Stem Cell Transplantation to Treat Severe Refractory Multiple Sclerosis
Athanasia Mouzaki*, Marios I Garyfallou and Kokona Chatzantoni
Division of Haematology, Department of Internal Medicine, Faculty of Medicine, University of Patras, Greece
*Corresponding author:
Athanasia Mouzaki, Division of Haematology, Department of Internal Medicine, Faculty of Medicine, School of Health Sciences, University of Patras, Rion, Patras GR-26500, Greece, Tel & Fax: +30-2610-969123, E-mail: mouzaki@upatras.gr
Int J Transplant Res Med,
IJTRM-1-008, (Volume 1, Issue 2),
Review Article
Received: July 09, 2015: Accepted: August 10, 2015: Published: August 13, 2015
Citation: Mouzaki A, Garyfallou MI, Chatzantoni K (2015) Stem Cell Transplantation to Treat Severe Refractory Multiple Sclerosis. Int J Transplant Res Med 1:008
Copyright: © 2015 Mouzaki A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
In severe, drug-resistant multiple sclerosis (MS), stem cell transplantation (SCT) was introduced as a treatment option about 20 years ago. Application of autologous SCT is based on the assumption that MS is not inherited in a Mendelian fashion and the auto reactive immune system can be replaced by a healthy one. Allogeneic SCT ensures that all leukocytes are of donor origin, but is applied rarely. Both approaches are fraught with problems resulting, mainly, from the high-intensity pre-transplant conditioning regimens. In certain reported cases, the patients' condition worsened significantly after SCT. Overall, SCT has been applied to relatively few patients worldwide with moderate success, and recent studies suggest that patients with early highly aggressive MS benefit most from this treatment approach. This article summarizes the salient points of the SCT therapy for MS and offers insight into promoting neural and glial differentiation of endogenous cells by alternative therapies.
Multiple Sclerosis (MS)
MS usually affects young adults aged between 20 to 40 years, although it can begin earlier or later in life. Women are affected almost twice as often as men. Most common symptoms include sensory disturbances in the limbs, optic nerve dysfunction, pyramidal tract dysfunction, bladder or bowel dysfunction, sexual dysfunction, ataxia and diplopia. The course of MS can follow 4 clinical patterns: (1) Relapsing-Remitting MS (RR-MS): It accounts for 80-90% of MS cases at onset. It is characterized by self-limited attacks of neurological dysfunction, which develop acutely, followed in most cases by a recovery of function that is often complete. In between attacks the patient is neurologically and symptomatically stable. (2) Secondary Progressive MS (SP-MS): It begins as RR-MS does, but at some point changes and the attack rate is reduced and the course becomes characterized by a steady deterioration in function, unrelated to acute attacks. Almost 80% of RR-MS patients follow this clinical course, and this type of MS causes the greatest amount of neurological disability. (3) Primary Progressive MS (PP-MS): It represents only about 10% of cases at onset, it is characterized by a steady decline in function from the beginning and complete lack of acute attacks. Such patients tend to have a later age of onset and a worse prognosis for ultimate disability. (4) Progressive Relapsing MS (PR-MS): It is the least common type of MS. It begins with a progressive course; patients experience occasional attacks, which are superimposed upon their steadily progressive disease course [1].
MS is a chronic disorder of the central nervous system. Derangement of cellular immunity is central in the pathophysiology of MS. MS is predominantly characterized by local T-cell and macrophage infiltration, leading to demyelination and loss of neurologic function. MS is an autoimmune disease triggered by T helper cells (mainly Th1 and Th17), with autoantibody involvement. Antigens within the myelin sheath, such as myelin basic protein, proteolipid protein and myelin oligodendrocyte glycoprotein, have been described as the main targets of self-reactive T helper cells and auto-antibodies [1-3].
Stem Cell Transplantation (SCT)
Stem cells can be isolated from the patient in autologous stem cell transplantation or a suitable donor in the case of allogeneic stem cell transplantation [4]. Stem cells are collected either from the bone marrow or the peripheral blood, after mobilisation using G-CSF as a growth factor or chemotherapy (usually cyclophosphamide). In allogeneic stem cell collection from a donor, chemotherapy is not used for mobilisation. The use of anti-CD34 antibody for ex vivo stem cell selection is optional. Stem cell selection based on CD34 positive selection usually involves efforts to deplete B and T cells from the marrow and the mobilised peripheral blood. A procedure called conditioning is used in order to eliminate autoreactive B and T cells in the patient. Conditioning includes chemotherapeutic agents, lymphodepleting antibodies and total body irradiation, which often deplete host haematopoietic stem cells. After this procedure stem cells are re-infused into the patient to reconstitute haematopoiesis. In the case of allogeneic stem cell transplantation, immunosuppression therapy is required after transplantation in order to avoid rejection reactions mediated by the host T cells, or the contaminating donor T cells in the case of Graft Versus Host Disease (GVHD) (Figure 1).
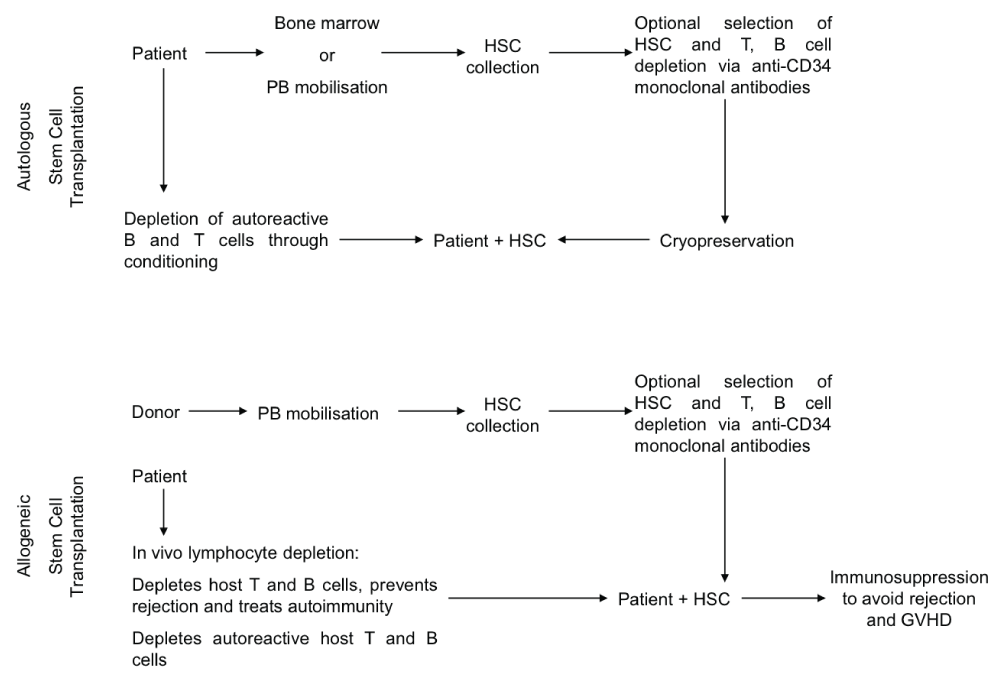
.
Figure 1: Outline of the methodologies used for autologous and allogeneic stem cell transplantation.
View Figure 1
SCT to Treat MS
The term "stem cell" includes many types, from completely undifferentiated progenitors to progenitors of specific lineages [5]. In an injured spinal cord model [6] it was demonstrated that transplanted adult neural stem cells survived, migrated and integrated with the injured spinal cord. The most prevalent theory about the way different types of stem cells approach the point of damage in myelin and lead to re-myelination is the chemoattractive hypothesis of stem cells homing [7] (Figure 2).
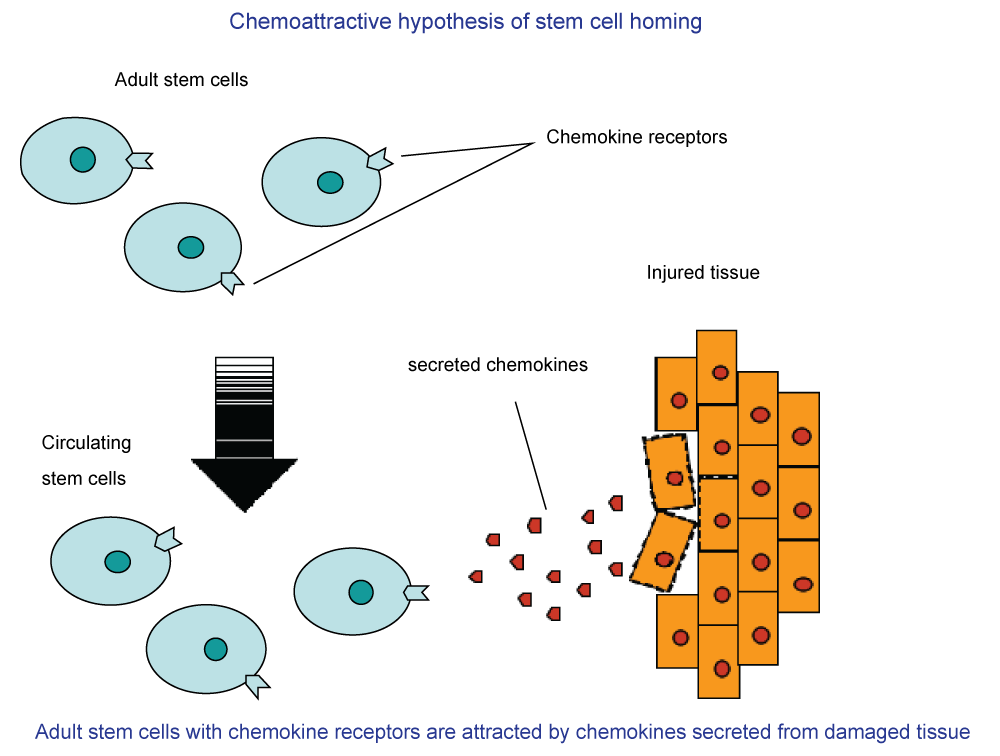
.
Figure 2: A schematic presentation of the chemoattractive hypothesis of stem cells' homing to the injured tissue.
View Figure 2
Results from autologous SCT applied to>600 MS patients between 1995 and 2012 showed improvement in more than 60% of the patients for at least 3 years post-transplantation, with a mortality risk of 2-10%. The accumulated experience from these trials helped clarify certain important points: (1) SCT works better with early aggressive MS whereas when this treatment is applied to patients with progressive MS and long disease duration, it has minor or moderate results and (2) intermediate-intensity pre-transplant conditioning regimens work equally well with toxic high-intensity regimens [8-13].
Results from heterologous SCT are relatively fewer. In 6 reported cases, SCT was performed to treat a hematological malignancy with concurrent MS. In 3 of these patients MS improved whereas in 3 patients it remained active [12,14].
A Glimpse to the Post-Autologous SCT Peripheral Blood Lymphocyte Populations
Very few studies have reported detailed analyses of peripheral blood populations of MS patients who underwent SCT. One study reported 7 patients with different types of MS (1 with RR-MS) who underwent autologous SCT and did not relapse during a 2-year follow-up [15]. Phenotypic analysis of their peripheral blood mononuclear cells revealed no significant differences from pre-SCT in the proportions of T cells, B cells, NK cells, NK T cells and monocytes. The T helper/T cytotoxic cell ratio was found significantly decreased during the first months, probably due to a greater impact of therapy on T helper cells, but reverted towards baseline at 1 year and reached pre-SCT levels at 2 years. There was also a doubling of the frequency of naive T helper cells at the expense of memory T helper cells.
Another study reported one patient with RR-MS, refractory to conventional treatments, who underwent autologous SCT and relapsed 1 year later [16]. Phenotypic analysis of his peripheral blood lymphocytes 7 years post-SCT, revealed that lymphocytes, B cells, T cells, cytotoxic T cells, memory T helper cells and regulatory T cells were within or at the low end of RR-MS control range. In contrast, his naive T helper and NK cell levels were extremely low (T helper cells 10% vs 38-55% of controls with RR-MS and NK cells 1% vs 6-13.5% of controls with RR-MS), and remained unchanged during a further 22-month study period.
Future Prospects
Experimental studies have shown that re-myelination of MS lesions or, in general, CNS lesions, can be achieved by myelinogenic or neural stem cells via transplantation or, even better, by promoting neural and glial differentiation of endogenous neural stem cells [6,17]. Three interesting studies [18-20], found increased numbers of circulating hematopoietic stem/progenitor cells that were chronically maintained, in MS patients treated with the antibody natalizumab. This antibody is administered to MS patients as second line treatment [6,18-21], and blocks lymphocyte trafficking to sites of inflammation, by adhering to the CD49d surface antigen of lymphocytes. Although the fate of these circulating hematopoietic stem/progenitor cells and their clinical relevance for MS patients remains to be determined, it is nevertheless encouraging to know that a conventional treatment for MS has a "bonus side-effect" which is the generation of new endogenous stem cells in the patients.
References-
Mouzaki A, Tselios T, Papathanassopoulos P, Matsoukas I, Chatzantoni K (2004) Immunotherapy for multiple sclerosis: basic insights for new clinical strategies. Curr Neurovasc Res 1: 325-340.
-
Mouzaki A, Deraos S, Chatzantoni K (2005) Advances in the treatment of autoimmune diseases; cellular activity, type-1/type-2 cytokine secretion patterns and their modulation by therapeutic peptides. Curr Med Chem 12: 1537-1550.
-
Hedegaard CJ, Krakauer M, Bendtzen K, Lund H, Sellebjerg F, et al. (2008) T helper cell type 1 (Th1), Th2 and Th17 responses to myelin basic protein and disease activity in multiple sclerosis. Immunology 125: 161-169.
-
Papanastasiou AD, Chatzantoni K, Mouzaki A (2008) Current therapeutic leads for the treatment of autoimmune diseases: stem cell transplantation and inhibition of post-translational modifications of autoantigens. Expert Opin Drug Discov 3: 1255-1265.
-
Blau HM1, Brazelton TR, Weimann JM (2001) The evolving concept of a stem cell: entity or function? Cell 105: 829-841.
-
Karimi-Abdolrezaee S, Eftekharpour E, Wang J, Morshead CM, Fehlings MG (2006) Delayed transplantation of adult neural precursor cells promotes remyelination and functional neurological recovery after spinal cord injury. J Neurosci 26: 3377-3389.
-
Martino G (2004) How the brain repairs itself: new therapeutic strategies in inflammatory and degenerative CNS disorders. Lancet Neurol 3: 372-378.
-
Fassas A, Mancardi GL (2008) Autologous hemopoietic stem cell transplantation for multiple sclerosis: is it worthwile? Autoimmunity 41: 601-610.
-
Fagius J, Lundgren J, Oberg G (2009) Early highly aggressive MS successfully treated by hematopoietic stem cell transplantation. Mult Scler 15: 229-237.
-
Chen JT, Collins DL, Atkins HL, Freedman MS, Arnold DL; Canadian MS/BMT Study Group (2008) Magnetization transfer ratio evolution with demyelination and remyelination in multiple sclerosis lesions. Ann Neurol 63: 254-262.
-
Shevchenko YL, Novik AA, Kuznetsov AN, Afanasiev BV, Lisukov IA, et al. (2008) High-dose immunosuppressive therapy with autologous hematopoietic stem cell transplantation as a treatment option in multiple sclerosis. Exp Hematol 36: 922-928.
-
Van Wijmeersch B, Sprangers B, Dubois B, Waer M, Billiau AD (2008) Autologous and allogeneic hematopoietic stem cell transplantation for Multiple Sclerosis: perspective on mechanisms of action. J Neuroimmunol 197: 89-98.
-
Saccardi R, Freedman MS, Sormani MP, Atkins H, Farge D, et al. (2012) A prospective, randomized, controlled trial of autologous haematopoietic stem cell transplantation for aggressive multiple sclerosis: a position paper. Mult Scler 18: 825-834.
-
Lu JQ, Storek J, Metz L, Yong VW, Stevens AM, et al. (2009) Continued disease activity in a patient with multiple sclerosis after allogeneic hematopoietic cell transplantation. Arch Neurol 66: 116-120.
-
Muraro PA, Douek DC, Packer A, Chung K, Guenaga FJ, et al. (2005) Thymic output generates a new and diverse tcr repertoire after autologous stem cell transplantation in multiple sclerosis patients. J Exp Med 201: 805-816.
-
Mouzaki A, Koutsokera M, Dervilli Z, Rodi M, Kalavrizioti D,et al. (2010) Remitting-relapsing multiple sclerosis patient refractory to conventional treatments and bone marrow transplantation who responded to natalizumab. Int J Gen Med 3: 313-320.
-
Yang J, Rostami A, Zhang GX (2009) Cellular remyelinating therapy in multiple sclerosis. J Neurol Sci 276: 1-5.
-
Bonig H, Wundes A, Chang KH, Lucas S, Papayannopoulou T (2008) Increased numbers of circulating hematopoietic stem/progenitor cells are chronically maintained in patients treated with the CD49d blocking antibody natalizumab. Blood 111: 3439-3441.
-
Zohren F, Toutzaris D, Klärner V, Hartung HP, Kieseier B, et al. (2008) The monoclonal anti-VLA-4 antibody natalizumab mobilizes CD34+ hematopoietic progenitor cells in humans. Blood 111: 3893-3895.
-
Jing D, Oelschlaegel U, Ordemann R, Hölig K, Ehninger G, et al. (2010) CD49d blockade by natalizumab in patients with multiple sclerosis affects steady-state hematopoiesis and mobilizes progenitors with a distinct phenotype and function. Bone Marrow Transplant 45: 1489-1496.
-
Dimisianos N, Rodi M, Kalavrizioti D, Georgiou V, Papathanasopoulos P, et al. (2014) Cytokines as Biomarkers of Treatment Response to IFN ? in Relapsing-Remitting Multiple Sclerosis. Mult Scler Int 2014: 436764.





