International Journal of Women's Health and Wellness
Use of Oral Contraceptives and Breast Cancer Survival
Emelie Nilsson1,2, Stina Olsson1,2, Henrik Thorlacius1,2 and Salma Butt Tuna1,2*
1Department of Surgery, Skåne University Hospital, Malmö, Sweden
2Department of Clinical Sciences, Lund University, Malmö, Sweden
*Corresponding author:
Butt Tuna Salma, Department of Surgery, Inga Maria Nilssons gata 47, plan 3, 20502 Malmö, Sweden, Tel: +46704804031, E-mail: salma.butt@med.lu.se
Int J Womens Health Wellness, IJWHW-2-039, (Volume 2, Issue 4), Cancer Epidemiology; ISSN: 2474-1353
Received: July 21, 2016 | Accepted: October 07, 2016 | Published: October 10, 2016
Citation: Nilsson E, Olsson S, Thorlacius H, Butt Tuna S (2016) Use of Oral Contraceptives and Breast Cancer Survival. Int J Womens Health Wellness 2:039. 10.23937/2474-1353/1510039
Copyright: © 2016 Emelie N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Oral contraceptives (OC) have been shown to give a transient increased breast cancer risk. However, studies on breast cancer survival after OC use are sparse and conflicting.
Aim: The aim of this study was to examine previous use of OC in relation to survival after breast cancer diagnosis.
Method: Data was collected from Malmö Diet and Cancer Study, with baseline examinations between 1991 and 1996. Out of 17035 women who completed all study parts, 765 women with incident breast cancer were included in this study. Kaplan Meier and Cox Proportional Hazards analyses, with 95% confidence intervals (CI), were used to study OC in relation to breast cancer-specific and overall survival. All analyses were stratified on age at diagnosis.
Results: Women who had ever used OC were younger and more often had grade III tumors than those who had never used OC. Breast cancer-specific survival in women who ever had used OC was better when adjusting for BMI, socioeconomic status as well as tumor characteristics. However, when adjusting for age at diagnosis, the results did not remain significant (0.68, CI 95% 0.39-1.18). Stratified analyses on age at diagnosis (40-55, 56-70 and ≥ 71) showed no statistically significant associations.
Conclusion: In this study we could not demonstrate a significant effect of OC use on breast cancer survival.
Keywords
Breast cancer, Oral contraceptives, Survival
Abbreviation
BMI: Body Mass Index; CI: Confidence Interval; ER: Estrogen Receptor; FISH: Fluorescence in Situ Hybridization; HER2: Human Epidermal Growth Factor Receptor2; HR: Hazard Ratio; ICD: International Statistical Classification of Disease and Related Health Problems; IHC: Immunohistochemistry; KM: Kaplan Meier; MDCS: Malmö Diet and Cancer Study; OC: Oral Contraceptives; PgR: Progesterone Receptor
Introduction
More than half a million women between the ages of 15 and 44 years use OC in Sweden today and more than 100 million worldwide [1,2]. Large-scale studies on OC have shown a significant association between use of OC and a slightly increased breast cancer risk [3-6]. In some studies, OC pose a higher risk of breast cancer recurrence, but there is still no conclusive evidence on whether OC affect survival after breast cancer [7-15].
The breast tissue develops under the influence of sex hormones. This process starts in fetal life and the tissue is fully developed after the first full-term pregnancy [16]. Characteristic of breast cancer is that regulatory systems of the sex hormone and other growth factors are usually mutated [17]. Consequently, some types of breast cancer grow faster under the influence of sex hormones. The normal cyclic pattern of breast tissue proliferation has been shown to be altered during use of OC suggesting OC to have a potential role in breast cancer differentiation and may influence the prognosis after breast cancer [18,19]. Furthermore, the level of proliferation in the breast cancer tissue has been seen to differ in OC users compared to never users, as do TNM-stage and grade (Elston-Ellis) at diagnosis [14,19,20].
A prospective population-based cohort, the Malmö Diet and Cancer Study (MDCS), provided information on use of OC, lifestyle factors and tumor characteristics such as expression of hormonal receptors, histological grade and vital status. Of 17035 women that participated in baseline examinations, 765 were diagnosed with incident breast cancer until end of follow-up for this study.
The aim of this study was to examine previous use of OC in relation to survival after breast cancer diagnosis. Our a priori hypothesis is that women who have used OC have a poorer breast cancer prognosis. This is based on previous research that suggests that OC users get a more aggressive type of cancer histologically speaking as well as a slightly increased, even though transient, breast cancer risk [3-6,14,19].
Material and Methods
The Malmö Diet and Cancer Study
The Malmö Diet and Cancer Study started in 1991 and residents in Malmö born between 1923 and 1950 were invited to participate. Both personal invitations and public advertisements were used in the recruitment strategy. The only predetermined exclusion criteria were mental retardation and language problems. A total of 68905 out of 74138 individuals participated in this prospective cohort study [21].
Between 1991 and 1996, the baseline examinations were conducted. One trained nurse performed the physical examination, measurements of weight and height and the questionnaire consisted of questions on dietary habits, other lifestyle factors and exposure to OC. Socioeconomic status was also collected at baseline and was defined as worker, civil servant and businessman. Of the 28098 participants who completed all study parts, 17035 were women and formed the basis of the female cohort in MDCS [21].
Oral contraceptives
Information on OC, i.e., whether they had ever or never used OC, was collected from the questionnaire. No information on type of OC or duration of use was available.
Follow-up and endpoints
All women were followed until 31st of December 2011. Information on vital status was retrieved from Swedish Cause of Death Registry. Tumor data were only available up until 31st of December 2007; hence all breast cancer cases diagnosed after this date were excluded from this study. Information on endpoint status was retrieved from The Swedish Cancer Registry and from The Southern Swedish Regional Tumor Registry, due to a delay in the national registry [22].
Two different endpoints were used i.e., breast cancer-specific mortality, and death from all causes (overall survival). Breast cancer-specific death, defined as breast cancer as cause of death and breast cancer as a part of a multiple cause of death, was used as the primary endpoint. International Statistical Classification of Disease and Related Health Problems (ICD) codes used for identification of cause of death were C50 (ICD 10) and 174 (ICD 9).
Histopathological analyses
Histopathological analyses were performed and described according to the WHO classification. All tumors were re-evaluated by one senior pathologist [23]. The tumors were graded according to Nottingham classification (Elston and Ellis) into a three-graded scale [24]. As described by Borgquist, et al. tissue micro arrays were constructed and used for immunohistochemical analyses (IHC). An evaluation was made twice, and in case of inconsistency a third survey was performed, and based on these a decision was made [23]. IHC analyses were performed to confirm the nuclear expression of Ki 67, PgR, ER and p27. These markers were all dichotomized, with ≤ 10% being the cut-off for low expression and > 10% being high expression.
As data of gene amplification of HER2 was not available, the classification of HER2 expression was made according to well-established clinical practice in Sweden with immunohistochemical analyses. The tumors were characterized as either HER2 moderate/strong (2+ and 3+) or negative/weak (0 and 1+). Tumors were considered positive when scored 3+ on IHC staining or when they received a score of 2+ and were additionally verified with Fluorescence in Situ Hybridization (FISH) described elsewhere [23,25].
Study population
Of a total of 1498 women with a breast cancer diagnosis, 476 cases had a prevalent breast cancer diagnosis, 11 missed necessary follow-up data and 246 were diagnosed after 31st of December 2007. All these cases were excluded from the study; hence our study population consisted of 765 women with incident breast cancer (Figure 1).
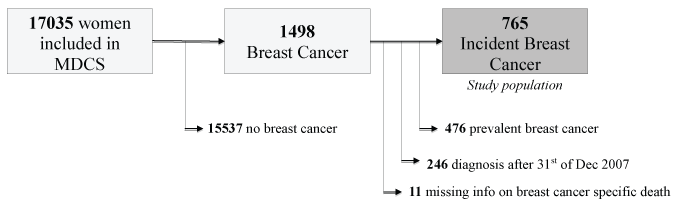
.
Figure 1: The malmö diet and cancer study (MDCS). The flow chart illustrates the selection of the study population. A total of 765 cases were further analyzed.
View Figure 1
Study population
Of a total of 1498 women with a breast cancer diagnosis, 476 cases had a prevalent breast cancer diagnosis, 11 missed necessary follow-up data and 246 were diagnosed after 31st of December 2007. All these cases were excluded from the study; hence our study population consisted of 765 women with incident breast cancer (Figure 1).
Statistical methods
For descriptive analyses, the Pearson Chi-Square test and Mann-Whitney U-test were used where appropriate.
Kaplan Meier curves were plotted to illustrate survival in relation to breast cancer specific death/overall death and use of OC. Complementary log-rank tests were made. If the assumption of proportionally was met, survival analyses with Cox Proportional Hazard analyses were performed to estimate hazard ratios (HR) on breast cancer-specific and overall survival with a 95% CI. The time scale used was time from breast cancer diagnosis until the previously described endpoints or end of follow-up. All survival analyses were adjusted for age at diagnosis, BMI, socioeconomic status, tumor size, lymph glandular involvement, tumor grade, ER, PgR Ki 67, HER2 and p27 in multivariate analyses. These are well-established or potential factors that could influence breast cancer survival [10,17,26,27]. The analyses were further stratified by age at diagnosis. Age at diagnosis was divided into three groups, 40-55 years (n = 116), 56-70 years (n = 471) and 71 years and older (n = 177) based on incidence and survival data. Studies have shown a high percentage of deaths attributed to breast cancer in women younger than 55 years, and age-specific incidence slightly decreases for women aged 70-74, motivating cut-offs accordingly [28,29].
All analyses were made in IBM SPSS version 22.
Ethical approval was obtained from the ethics committee of Lund University. Ethical approval number: LU51-90,DNR652/2005.
Results
Descriptive analyses
Age at diagnosis ranged from 44 to 73 years. At follow-up, a total of 572 women were alive, 191 had died and two were lost to follow-up. Of these, 115 deaths were attributed to either breast cancer directly or breast cancer as part of a multiple cause of death.
Women who ever had used OC were significantly younger at diagnosis. The BMI scores were significantly lower amongst women who had ever used OC. The socioeconomic status differed significantly, with lower educational status amongst women who had never used OC. In the group of women who had used OC, the expression of PgR was more frequently low and they had more grade III tumors than the women who had never used OC (Table 1).
![]()
Table 1: Distribution of potential risk factors, prognostic and predictive factors in ever and never use of oral contraceptives.
View Table 1
![]()
Table 2A: Breast cancer-specificsurvival in relation to OC use.
View Table 2A
Survival analyses
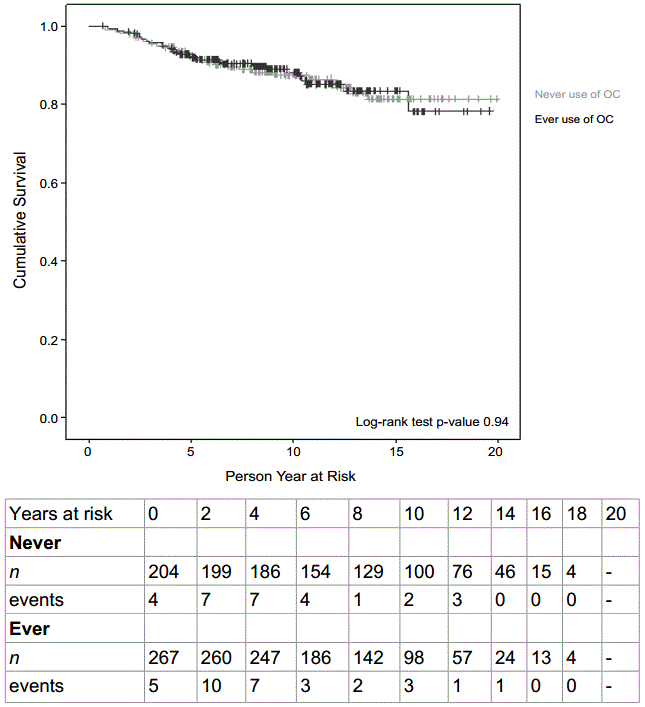
Kaplan Meier curves: In Kaplan Meier curves on ever/never use of OC assumption of proportionality were met (Figure 2). Moreover, a statistically significant difference in breast cancer-specific survival was found (log-rank, p value 0.01). After stratification on age at diagnosis another three Kaplan Meier curves were performed. In the age group 40-55 there is a crossing of lines after approximately 12 years of follow-up (Figure 3A). Prior to this point the ever users' graph had a less incline, indicating a better survival, though not significant with a log-rank test (p value 0.45). In the age group 56-70 no difference in survival could be observed (Figure 3B). The oldest women at diagnosis had better survival amongst the ever users of OC (log-rank, p value 0.03) (Figure 3C). Kaplan Meir Curves were plotted on overall survival and did not differ from breast cancer-specific death (data not shown).
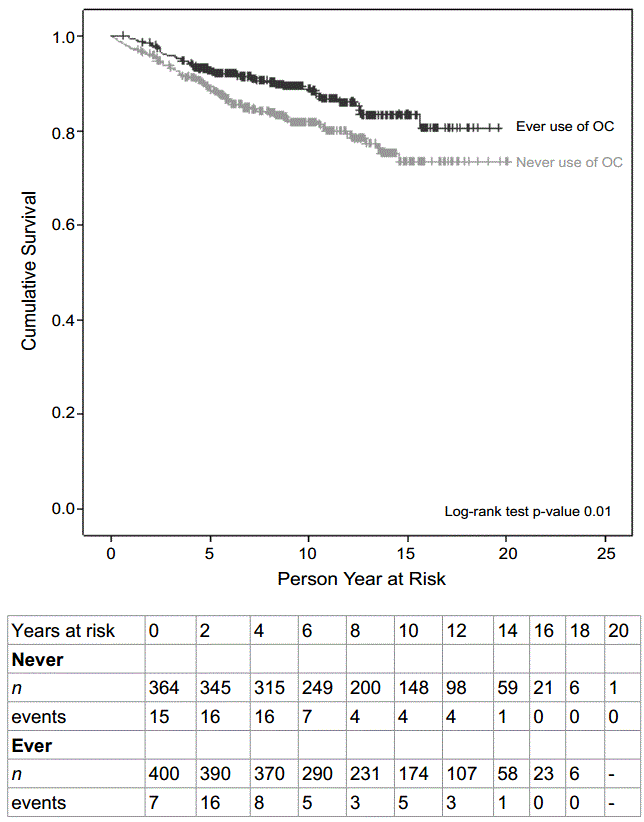
.
Figure 2: Kaplan meier curve showing breast cancer-specific survival in ever and never users of oral contraceptives. Beneath, a life table demonstrates the cohort during the different intervals. “n”, the number of women entering the specific interval. Events, deaths during that interval.
View Figure 2
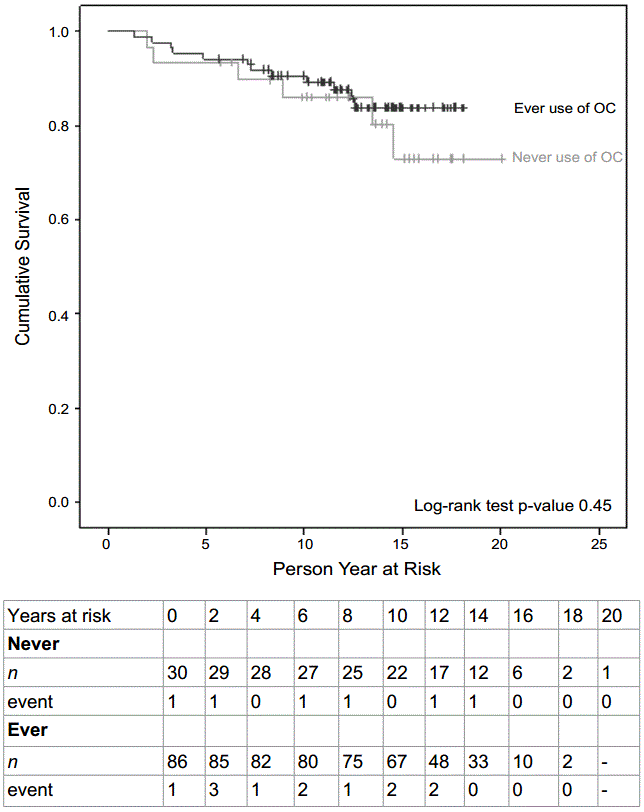
.
Figure 3a: Kaplan meier curve showing breast cancer-specific survival amongst women aged 55 or younger. Beneath, a life table demonstrates the cohort during the different intervals. “n”, the number of women entering the specific interval. Events, deaths during that specific interval.
View Figure 3a
Cox proportional hazard: In Cox analyses for breast cancer-specific and overall survival, a better survival was noted in the crude analyses for ever users of OC. However, in the multivariate analyses this association did not remain statistically significant (Table 2A and Table 2B).
.
Figure 3b: Kaplan meier curve showing breast cancer-specific survival amongst women aged 56 to 70. Beneath, a life table demonstrates the cohort during the different intervals. “n”, the number of women entering the specific interval. Events, deaths during that specific interval.
View Figure 3b
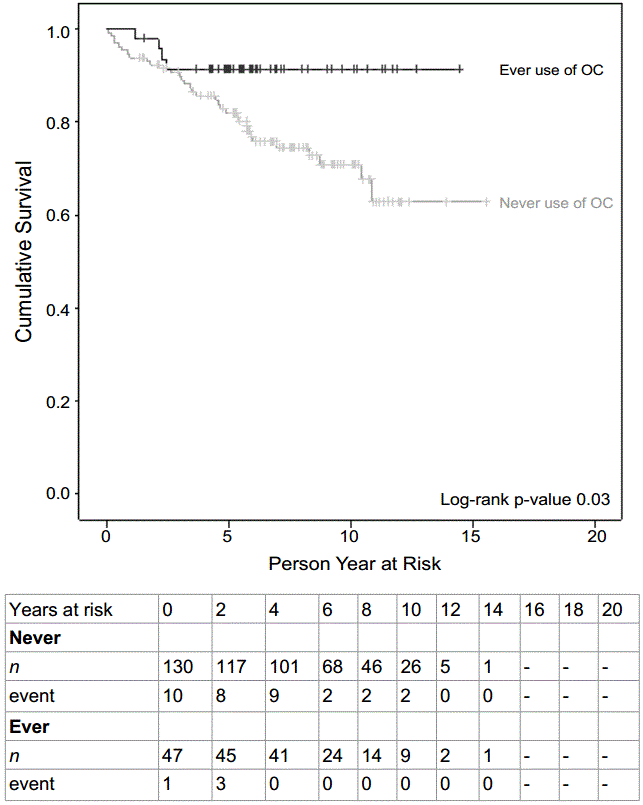
.
Figure 3c: Kaplan meier curve showing breast cancer-specific survival amongst women aged 71 and above. Beneath, a life table demonstrates the cohort during the different intervals. “n”, the number of women entering the specific interval. Events, deaths during that specific interval.
View Figure 3c
Age was the factor affecting the HR the most; hence, stratification on age was made. In age-stratified survival analysis the oldest women who had ever used OC showed significantly better breast cancer-specific survival, these results did not remain significant in multivariable analyses (Table 3A). The same pattern was seen when analyzing overall survival (Table 3B).
![]()
Table 2B: Breast cancer-specific survival in relation to OC use, stratified by age at diagnosis.
View Table 2B
![]()
Table 3A: Overall survival in relation to OC use.
View Table 3A
Even though not significant, the HR in all stratified groups measured less than one. This was most distinctive in the youngest and the oldest age groups.
![]()
Table 3B: Overall survival in relation to OC use, stratified by age at diagnosis.
View Table 3B
Discussion
The aim of this study was to examine if OC had an impact on breast cancer survival. We could not find significant associations between OC use and breast cancer survival in multivariate analyses.
Methodological considerations
Information on OC use was collected as either never or ever use in the questionnaire. Information such as type of OC, age at first use, duration or time since cessation, were not collected and could hence not be obtained. However, a previous study has shown that neither type nor duration of OC use was associated with risk of death from breast cancer [8]. A recently published report from the Nurses' Health Study shows a higher risk of breast cancer death with increased duration [30]. Yet another from Holmberg, et al. demonstrate a lower risk of death in short-term users compared to never users [9]. We suggest that the matter is not yet settled.
Breast cancer risk factors seem different from factors affecting the breast cancer prognosis. Important risk factors such as age at first use and use before first childbirth do not seem to affect the breast cancer prognosis [8,9]. This is information that could not be retrieved in this study and was not adjusted for. Considering recent research on use patterns we acknowledge duration of OC use as a potential confounding factor, thus, a shortage of information is a limitation in our study and should of course be taken into account when interpreting our findings.
The variable ever/never use of OC used in our study was complete for the entire studied group of breast cancer cases with one exception only, and previous studies have shown self-reported information on OC to be valid [31].
Another important limitation in this study is the lack of information on metastatic disease and breast cancer treatment. However, tumor size, lymph node involvement and tumor grade i.e., known important prognostic factors, were adjusted for in the multivariate analyses.
Representativeness and validity of endpoints
Approximately 40% of the invited women in the MDCS completed the study. Ethnic Swedes and women with higher socioeconomic status may be overrepresented [21]. This may of course have caused a selection bias and an unrepresentative study population. However, since this study is of relative survival and not absolute survival, we believe our results to be less affected by this bias. All tumor endpoints were collected with linkage of personal identity number to the Swedish Cancer Registry and the regional branch. The data are considered valid because of the completeness of the registries [32,33]. Vital status was obtained from the Swedish Cause of Death Registry, and due to the individual personal identity number that all residents in Sweden are given, the completeness is close to 100% [34]. We chose to use both breast-specific death and death from all causes as endpoints to identify potential misclassification of breast cancer-specific death.
Previous studies
According to Russo, et al. the breast tissue is not fully developed until the first full-term pregnancy. Carcinogens have greater impact on undifferentiated breasts; consequently breast tissue following a pregnancy becomes less sensitive to malignant transformation [16]. Could OC interfere with this sensitivity, and in that case contribute to a type of breast cancer that differs in histological expression and/or breast cancer prognosis? Since we had no information on time of first use, this aspect could not be examined in this study. Studies have shown a different pattern and higher proliferation in healthy breast tissue in women using OC compared to women not using OC [18,35]. Garcia y Narvaiza, et al. showed that the breast tissue proliferation was significantly higher during the first week of the menstrual cycle in healthy OC users, whereas normally the proliferation rate is highest during the luteal phase [18]. A difference in Ki67 density in the breast cancer tissue due to exposure to OC could not be demonstrated in this study; however, previous research has shown that early exposure to OC could lead to a more aggressive disease i.e., a higher proliferation rate of the tumor cells [19].
In this study, women who had used OC were younger. Sauerbrei, et al. demonstrate the same pattern; however, their study was made on node positive breast cancer only and hence is not directly comparable to our results [11]. In accordance with this study, Sauerbrei, et al. could not see any difference in tumor size between users and non-users of OC [11]. Schönborn, et al. found OC caused women to more often get grade III tumors in conformity with our results [14]. As grade III is the most aggressive tumor type histologically, one could expect a higher death rate amongst these patients.
This does not seem to be the case, based on results from this study and earlier research [14]. At least one study to our knowledge reported more advanced disease (according to TNM-stage) in women who had never used OC compared to those who had ever used it [20]. Hence, there is not yet evidence for definite co-variation of tumor characteristics and OC use.
As to the risk of death from breast cancer there are conflicting results in the research field. Trivers, et al. demonstrate results indicating that OC use could influence survival negatively [13]. Other studies found no difference in survival between exposed and non-exposed patients [8,9,11,12,15]. Better survival has also been described in current long-term OC users [14]. According to Reeves, et al. the risk of dying decreases with increasing time after cessation of OC [10]. Saxe, et al. showed interesting results, where OC was associated with increased recurrence but not death [7]. These studies were not all homogenous. Some of them studied mostly premenopausal women and therefore the results cannot be directly compared with our study. In this study, univariate analysis shows a significantly better survival in women who ever had used OC. This could not be confirmed in multivariate analyses, suggesting no effect of OC use on breast cancer survival. Still, in several of the multivariate subgroup analyses the HR showed protective associations, suggesting a difference in survival, though this was not statistically significant. This indicates a possibility of type II errors in these analyses considering the low numbers of deaths and the total number of participants in these subgroups.
To our knowledge it is not yet settled whether duration of use influences survival in breast cancer [8,9,30]. However, duration could have a confounding effect on breast cancer-specific survival. If the duration pattern of use has changed over time, this could perhaps partially explain some of the variations in the different age groups. However, as already mentioned, whether duration of use affects survival after breast cancer diagnosis is still uncertain. Another factor that could contribute to explaining the potential survival difference between age groups is that older women die from illnesses other than their breast cancer.
Hypothetically, the difference in biological treatment between older and younger women could contribute. Decreased recurrence has been observed in OC users with aromatase inhibitors [36]. To our knowledge there is no study on OC´s impact on breast cancer-specific survival after hormonal therapy.
Whether OC affects the breast cancer-specific mortality is not yet settled. Our results point towards no survival difference, supporting the conclusions from the majority of previous studies.
Conclusion
This study found no statistically significant difference between ever and never use of oral contraceptives on breast cancer survival when adjusting for potential confounders.
Conflicts of Interest
All authors declare that they do not have competing interests or conflicts of interest that could influence the study.
Authors' Contribution
Emelie Nilsson performed all statistical analyses, drafted the manuscript and was involved in the study design. Stina Olsson supported some statistical analyses and critically revised the manuscript. Henrik Thorlacius critically revised the statistical analyses and manuscript. Salma Tunå Butt designed the study and supervised all statistical analyses and critically revised the manuscript.
Acknowledgement
Thanks to all the people participating in the work of the malmö diet and cancer study.
Novelty and Impact Description
Previous studies have shown that use of oral contraceptives gives a transient higher risk of getting breast cancer. However, whether use of oral contraceptives could affect breast cancer survival is sparsely investigated. The studies that do exist show conflicting results. The results from this research show that use of oral contraceptives does not influence breast cancer survival.
References
-
Dhont M (2010) History of oral contraception. Eur J Contracept Reprod Health Care 15: S12-18.
-
http://www.socialstyrelsen.se/Lists/Artikelkatalog/Attachments/18278/2011-3-30.pdf.
-
Collaborative Group on Hormonal Factors in Breast C (1996) Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53 297 women with breast cancer and 100 239 women without breast cancer from 54 epidemiological studies. Lancet 347: 1713-1727.
-
Kahlenborn C, Modugno F, Potter DM, Severs WB (2006) Oral contraceptive use as a risk factor for premenopausal breast cancer: a meta-analysis. Mayo Clin Proc 81: 1290-1302.
-
Beaber EF, Buist DS, Barlow WE, Malone KE, Reed SD, et al. (2014) Recent oral contraceptive use by formulation and breast cancer risk among women 20 to 49 years of age. Cancer Res 74: 4078-4089.
-
Beaber EF, Malone KE, Tang MT, Barlow WE, Porter PL, et al. (2014) Oral contraceptives and breast cancer risk overall and by molecular subtype among young women. Cancer Epidemiol Biomarkers Prev 23: 755-764.
-
Saxe GA, Rock CL, Wicha MS, Schottenfeld D (1999) Diet and risk for breast cancer recurrence and survival. Breast Cancer Res Treat 53: 241-253.
-
Wingo PA, Austin H, Marchbanks PA, Whiteman MK, Hsia J, et al. (2007) Oral contraceptives and the risk of death from breast cancer. Obstet Gynecol 110: 793-800.
-
Holmberg L, Lund E, Bergstrom R, Adami HO, Meirik O (1990) Oral contraceptives and prognosis in breast cancer: effects of duration, latency, recency, age at first use and relation to parity and body mass index in young women with breast cancer. Eur J Cancer 30: 351-354.
-
Reeves GK, Patterson J, Vessey MP, Yeates D, Jones L (2000) Hormonal and other factors in relation to survival among breast cancer patients. International journal of cancer. Int J Cancer 89: 293-299.
-
Sauerbrei W, Blettner M, Schmoor C, Bojar H, Schumacher M (1998) The effect of oral contraceptive use on the prognosis of node positive breast cancer patients. German Breast Cancer Study Group. Eur J Cancer 34: 1348-1351.
-
Schouten LJ, Hupperets PS, Jager JJ, Volovics L, Wils JA, et al. (1997) Prognostic significance of etiological risk factors in early breast cancer. Breast Cancer Res Treat 43: 217-223.
-
Trivers KF, Gammon MD, Abrahamson PE, Lund MJ, Flagg EW, et al. (2007) Oral contraceptives and survival in breast cancer patients aged 20 to 54 years. Cancer Epidemiol Biomarkers Prev 16: 1822-1827.
-
Schönborn I, Nischan P, Ebeling K (1994) Oral contraceptive use and the prognosis of breast cancer. Breast Cancer Res Treat 30: 283-292.
-
Lu Y, Ma H, Malone KE, Norman SA, Sullivan-Halley J, et al. (2011) Oral contraceptive use and survival in women with invasive breast cancer. Cancer Epidemiol Biomarkers Prev 20: 1391-1397.
-
Russo J, Hu YF, Silva ID, Russo IH (2001) Cancer risk related to mammary gland structure and development. Microsc Res Tech 52: 204-223.
-
Jayant S, Vaidya DJ (2014) Fast Facts: Breast Cancer. Health Press Limited, Oxford.
-
Garcia y Narvaiza D, Navarrete MA, Falzoni R, Maier CM, Nazário AC (2008) Effect of combined oral contraceptives on breast epithelial proliferation in young women. Breast J 14: 450-455.
-
Olsson H, Ranstam J, Baldetorp B, Ewers SB, Fernö M, et al. (1991) Proliferation and DNA ploidy in malignant breast tumors in relation to early oral contraceptive use and early abortions. Cancer 67: 1285-1290.
-
Vessey M, Baron J, Doll R, McPherson K, Yeates D (1983) Oral contraceptives and breast cancer: final report of an epidemiological study. Br J Cancer 47: 455-462.
-
Manjer J, Carlsson S, Elmstahl S, Gullberg B, Janzon L, et al. (2001) The Malmo Diet and Cancer Study: representativity, cancer incidence and mortality in participants and non-participants. Eur J Cancer Prev 10: 489-499.
-
Butt S, Harlid S, Borgquist S, Ivarsson M, Landberg G, et al. (2012) Genetic predisposition, parity, age at first childbirth and risk for breast cancer. BMC Res Notes 5: 414.
-
Borgquist S, Anagnostaki L, Jirstrom K, Landberg G, Manjer J (2007) Breast tumours following combined hormone replacement therapy express favourable prognostic factors. International journal of cancer 120: 2202-2207.
-
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19: 403-410.
-
Dowsett M, Bartlett J, Ellis IO, Salter J, Hills M, et al. (2003) Correlation between immunohistochemistry (HercepTest) and fluorescence in situ hybridization (FISH) for HER-2 in 426 breast carcinomas from 37 centres. The Journal of pathology 199: 418-423.
-
Bouchardy C, Verkooijen HM, Fioretta G (2006) Social class is an important and independent prognostic factor of breast cancer mortality. International journal of cancer 119: 1145-1151.
-
Sun X, Nichols HB, Robinson W, Sherman ME, Olshan AF, et al. (2015) Post-diagnosis adiposity and survival among breast cancer patients: influence of breast cancer subtype. Cancer causes control 26: 1803-1811.
-
http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/breast-cancer/incidence-invasive - heading-One.
-
McPherson K, Steel CM, Dixon JM (2000) ABC of breast diseases. Breast cancer-epidemiology, risk factors, and genetics. BMJ 321: 624-628.
-
Charlton BM, Rich-Edwards JW, Colditz GA, Missmer SA, Rosner BA, et al. (2014) Oral contraceptive use and mortality after 36 years of follow-up in the Nurses' Health Study: prospective cohort study. BMJ 349: g6356.
-
Norell SE, Boethius G, Persson I (1998) Oral contraceptive use: interview data versus pharmacy records. Int J Epidemiol 27: 1033-1037.
-
Garne JP, Aspegren K, Balldin G (1996) Breast cancer as cause of death--a study of the validity of the officially registered cause of death of 2631 breast cancer patients dying in Malmo, Sweden 1964-1992. Acta oncologica 35: 671-675.
-
Ludvigsson JF, Almqvist C, Bonamy AK, Ljung R, Michaëlsson K, et al. (2016) Registers of the Swedish total population and their use in medical research. Eur J Epidemiol 31: 125-136.
-
http://www.socialstyrelsen.se/publikationer2015/2015-8-1.
-
Isaksson E, von Schoultz E, Odlind V, Söderqvist G, Csemiczky G, et al. (2001) Effects of oral contraceptives on breast epithelial proliferation. Breast Cancer Res Treat 65: 163-169.
-
Huzell L, Persson M, Simonsson M, Markkula A, Ingvar C, et al. (2015) History of oral contraceptive use in breast cancer patients: impact on prognosis and endocrine treatment response. Breast Cancer Res Treat 149: 505-515.





