Clinical Medical
Reviews and Case Reports
Stevens - Johnson Syndrome and Intestinal Perforation following regorafenib Administration for Metastatic Rectal Cancer: A Case Report
Tetsuhito Muranaka1,2, Yoshito Komatsu2*, Kentaro Sawada1, Hiroshi Nakatsumi2, Yasuyuki Kawamoto2, Satoshi Yuki1, Riichiro Abe3, Kanako C Hatanaka4 and Naoya Sakamoto1
1Department of Gastroenterology and Hepatology, Hokkaido University Hospital, Hokkaido, Japan
2Cancer Chemotherapy, Hokkaido University Hospital, Hokkaido, Japan
3Division of Dermatology, Niigata University Graduate School of Medical and Dental Sciences, Niigata, Japan
4Department of Surgical Pathology, Hokkaido University Hospital, Hokkaido, Japan
*Corresponding author: Yoshito Komatsu, Cancer Chemotherapy, Hokkaido University Hospital Cancer Center, Hokkaido University, Kita-14-Jyou Nishi-5-Choume, Kita-ku, Sapporo-City, Hokkaido, Japan, E-mail: ykomatsu@ac.cyberhome.ne.jp
Clin Med Rev Case Rep, CMRCR-3-088, (Volume 3, Issue 2), Case Report; ISSN: 2378-3656
Received: January 06, 2016 | Accepted: February 06, 2016 | Published: February 09, 2016
Citation: Muranaka T, Komatsu Y, Sawada K, Nakatsumi H, Kawamoto Y, et al. (2016) Stevens - Johnson Syndrome and Intestinal Perforation following regorafenib Administration for Metastatic Rectal Cancer: A Case Report. Clin Med Rev Case Rep 3:088. 10.23937/2378-3656/1410088
Copyright: © 2016 Muranaka T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Regorafenib monotherapy is useful for the treatment of colorectal cancer (CRC), which is refractory to multiple anti-cancer agents. However, it is associated with a high incidence of adverse events such as hand-foot skin reactions, elevated liver enzymes, etc. Although there are few reports of Stevens-Johnson syndrome (SJS) or intestinal perforation associated with regorafenib, such events should be taken into consideration, as with various other anti-cancer therapies, including bevacizumab. Our patient was a 61-year-old Japanese woman who underwent low anterior resection for stage IIIA rectal cancer in July 2012. Following surgery, the patient received S-1 monotherapy for 6 months. In May 2013, metastasis was observed in the peritoneal and lymph nodes. She received both oxaliplatin- and irinotecan-based chemotherapies, including bevacizumab; however, her disease continued to progress. A mutation of KRAS exon 2 precluded anti-EGFR therapy; thus, we administered regorafenib from January 2014. A whole-body, pruritic, skin rush developed on day 12 and continued to worsen. In addition, the mucosae of her palpebral conjunctiva, mouth and anus became reddish and edematous with pain on day 14. She was diagnosed with SJS based on the symptoms and high serum granulysin levels (10 ng/ml); she received steroid therapy from day 14. Severe abdominal pain developed on day 16. Computed tomography revealed free air in the abdominal cavity. Her condition worsened. Because her cancer was advanced and refractory, we could not perform surgery. She received best supportive care and died on day 65. Regorafenib is useful for refractory and metastatic CRC; however, it is associated with a high incidence of adverse events. This is the first report of intestinal perforation and SJS following regorafenib administration. Therefore, regorafenib should be carefully monitored for treatment of CRC.
Keywords
Regorafenib, Stevens-Johnson syndrome, Intestinal perforation, Colorectal cancer, Adverse events
Abbreviations
SJS: Stevens-Johnson Syndrome, CRC: Colorectal Cancer, JCOG: Japan Clinical Oncology Group, Cape-Ox + Bv: Chemotherapy for CRC consisting of oxaliplatin at 130 mg/m2 on day 1, capecitabine at 1000 mg/m2 twice daily on days 1–14, plus bevacizumab 7.5 mg/kg on day 1 every 3 weeks, IRIS + Bv: Chemotherapy for CRC consisting of irinotecan at 100 mg/m2 on days 1 and 15; S-1 at 60 mg per body, twice daily on days 1-14; plus bevacizumab at 5 mg/kg on day 1 every 4 weeks, EGFR: Epidermal Growth Factor Receptor, VEGF: Vascular Endothelial Growth Factor, FDA: The US Food and Drug Administration, ALDEN: Algorithm for Assessment of Causality in SJS and Toxic Epidermal Necrolysis, SCAR: Severe Cutaneous Adverse Reactions.
Introduction
Recent advances in chemotherapy for colorectal cancer (CRC) have contributed to remarkable improvements in the prognosis of this disease. Regorafenib, which is an anti-vascular endothelial growth factor (VEGF) therapy, was approved by the US Food and Drug Administration (FDA) in September 2012 for the treatment of patients with metastatic CRC who were previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy [1,2]. However, regorafenib is associated with a high incidence of adverse events such as hand-foot skin reactions and elevated liver enzymes; thus, it must be administered cautiously. On the other hand, bevacizumab is an anti-VEGF agent that reportedly causes intestinal perforation in approximately 1% of patients [3]. Despite the similar actions of regorafenib and bevacizumab on the VEGF receptor, there are few reports of intestinal perforation or Stevens-Johnson syndrome (SJS) associated with regorafenib use. Here, we report our experience with a patient who developed intestinal perforation and SJS following regorafenib administration for recurrent rectal cancer.
Case Report
Our patient was a 61-year-old Japanese woman who underwent low anterior resection for rectal cancer in July 2012. The pathological diagnosis was adenocarcinoma (tub2 > por2), and it was classified as T4aN1M0 p Stage III based on the criteria of the Union for International Cancer Control (7th edition). After surgery, she received adjuvant chemotherapy with S-1(tegafur, gimeracil and oteracil potassium) for 6 months, according to the findings of the JCOG0910 clinical trial registered as UMIN000003272 conducted by the Japan Clinical Oncology Group (JCOG), which was a randomized study of capecitabine vs. S-1 for stage III CRC as an adjuvant chemotherapy. In May 2013, computed tomography revealed the recurrence of peritoneal and lymph node metastasis. As an initial treatment, the patient received six cycles of Cape-Ox + Bv and two cycles of IRIS + Bv [4,5]. However, these treatment regimens failed and the disease progressed. Because of a mutation in KRAS exon 2, we did not administer anti-EGFR therapy but rather administered regorafenib monotherapy as a tertiary chemotherapy from January 2014. Her Eastern Cooperative Oncology Group performance status was grade 0. Laboratory data are shown in table 1. She had no history of allergy or smoking. Her medication history included esomeprazole (20 mg/day) for gastroesophageal reflux disease and magnesium oxide (500 mg, three times perday) for constipation. On day 12 of the first cycle of regorafenib monotherapy, a pruritic skin rash developed. The skin rash worsened and the mucosae of her palpebral conjunctiva, mouth and anus became reddish and edematous with pain. She presented to our hospital on day 14 with diffuse erythema and a whole-body rash with a small part of skin detachment and bullous lesions (Figure 1). Laboratory results revealed serum granulysin levels > 10 ng/ml and other laboratory data were shown at table 2. She was admitted to our hospital diagnosed of SJS. Steroid pulse therapy with methylprednisolone (1000 mg/day i.v.) was started from day 14. On day 16, the patient developed abdominal pain with symptoms of peritoneal irritation. Computed tomography revealed free air in the abdominal cavity (Figure 2). Although symptoms of the skin and mucous membrane tended to improve and serum granulysin levels decreased to < 10 ng/ml, her condition worsened and her cancer was advanced and refractory, we could not perform surgery. Therefore, she received best supportive care and died on day 65 of the first administration of regorafenib monotherapy.
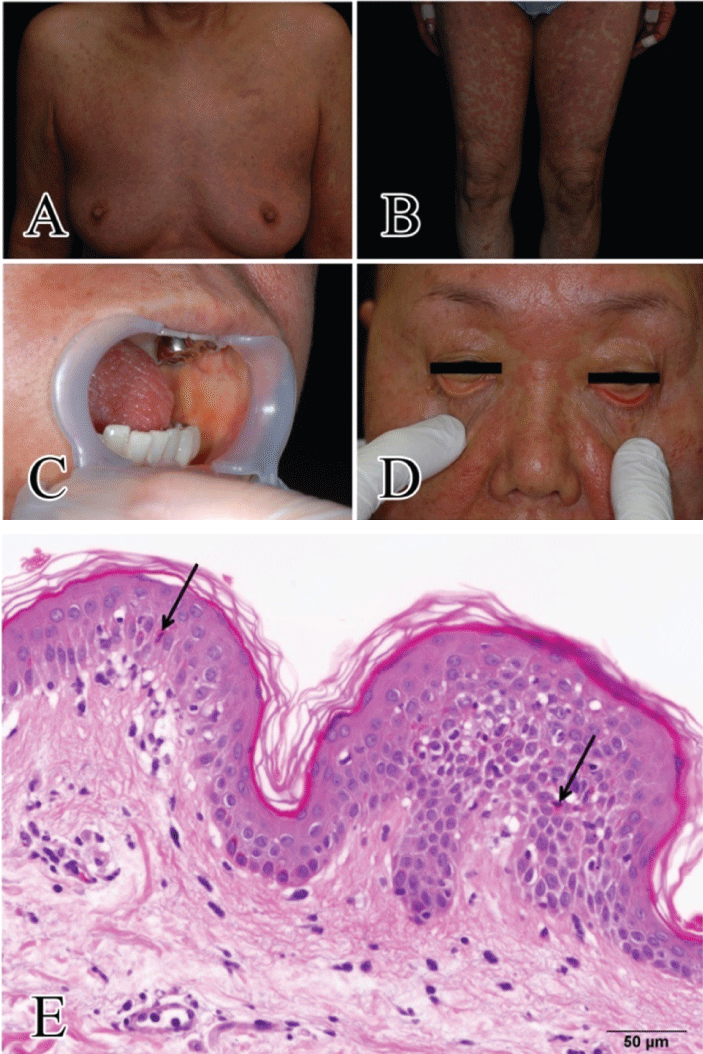
.
Figure 1: Erythema multiforme and mucositis of this patient and histopathological features of this patient. Erythema multiforme seen on the breast (A) and legs (B). Oral and palpebral conjunctiva mucositis (C and D) of this patient. Systemic eruptions and mucositis developed on day 13. Individual necrotic keratinocytes (arrows) and lymphocytic infiltration of the epidermis were observed in a biopsy specimen by erythema of the left arms (E). Hematoxylin and eosin stain; × 400.
View Figure 1
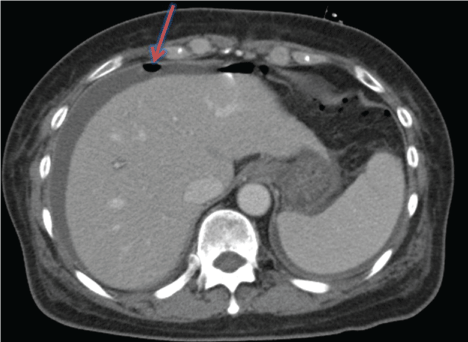
.
Figure 2: Computed tomography on the third hospital day. Free air (arrow) in the abdominal cavity was revealed by computed tomography.
View Figure 2
![]()
Table 1: Laboratory data before regorafenib administration.
View Table 1
![]()
Table 2: Laboratory data on the first day of the hospitalization.
View Table 2
Discussion
Our patient received regorafenib for metastatic rectal cancer. However, she developed intestinal perforation and died. Such an adverse event is rare because only two cases of intestinal perforation using regorafenib have been reported to date [6], but the FDA has indicated an association between regorafenib and gastrointestinal perforations through the action mechanism of regorafenib. However, it should be noted that our patient received corticosteroids before abdominal symptoms precipitated by intestinal perforation. It was reported that corticosteroid use is associated with increased risk of gastrointestinal bleeding and perforation, thus corticosteroid use may have caused intestinal perforation [7]. Intestinal perforation could be due to corticosteroid use, colorectal cancer metastasis and administration of regorafenib. Consequently, it is difficult to define causality with other possible factors being involved. On the other hand, SJS has been widely reported in the literature since the 1940s and is classified as a severe form of erythema multiforme [8,9]. We arrived at a diagnosis of SJS based on the diagnostic criteria of the Ministry of Health, Labour and Welfare in Japan according to symptoms of erythema multiform with fever and involvement of the mucous membranes. There is only one report of SJS arising from regorafenib treatment [10]. The algorithm for assessment of causality in SJS and toxic epidermal necrolysis (ALDEN) score of this patient was three (unlikely). However, this novel drug induced SJS or toxic epidermal necrolysis and thus cannot affect the ALDEN score enough [11]. Her concomitant medications were esomeprazole and magnesium oxide but esomeprazole has been known as a non-significant risk of severe cutaneous adverse reactions (SCAR), and the risk of SCAR after magnesium oxide treatment has not been assessed [12]. Furthermore, in this case, Naranjo score associated with regorafenib and SJS is five (defined as a probable adverse drug reaction) [13]. Serum granulysin is useful for the diagnosis of drug-induced skin reactions occurring in SJS and toxic epidermal necrolysis [14]. Indeed there was still no consensus on treatment of SJS, in the present case, we administered steroid therapy for SJS. Before administration of regorafenib, there was no indication of SJS or intestinal perforation. Both SJS and intestinal perforation are severe adverse events; thus, it is important to identify patients at risk for SJS or intestinal perforation because of the use of anti-VEGF receptor agents. Indeed, although SJS and intestinal perforation are rare adverse events of anti-cancer drugs, they can be lethal. Therefore, the use of regorafenib as an anti-VEGF receptor agent should be carefully monitored.
Acknowledgement
The authors would like to thank this patient, her family, and the staff members of the departments of gastroenterology and hepatology, pathology and dermatology of Hokkaido University hospital. Additionally, they would like to thank Enago (www.enago.jp) for the English language review.
References
-
Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, et al. (2013) Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381: 303-312.
-
Yoshino T, Komatsu Y, Yamada Y, Yamazaki K, Tsuji A, et al. (2015) Randomized phase III trial of regorafenib in metastatic colorectal cancer: analysis of the CORRECT Japanese and non-Japanese subpopulations. Invest New Drug 33: 740-750.
-
Hapani S, Chu D, Wu S (2009) Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol 10: 559-568.
-
Komatsu Y, Yuki S, Sogabe S, Fukushima H, Nakatsumi H, et al. (2012) Phase II study of combined chemotherapy with irinotecan and S-1 (IRIS) plus bevacizumab in patients with inoperable recurrent or advanced colorectal cancer. Acta Oncol 51: 867-872.
-
Kato S, Andoh H, Gamoh M, Yamaguchi T, Murakawa Y, et al. (2012) Safety verification trials of mFolfiri and sequential IRIS + bevacizumab as first- or second-line therapies for metastatic colorectal cancer in Japanese patients. Oncology 83: 101-107.
-
Adenis A, Kotecki N, Decanter G, Clisant S, Penel N (2013) Regorafenib use as a possible cause of intestinal perforation. Acta Oncol 52: 1789-1790.
-
Narum S, Westergren T, Klemp M (2014) Corticosteroids and risk of gastrointestinal bleeding: a systematic review and meta-analysis. BMJ Open 4: e004587.
-
Keil H (1940) Erythema multiforme exudativum (Hebra): A clinical entity associated with systemic features. Ann Intern Med 14: 449-494.
-
Carroll OM, Bryan PA, Robinson RJ (1966) Stevens-Johnson syndrome associated with long-acting sulfonamides. JAMA 195: 691-693.
-
Mihara Y, Yamaguchi K, Nakama T, Nakayama G, Kamei H, et al. (2015) Stevens-Johnson syndrome induced by regorafenib in a patient with progressive recurrent rectal carcinoma. Gan To Kagaku Ryoho 42: 233-236.
-
Sassolas B, Haddad C, Mockenhaupt M, Dunant A, Liss Y, et al. (2010) ALDEN, an algorithm for assessment of drug causality in Stevens-Johnson Syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther 88: 60-68.
-
Mockenhaupt M, Viboud C, Dunant A, Naldi L, Halevy S, et al. (2008) Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. J Invest Dermatol 128: 35-44.
-
Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, et al. (1981) A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 30: 239-245.
-
Abe R, Yoshioka N, Murata J, Fujita Y, Shimizu H (2009) Granulysin as a marker for early diagnosis of the Stevens-Johnson syndrome. Ann Intern Med 151: 514-515.





