International Journal of Surgery Research and Practice
A Comparative Study of Electrofulguration and Formocresol Pulpotomy Techniques in Primary Teeth
Omar A.S. El Meligy1,2*, Sawsan H Mahmoud2
1Faculty of Dentistry, King Abdulaziz University, Saudi Arabia
2Faculty of Dentistry, Alexandria University, Egypt
*Corresponding author: Omar Abd El Sadek El Meligy, Professor of Pediatric Dentistry, Faculty of Dentistry, King Abdulaziz University, Kingdom of Saudi Arabia, Tel: 0122871660, Fax: 0126403316, E-mail: omeligy@kau.edu.sa
Int J Surg Res Pract, IJSRP-1-012, (Volume 1, Issue 1), Research Article; ISSN: 2378-3397
Received: August 25, 2014 | Accepted: November 18, 2014 | Published: November 22, 2014
Citation: El Meligy OAS, Mahmoud SH (2014) A Comparative Study of Electrofulguration and Formocresol Pulpotomy Techniques in Primary Teeth. Int J Surg Res Pract 1:012. 10.23937/2378-3397/1410012
Copyright: © 2014 El Meligy OAS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
While the formocresol pulpotomy has enjoyed long-term clinical use and success, concerns over its toxicity and mutagenicity have prompted research into other pulpotomy techniques. The purpose of this study was to prospectively compare electrofulguration pulpotomies versus formocresol pulpotomies in children vital primary molar teeth. Electrofulguration and formocresol pulpotomies were completed on 40 primary molars in 20 children aged 4 to 8 years. Each child had one molar treated by electrofulguration pulpotomy and another molar by formocresol pulpotomy. Teeth were evaluated clinically and radiologically after 1, 3, and 6 months. After 6 months postoperative observation time, the clinical and radiographic success rates for the electrofulguration group were 95% and 85%, respectively, and for the formocresol group, 100% and 90%, respectively. There was no statistically significant difference between the two groups, either clinically or radiographically.
Keywords
Formocresol, Electrofulguration, Pulpotomy, Children
Introduction
Vital pulpotomy is considered a one-stage procedure and is defined as �the surgical amputation of the coronal portion of an exposed vital pulp as a means of preserving the vitality and function of the remaining radicular portion�. Many pharmacotherapeutic agents have been used when performing pulpotomies of primary teeth. Formocresol has been a popular material of choice for use in the pulpotomy procedure, mainly because of its ease in use and excellent clinical success. Yet, despite its excellent clinical success rate, the formocresol pulpotomy has come under close observation because of safety consideration [1-5]. Other medicaments, such as glutaraldehyde, calcium hydroxide, collagen and ferric sulfate, have been suggested as possible replacements for formocresol. Success rates have varied with the agent used and the particular study, with several medicaments contributing to favorable results [1,6].
The use of electrosurgery as a nonpharmacological pulpotomy technique has been well-documented and has proven to have merit [1,2,7]. The self-limiting, pulpal penetration is only a few cell layers deep. There is good visualization and homeostasis without chemical coagulation or systemic involvement. It is less time consuming than the formocresol approach [8].
Aim of the Work
The aim of this study was to compare the clinical and radiographic success of the electrofulguration and formocresol pulpotomy technique used on human primary molar teeth requiring vital pulp therapy secondary to carious involvement.
Materials and Methods
This study was carried out on forty primary molar teeth of twenty children, with an age that ranged from 4 to 8 years, and a mean of 6.2 years. The children were selected from the Pediatric Dental Clinic, Faculty of Dentistry, Alexandria University, Egypt. Each child had two lower or upper primary molars indicated for pulpotomy.
Teeth were selected based upon the following criteria:
i) Symptomless, carious exposure of the vital pulp.
ii) No clinical or radiographic evidence of pulpal degeneration.
iii) Possibility of proper restoration of primary molars.
The children were free from any systemic diseases.
This study was conducted in compliance with all policies of appropriate patient care at Alexandria University. Prior to treatment, a written informed consent was obtained from the parents.
The materials used in this study were:
� Formocresol (Cavex Holland, P.O. Box 852, 2003 RW Harlem (Holland))
� Electrosurgical unit (Ellman 90 FFP Dento-Surg., Ellman International, 1135 Railroad Av., Hawlett, N.Y. USA).
The teeth were divided into two groups:
Group I: Conventional formocresol pulpotomy (control group) (20 teeth).
Group II: Electrofulguration pulpotomy (experimental group) (20 teeth).
Clinical Procedure
Following profound local anesthesia, rubber dam isolation, occlusal preparation and removal of caries, the roof of the pulp chamber was then removed.
Group I (Formocresol pulpotomy): The coronal pulp was amputated with a sharp excavator and/or with a large slow-speed bur. Hemorrhage was controlled using dry sterile cotton pellets, then a five-minute application of formocresol was done. Zinc oxide eugenol (ZOE) dressing was placed directly on the radicular pulp stumps.
Group II (Electrofulguration pulpotomy): The procedures for pulp exposure and amputation were identical to the formocresol approach. Once hemostasis was accomplished, the electrosurgery dental electrode was immediately placed 0.5 mm above the tissue. Fulguration current was used at an intensity setting of nine. When activated, a spark will jump from the electrode tip to the surface of the tissue causing coagulation. The current was applied for 1-2 seconds over each pulpal stump. If additional fulguration was required, 5 seconds elasped prior to subsequent application of the current. ZOE dressing was then placed on the radicular pulp stumps. All the treated teeth in both groups were restored with stainless steel crowns.
Postoperative periapical radiographs of the treated teeth were taken and considered as a baseline. Then, the patients were recalled for follow-up at 1, 3 and 6 months, for clinical and radiographic evaluation. Two examiners, who were blinded to treatment type, evaluated the teeth clini�cally and radiographically. The examiners were faculty from the Department of Pediatric Dentistry and Dental Public Health, Alexandria University.
Evidence of Success
� No prolonged adverse clinical signs and symptoms such as prolonged sensitivity, pain, swelling or mobility.
� No premature loss or extraction of the treated teeth (before the normal exfoliation time), secondary to root or bone resorption.
� Lack of radiographic evidence of pathological internal or external root resorption and periapical or furcal radiolucency
The overall success of the treatment was then evaluated using both clinical and radiographic findings. If either was judged to be a failure, treatment then was assessed to be a failure.
Results
The results of this work are divided into clinical and radiographic findings.
Clinical findingsTable 1 shows the clinical findings of the two groups during the follow-up period. After the first month, no clinical signs or symptoms of failure were observed in any of the two groups. After three months, no signs of failure were observed in both groups I and II. After six months follow-up, no signs of failure were observed in group I, while one case in group II complained of pain and tenderness to percussion. At the end of the study, one case in group II was considered to be clinically failure.
![]()
Table 1: Number of teeth exhibiting clinical responses of the two groups.
View Table 1
Radiographic findings
Table 2 shows the radiographic findings of the two groups during the follow-up period. After one month, no radiographic findings were observed in any of the two groups. After three months, no signs of failure were observed in both groups I and II. After six months follow-up, two teeth in group I showed widening of lamina dura. In group II, one tooth showed widening of lamia dura and periapical radiolucency and two teeth showed abnormal root resorption. At the end of the study, two teeth in group I and three teeth in group II, were considered to be failure.
![]()
Table 2: Number of teeth exhibiting Radiographic Findings of the two groups.
View Table 2
Clinical and radiographic success rates
The clinical and radiographic success rates for the electrofulguration group were 95% and 85%, respectively, and for the formocresol group, 100% and 90%, respectively. Figures 1-4 show radiographs of one successful case for each of the two groups. Although the electrofulguration group radiographic failure rate was more than that of the formocresol group, this difference was not statistically significant at the P > 0.05 level using Fisher�s exact test.
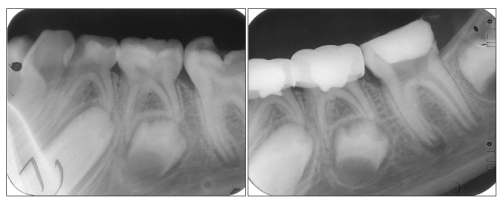
Figure 1: Pre (a) and immediate postoperative (b) radiographs of an electrofulguration pulpotomized tooth (mandibular
left second primary molar) and a formocresol pulpotomized tooth (mandibular left first primary molar).
View Figure 1
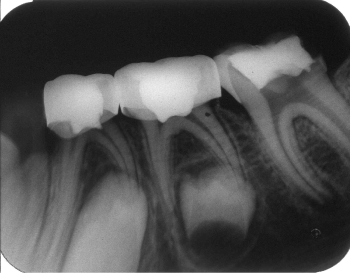
Figure 2: One-month postoperative radiograph of the same case showing no
signs of failure.
View Figure 2
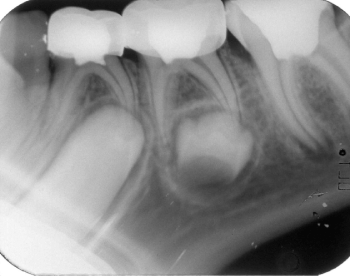
Figure 3: Three-months postoperative radiograph of the same case considered
to be successful.
View Figure 3
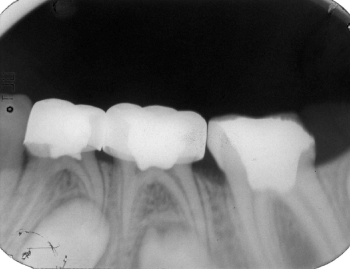
Figure 4: Six-months postoperative radiograph of the same case that was
normal at the last observation.
View Figure 4
Discussion
Concern over the use of formocresol in the pulpotomy for primary teeth has promoted the investigation of several alternatives to this medicament [9].
Several authors seeking to avoid the use of medicaments, have suggested electrosurgery for pulpotomies [8,10]. Even though, the electrosurgical pulpotomy technique has been advocated for years, there was very little data to support its use in human primary teeth. In this study, the Ellman-Dento-Surg Electrosurgical Unit was used as it does not cause any adverse side effects and it allowed current selection [11].
For coagulation, fulguration current was used with intensity setting of nine, which did not cause any tearing of tissues. A cooling period of 5 seconds elasped between successive electrocoagulations has been allowed to decrease possible lateral heat accumulation [12].
ZOE was used as a base over the pulpal stumps, as it is traditionally recommended, to compare more directly the electrosurgical technique to the widely used formocresol technique. A question of whether the pulp response following pulpotomies was due to the technique or to the base, was raised by many authors [2,3,8,13]. However, Ruemping et al. [3] speculated that a coagulation layer produced by electrosurgery might limit the actions of ZOE on the pulp.
Alternative biocompatible base might provide better results, eliminating the inevitable tissue response seen with ZOE.
Although no statistically significant difference between the two groups was found, the electrofulguration pulpotomy technique had two advantages, which are faster application and no risk of formocresol side effects. This finding coincides with the results reported in a previous study with electrosurgery on primates [8].
Remarkable similarities have been seen between the formocresol results in the present study and other previous studies [14,15]. The findings of the electrofulguration group (group II) compared favorably with those from many previous human clinical studies [1,4,6,16].
On the other hand, our findings disagree with Fishman et al. [17], who found a clinical success after only 6 months to be 77% and a radiographic success to be 55%. Compared with the present study, their success rate is low. Fishman et al. [17] concluded that it would be premature to recommend use of the electrosurgical pulpotomy technique. This conclusion may be inappropriate as the investigation was specifically designed to compare two different pulpotomy covering medicaments following electrosurgical pulpotomies, not the success rate of the electrosurgical pulpotomy versus the formocresol pulpotomy. The results of our study indicate that the electrosurgical pulpotomy appears comparable to the formocresol pulpotomy for human primary molars for a postoperative period of at least 6 months.
Most of the previous studies used caries-free teeth [8,18]. Shaw et al. [8] stated that having a healthy radicular pulp would be necessary for success. Contaminated pulp tissue might not promote adequate penetration of the current. However, the relatively high success rate reported by the present study and the study of Mack and Dean [1], in cariously exposed human teeth seem to negate that hypothesis.
The relatively benign nature of electrosurgical pulpotomy treatment as compared to pharmacotherapeutic pulpotomy procedures is encouraging.
Long-term, prospective human clinical studies are now required to confirm the optimistic results of this study.
Conclusion
The clinical and radiographic success rates for the electrofulguration group were 95% and 85%, respectively, and for the formocresol group, 100% and 90% respectively. There was no statistically significant difference between electrofulguration and formocresol pulpotomy clinical and radiographic success rates at the P>0.05 level.
Ethical statement
The Ethical Committee of Pediatric and Dental Public Health Department at Faculty of Dentistry, Alexandria University, Egypt approved the research protocol.
References
-
Mack RB, Dean JA (1993) Electrosurgical pulpotomy: a retrospective human study. ASDC J Dent Child 60: 107-114.
-
Shulman ER, McIver FT, Burkes EJ Jr (1987) Comparison of electrosurgery and formocresol as pulpotomy techniques in monkey primary teeth. Pediatr Dent 9: 189-194.
-
Ruemping DR, Morton TH Jr, Anderson MW (1983) Electrosurgical pulpotomy in primates--a comparison with formocresol pulpotomy. Pediatr Dent 5: 14-18.
-
Sheller B, Morton TH Jr (1987) Electrosurgical pulpotomy: a pilot study in humans. J Endod 13: 69-76.
-
Oztas N, Ulusu T, Oyg�r T, Cokpekin F (1994) Comparison of electrosurgery and formocresol as pulpotomy techniques in dog primary teeth. J Clin Pediatr Dent 18: 285-289.
-
Nadin G, Goel BR, Yeung CA, Glenny AM (2003) Pulp treatment for extensive decay in primary teeth. Cochrane Database Syst Rev : CD003220.
-
Yadav P, Indushekar K, Saraf B, Sheoran N, Sardana D (2014) Comparative evaluation of Ferric Sulfate, Electrosurgical and Diode Laser on human primary molars pulpotomy: an "in-vivo" study. Laser Ther 23: 41-47.
-
Shaw DW, Sheller B, Barrus BD, Morton TH Jr (1987) Electrosurgical pulpotomy--a 6-month study in primates. J Endod 13: 500-505.
-
El Tekeya MM, Abd el-Rahman AM (1997) Comparison of electrosurgery and formocresol as pulpotomy techniques in primary teeth. ADJ 22: 69-83.
-
El-Meligy O, Abdalla M, El-Baraway S, El-Tekya M, Dean JA (2001) Histological evaluation of electrosurgery and formocresol pulpotomy techniques in primary teeth in dogs. J Clin Pediatr Dent 26: 81-85.
-
Kalkwarf KL, Krejci RF, Edison AR, Reinhardt RA (1983) Subjacent heat production during tissue excision with electrosurgery. J Oral Maxillofac Surg 41: 653-657.
-
Dean JA, Mack RB, Fulkerson BT, Sanders BJ (2002) Comparison of electrosurgical and formocresol pulpotomy procedures in children. Int J Paediatr Dent 12: 177-182.
-
Nematollahi H, Sahebnasagh M, Parisay I (2011) Comparison of electrosurgical pulpotomy with zinc oxide eugenol or zinc polycarboxylate cements sub-base. J Clin Pediatr Dent 36: 133-137.
-
Hicks MJ, Barr ES, Flaitz CM (1986) Formocresol pulpotomies in primary molars: a radiographic study in a pediatric dentistry practice. J Pedod 10: 331-339.
-
Bahrololoomi Z, Moeintaghavi A, Emtiazi M, Hosseini G (2008) Clinical and radiographic comparison of primary molars after formocresol and electrosurgical pulpotomy: a randomized clinical trial. Indian J Dent Res 19: 219-223.
-
Khorakian F, Mazhari F, Asgary S, Sahebnasagh M, Alizadeh Kaseb A, et al. (2014) Two-year outcomes of electrosurgery and calcium-enriched mixture pulpotomy in primary teeth: a randomised clinical trial. Eur Arch Paediatr Dent 15: 223-228.
-
Fishman SA, Udin RD, Good DL, Rodef F (1996) Success of electrofulguration pulpotomies covered by zinc oxide and eugenol or calcium hydroxide: a clinical study. Pediatr Dent 18: 385-390.
-
Yahushiji M (1975) Pulpotomy of primary teeth by means of electrosurgery. Shoni Shkiagaku Zasski 13: 213-219.





