Clinical Medical
Reviews and Case Reports
Prognostic Significance of Tumor Regression in Locally Advanced Rectal Cancer after Preoperative Radiochemotherapy
Potisek M1, Velenik V2 and Omejc M1*
1University Medical Centre, Slovenia
2Institute of Oncology, Slovenia
*Corresponding author:
Omejc M, University Medical Centre, Zaloska 7, 1000 Ljubljana, Slovenia, E-mail: mirko.omejc@mf.uni-lj.si
Clin Med Rev Case Rep, CMRCR-2-054, (Volume 2, Issue 9), Research Article; ISSN: 2378-3656
Received: June 30, 2015 | Accepted: September 03, 2015 | Published: September 06, 2015
Citation: Potisek M, Velenik V, Omejc M (2015) Prognostic Significance of Tumor Regression in Locally Advanced Rectal Cancer after Preoperative Radiochemotherapy. Clin Med Rev Case Rep 2:054. 10.23937/2378-3656/1410054
Copyright: © 2015 Potisek M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Pathologic response of tumor to therapy is an important prognostic factor in predicting long-term survival of patients with locally advanced rectal cancer. Pathologic complete response indicates excellent surival. The majority of rectal cancers are discovered in locally advanced forms (UICC stage II, III), which require agressive treatment. Usually it consists of preoperative radiochemotherapy, followed by surgery 6-8 weeks later and finally by postoperative chemotherapy. After preoperative radiochemotherapy complete pathologic response of tumor (no tumor cells in rectal wall or perirectal lymph nodes in resected specimen) occurs in 7-30%.
Aim: We wanted to determine the effect of tumor regression on long-term survival of patients treated with preoperative radiochemotherapy.
Methods: Patients with rectal cancer stage II or III, treated surgically at Clinical department of abdominal surgery, University Medical Centre Ljubljana, between 2006 and 2010, were included in this retrospective study. Clinical and pathohistologic data were acquired from computer databases and information about survival from Cancer Registry. Survival was estimated according to Kaplan-Meier method. Significance of prognostic factors was evaluated in univariate analysis, comparison was carried out with log-rank test. The multivariate analysis was performed according to the Cox regression model; statistically significant variables from univariate analysis were included.
Results: 202 patients met inclusion criteria. Median follow-up was 53,2 months. Pooperative stage ypT0N0 (pathologic complete response, pCR) was observed in 14,8% of patients. Pooperative stage has statistically significant impact on survival (p = 0,001). 5-year survival in patients with pCR was >90%. Pooperative T and N status were also found to be statistically significant (p = 0,011 for ypT and p < 0,001 for ypN). According to multivariate analysis, tumor response to neoadjuvant therapy was the only independent prognostic factor (p = 0,003).
Conclusions: Pathologic response of tumor to preoperative radiochemotherapy is an important prognostic factor for prediction of long-term survival of patients with locally advanced rectal cancer. Achieving pCR was proved to be a sign of excellent prognosis, as 5-year survival of these patients exceeded 90%.
Keywords
Rectal cancer, Tumor regression, Preoperative radiochemotherapy, Prognosis
Introduction
Colorectal cancer is one of the most important oncological issues worldwide. It is estimated that in 2012 1,4 millions of colorectal cancer patients were newly discovered, which accounts for 9,7% of newly diagnosed cancers. Almost 700 000 people died because of colorectal cancer in 2012, that is 8,5% of cancer-related deaths. Rectal cancer represents approximately one third of colorectal cancer [1]. The main problem with rectal cancer is that it is usually discovered in locally advanced form (UICC stage II, III), which is why it mostly requires agressive treatment. Standard approach to patient with locally advanced rectal cancer consists of neoadjuvant radiotherapy with or without chemotherapy, followed by surgery (usually total mesorectal excision) and adjuvant chemotherapy [2-4].
It is well known that pathologic tumor response to therapy is an important prognostic factor for long-term prognosis. Moreover, patients with complete pathologic response to neoadjuvant treatment have much better prognosis than patients with less or no response.
The aim of this study was to find out if tumor regression affected long-term survival in patients with localy advanced rectal cancer, treated with neoadjuvant radiochemotherapy.
Materials and Methods
This retrospective research included patients with locally advanced rectal cancer (stage II, III), treated in Clinical department of abdominal surgery, University Medical Centre Ljubljana between 2006 and 2010. Totally 501 patients with rectal carcinoma were treated: 287 only surgically (therefore they were excluded from the study) and 214 with preoperative radiotherapy or radiochemotherapy. After analysing available medical documentation and considering exclusion criteria, 202 patients from the second group were selected for the research.
Exclusion criteria were following
Stage I or IV at diagnosis; noninvasive tumors, tumors in situ, inoperable tumors (no resection was performed, only colo- or ileostomy was created), nonradical resection (R1, R2), reoperation because of tumor relapse.
Relevant patients' data were
Age, genre, type of operation, survival, preoperative stage established by MRI (cTNM), type of neoadjuvant therapy and pathohistological findings. The latter allowed for a classification of the anatomical extent of the disease according to the Seventh edition of the UICC TNM classifiation [5]. Histopathological regression grade of the primary tumor after neoadjuvant radiochemotherapy, was assessed according to Dworak regression scale [6]: grade 0 - no regression, grade 1 - dominant tumor mass with obvious fibrosis and/or vasculopathy, grade 2 - few tumor cells or groups and dominantly fibrotic changes, grade 3 - very few tumor cells in fibrotic tissue with or without mucous substance and grade 4 - no tumor cells, only fibrotic mass. Grade 4 means pathological complete regression.
Statistics
In the research, survival of patients, treated with neoadjuvant therapy and surgery was observed. Some of the patients received radiotherapy only, others received radiochemotherapy, but they were not considered as two different groups as our point of interest was overall survival. We know that addition of chemotherapy to radiation lowers chance of local recurrence but it does not affect overall survival [7,8]. As the beginning of the follow up we considered the date of operation. Survival data were provided by Cancer registry. Kaplan-Meier method was used to analyse survival. Significance of prognostic factors was evaluated with univariate analysis an log-rank test. Statistically sigificant variables from univariate analysis were used in multivariate analysis; with Cox regression model independent variables with effect on long-term survival of rectal cancer patients were pointed out.
All statistical analyses were carried out with statistical program SPSS 19.0.0 (SPSS Inc, Chicago, USA). A p value < 0,05 was considered statistically significant.
Results
202 rectal cancer patients were included in the research. 114 (56.4%) were male and 88 (43.6%) female. The median age was 62.5 years (range 33-86). Median follow up was 53.2 months (range 29-88). According to preoperative diagnostics (physical examination, laboratory tests, chest radiography, ultrasound of abdomen and MRI of pelvis) TNM stage was established. 38 patients (18.5%) had stage II and 164 (81.5%) stage III of the disease.They all received neoadjuvant treatment: long-course radiotherapy (radiation of totally 50,4 - 54 Gy) and most of them additional chemotherapy (5-fluorouracil or capecitabine). 6-8 weeks after finishing preoperative treatment all patients underwent TME surgery. 152 (75%) patients had low anterior resection, of which 2 were without creating anastomosis (Hartmann resection) and 1 was laparoscopic. 52( 25%) patients underwnet abdominoperineal excision. 168 (83%) patients received postoperative 5-FU based chemotherapy. The rest 17% of patients did not receive adjuvant therapy because of postoperative complications, preexisting comorbidities or favourable patohistological results.
Pathohistological findings of resected specimens revealed
31 patients (15.3%) with complete tumour response in rectal wall (ypT0). Other results were: ypT1 in 13 patients (6%), ypT2 in 46 (23%), ypT3 in 104 (52%) and ypT4 in 7 patients (4%).
Lymph nodes in resected specimens
In 133 patients (66%) no tumor cells were found in them (ypN0) and in the 69 patients (34%), the lymph nodes were positive.
For 103 patients (51%) information about pathological response according to Dworak grade were also described. Grade 4 or complete pathological regression was observed in 12 patient (5.9%). Grade 3 was observed in 15 patients (14.3%), grade 2 in 37 (17.3%), grade 1 in 35 (18.3%), while no regression was observed in 4 patients (2%).
After neoadjuvant therapy, TNM stage was reassessed. 30 patients (14.8%) achieved final stage 0 (ypT0N0), which means complete pathologic response to preoperative treatment. Other tumors responded as follows: pooperative stage I was achieved in 45 patients (22.3%), stage II in 52 (25.8%), stage III in 63 (31.2%) and stage IV in 12 patients (5.9%).
Analysing closely the group of patients with complete pathological response (ypT0N0), 17 of them (57%) had preoperatively stage II disease and 13 (43%) stage III. Preoperative stage T was following: cT2 in 6 patients (20%), cT3 23 (77%) and cT4 1 patient (3%). Lymh nodes were preoperatively negative in 17 patients (57%) and cN1 was established in 13 (43%). In none of the patients with pathological complete response cN2 was detected preoperatively.
Survival analysis
The Kaplan-Meier method was used for analysis of survival. Significance of prognostic factors was evaluated with univariate analysis and statistical significance was tested with log-rank test. Results are gathered in table 1 and shown in figure 1.
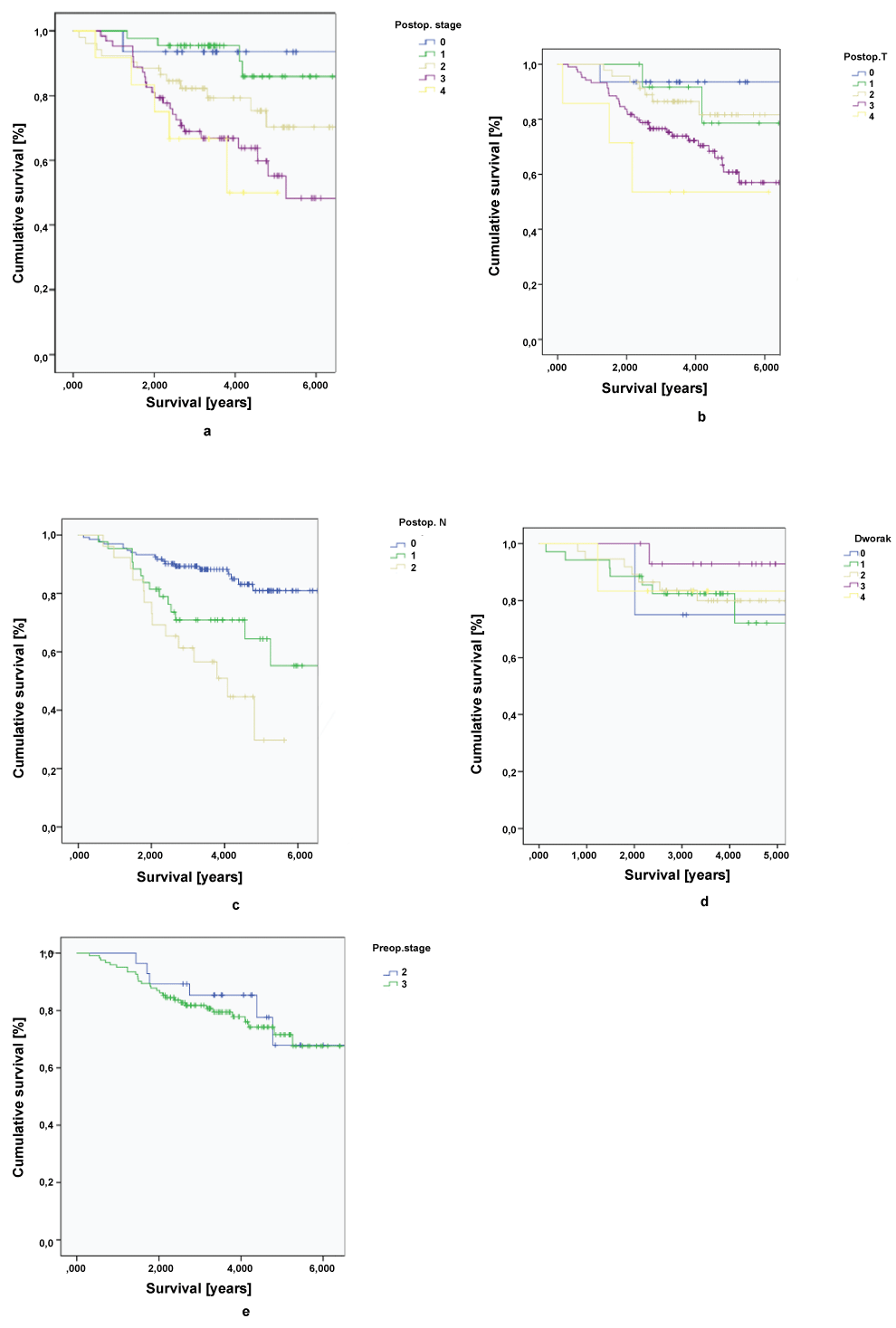
.
Figure 1: Survival according to: a - pooperative stage, b - pooperative T (ypT), c - pooperative N (ypN), d - Dworak regression grade, e - preoperative stage.
View Figure 1
![]()
Table 1: Results of survival analysis
View Table 1
The results show that patients with complete pathological response (ypT0N0) have excellent prognosis, as 5-year survival rate exceeds 90% (72% in postoperative stage II ad 57% in postoperative stage III). Statistically significant are also differences in survival according to preoperative T stage (p = 0.011) and preoperative N stage (p < 0.001). If tumor cells are found in resected specimens, it means worse prognosis, as 5-year survival rate falls from 80 % in ypN0 to 65% in ypN1 and only 30% in ypN2.
There are no statistically significant differences in survival according to Dworak tumor regression grade and preoperative stage (p = 0.513 and 0.389, respectivelly).
Cox regression - multivariate analysis
According to univariate analysis, statistically important variables are pooperative stage and pooperative T and N. We used proportional hazards model or the Cox regression to check, if any of aforementioned variables, including response to preoperative therapy (considered as postoperative downstaging), act as independent prognostic factors in predicting survival in patients after neoadjuvant therapy. The results are shown in table 2. ypT, ypN and postoperative stage do not act as independent variables. The only statistically significant independent prognostic factor is the response to neoadjuvant therapy (p < 0.003).
![]()
Table 2: Results of multivariate analysis
View Table 2
Figure 2 shows differences in survival according to response to neoadjuvant therapy in group of patients with preoperative stage II, compared to group of patients with preoperative stage III. To sum up, survival is statistically significantly better if patients respond to neoadjuvant therapy.
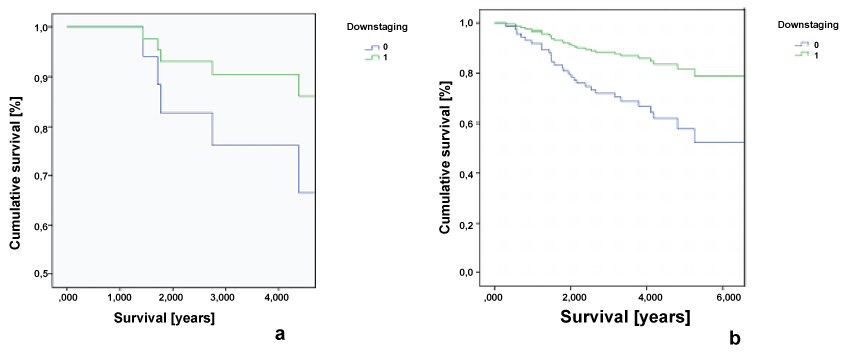
.
Figures 2: Survival according to response to neoadjuvant therapy (0: no response, 1: response): a - group of patients with preoperative stage II, b - group of patients with preoperative stage III.
View Figures 2
Discussion
So far, various researches tried to answer the question, whether pathologic response to preoperative therapy means better prognosis in terms of survival or not. They mostly comprised smaller groups of patients (100, rarely up to 170) and median follow up was mostly 30-40 months. In our research, we wanted to confirm results of foreign studies on a larger group of patients with longer follow up and evaluate their clinical implications. Our research group consisted of 202 patients and median follow up was 53 months, which is why we consider our results relevant.
Complete pathologic response (pCR), which means stage ypT0N0 or in other words no tumor cells in resected surgical specimen, can be detected in 7-24% of patients with locally advanced rectal cancer, treated with neoadjuvant therapy [9-26]. Some newer researches report up to 30% of pCR [27]. Our results are comparable with those studies, as we detected 14.8% of pCR. Using statistical analysis, we found out that pCR means excellent prognosis, as 5-year survival rate turned out to be >90% (p = 0.001). Similar results can be found in the literature: meta analysis of 12 larger researches worldwide reports 90.2% 5-year survival rate in pCR patients (p = 0.0001) [27]; similar percentage (90% or more) is mentioned in various other studies [12,25,26,28,29], while others failed to prove relation between pCR and better survival [15]. In the literature, strong evidence exists that patients with pCR have very few local recurrences (2-5% in 5 years) and that there are statistically significant differences, if groups of patients with pCR are compared to those who failed to respond to preoperative treatment [12,26,28]. It is important to state that some researches detected no local recurrences at all in groups of pCR patients [15,15]. Nevertheless, regardless of no local recurrences, chance of distant metastases still exists. Primary tumor can completely respond to neoadjuvant therapy, but the problem are distant micrometastatic focuses, which can stay undetected in the time of primary diagnostics. They can respond to neoadjuvant therapy or not, in the latter case they remain the source of tumor cells even after successful neoadjuvant treatment at the site of primary tumor [27].
According to our research, pT, pN and postoperative stage all importantly affect survival. Lower pT, no tumor cells in resected lymph nodes and lower postoperative stage mean better prognosis (p = 0.011; < 0.001 and 0.001 for pT, pN and postoperative stage, respectivelly). Nevertheless, none of mentioned variables proved to be statistically significant in multivariate analysis. The only prognostic factor, which acts as independend variable, was response to neoadjuvant therapy, in other words downstaging (p = 0.003). Many researches so far tried to figure out what happens to patients that respond to neoadjuvant therapy, but the response is not complete. This type of response is called partial pathologic response. As in our case, they demonstrated better prognosis in group of patients with partial response compared to group of patients with no response [16,25,30]. Literature also reports effects of nodal status on survival and local recurrence. Tumor deposits in local lymph nodes almost invariably mean worse prognosis. In those cases, postoperative chemotherapy is of undoubtable benefit [30].
An interesting finding is that by approximately 17% of patients with ypT0 tumor cells in perirectal lymph nodes can still be found. These patients act similar as group of patients with no response to neoadjuvant therapy [15,30].
It is important to put stress on the fact that evaluating tumor response to neoadjuvant radiochemotherapy only on the basis of downstaging can be misleading. Tumor can decrease in size significantlly (for example from preoperative T3 to postoperative T2), but there may be no evident tumor regression, which means considerable mass of tumor cells in macroscopically small tumor. On the other hand, despite of no downsizing after neoadjuvant therapy, there may be good regression and very few or no tumor cells are found in the resected surgical specimen. According to these findings, researchers propose to include regression grade (on cellular level) as prognostic factor [22,31].
Our research however failed to prove regression grade an important prognostic factor (p = 0.513), which is in contrast with most of foreign studies. The reason could be that at the beginning of the research (in 2006) in Slovenia regression grade was not evaluated and between 2007 and 2008 it was evaluated inconsistently. Only after 2008 it became mandatory part of pathologic report. Consequently we have information about Dworak regression grade for only 51% of patients, which is why they are not reliable.
Nevertheless, comparisson with other studies may be difficult from another reason. Namely, there is no uniform way of determining regression grade. Some of the studies used Mandard's recommendations [19,23,29,31-33], others Dworak's [21,22] and some used RCRG (»Rectal cancer regression grade«) classification [20]. In all of those classifications, one of the grades means complete regression without detectable tumor cells and one no regression, while definitons of intermediate grades differ. In spite of this inconsistency most researches proved that good response to chemoradiotherapy (complete or partial tumor regression) acts as independent prognostic factor and usually means less locoregional recurrences and longer overall survival [19-21,23,29,31-33]. Polish researchers pointed out that complete pathological regression can act as prognostic factor, but in case of any residual tumor regression grade has no independent effect on overall survival because of probability of tumor focuses in regional lymph nodes [24]. French trial failed to prove improved survival in a group of patients with complete and almost complete tumor regression compared to the group of patients with no regression [34].
According to our and some other studies it is evident that in the population there are patients who respond very well to preoperative treatment or even achieve complete pathological response, which means excellent prognosis. It prompted researches to search possible predictive factors for good response to radiochemotherapy. Korean trial showed correlation between pCR and 3 factors: preoperatively low serum CEA ( < 5ng/ml), sufficient negative circumferential margin and no microscopic ulcerations (ulceration means inflammation, tissue hypoxia and worse oxygenation, which causes tumor to respond poorly to radiochemotherapy) [18]. Canadian research proved interval between the end of neadjuvant treatment and operation to be the only independent prognostic factor for pCR. According to their results interval, longer than 8 weeks, means bigger chances for complete response [17]. American trial confirmed the importance of larger negative circumferential margin and low serum CEA ( < 2,5 ng/ml) and moreover, it was proven that smaller distance to anocutaneus margin enchances ocurrence of pCR. This research failed to prove the importance of preoperative tumor stage, eventhough the authors stated that it could prove important if uniform measuring methods were introduced [35].
French research emphasized the importance of vascular tumor invasion as negative predictive factor for pCR [34]. Next, researchers proved the correlation between good response to neadjuvant chemoradiotherapy and lower 18F-FDG standardised uptake values (»SUV«) on 18F-FDG PET. Therefore they suggested that 18F-FDG PET could be used to distinguish the patients with no response to neoadjuvant therapy and those, in whom good response may be more probable [36].
There remains the question why achieving pCR means good prognosis. The answer might be simple: pCR is achieved in tumors, which themselves have a favourable biological profile with lesser susceptibility to local recurrences or distant metastases. It off course means better survival rates [28]. Various trials tried to find possible biological markers for pCR. Spanish researchers found out that expression of Epidermal Growth Factor Receptor (EGFR) implicates worse response to preoperative therapy. Moreover, searching for patients with expression of this receptor could be useful, if new chemotherapeutics-monoclonal antibodies against EGFR were developed [37]. Worse response to neoadjuvant terapy can be expected in patients with enzyme cyclooxygenase-2 overexpression and low levels of spontaneous apoptosis in pretreatment biopsies. Researchers proposed treatment with selective cyclooxygenase-2 inhibitors in these patients, which could improve pathological response [38]. Another research group investigated whether gene expression profiling could contribute to selection of patients that respond well to neoadjuvant chemotherapy. They established that level of expression of certain 54 genes can offer relativelly reliable prediction of non-responders to treatment [39].
Considering data aboud excellent prognosis in patients with pCR, a question about most appropriate therapy appeared. Could neoadjuvant radiochemotherapy without surgery suffice or might less extensive operation, for example transanal local excision be a better option for them [3,28,40]? There are many reasons against TME: it is a mutilating procedure with significant mortality and many long-term consequences (fecal incontinence, urinary and sexual dysfunction). But on the other hand, without surgery we cannot reliably assess pCR as accuracy of other methods for response to preoperative treatment is small [41].
Is there any possibility to assess preoperatively, whether patients responded to treatment completely and all tumor cells were destroyed? One of the options is analysis of prognostic factors and biological markers of pCR as mentioned in previous paragraph, but unfortunately, this is not a routine clinical practice. Brasilian research group first defined a possible solution to this question: complete clinical response (cCR), which led them to furhter treatment decisions. cCR stands for a list of clinical and endoscopic characteristics: whitening of rectal wall mucosa, telangiectasias within mucosa, scars in rectal wall, seen as light stiffness of the wall during the insuflation. If an ulceration, palpable node or stenosis are found during examination, it means incomplete clinical response. Two different terms are used: initial cCR, which is assessed immediately after neoadjuvant therapy, and sustained cCR, when cCR is mantained for 10 weeks-12 months after completing chemoradiotherapy. Whenever incomplete cCR is detected, transanal excission may be performed. The problem of this approach is that we do not know anything about nodal status. Namely, in lymph nodes residual tumor cells may still be present [42]. Brasilian researchers were the first to introduce so-called "wait-and-see" approach in selected group of patients. Those patients were not operated, yet were closely followed. Regular control examinations were performed once monthly in the first year, once in two months in the second year, once in three months in the third year, once in a half year in the fourth year and once per every following year. They consisted of clinical examination, rigid proctoscopy, biopsies and measurements of serum CEA levels. In this trial only 99 patients with sustained cCR were included. 5-year overall survival was 92,7% and 5-year disease free survival 85%, which is comparable with results in operated patients. According to the results of exsistent trial they concluded that "wait-and-see" is safe and successfull method, but only in carefully selected patients with low-rectal carcinoma and good response to neadjuvant therapy [41].
In addition to Brasilian trial there exist several other trials, which studied this type of approach to rectal cancer patients. Dutch research group defined cCR on the basis of MRI and endoscopy as follows: on MRI no residual tumor is detected or only fibrosis is present; there are no suspicious lymh nodes; endoscopically there can be no residual tumor seen; biopsy must be negative; if in the beginning tumor is palpable at the digitorectal examination, it should be undetectable at the same examination after neoadjuvant therapy. Their testing group numbered 21 patients: oncological outcome was comparable to the outcome in operated patients, 2-year survival was 100%, local recurrence was detected in 2%. Moreover, unoperated patients had significantly less functional complications. Researchers put stress on the importance of assessing nodal status after neoadjuvant therapy when making a decision whether certain patient is appropriate for "wait-and-see" approach. They used MRI to assess nodal status, which was not the case in Brasilian trial. Consequently the latter included more patients with undetected residual tumor cells in lymph nodes. It might be the reason why oncological outcome in brasilian trial is worse than in the Dutch one [43]. Other trials did not present such good results of "wait-and-see" approach, in fact they noted significantly more local recurrences (23-83%) while long term survival could be compared to long term survival in operated patients.
One must point out the limitations of current researches: they are small, retrospective studies with short follow-up, therefore more extensive trial should be carried out in the future [44]. The most appropriate would be a prospective randomised clinical trial to compare »wait-and-see« approach to standard neoadjuvant radiochemotherapy with total mesorectal excision of rectal cancer. However, random patient assignment to either of research groups could be questionable [45]. At the end, let us consider American retrospective trial, which assessed the percentage of patients with preoperatively determined cCR that actually achieved pCR, determined postoperatively. Only a fourth of cCR patients achieved also pCR, which reminds us again, how important is careful selection of patients, suitable for nonoperative treatment [46].
Another approach to rectal cancer patients should be mentioned. Various researches were carried out to compare effectiveness of local excision to effectivenes of total mesorectal excision in smaller tumors (T3) that responded well to preoperative radiochemotherapy. The results show that local recurrences were encountered more frequently in local excision group, but overall survival was comparable. Local excision proved to be a good treatment option with lesser probability of worse functional outcome in smaller tumors with good response to neodjuvant therapy, but only when strict criteria were considered. It must be stated that existent studies are small and with short follow up, which requires caution when interpreting results [47,48].
As it was mentioned before, our research allowed us to demonstrate that patients with good response to preoperative radiochemotherapy have better prognosis and less recurrences or distant metastases. For them, benefits of neoadjuvant therapy are indisputable. Existent research should be a basis for further researches, with which predictive factors of good or poor respose to radiochemotherapy in a population of patients with locally advanced rectal cancer could be defined.
At last, another issue should be mentioned. In a population there are always patients with poor or no response to neodjuvant therapy. Do they benefit from radiochemotherapy at all? It is proven that preoperative radiochemotherapy generally (except for patients with pCR) does not improve overall survival. It certainly diminishes possibility of local recurrences, but the main cause of death in rectal cancer patients are usually distant metastases, which can not always be prevented by neoadjuvant therapy [49,50]. Many studies show that high quality of radical total mesorectal excisions overweights multimodal treatment. The question remains whether chemotherapeutics and radiation are really so vital for rectal cancer patients. The fact is that with quality radical mesorectal excision all tumor tissue and lymph nodes are removed [51]. There is no doubt that TME is mutilating procedure which causes many functional disabilities, but on the other hand radiochemotherapy also has its side effects. One of them are long-term effects because of nerve and vascular damage in perirectal area, which means worsening of anorectal function. It can be much worse after radiochemotherapy than after TME alone [52]. We do not have enough surveys on posttreatment life quality, which is why we cannot compare both treatment approaches objectively. In the future this type of surveys would be mandatory to define most appropriate approach with best oncological and functional outcome in patients, who respond to treatment poorly or do not respond at all.
References
-
Ferlay J, Soerjomataram I, Ervik M (2014) GLOBOCAN 2012, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 11. Lyon, France: International Agency for Research on Cancer.
-
Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, et al. (2010) Colorectal cancer. Lancet 375: 1030-1047.
-
Trakarnsanga A, Ithimakin S, Weiser MR (2012) Treatment of locally advanced rectal cancer: controversies and questions. World J Gastroenterol 18: 5521-5532.
-
Dipetrillo T1, Pricolo V, Lagares-Garcia J, Vrees M, Klipfel A, et al. (2012) Neoadjuvant bevacizumab, oxaliplatin, 5-fluorouracil, and radiation for rectal cancer. Int J Radiat Oncol Biol Phys 82: 124-129.
-
Sobin LH, Gospodarowicz MK, Wittekind C (2009) TNM classification of malignant tumours 7th edition. New York: John Wiley & Sons.
-
Dworak O, Keilholz L, Hoffmann A (1997) Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 12: 19-23.
-
Engels B, Gevaert T, Sermeus A, De Ridder M (2012) Current status of intensified neo-adjuvant systemic therapy in locally advanced rectal cancer. Front Oncol 2: 47.
-
Bosset JF, Collette L, Calais G, Mineur L, Maingon P, et al. (2006) Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med 355: 1114-1123.
-
Sauer R, Becker H, Hohenberger W, Rödel C, Wittekind C, et al. (2004) Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 351: 1731-1740.
-
Duldulao MP, Lee W, Le M, Wiatrek R, Nelson RA, et al. (2011) Surgical complications and pathologic complete response after neoadjuvant chemoradiation in locally advanced rectal cancer. Am Surg 77: 1281-1285.
-
Peeters KC, Van de Velde CJH, Leer JW, Junggeburt JM, Kranenbarg EK, et al. (2005) Late side effects of short-course preoperative radiotherapy combined with total mesorectal excision for rectal cancer: increased bowel dysfunction in irradiated patients - a Dutch colorectal cancer group study. J Clin Oncol 23: 6199-6206.
-
Guillem JG, Chessin DB, Cohen AM, Shia J, Mazumdar M, et al. (2005) Long-term oncologic outcome following preoperative combined modality therapy and total mesorectal excision of locally advanced rectal cancer. Ann Surg 241: 829-36.
-
Kaminsky-Forrett MC, Conroy T, Luporsi E, Peiffert D, Lapeyre M, et al. (1998) Prognostic implications of downstaging following preoperative radiation therapy for operable T3-T4 rectal cancer. Int J Radiat Oncol Biol Phys 42: 935-941.
-
García-Aguilar J, Hernandez de Anda E, Sirivongs P, Lee SH, Madoff RD, et al. (2003) A pathologic complete response to preoperative chemoradiation is associated with lower local recurrence and improved survival in rectal cancer patients treated by mesorectal excision. Dis Colon Rectum 46: 298-304.
-
Hughes R, Glynne-Jones R, Grainger J, Richman P, Makris A, et al. (2006) Can pathological complete response in the primary tumour following pre-operative pelvic chemoradiotherapy for T3-T4 rectal cancer predict for sterilisation of pelvic lymph nodes, a low risk of local recurrence and the appropriateness of local excision? Int J Colorectal Dis 21: 11-17.
-
Huebner M, Wolff BG, Smyrk TC, Aakre J, Larson DW (2012) Partial pathologic response and nodal status as most significant prognostic factors for advanced rectal cancer treated with preoperative chemoradiotherapy. World J Surg 36: 675-683.
-
Kalady MF, de Campos-Lobato LF, Stocchi L, Geisler DP, Dietz D, et al. (2009) Predictive factors of pathologic complete response after neoadjuvant chemoradiation for rectal cancer. Ann Surg 250: 582-589.
-
Huh JW, Kim HR, Kim YJ (2013) Clinical prediction of pathological complete response after preoperative chemoradiotherapy for rectal cancer. Dis Colon Rectum 56: 698-703.
-
Suárez J, Vera R, Balén E, Gómez M, Arias F, et al. (2008) Pathological response assessed by Mandard grade is a better prognostic factor than down staging for disease-free survival after preoperative radiochemotherapy for advanced rectal cancer. Colorectal Dis 10: 563-568.
-
Wheeler JMD, Dodds E, Warren BF, Cunningham C, George BD, et al. (2004) Preoperative chemoradiotherapy and total mesorectal excision surgery for locally advanced rectal cancer: correlation with rectal cancer regression grade. Dis Colon Rectum 47: 2025-2031.
-
Hermanek P, Merkel S, Hohenberger W (2013) Prognosis of rectal carcinoma after multimodal treatment: ypTNM classification and tumor regression grading are essential. Anticancer Res 33: 559-566.
-
Losi L, Luppi G, Gavioli M, Iachetta F, Bertolini F, et al. (2006) Prognostic value of Dworak grade of regression (GR) in patients with rectal carcinoma treated with preoperative radiochemotherapy. Int J Colorectal Dis 21: 645-651.
-
Santos MD, Silva C, Rocha A, Matos E, Nogueira C, et al. (2013) Tumor regression grades: can they influence rectal cancer therapy decision tree? Int J Surg Oncol 2013: 572149.
-
Bujko K, Kolodziejczyk M, Nasierowska-Guttmejer A, Michalski W, Kepka L, et al. (2010) Tumour regression grading in patients with residual rectal cancer after preoperative chemoradiation. Radiother Oncol 95: 298-302.
-
Theodoropoulos G, Wise WE, Padmanabhan A, Kerner BA, Taylor CW, et al. (2002) T-level downstaging and complete pathologic response after preoperative chemoradiation for advanced rectal cancer result in decreased recurrence and improved disease-free survival. Dis Colon Rectum 45: 895-903.
-
Kuo LJ, Liu MC, Jian JJ, Horng CF, Cheng TI, et al. (2007) Is final TNM staging a predictor for survival in locally advanced rectal cancer after preoperative chemoradiation therapy? Ann Surg Oncol 14: 2766-2772.
-
Martin ST, Heneghan HM, Winter DC (2012) Systematic review and meta-analysis of outcomes following pathological complete response to neoadjuvant chemoradiotherapy for rectal cancer. Br J Surg 99: 918-928.
-
Maas M, Nelemans PJ, Valentini V, Das P, Rödel C, et al. (2010) Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol 11: 835-844.
-
Dhadda AS, Bessell EM, Scholefield J, Dickinson P, Zaitoun AM (2014) Mandard tumour regression grade, perineural invasion, circumferential resection margin and post-chemoradiation nodal status strongly predict outcome in locally advanced rectal cancer treated with preoperative chemoradiotherapy. Clin Oncol 26: 197-202
-
Lee YC, Hsieh CC, Chuang JP (2013) Prognostic significance of partial tumor regression after preoperative chemoradiotherapy for rectal cancer: a meta-analysis. Dis Colon Rectum 56: 1093-1101.
-
Dhadda AS, Dickinson P, Zaitoun AM, Gandhi N, Bessell EM (2011) Prognostic importance of Mandard tumour regression grade following pre-operative chemo/radiotherapy for locally advanced rectal cancer. Eur J Cancer 47: 1138-1145.
-
Santos MD, Silva C, Rocha A, Matos E2, Nogueira C, et al. (2014) Prognostic value of mandard and dworak tumor regression grading in rectal cancer: study of a single tertiary center. ISRN Surg 2014: 310542.
-
Bouzourene H, Bosman FT, Seelentag W, Matter M, Coucke P (2002) Importance of tumor regression assessment in predicting the outcome in patients with locally advanced rectal carcinoma who are treated with preoperative radiotherapy. Cancer 94: 1121-1130.
-
Rullier A, Laurent C, Capdepont M, Vendrely V, Bioulac-Sage P, et al. (2010) Impact of tumor response on survival after radiochemotherapy in locally advanced rectal carcinoma. Am J Surg Pathol 34: 562-568.
-
Das P, Skibber JM, Rodriguez-Bigas MA, Feig BW, Chang GJ, et al. (2007) Predictors of tumor response and downstaging in patients who receive preoperative chemoradiation for rectal cancer. Cancer 109: 1750-1755.
-
Cascini GL, Avallone A, Delrio P, Guida C, Tatangelo F, et al. (2006) 18F-FDG PET is an early predictor of pathologic tumor response to preoperative radiochemotherapy in locally advanced rectal cancer. J Nucl Med 47: 1241-1248.
-
Jordi Giralta, Manuel de, las Herasb, Laura Cerezoc, Aranzazu Erasoa, et al. (2005) The expression of epidermal growth factor receptor results in a worse prognosis for patients with rectal cancer patients treated with preoperative radiotherapy: a multicenter, retrospective analysis. Radiother Oncol 74: 101-118.
-
Smith FM, Reynolds JV, Kay EW, Crotty P, Murphy JO, et al. (2006) COX-2 overexpression in pretreatment biopsies predicts response of rectal cancers to neoadjuvant radiochemotherapy. Int J Radiat Oncol Biol Phys 64: 466-472.
-
Ghadimi BM, Grade M, Difilippantonio MJ, Varma S, Simon R, et al. (2005) Effectiveness of gene expression profiling for response prediction of rectal adenocarcinomas to preoperative chemoradiotherapy. J Clin Oncol 23: 1826-1838.
-
Kim NK, Kim MS, Al-Asari SF (2012) Update and debate issues in surgical treatment of middle and low rectal cancer. J Korean Soc Coloproctol 28: 230-240.
-
Habr-Gama A, Perez RO, Proscurshim I, Campos FG, Nadalin W, et al. (2006) Patterns of failure and survival for nonoperative treatment of stage c0 distal rectal cancer following neoadjuvant chemoradiation therapy. J Gastrointest Surg 10: 1319-1328.
-
Habr-Gama A, Perez RO, Wynn G, Marks J, Kessler H, et al. (2010) Complete clinical response after neoadjuvant chemoradiation therapy for distal rectal cancer: characterisation of clinical and endoscopic findings for standardization. Dis Colon Rectum 53: 1692-1698.
-
Maas M, Beets-Tan RG, Lambregts DM, Lammering G, Nelemans PJ, et al. (2011) Wait-and-see policy for clinical complete responders after chemoradiation for rectal cancer. J Clin Oncol 29: 4633-4640.
-
Glynne-Jones R, Hughes R (2010) Critical appraisal of "wait and see" approach in rectal cancer for clinical complete responders after chemoradiation. Br J Surg 99: 897-909.
-
Smith JD, Ruby JA, Goodman KA, Saltz LB, Guillem JG, et al. (2012) Nonoperative management of rectal cancer with complete clinical response after neoadjuvant therapy. Ann Surg 256: 965-972.
-
Hiotis SP, Weber SM, Cohen AM, Minsky BD, Paty PB, et al. (2002) Assessing the predictive value of clinical complete response to neoadjuvant therapy for rectal cancer: an analysis of 488 patients. J Am Coll Surg 194: 131-135.
-
Callender GG, Das P, Rodriguez-Bigas MA, Skibber JM, Crane CH, et al. (2010) Local excision after preoperative chemoradiation results in an equivalent outcome to total mesorectal excision in selected patients with T3 rectal cancer. Ann Surg Oncol 17: 441-447.
-
Pucciarelli S, De Paoli A, Guerrieri M, La Torre G, Maretto I, et al. (2013) Local excision after preoperative chemoradiotherapy for rectal cancer: results of a multicenter phase II clinical trial. Dis Colon Rectum 56: 1349-1356.
-
Voelter V (2009) Preoperative chemoradiotherapy allows for local control in rectal cancer - but distant metastases remain an unsolved problem 4:389-411.
-
Peeters KC, Marijnen CA, Nagtegaal ID, Kranenbarg EK, Putter H, et al. (2007) The TME trial after a median follow-up of 6 years: increased local control but no survival benefit in irradiated patients with resectable rectal carcinoma. Ann Surg 246: 693-701.
-
Chang KH, Smith MJ, McAnena OJ, Aprjanto AS, Dowdall JF (2012) Increased use of multidisciplinary treatment modalities adds little to the outcome of rectal cancer treated by optimal total mesorectal excision. Int J Colorectal Dis 27: 1275-1283.
-
Loos M, Quentmeier P, Schuster T, Nitsche U, Gertler R, et al. (2013) Effect of preoperative radio(chemo)therapy on long-term functional outcome in rectal cancer patients: a systematic review and meta-analysis. Ann Surg Oncol 20: 1816-1828.





