Clinical Medical
Reviews and Case Reports
Non-operative Treatment for Extensive Skin Necrosis of a Neonatal Dorsal Foot caused by Extravasation
Hiromu Masuoka1,2*, Susumu Saito1, Yoko Nakamura1 and Shigehiko Suzuki1
1Department of Plastic and Reconstructive Surgery, Graduate School of Medicine, Kyoto University, Japan
2Department of Plastic and Reconstructive Surgery, National Hospital Organization Kyoto Medical Center, Japan
*Corresponding author: Hiromu Masuoka, Department of Plastic and Reconstructive Surgery, National Hospital Organization Kyoto Medical Center,1-1 Fukakusa mukaihata-cho, Fushimi-ku, Kyoto, 612-8555, Japan, Tel: +81-75-641-9161, E-mail: rommasu@kuhp.kyoto-u.ac.jp
Clin Med Rev Case Rep, CMRCR-3-082, (Volume 3, Issue 1), Short Communication; ISSN: 2378-3656
Received: August 28, 2015 | Accepted: January 06, 2016 | Published: January 09, 2016
Citation: Masuoka H, Saito S, Nakamura Y, Suzuki S (2016) Non-operative Treatment for Extensive Skin Necrosis of a Neonatal Dorsal Foot caused by Extravasation. Clin Med Rev Case Rep 3:082. 10.23937/2378-3656/1410082
Copyright: © 2016 Masuoka H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We herein describe two cases of non-surgical treatment for skin necrosis of the dorsum of the neonatal foot caused by extravasation. In both the cases, closure of the defects was achieved after 3 months of conservative treatment without any complications. Although the defects occupied almost all of the dorsal skin of the foot, no significant contracture disabling the toe or ankle occurred during the maximum follow-up period of 9 years. Non-surgical treatments could be an alternative treatment for neonatal skin necrosis caused by extravasation.
Keywords
Extravasation, Skin necrosis, Neonatal intensive care, Conservative therapy, Foot
Introduction
In neonatal care, extravasation is a clinical issue in which the accidental leakage of intravenous infusion fluid into the subcutaneous space causes damage to the skin around the infusion site. Both the skin and the subcutaneous tissues can be severely damaged, especially if cytotoxic drugs such as calcium, potassium or bicarbonate solutions leak out of the vein. Total skin necrosis is occasionally observed in serious cases [1]. If the necrosis is extensive, then immediate debridement followed by skin coverage would be indicated to avoid associated infection or to rescue the underlying organs from tissue dehydration. In addition, physicians usually consider skin resurfacing to be necessary in order to prevent future deformities or functional losses due to subsequent scar contracture [1-3]. Nevertheless, the possibility that surgery will worsen a neonate's general conditions often limits surgical indications.
Respecting the priority of conservative skin care for neonates, we treated two Japanese patients with extensive extravasation-associated skin necrosis of the dorsum of the foot without surgery. In both the cases, we were able to achieve closure of the skin defects that were caused by bedside debridement, after 3 months of conservative treatment and without any tendon exposure or infection. Although the defects occupied almost all of the skin of the dorsum of the foot, no significant contracture occurred during the maximum follow-up period of 9 years. The purpose of this paper is to describe the detailed healing courses of the two cases and to provide a guide for the non-surgical treatment of neonatal cases involving extensive extravasation-associated skin necrosis.
Patients and Methods
Case 1
A 5-day-old female infant (gestational age: 41 weeks, birth weight: 3512 g) was hospitalized in a neonatal intensive care unit due to meconium aspiration syndrome. An intravenous line was placed in the dorsum of the right foot to administer calcium gluconate. Extravasation occurred and resulted in skin necrosis covering almost all of the dorsal skin of the foot to reach the inter digital space distally and the lateral malleolus proximally (Figure 1). Eleven days after the extravasation, the patient was moved to our hospital. Although there was extensive damage to the skin of the dorsum, skin grafting was not performed. Instead, the patient was treated conservatively because the underlying tendons and bones were not exposed. The initial therapies performed were daily rinsing with saline solution, the daily application of gentamicin-containing ointment, and the covering of the affected area with a non-adherent gauze. The necrotic tissues were removed manually. Trafermin agent (Fiblast spray; Kaken Pharmaceutical Co, Ltd, Tokyo, Japan) was also applied topically to the raw area that resulted from the removal of the necrotic tissue. Debridement of almost all of the affected area was completed after 3 weeks. Around Week 5, the superficial layer was covered by fibrotic tissue; however, only partial granulation changes were observed. On day 75 after admission, the patient was discharged to receive home treatment from her mother. Complete wound closure was confirmed at 15 weeks after the extravasation. One month after the wound closure, hypertrophic changes occurred in the scar, especially in the middle of the dorsum of the foot. This hypertrophy showed regression during follow-up examinations, however, we performed no additional treatments (such as moisturization or compression). At one year after the extravasation, the scar was observed to have softened and flattened. At 9 years after the extravasation, the skin of the affected area had regained the physiological texture of the dorsum of the foot (Figure 2). There was no limitation to the range of the ankle joint. Although the skin was slightly tense in comparison to the contralateral foot, toe flexion was not impaired.

.
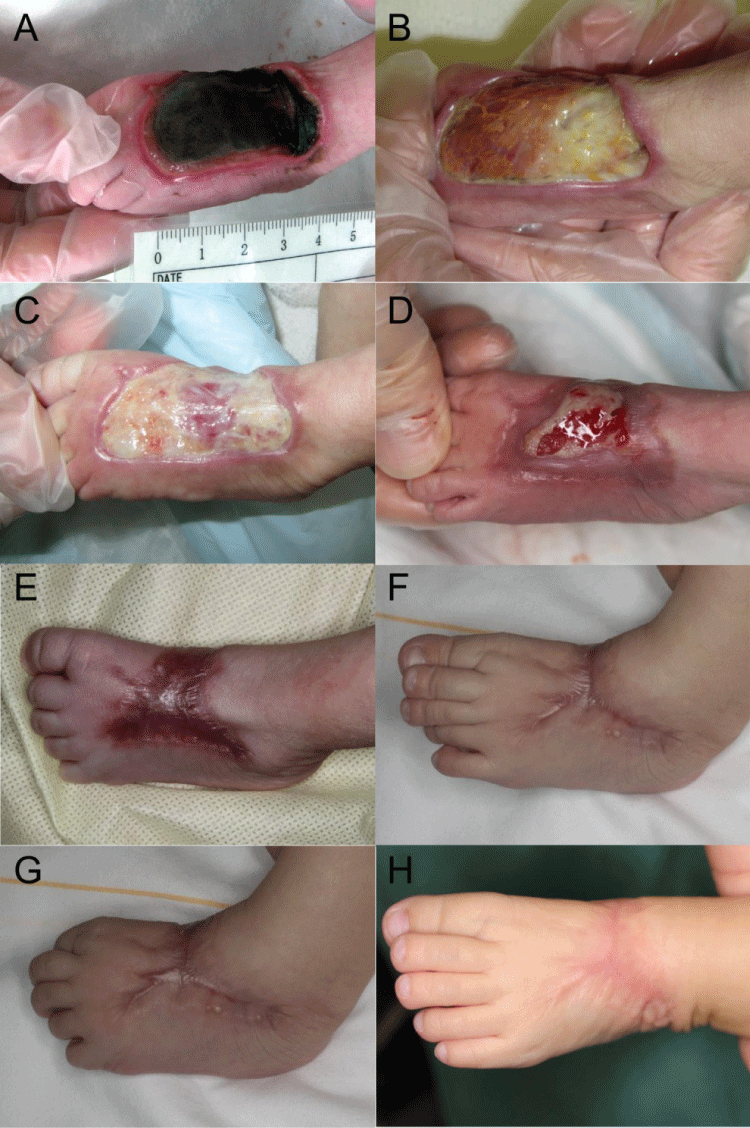
Figure 1: Case1: The wound healing and scar formation process. (A) Full-thickness skin necrosis 9 days after extravasation injury; (B) Week 3; (C) Week 5 Note that there are reddish granulations under the fibrous tissue; (D) Week 10; (E) Week 16; (F, G) The scar was the most hypertrophic at 7months after injury; (H) The scar was flattened and linearized one year after injury.
View Figure 1
.
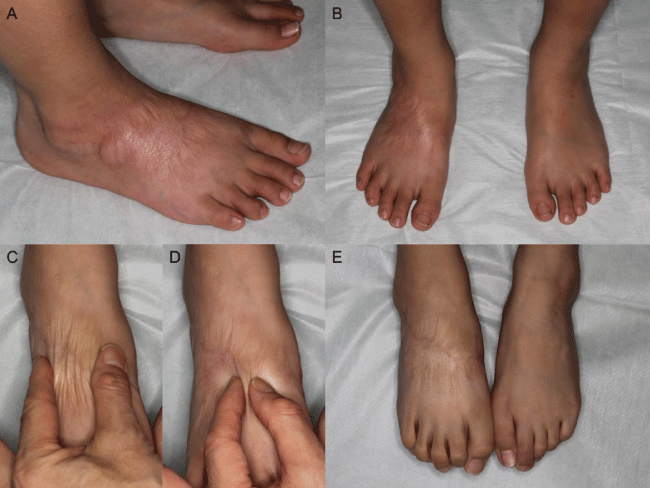
Figure 2: Nine years after injury in Case 1 (A) Oblique view; (B) Frontal view; (C) The scar is wrinkled by a wide pinch; (D) The scar is thinned by a narrow pinch; (E) Toe flexion.
View Figure 2
Case 2
Extravasation occurred during cardiac surgery to repair an atrioventricular septal defect in an18-day-old female infant (gestational age: 40 weeks, birth weight: 3082 g). The infusion fluid contained fentanyl, midazolam, and vecuronium bromide. The patient was referred to us 21 days after the injury. Although almost all of the dorsal skin of the left foot had already necrotized, the underlying tissues were not exposed. Similarly to Case 1, we chose a conservative treatment. The course of wound healing was almost the same as in Case 1 (Figure 3). The wound had completely closed at 14 weeks after the extravasation, leaving slight hypertrophic scarring. As was seen in Case 1, a gradual improvement of hypertrophic scar was observed during the 1-year follow-up examination.
.
Figure 3: Case2: (A) Full-thickness skin necrosis on day 21 after the extravasation injury; (B) Week 4; (C) Week 5; (D) Week 10; (E) Week 15; (F, G) Month 5. No functional disorder occurred, in spite of the distortion of the skin; (H) Nine months after the injury.
View Figure 3
Discussion
Extravasation is an inevitable issue in neonatal care. The rate of extravenous fluid leakage in the routine care of infants has been estimated to range from 23-63% [4]. Wilkins et al. reported that 3.8% of extravasations caused skin necrosis, which mostly occurred during parenteral nutrition infusion for premature neonates of 26 weeks gestation or less [5]. Small areas of skin necrosis can heal smoothly by maintaining a moisturized environment. When skin necrosis is extensive, however, surgical treatments such as skin grafting must be considered in order to avoid infection, shorten the treatment period and prevent subsequent scar contracture.
Skin necrosis on the dorsum of the hand or foot, which are the most common sites for infusion, has a risk of causing tendon exposure. The resection of the tendons may be necessary if infection or desiccation of the exposed tendons occur. To avoid this risk, a nearly debridement followed by the immediate covering of the affected area with a skin graft or flap is indicated, especially when a skin defect is relatively wide. Such surgical treatments, however, may be complicated by donor-site morbidity. Moreover, surgical stresses and anesthesia can become burdens to the health of neonates and worsen their general condition. In both of the above reported cases, the necrotic sites healed successfully within 4 months of the initial injury following conservative treatment, which included daily bedside debridement, followed by the application of wet dressings. As a result of this treatment, the necrotic tissues were gradually replaced by granulation. Several authors have emphasized the importance of moisturization for infant skin care because of the immaturity of the barrier function of infant skin [6,7]. Acknowledging this importance, Moon et al. employed wet dressing in the treatment of their extravasation cases [8].
Trafermin (Fiblast®) is a recombinant human basic fibroblast growth factor (bFGF), a cytokine that is known to stimulate the proliferation of fibroblasts and capillary endothelial cells and to promote angiogenesis during the wound healing process [9,10]. In Japan, it has already been used clinically to treat skin ulcers. We therefore expected bFGF to have a potent effect in extensive skin defects of the dorsal foot. bFGF was administered once a day until wound closure was almost complete.
Interestingly, the time-course of the granulation changes of our two cases was almost the same (week 5). During the replacement process, no exposure or infection of the extensor tendons was encountered. Although the mechanism by which these results were achieved is unclear, these two successful experiences demonstrate that the conservative treatment of extravasation wounds is possible, even when the affected area is relatively large. Even in cases with a high risk of tendon exposure, such as the cases of extensive total skin necrosis that we experienced careful daily observation of the wound should be performed until the completion of granulation as long as the tendons are not exposed.
Even if skin defects can be closed through conservative treatment, a possibility exists that contracture due to wound constriction will affect the functional outcome. In spite of the presence of extensive areas of necrosis (over 80% of the dorsal surface of the foot), the present cases showed no functional complications, which could include limitation in the range of motion of the ankle or toe joint. Such complications are frequently seen in post-burn conditions of the foot. The sequential photos of the healing processes in the contraction of the wound led us to hypothesize that the high extensibility of the skin around the wound could contribute to lessening the risk of the limitation of joint motions. Indeed, the recovered dorsal skin just proximal to the toes showed prominent extension in the most recent photos (Figure 2). In Case 1, there was no contracture requiring surgical treatment in the 9-year follow-up period. If scar contracture were to occur during further body growth, we would have to consider surgical treatments such as scar plasty or skin graft.
Thus far, no reports have so far demonstrated the long-term success of conservative treatment for extensive necrosis of the dorsum of the foot due to extravasation. This case report could therefore provide guidance to help medical staff members to manage cases of skin necrosis after intravenous leakage.
In conclusion, conservative treatment could be an alternative to surgery for treating extensive skin necrosis caused by extravasation in neonates. Meticulous wound care, including rinsing, daily debridement and careful observation could contribute to enhancing the healing potential of the skin of neonates. Further research is needed to establish the best treatment protocol for extravasation injury, including the prevention of extravasation itself, to prevent infection or invasive damage of the underlying tissues and to reduce the incidence of sequelae.
References
-
Gault DT (1993) Extravasation injuries. Br J Plast Surg 46: 91-96.
-
Casanova D, Bardot J, Magalon G (2001) Emergency treatment of accidental infusion leakage in the newborn: report of 14 cases. Br J Plast Surg 54: 396-399.
-
Kumar RJ, Pegg SP, Kimble RM (2001) Management of extravasation injuries. ANZ J Surg 71: 285-289.
-
Franck LS, Hummel D, Connell K, Quinn D, Montgomery J (2001) The safety and efficacy of peripheral intravenous catheters in ill neonates. Neonatal Netw 20: 33-38.
-
Wilkins CE, Emmerson AJ (2004) Extravasation injuries on regional neonatal units. Arch Dis Child Fetal Neonatal Ed 89: F274-275.
-
Afsar FS (2009) Skin care for preterm and term neonates. Clin Exp Dermatol 34: 855-858.
-
Fox MD (2011) Wound care in the neonatal intensive care unit. Neonatal Netw 30: 291-303.
-
Moon HS, Burm JS, Yang WY, Kang SY (2012) Prognosis of Full-Thickness Skin Defects in Premature Infants. Arch plast Surg 39: 463-468.
-
McGee GS, Davidson JM, Buckley A, Sommer A, Woodward SC, et al. (1988) Recombinant basic fibroblast growth factor accelerates wound healing. J Surg Res 45: 145-153.
-
Gospodarowicz D, Ferrara N, Schweigerer L, Neufeld G (1987) Structural characterization and biological functions of fibroblast growth factor. Endocr Rev 8: 95-114.





