Trauma Cases and Reviews
A Metabolic Stress Fracture of the Mandibular Condylar Head
Sabah Kalamchi* and Trent C Clifton
Department of Oral and Maxillofacial Surgery, Arizona School of Dentistry and Oral Health, USA
*Corresponding author:
Sabah Kalamchi, Department of Oral and Maxillofacial Surgery, Arizona School of Dentistry and Oral Health, 5850 E Still Circle, Mesa, AZ 85206, USA, Tel: 480-248-8156, Fax: 480-941-1362, E-mail: skalamchi@atsu.edu
Trauma Cases Rev, TCR-1-002 (Volume 1, Issue 1), Case Report; ISSN: 2469-5777
Received: February 26, 2015 | Accepted: April 30, 2015 | Published: May 02, 2015
Citation: Kalamchi S, Clifton TC (2015) A Metabolic Stress Fracture of the Mandibular Condylar Head. Trauma Cases Rev 1:002. 10.23937/2469-5777/1510002
Copyright: © 2015 Kalamchi S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Fractures of the mandibular condyle and neck are very common with well recognized anatomical features. This patient sustained an anatomically unusual right condylar head fracture with subluxation secondary to ictal myocontraction where there was no fall or facial trauma during or after the seizure. Investigation suggested an underlying disturbance in bone metabolism. We found one case report of mandibular stress fracture as a result of reflexive myo contraction of the temporalis and lateral pterygoid muscles in the literature. We conclude stress fractures resultant from ictal myo contractions are exceptionally rare without a covert disturbance in bone density. This report also applies to the recognition and treatment of patients at risk for bone fractures due to seizures: such as patients with osteomalacia, osteoporosis, chronic alcoholics, and elderly stroke victims.
Keywords
Condylar head, Mandible, Fracture, Stress fracture, Seizure
Introduction
Stress fractures of the human skeleton are categorized by (1) over-use injuries and (2) insufficiency fractures. Over-use fractures result from non-physiologic or repetitive forces on normal, healthy bone, and primarily effect the tibia, tarsals, and metatarsals [1]. Insufficiency fractures are caused by physiologic forces on weakened bone such as in metabolic bone disease [2]. They are most commonly reported in the pelvic girdle, proximal femur, and vertebral bodies of the lumbar spine [3]. One group of patients at risk for insufficiency fractures are those taking anti-epileptic drugs (AED) since AED are known to induce metabolic bone disease by interfering with both vitamin D and parathyroid hormone metabolism reducing bone mineral density (BMD) [4-6].
The oral and maxillofacial surgery literature documents stress fractures at donor sites such as iliac crest and long bones following bone graft harvesting [7]. However, the literature on metabolic bone disease in the maxillofacial region is mostly limited to radiography techniques used to identify osteoporosis [8]. There are few reports of insufficiency fractures in the oral and maxillofacial literature. We found one case where reflex myocontraction of the temporalis muscles caused bilateral coronoid process fracture. Underlying metabolic bone disease in this case was not clear [9]. We did not find any published reports of AED use underlying a maxillofacial fracture.
The present case report is of an anatomically unusual insufficiency fracture of the right condylar head secondary to ictal myocontraction with evidence of underlying metabolic bone disease induced by an AED.
Case Report
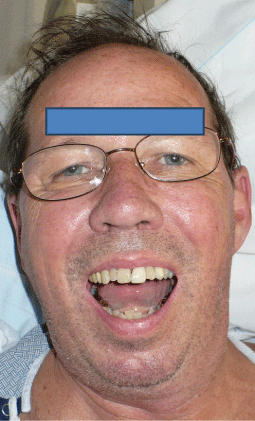
A 64-year-old man presented to the emergency room with his wife complaining of pain in his jaw and difficulty closing his mouth following a seizure. The patient reported they were discussing dinner options when he suddenly yelled, contorted his face, and became noticeably tense for approximately 30 seconds. No tonic clonic activity was observed. His wife confirmed that he did not fall during or after the seizure, and he had no evidence of external trauma (Figure 1).
.
Figure 1: The patient reported to the hospital unable to close his jaw. He had no signs of facial trauma following his seizure.
View Figure 1
Prior to the seizure the patient had no signs or symptoms of abnormal jaw function or pain. Following the seizure the patient was unable to close his mouth, and reported to the hospital for evaluation. He reported never having had a previous seizure or facial trauma.
The patient�s medical history includes gastroesophageal reflux disease, hypertension, and cerebrovascular accident (CVA) five-and-a-half months prior to hospital admission. At the time of the CVA the patient experienced a right frontal cerebral hemorrhage resulting in weakness and loss of coordination that resolved during two months of physical therapy. He began taking levetiracetam (Keppra�), which was discontinued along with the physical therapy and is possibly another potential etiological contributor to the seizure. Levetiracetam is also implicated in the patient�s reduced BMD [4-6].
The patient�s social history includes drinking at least 3-4 Vodka and tonics per day. Three days prior to the seizure episode the patient had discontinued drinking alcohol, which may also be a contributing factor in the seizure�s etiology [10].
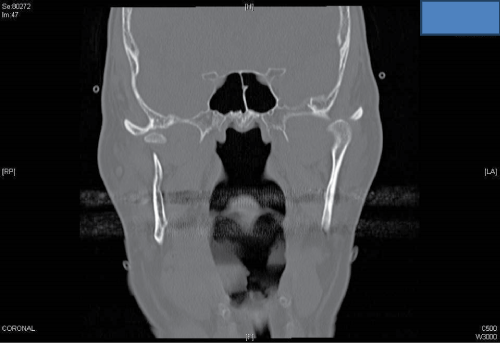
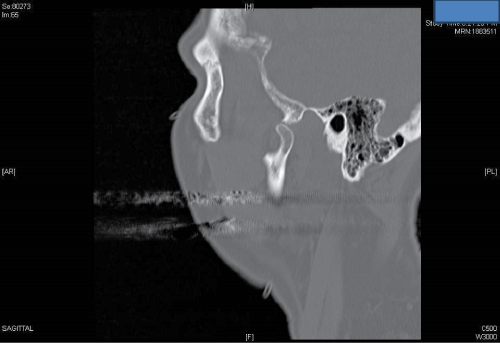
A C.T. scan revealed left condylar head subluxation and right condylar head fracture with medial displacement. The margins of the fracture were fresh according to the radiologist�s report, which excludes the possibility of a pre-existing fracture (Figure 2a,2b).
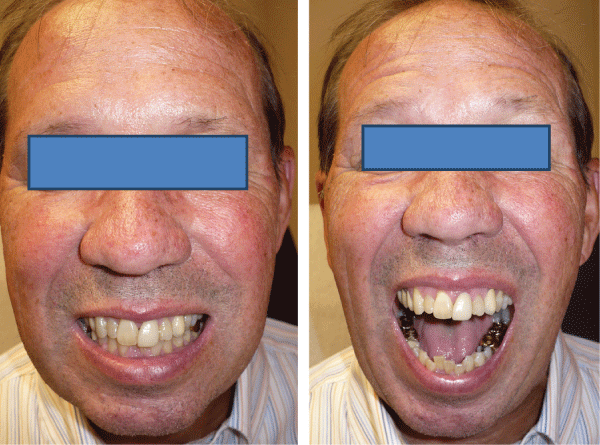
The subluxated left condyle was reduced a day later under general anesthesia after the correction of a low serum potassium. The malocclusion was treated with upper and lower arch bars in maxilla mandibular fixation. Intermaxillary training elastics were used two-weeks postoperatively to stabilize the jaw until full mandibular function was achieved without pain (Figure 3).
.
Figure 3: The patient returned to normal function following treatment with full range of motion and interincisal opening of 42 m.mand normal occlusal alignment.
View Figure 3
Suspecting covert metabolic bone disease, appropriate tests were ordered. Serum 25-hydroxy vitamin D [25 (OH) D] levels were 21.1ng/mL (normal 30.0ng/mL) [11]. Dual energy x-ray absorptiometry scans of the lumbar spine, left pelvis, and left femoral neck revealed BMD T-scores-which represent standard deviations (SD) away from peak bone mass of young adults of the same gender-were, respectively, -2.3, -1.6, and -1.9-1.0. Osteopenia is defined by a t-score between -1.0 and -2.5 SD; osteoporosis is defined by a t-score -2.5 SD [12].
He was diagnosed with Vitamin D deficiency and osteopenia with borderline osteoporosis. A metabolic bone disease pharmaceutical regimen was started by his physician.
Summary and Conclusion
Most seizure-related facial fractures are sustained when the face of the seizing patient strikes a hard surface during the uncontrolled and spastic myocontractions characteristic of ictal events. Such mandibular fractures secondary to seizures are well recognized [13,14].
We believe this is the first documented case in the literature of ictal myocontraction-induced fracture of the condylar head secondary to metabolic bone disease. Like most uncommon lesions it may be anatomically unrecognized and clinically under investigated. This report is also significant in the light of the patient�s history of daily alcohol consumption, which is associated with reduced bone mineralization [15].
The role of levetiracetam in lowering bone mineral density, BMD, by increasing bone turnover with a net increase in bone resorption over bone formations although was taken for around five months cannot be underestimate metabolic effect on bone [16].
Suppliementaryfile
References
-
Snyder RA, DeAngelis JP, Koester MC, Spindler KP, Dunn WR (2009) Does shoe insole modification prevent stress fractures? A systematic review. HSS J 5: 92-98.
-
Ruohola JP, Laaksi I, Ylikomi T, Haataja R, Mattila VM, et al. (2006) Association between serum 25(OH)D concentrations and bone stress fractures in Finnish young men. J Bone Miner Res 21: 1483-1488.
-
Krestan CR, Nemec U, Nemec S (2011) Imaging of insufficiency fractures. Semin Musculoskelet Radiol 15: 198-207.
-
Andress DL, Ozuna J, Tirschwell D, Grande L, Johnson M, et al. (2002) Antiepileptic drug-induced bone loss in young male patients who have seizures. Arch Neurol 59: 781-786.
-
Bischof F, Basu D, Pettifor JM (2002) Pathological long-bone fractures in residents with cerebral palsy in a long-term care facility in South Africa. Dev Med Child Neurol 44: 119-122.
-
Drezner MK (2004) Treatment of anticonvulsant drug-induced bone disease. Epilepsy Behav 5: S41-47.
-
Westesson PL, Wandtke JC (1999) Stress fracture of the tibia: an unusual complication of reconstructive surgery of the mandible. J Oral Maxillofac Surg 57: 70-74.
-
Li N, Jing H, Li J, Zhou F, Bu L, et al. (2011) Study of mandible bone mineral density of Chinese adults by dual-energy X-ray absorptiometry. Int J Oral Maxillofac Surg 40: 1275-1279.
-
Philip M, Sivarajasingam V, Shepherd J (1999) Bilateral reflex fracture of the coronoid process of the mandible. A case report. Int J Oral Maxillofac Surg 28: 195-196.
-
Matti H, Ilkka P, Maurizio L (2003) Seizures in Alcohol-Dependent Patients: Epidemiology, Pathophysiology and Mangement., CNS Drugs 17: 1013-1030.
-
Sunil J (2012) Vitamin D in the new millennium. Curr Osteoporos Rep: 1-12.
-
Lim LS, Hoeksema LJ, Sherin K; ACPM Prevention Practice Committee (2009) Screening for osteoporosis in the adult U.S. population: ACPM position statement on preventive practice. Am J Prev Med 36: 366-375.
-
Aragon CE, Burneo JG, Helman J (2001) Occult maxillofacial trauma in epilepsy. J Contemp Dent Pract 2: 26-32.
-
Buck D, Baker GA, Jacoby A, Smith DF, Chadwick DW (1997) Patients' experiences of injury as a result of epilepsy. Epilepsia 38: 439-444.
-
Tucker KL, Jugdaohsingh R, Powell JJ, Qiao N, Hannan MT, et al. (2009) Effects of beer, wine, and liquor intakes on bone mineral density in older men and women. Am J Clin Nutr 89: 1188-1196.
-
Sato Y, Kondo I, Ishida S, Motooka H, Takayama K, et al. (2001) Decreased bone mass and increased bone turnover with valproate therapy in adults with epilepsy. Neurology 57: 445-449.