
Limitations of this study are firstly poor quality of two of the laterally obtained specimens. This might have been caused by inadequate Formalin fixation during transport. Secondly, due to an inadequate effect of the slow decalcification process a rapid decalcifier was used, which rendered immunohistochemistry impossible. Thirdly, the specimens obtained from the lateral as well as the medial part of the metaphysis differs in the amount of cortical and trabecular bone. Although, from each patient specimens were obtained at the same anatomic location. Fourthly, an attempt was made to quantify the cells with specially designed software (Cellprofiler), which was not possible due to a lack of contrast between cells. Finally, blood sample testing to rule out other causes for tibia vara than Blount's disease like rickets was not performed. However, the clinical presentation and roentgenologic morphology as described by Langenskiold [8] were all typical for Blount's disease.
Little literature on histopathological changes in infantile Blount's disease is available. Recommendations for future research include, obtaining blood samples from each patient to rule out other underlying causes for tibia vara and slowly decalcifying specimens for subsequent immunohistochemistry in addition to histochemistry. The biomechanical theory has generally been accepted in literature, however genetic predispositions seems plausible since it is seen amongst twins, more common amongst siblings and the prevalence is proportionally higher in the Afro-Caribbean race [22,23].
In conclusion, in patients with infantile Blount's disease endochondral ossification was only found in the medial part of the metaphysis. Normal cortical and trabecular bone was found in all lateral specimens. This may reflect the clinical tibial vara deformity in Blount's disease in which increased mechanical load on the medial metaphysis causes failure of normal ossification.
Acknowledgements
We thank Dr. J. Korpisah for providing the possibility to harvest specimens from his patients with infantile Blount's disease.
Conflict of Interest
Each author certifies that he or she has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
References
-
Carter JR, Leeson MC, Thompson GH, Kalamchi A, Kelly CM, et al. (1988) Late-onset tibia vara: a histopathologic analysis. A comparative evaluation with infantile tibia vara and slipped capital femoral epiphysis. J Pediatr Orthop 8: 187-195.
-
Ingvarsson T, Hägglund G, Ramgren B, Jonsson K, Zayer M (1998) Long-term results after infantile Blount's disease. J Pediatr Orthop B 7: 226-229.
-
de Sanctis N, Della Corte S, Pempinello C, Di Gennaro G, Gambardella A (1995) Infantile type of Blount's disease: considerations concerning etiopathogenesis and treatment. J Pediatr Orthop B 4: 200-203.
-
Sabharwal S (2009) Blount disease. J Bone Joint Surg Am 91: 1758-1776.
-
Shinohara Y, Kamegaya M, Kuniyoshi K, Moriya H (2002) Natural history of infantile tibia vara. J Bone Joint Surg Br 84: 263-268.
-
Cook SD, Lavernia CJ, Burke SW, Skinner HB, Haddad RJ Jr (1983) A biomechanical analysis of the etiology of tibia vara. J Pediatr Orthop 3: 449-454.
-
Arkin AM, Katz JF (1956) The effects of pressure on epiphyseal growth; the mechanism of plasticity of growing bone. J Bone Joint Surg Am 38: 1056-1076.
-
Langenskiöld A (1981) Tibia vara: osteochondrosis deformans tibiae. Blount's disease. Clin Orthop Relat Res 158: 77-82.
-
Ho-Fung V, Jaimes C, Delgado J, Davidson RS, Jaramillo D (2013) MRI evaluation of the knee in children with infantile Blount disease: tibial and extra-tibial findings. Pediatr Radiol 43: 1316-1326.
-
Kawu AA, Salami OO, Olawepo A, Ugbeye MA, Yinusa W, et al. (2012) Outcome analysis of surgical treatment of Blount disease in Nigeria. Niger J Clin Pract 15: 165-167.
-
Golding JSR, McNeil-Smith JDG (1963) Observations on the etiology of tibia vara. J Bone Joint Surg 45B: XO.
-
Langenskiold A (1952) Tibia vara; (osteochondrosis deformans tibiae); a survey of 23 cases. Acta Chir Scand 103: 1-22.
-
Blount WP (1937) Tibia vara: Osteochondrosis deformans tibiae. J Bone Joint Surg 19: l.
-
Langenskiöld A (1989) Tibia vara. A critical review. Clin Orthop Relat Res 246: 195-207.
-
Thompson GH, Carter JR, Smith CW (1984) Late-onset tibia vara: a comparative analysis. J Pediatr Orthop 4: 185-194.
-
Wenger DR, Mickelson M, Maynard JA (1984) The evolution and histopathology of adolescent tibia vara. J Pediatr Orthop 4: 78-88.
-
Claes LE, Heigele CA (1999) Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. J Biomech 32: 255-266.
-
Augustin G, Antabak A, Davila S (2007) The periosteum. Part 1: Anatomy, histology and molecular biology. Injury 38: 1115-1130.
-
Scoles PV, Latimer BM, DigIovanni BF, Vargo E, Bauza S, et al. (1991) Vertebral alterations in Scheuermann's kyphosis. Spine 16: 509-515.
-
Lacroix P, Verbrugge J (1951) Slipping of the femoral epiphysis A pathological study. J Bone Joint Surg [Am] 33: 371-381.
-
Jamil K, Abdul Rashid AH, Ibrahim S (2015) Tibia vara and slipped upper femoral epiphysis: is there an association? J Pediatr Orthop B 24: 46-49.
-
Bathfield CA, Beighton PH (1978) Blount disease. A review of etiological factors in 110 patients. Clin Orthop Relat Res 135: 29-33.
-
Yazici M, Atilla B (2001) Unilateral late-onset tibia vara associated with bilateral proximal femoral growth disturbance in monozygotic twins: case report. Turk J Pediatr 43: 155-158.





