Clinical Medical
Reviews and Case Reports
Gastrointestinal Stromal Tumor near Gastroesophageal Junction with Concomitant Hiatal Herni
Amir Aryaie*, Yuxiang Wen and Leena Khaitan
Department of surgery, University Hospitals Case Medical Center, Cleveland, OH, USA
*Corresponding author: Amir Aryaie, Department of surgery, University Hospitals Case Medical Center, Cleveland, OH, USA, E-mail: Amir.Aryaie@UHhospitals.org
Clin Med Rev Case Rep, CMRCR-2-069, (Volume 2, Issue 11), Case Series; ISSN: 2378-3656
Received: September 02, 2015 | Accepted: November 10, 2015 | Published: November 12, 2015
Citation: Aryaie A, Wen Y, Khaitan L (2015) Gastrointestinal Stromal Tumor near Gastroesophageal Junction with Concomitant Hiatal Hernia. Clin Med Rev Case Rep 2:069. 10.23937/2378-3656/1410069
Copyright: © 2015 Aryaie A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Gastrointestinal stromal tumors, GIST, Esophagogastric junction, Hiatal hernia
Case Summary
The patient is a 71 years old lady who initially presents with significant gastroesophageal reflux, chronic blood loss anemia, and mild dysphagia. Her reflux symptoms have been refractory to optimal medical treatments. She was initially afebrile with unremarkable vital signs, and physical exam was otherwise normal: abdomen was non-tender, non-distended, and no hepatosplenomegaly.
Despite the fact that she denied recent weight gain or weight loss, her hemoglobin and hematocrit levels were 8.6 and 27.1 respectively at initial office visit.
She then underwent upper endoscopy due to ongoing symptoms. Endoscopic evaluation revealed a gastric mass located in the fund us near the gastroesophageal (GE) junction, which appeared localized at the submucosal layer. The tumor was found to be mildly ulcerated with minimal bleeding on the endoscopy (Figure 1). Multiple biopsies were taken, and pathology only revealed normal reactive changes.
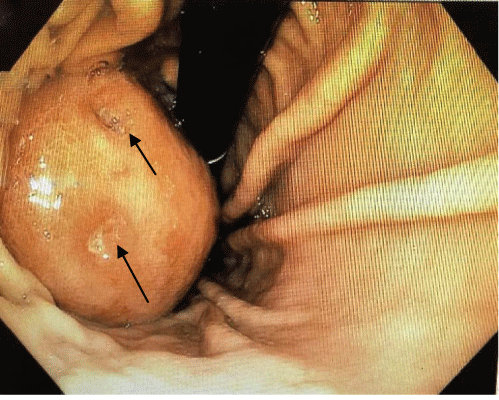
.
Figure 1: Retrograde endoscopic view of submucosal mass with ulcerations at GE junction (Arrow: ulcerations)
View Figure 1
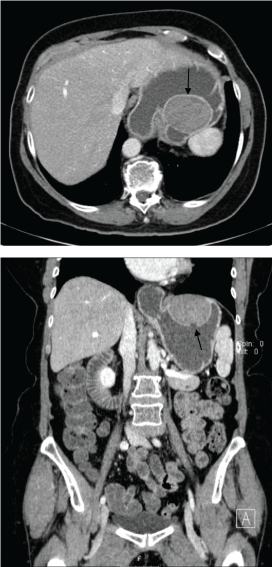
Patient further had a CT enterography of the abdomen and pelvis as a follow-up for this submucosal gastric fundal mass which demonstrate a gastric mass near GE junction measuring 5.2 × 4.6 cm in diameter (Figure 2). Moreover, there were also at least 2 small mucosal ulcers suspected along the caudal margin of the mass and a small sliding-type hiatus hernia.
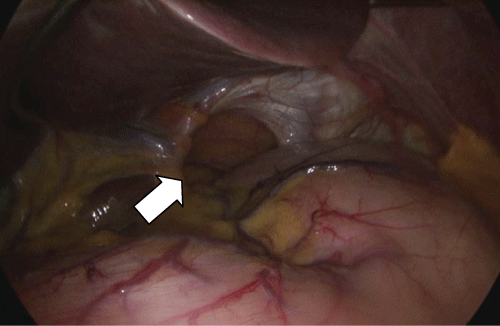
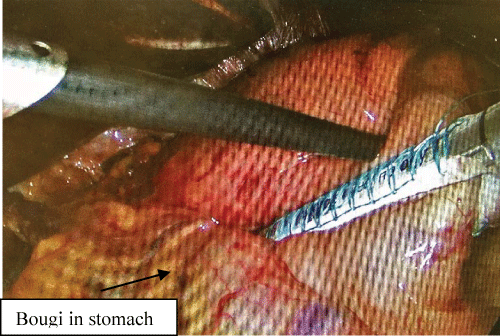
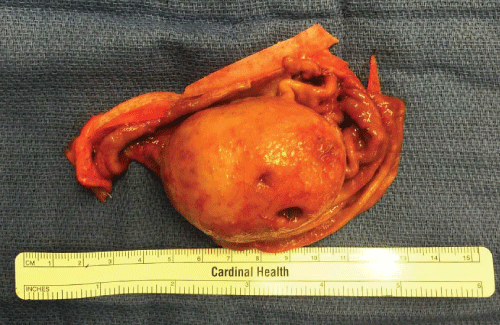
She was scheduled for laparoscopic surgery for simultaneous tumor resection and hiatal hernia repair. After standard trocar placement and placement of a liver retractor a large mass and a moderate size hiatal hernia were identified (Figure 3). No other gross abnormalities were noted. Hiatal hernia was reduced in standard fashion and closed over a 56 F boogie in esophagus. Intraoperative endoscopy was performed to evaluate intraluminal location of this mass which appeared to be near GE junction on greater curvature of stomach. Then, a56 F boogie was re-inserted and the gastric mass was resected hugging the bougie with laparoscopic 60 mm stapler in order to prevent narrowing at the GE junction (Figure 4). The specimen was placed in an EndoCatch bag and removed from the peritoneal cavity. The specimen was opened on the back table and there was at least 1 cm margin all the way around the base (Figure 5).
The specimen was sent for frozen which it was reported as a gastrointestinal stromal tumor. Final pathology reported an 8 cm low grade gastrointestinal stromal tumor with mitotic rate ≤ 5/50. All incisal margins were negative circumferentially by at least 1 cm (Figure 6a and Figure 6b).
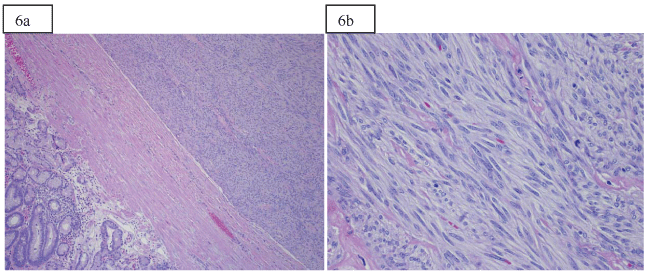
.
Figure 6: a) View of the hematoxylin and eosin stained section of the tumor taken at 100x original magnification showing the tumor in the upper right and the overlying gastric mucosa in the lower left. The tumor is well circumscribed, moderately cellular and composed of spindle cells with moderate amounts of indistinct eosinophilic cytoplasm. Necrosis is not identified.
b) High power shows the hematoxylin and eosin stained section at 400x original magnification. The higher power tumor shows spindled cells with elongated, blunt-ended nuclei and scant to moderate amounts of gray-pink cytoplasm. There is a mitotic figure to the right of the center of the image. Overall, there were up to 4 mitotic figures per 50 high power fields. Immunohistochemistries (not shown) with antibodies directed against KIT (CD117) and DOG1 were reactive with the tumor cells.
View Figure 6
Patient did very well postoperatively. She was reflux free after surgery. While tolerating soft diet and adequate pain control, she was discharged in stable condition.
Discussion
Gastrointestinal stromal tumors (GIST) are uncommon tumors arising in the submucosal layer of anywhere in the gastrointestinal tract (0.1% to 3% of all gastrointestinal malignancies) [1]. Thought to originate from the interstitial cells of Canal, an intestinal pacemaker cell. They are most commonly found in the stomach and small intestines [2-4]. About 10%-30% GISTs are malignant [4]. According to the 2002 classification criteria, the aggressive behavior of GIST is based on 2 parameters: tumor size and mitotic index. Tumors are classified using a ranking system, grouping tumors into very low-, low-, intermediate-, and high-risk categories based on size (< 2 cm, 2-5 cm, 5-10 cm, and > 10 cm) and on number of mitoses within 50 high-power fields (HPFs); such measurements typically being reported as less than 5, 5 to 10, or greater than 10 [5-7].
GIST at GE junction is extremely rare and the treatment options are still in debate [6]. Wedge resection, intra-gastric tumor resection, and combined endoscopic and laparoscopic resection have all been previously described [8-15]. Considering oncologic safety; the GIST Consensus Conference had defined in 2004 that the primary goal for surgical resection is to achieve negative margins [16]. Further, the 2007 NCCN guideline established a set of criteria which also noted negative microscopic margins being the key objective in surgical management [17].
In this case, we have shared our experience to resect a GIST at GE junction with laparoscopic wedge resection and assist of a bougi to prevent stenosis and obtain adequate margin. The laparoscopic approach allows full-thickness resection of the tumor within the gastric wall and achieving negative margins with minimally invasive method [17,18]. It is safe and effective, with no intra- and post-operative morbidity.
References
-
Lewis JJ, Brennan MF (1996) Soft tissue sarcomas. Curr Probl Surg 33: 817-872.
-
Pidhorecky I, Cheney RT, Kraybill WG, Gibbs JF (2000) Gastrointestinal stromal tumors: current diagnosis, biologic behavior, and management. Ann Surg Oncol 7: 705-712.
-
Miettinen M, Majidi M, Lasota J (2002) Pathology and diagnostic criteria of gastrointestinal stromal tumors (GISTs): a review. Eur J Cancer 38 Suppl 5: S39-51.
-
Greenson JK (2003) Gastrointestinal stromal tumors and other mesenchymal lesions of the gut. Mod Pathol 16: 366-375.
-
Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, et al. (2002) Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol 33: 459-465
-
Hueman MT, Schulick RD (2008) Management of gastrointestinal stromal tumors. Surg Clin North Am 88: 599-614.
-
Coccolini F, Catena F, Ansaloni L, Lazzareschi D, Pinna AD (2010) Esophagogastric junction gastrointestinal stromal tumor: resection vs enucleation. World J Gastroenterol 16: 4374-4376.
-
Tagaya N, Mikami H, Kogure H, Kubota K, Hosoya Y, et al. (2002) Laparoscopic intragastric stapled resection of gastric submucosal tumors located near the esophagogastric junction. Surg Endosc 16: 177-179.
-
Nguyen SQ, Divino CM, Wang JL, Dikman SH (2006) Laparoscopic management of gastrointestinal stromal tumors. Surg Endosc 20: 713-716.
-
Otani Y, Furukawa T, Yoshida M, Saikawa Y, Wada N, et al. (2006) Operative indication for relatively small (2-5 cm) gastrointestinal stromal tumor of the stomach based on analysis of 60 operated cases. Surgery 139: 484-492.
-
Hiki N, Yamamoto Y, Fukunaga T, Yamaguchi T, Nunobe S, et al. (2008) Laparoscopic and endoscopic cooperative surgery for gastrointestinal stromal tumor dissection. Surg Endosc 22: 1729-1735.
-
Novitsky YW, Kercher KW, Sing RF, Heniford BT (2006) Long-term outcomes of laparoscopic resection of gastric gastrointestinal stromal tumors. Ann Surg 243: 738-745.
-
Nishimura J, Nakajima K, Omori T, Takahashi T, Nishitani A, et al. (2007) Surgical strategy for gastric gastrointestinal stromal tumors: laparoscopic vs. open resection. Surg Endosc 21: 875-878.
-
Matsui H, Nabeshima K, Okamoto Y, Nakamura K, Kondoh Y, et al. (2011) Fundic rotation technique: a useful procedure for laparoscopic exogastric resection of gastric submucosal tumor located on the posterior wall near the esophagogastric junction. Tokai J Exp Med 36: 152-158.
-
Tsujimoto H, Yaguchi Y, Kumano I, Takahata R, Ono S, et al. (2012) Successful gastric submucosal tumor resection using laparoscopic and endoscopic cooperative surgery. World J Surg 36: 327-330.
-
Blay JY, Bonvalot S, Casali P, Choi H, Debiec-Richter M, et al. (2005) Consensus meeting for the management of gastrointestinal stromal tumors. Report of the GIST Consensus Conference of 20-21 March 2004, under the auspices of ESMO. Ann Oncol 16: 566-578.
-
Demetri GD, Benjamin RS, Blanke CD, Blay YJ, Casali P, et al. (2007) NCCN Task Force report: optimal management of patients with gastrointestinal stromal tumor GIST: Update of NCCN clinical practice guideline. J Natl Compr Canc Netw 5: 1-28.
-
De Vogelaere K, Van Loo I, Peters O, Hoorens A, Haentjens P, et al. (2012) Laparoscopic resection of gastric gastrointestinal stromal tumors (GIST) is safe and effective, irrespective of tumor size. Surg Endosc 26: 2339-2345.