Trauma Cases and Reviews
18-Fluordesoxiglucose Positron Emission Tomography (PET) in Evaluating Neurological Prognosis after Cardiac Arrest Resulting from Hanging: A Case Report
Marco Ciapetti1, Andrea Cecchi1, Marco Mangini2*, Valentina Berti3, Matteo Bartolini3, Alberto Pupi3 and Adriano Peris1
1Anesthesia and Intensive Care Unit of Emergency Department, Careggi Teaching Hospital, Largo Brambilla 3, 50139 Florence, Italy
2Postgraduate School of Anesthesia and Intensive Care, Careggi Teaching Hospital, Largo Brambilla 3, 50139 Florence, Italy
3Biomolecular Nuclear Medicine, Careggi Teaching Hospital, Largo Brambilla 3, 50139 Florence, Italy
*Corresponding author: Marco Mangini, MD, Postgraduate School of Anesthesia and Intensive Care, Careggi Teaching Hospital, Largo Brambilla 3, 50139, Florence, Italy, E-mail: eat83@hotmail.com
Trauma Cases Rev, TCR-2-029, (Volume 2, Issue 1), Case Report; ISSN: 2469-5777
Received: September 15, 2015 | Accepted: January 28, 2016 | Published: January 30, 2016
Citation: Ciapetti M, Cecchi A, Mangini M, Berti V, Bartolini M, et al. (2016) 18-Fluordesoxiglucose Positron Emission Tomography (PET) in Evaluating Neurological Prognosis after Cardiac Arrest Resulting from Hanging: A Case Report. Trauma Cases Rev 2:029. 10.23937/2469-5777/1510029
Copyright: © 2016 Ciapetti M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We report clinical and 18 - FDG PET features of a 58 years old woman found in cardiac arrest after an attempt of hanging. The patient was resuscitated and transferred to our Emergency Intensive Care Unit (ICU), where she was treated for 23 days and then she was discharged to a neuro rehabilitation long term care center with a Glasgow Coma Scale (GCS) of 6. A 18 F - FDG PET with semi quantitative glucose metabolism measurements was performed showing a global reduction in supra tentorial areas, in particular in cortical and thalamic nucleus. These data were confirmed by a second 18F FDG PET performed one year later.
Keywords
Hanging, Cardiac arrest, Cardiopulmonary resuscitation, Glasgow coma scale, 18 fdg pet
Abbreviations
18F-FDG PET: 18- fluordesoxiglucose positron emission tomography, GCS: Glasgow Coma Scale, CT: Computed Tomography, MRI: Magnetic Resonance Imaging.
Background
Hanging is an increasingly common way of attempting suicide [1]. Death by hanging is induced by neck airway obstruction due to a ligature that is tightened by victim's own body weight, ultimately causing cardiac arrest. Hanging suicide with cardiac arrest induces acute hypoxic-ischemic encephalopathy [2]. Although several CT scan and MRI studies of the head are present in international literature [3-5] no brain 18F-FDG PET study in hanging has been reported. Here we present a case of a 58 year-old woman found in cardiac arrest after a suicide attempt by hanging who underwent brain 18F-FDG PET examination both in subacute phase and at 1-year follow-up.
Case Presentation
A 58 years old woman (58 Kg of body weight and 160 cm in height) with bipolar mental illness was admitted to the Toxicology Unit of Careggi Teaching Hospital in Florence, Italy, on August 22nd, 2008 after self-intoxication resulting from ingestion of 10 mg of alprazolam. On the first day from the admission the staff found the patient hanged, in cardiac arrest, with a bathrobe belt attached to a water pipe, after about 20 minutes from the moment she entered the bathroom. The patient was carefully placed on the floor and basic life support manoeuvres were immediately started. The return of spontaneous circulation (ROSC) occurred after 10 minutes of resuscitation and at the arrival of the Hospital Emergency Team (HET) the woman was still unconscious and in respiratory arrest, with presence of carotid pulses and sinusal rhythm on the monitor. The intensivist of the HET proceeded to tracheal intubation, started to ventilate the patient, placed a neck collar and began the administration of cold fluids to induce therapeutic hypothermia. After a few minutes the patient started to breath spontaneously and was then transported to the Emergency Intensive Care Unit (ICU) of the same hospital. Hemodynamic vital sign were in normal range and subsequently, before admission to ICU, was a computed tomograohy (CT) scan of the head, neck and chest with and without contrast medium was performed. The scan excluded cervical spinal cord brain and skeletal lesions. At the admission in the ICU the patient was unconscious with a Glasgow Coma Scale (GCS) of 3/15 without cardiovascular or respiratory derangement but with metabolic acidosis (bpm: 80, arterial blood pressure: 128/82 mmHg without any vasoactive drug support; SatO2 0.98; timpanic temperature 35.2°C; arterial lactate 6.2 mmol/L)). Pupils were isochoric with a normal response to light. A central venous line was placed. An echocardiogram was performed showing an increased septum (13 mm), and mild mitral regurgitation. In order to continue patient neuroprotection, therapeutic hypothermia was continued with a cooling endovascular system (Alsius®, Zoll Medical Corporation) and a target temperature of 33°C was reached within one hour after ICU admission and mantained for 24 hours. Propofol 80 mg/h and atracurium besylate 80 mg/h were used in continuous perfusion for neuroprotection and the maintenance of a stable hypothermia.
After 6 hours from the time of admission in the ICU, according to the internal protocol, an electroencephalogram (EEG) and somatosensory evocated potential (SEPs) were performed, even if in the presence of sedation and hypothermia. EEG documented an almost isoelectric pattern and SEPs showed a reduced amplitude in n20 - n25 potential on the right hemisphere, while the intermediate amplitude potentials were not present.
After 24 hours hypothermia was discontinued and passive rewarming was started while the pharmacological treatment of neuroprotection with propofol was kept for 72 hours.
A second EEG was performed two days after the admission, recording a continuous spiking associated with facial myoclonus when sedation with propofol was stopped. For this reason, treatment with leviteracetam 1000 mg once a day was started, without a complete resolution of the facial myoclonic crisis.
On the fifth day a percutaneous tracheostomy was performed for the prediction of prolonged coma, and sedation with propofol was stopped, while the antiepileptic therapy with diazepam (6 mg/day) and leviteracetam (on the basis of plasma level) was continued. Mechanical ventilation was stopped on the sixth day. On the seventh day head MRI was done, showing a signal alteration in basal nucleus and on fronto-parietal-occipital cortex bilaterally due to anoxic ischemia.
On the eleventh day an 18F-FDG PET scan was performed, showing a global decrease of brain metabolism. Semi-quantitative measurements of brain glucose metabolism were performed, using white matter metabolic consumption as reference value. The reduction of supratentorial glucose metabolism was homogeneous, at an average of 64% of the normal range, but more accentuated in cortical regions as compared to basal ganglia and thalamus. Cerebellar and brainstem metabolism was substantially preserved, being more than 80% of the normal range.
The twelfth day the patient was discharged to high dependency unit with a GCS of 6 ( E4 - V1t - M1) in the absence of myoclonus and after 23 days the patient was transferred to neuro-rehabilitation long-term care centre with a Glasgow Outcome Scale (GOS) of 2/5 with tracheostomy and percutaneous gastrostomy.
The patient was enrolled in our program of post-intensive care follow-up. After 6 months her GCS was 6 and Glasgow Outcome Scale was 2. After 12 month, the GCS was unchanged, showing a value of 6, characterized by a minimally conscious state.
After 12 months, the patient underwent a second 18F-FDG PET scan; the follow-up examination showed persistence of the global reduction of brain metabolism, involving in particular cortical regions and thalamic nuclei, with metabolic values at 61% of the normal range. Differences in the metabolic pattern were observed, with greater impairment of metabolic activity in precuneus and posterior cingulate cortex, motor, somatosensory and visual cortices (All images are showed in figure 1).
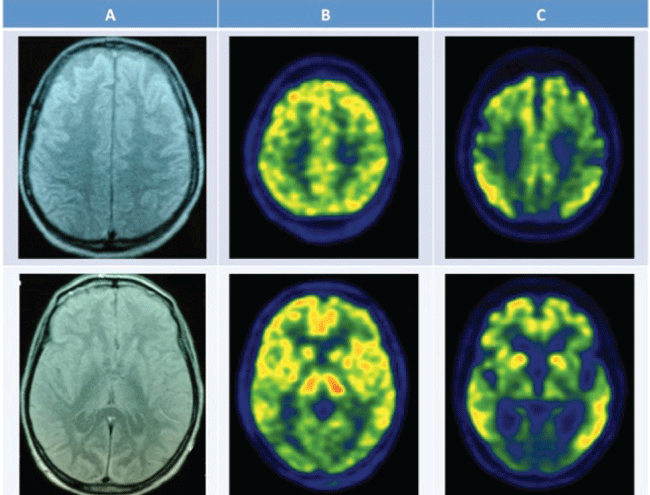
.
Figure 1: Neuroimaging results. A) head MRI (7th day), showing signal alterations due to anoxic ischemia in basal nuclei and fronto-parietal cortex; B) 18F-FDG PET scan (11th day), demonstrating global decrease of brain metabolism, in particular in cortical areas; C) follow-up 18F-FDG PET scan (1 year), showing global reduction of cerebral metabolism, with greater impairment of metabolic activity in precuneus and posterior cingulate cortex, motor, somatosensory and visual cortices.
View Figure 1
As compared to the 18F-FDG PET scan performed on day 11 during the ICU stay, the 1-year follow-up examination showed metabolic decrease involving thalamus, precuneus/posterior cingulate, motor, somatosensory, and visual cortices (respectively 20%, 15%, 13%, 16%, and 15% reduction). Metabolic values in striata, in frontal, parietal, temporal and occipital associative regions showed only a slight decrease as compared to the previous examination, remaining below the normal limits. Besides, metabolic activity in cerebellum and brainstem remained stable over time, at levels greater than 80% of the normal range.
Near fatal hanging results in respiratory, cardiac, psychiatric, metabolic and neurological sequelae. Several studies have reported specific brain lesions detected by CT head scan and MRI [1,4,5]. This lesions are located in areas with large metabolic requirements but poor vascularity, like basal ganglia and thalamus, due to their position in boundary zones of perfusion, where hypoxia can develop easily [6,7]. While PET has been already used to study the brain and other organs metabolism changes after various diseases [8,9], to our knowledge at present no brain 18F-FDG PET study has been performed after hanging. In our patient brain CT examination failed to show any pathological sign, while MRI and 18F-FDG PET proved to be more sensitive in the detection of post-anoxic alterations. Both MRI and 18F-FDG PET demonstrated diffuse alterations involving the entire encephalon: MRI detected macroscopically-visible structural anoxic damage in basal ganglia and cortex, while 18F-FDG PET highlighted underlying functional impairment of cerebral activity following the anoxic insult. Day-11 18F-FDG PET findings of relative sparing of metabolic activity in cerebellum and brainstem are consistent with several lines of evidence, both with international literature results and with hanging neurophysiopathologic mechanism [2,5,10,11]. Firstly, as showed in previous 18F-FDG PET and MRI studies [2,5,10,11] cerebellum and brainstem demonstrated relative sparing of metabolic activity in post anoxic encephalopathy by all causes, due to less sensitivity of these tissues to hypoxia. Besides, lower metabolic impairment of cerebellum and brainstem in hanging is supported by mechanism and timing, in this specific type of injury. In this case of hanging the neck vessels compression involved only carotid arteries, causing hypoperfusion in supra tentorial brain regions. In these areas hypoxic injuries develop earlier than in other regions, where the anoxic damage start with hypoxia due to cardiac arrest.
18F-FDG PET results of a global metabolic reduction at 60% of the normal range were consistent with previous studies obtained in coma patients. In particular, 18F-FDG PET examination showed a metabolic impairment encompassing precuneus, posterior cingulate cortex, medial frontal, parieto-temporal associative cortices, suggesting poor prognosis, which was confirmed by the follow-up GOS score of 2.
The 1-year 18F-FDG PET scan confirmed the diffuse impairment of brain metabolic activity, which was correlated with the persistent clinical impairment. As compared to baseline examination, follow-up 18F-FDG PET demonstrated slight global metabolic reduction in both cortical and subcortical regions, suggesting a continuation of the post-anoxic damage even after the subacute phase. Indeed, the anoxic insult triggers a series of pathological events [12] (environmental changes, lack of metabolic and trophic supply and altered gene transcription) which keep damaging the brain for several weeks. Interestingly, in 18F-FDG PET follow-up examination the metabolic reduction was more significant in thalamus, precuneus/posterior cingulate, primary sensory-motor cortices. This finding suggests further impairment of cortical networks involved in the awareness of the self and the environment [13], which is correlated with the persistent clinical impairment of the patient and underline the importance of follow - up to correlate clinical and nuclear imaging outcome.
One year follow-up results confirmed that reduced SEPs and epileptic EEG pattern are good outcome indicators in hypoxic - ischaemic coma, as indicate in other research [14-16].
The lack of one year follow up SEPs and MRI are limits of this study. Moreover, the design of the study as a case report, and therefore retrospective, do not allow to suggest strong evidence and lack the power of prospective study. If the results will be confirmed in a larger patient population 18F-FDG PET could be included in post anoxic patient evaluation protocols for sub-acute and follow-up examinations.
Conclusion
The present case report of post-anoxic encephalopathy after hanging suicide showed the high sensitivity of 18F-FDG PET brain imaging in detecting post-anoxic functional impairment in addition to MRI brain imaging identifying the structural damage. 18F-FDG PET examination showed early functional impairment, suggesting poor prognosis, associated with persistent reduction of metabolic activity after 1 year.
Written informed consent was obtained from the next of kin of the patient for the publication of this care report and of any accompanying images. A copy of the written consent is available for review for the Editor-in-Chief of this journal.
Authors Contribution
Marco Ciapetti, MD, proposed the study and collaborated in data collection.
Andrea Cecchi, MD, collaborated in follow-up data collection and in writing the manuscript.
Marco Mangini, MD, collaborated in data collection and in writing the manuscript.
Valentina Berti, MD, collaborated in nuclear medicine data collection and in writing the manuscript.
Matteo Bartolini, MD, collaborated in data collection and data analysis.
Alberto Pupi, MD, propose the study, collaborate in data collection and review the manuscript.
Adriano Peris, MD, propose the study, collaborate in data collection and review the manuscript.
References
-
Brancatelli G, Sparacia G, Midiri M, D'Antonio V, Sarno C, et al. (2000) Brain damage in hanging: a new CT finding. Neuroradiology 42: 209-210.
-
Singhal AB, Topcuoglu MA, Koroshetz WJ (2002) Diffussion MRI in three types of anoxic encephalopathy. J Neurol Sci 196: 37-40.
-
Bianco F, Floris R (1987) Computed tomography abnormalities in hanging. Neuroradiology 29: 297-298.
-
Ohkawa S, Yamadori A (1993) CT in hanging. Neuroradiology 35: 591.
-
Matsuyama T, Okuchi K, Seki T, Higuchi T, Ito S, et al. (2006) Magnetic resonance images in hanging. Resuscitation 69: 343-345.
-
McHugh TP, Stout M (1983) Near-hanging injury. Ann Emerg Med 12: 774-776.
-
Nemoto EM (1978) Pathogenesis of cerebral ischemia-anoxia. Crit Care Med 6: 203-214.
-
Li WL, Fu C, Xuan A, Shi DP, Gao YJ, et al. (2015) Preliminary study of brain glucose metabolism changes in patients with lung cancer of different histological types. Chin Med J (Engl) 128: 301-304.
-
Malhotra S, Fernandez SF, Fallavollita JA, Canty JM Jr (2015) Prognostic Significance of Imaging Myocardial Sympathetic Innervation. Curr Cardiol Rep 17: 62.
-
Tommasino C, Grana C, Lucignani G, Torri G, Fazio F (1995) Regional cerebral metabolism of glucose in comatose and vegetative state patients. J Neurosurg Anesthesiol 7: 109-116.
-
Laureys S, Owen AM, Schiff ND (2004) Brain function in coma, vegetative state, and related disorders. Lancet Neurol 3: 537-546.
-
Kermer P, Klocker N, Bahr M (1999) Neuronal death after brain injury. Models, mechanisms, and therapeutic strategies in vivo. Cell Tissue Res 298: 383-395.
-
Laureys S, Schiff ND (2012) Coma and consciousness: paradigms (re)framed by neuroimaging. Neuroimage 61: 478-491.
-
Amantini A, Amadori A, Fossi S (2008) Evoked potentials in the ICU. Eur J Anaesthesiol Suppl 42: 196-202.
-
Robinson LR, Micklesen PJ, Tirschwell DL, Lew HL (2003) Predictive value of somatosensory evoked potentials for awakening from coma. Crit Care Med 31: 960-967.
-
Fahn S (1979) Posthypoxic action myoclonus: review of the literature and report of two new cases with response to valproate and estrogen. Adv Neurol 26: 49-84.





