Clinical Medical
Reviews and Case Reports
Chemotherapeutic Agents for Brain Metastases in Non-Small Cell Lung Cancer: A Case Report with Eribulin Mesylate and Review of the Literature
Alejandra C Fuentes1, Reordan O De Jesus2 and David N Reisman3*
1Department of Internal Medicine, University of Florida, Gainesville, FL, USA
2Department of Radiology, University of Florida, Gainesville, FL, USA
3Department of Medicine, Division of Hematology/Oncology, University of Florida, Gainesville, FL, USA
*Corresponding author: David N Reisman M.D., PhD, Department of Medicine, Division of Hematology/Oncology, University of Florida, PO Box 100278, Gainesville, FL 32610-0278, USA, Tel: 352-273-7832, E-mail: dnreisman@ufl.edu
Clin Med Rev Case Rep, CMRCR-3-107, (Volume 3, Issue 5), Case Report; ISSN: 2378-3656
Received: March 15, 2016 | Accepted: May 28, 2016 | Published: May 30, 2016
Citation: Fuentes AC, Jesus ROD, Reisman DN (2016) Chemotherapeutic Agents for Brain Metastases in Non-Small Cell Lung Cancer: A Case Report with Eribulin Mesylate and Review of the Literature. Clin Med Rev Case Rep 3:107. 10.23937/2378-3656/1410107
Copyright: © 2016 Fuentes AC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Lung cancer accounts for the majority of cases of brain metastases, resulting in higher morbidity and mortality. Surgery and radiation are the current standard of care for the treatment of brain metastases. However, when brain metastases recur despite these treatments, the management options are limited, especially when recurrent metastatic events occur. The role of systemic chemotherapy for brain metastases remains undefined, with advances in drug delivery and ongoing studies using targeted agents showing promising results. We describe the case of a patient with non-small cell lung cancer who had recurrent brain metastases, which eventually became untreatable due to prior radiation. After using a combination of eribulin mesylate, bevacizumab, and erlotinib, the patient's brain metastases exhibited a remarkable response. This case highlights the potential of combined agents with low profiles of toxicity in achieving a notable intracranial response. It also suggests that the activity of eribulin mesylate, a novel treatment currently being studied in non-small cell lung cancer, should be further studied on brain metastases. We also review the current literature on the treatment of non-small cell lung cancer, focusing on the treatments' reported response rates on brain metastases.
Keywords
Non-small cell, Lung cancer, Brain metastases, Eribulin, Chemotherapy, Systemic therapy
Introduction
Lung cancer is the leading cause of cancer-related deaths, accounting for nearly 27% in the United States and 20% worldwide [1,2]. It also accounts for the majority of cases of brain metastases (30-50%), which are associated with a poor prognosis and a higher morbidity and mortality [3,4]. The reported incidence of brain metastases (BM) is growing due to increased utilization of magnetic resonance imaging (MRI) during the staging work-up, as well as the advances in therapy leading to a prolonged overall survival with enhanced opportunity for dissemination into the central nervous system (CNS), where systemic chemotherapy has had poor penetration.
The standard treatment options for BM include surgical resection or stereotactic radio surgery (SRS) for patients with a limited number of lesions (3-4), and whole brain radiation therapy (WBRT) for multiple lesions. What has not been well defined in the treatment of BM is the role of systemic chemotherapy [5]. Systemic chemotherapy has been found in some evidence-based studies to show no survival benefit in the treatment of BM [5,6].
BM that is recurrent can become a challenge. Radiation therapy to the brain is limited by normal tissue tolerance, the threshold at which significant toxicity from radiation therapy may occur. Severe toxicity is rarely seen due to limitations in radiation dosage, as well as the short overall survival of NSCLC patients, but severe forms of toxicity can include brain tissue necrosis, leukoencephalopathy, and dementia [7-10]. Thus, once a patient with recurrent BM has received the maximum radiation dose, further treatment options for BM are scarce at best. As a result, the scientific community continues to study the barriers affecting the use of chemotherapy for BM.
The blood brain barrier (BBB) has been the main proposed mechanism hindering the entry of systemic chemotherapeutic agents. Understanding BBB physiology has been the target of multiple studies, and novel drugs with augmented BBB penetration are beginning to show promising results. The new generations of targeted molecular drugs have demonstrated some activity in the CNS. These include tyrosine kinase inhibitors such as erlotinib (Tarceva) or monoclonal antibodies such as bevacizumab (Avastin), which have been approved in the US for the treatment of non-small cell lung cancer (NSCLC). Multiple targeted agents are currently under investigation; however, there is no consensus on the best agent or combination of agents for BM.
This article will discuss a patient with NSCLC who had recurrent brain metastases that were not treatable with radiation or surgery. This patient, after triple treatment with erlotinib, bevacizumab, and eribulin mesylate, showed first regression and then prolonged stabilization of brain metastases. While erlotinib and bevacizumab have been studied on BM in NSCLC with modest results, no such studies have been done on eribulin. Eribulin mesylate, a novel agent with a low toxicity profile that inhibits microtubule dynamics, is currently FDA-approved for refractory metastatic breast cancer. Eribulin mesylate has shown promising activity in a phase II trial on patients with advanced NSCLC previously treated with a taxane, with 50% of patients achieving stable disease and a clinical benefit rate (defined as the summation of complete remission, partial remission, and stable disease for at least three months) of 27% [11]. There has also been one reported case with effective BM regression in breast cancer after treatment with eribulin mesylate [12].
We will also review the current systemic and targeted agents and their documented activity on BM, with the aim of further highlighting the need for more clinical trials to help establish a consensus on a chemotherapeutic approach to BM.
Case Presentation
A 58 year-old female with a past medical history of hypertension and tobacco abuse (20 pack-year) was diagnosed with Stage IIIa adenocarcinoma of the lung. She initially presented in January 2013 at another institution with a complaint of mildly progressive shortness of breath and cough. Computed tomography (CT) of the chest showed mediastinal lymphadenopathy. Follow-up positron emission tomography (PET) scan on February 12th showed a fluorodeoxyglucose (FDG)-avid, left-sided level III cervical lymph node (sub-centimeter in size) with a maximum standardized uptake value (SUV) of 3.3 g/mL, as well as a single FDG-avid mediastinal node in the aorto-pulmonary window (1.8 × 1.4 cm) with maximum SUV of 10.2 g/mL. This left cervical node was biopsied and found to be negative. Hence, on March 4th, 2013 she underwent an aortal pulmonic lymph node excision (2.5 × 2 × 1.5 cm). Immuno-peroxidase studies were positive for cytokeratin 7, monoclonal CEA, vimentin, and TTF1, and negative for cytokeratin 20, CDX2, estrogen receptors and mammaglobin, consistent with lung adenocarcinoma stage IIIA. EGFR or ALK mutations were not tested. She received one cycle of cisplatin/etoposide with one week of radiation at the outside institution. Her care was then delayed as she lost her job and insurance and moved to Florida. She presented to our institution in late April 2013, at which time a PET scan showed worsening disease with FDG-avid lymphadenopathy in the anterior mediastinal (1.2 cm, SUV 3.9 g/mL), left hilar (sub-centimeter, SUV 1.9 g/mL), and left cervical (1 cm, SUV 2.2 g/mL) nodes. Brain MRI on May 8th, 2013 showed multiple enhancing lesions, with the largest ones in the left parietal region (1.7 cm), left basal ganglia (1.3 cm), and right cerebellum (1.3 cm), confirming stage IV disease. She was otherwise neurologically intact. On May 15th she underwent WBRT for a total of 30 gray (Gy) at 3 Gy/fraction. On May 31st she began concurrent chemotherapy and radiation, with carboplatin (AUC 2) and paclitaxel (45 mg/m2) weekly for 6 weeks, and a total of 50 Gy (2.5 Gy/fraction) of radiation to the mediastinum. A follow-up PET scan 3 weeks after completion of radiation on August 9th demonstrated treatment response, with decreased mediastinal lymphadenopathy.
An MRI scan of her brain in September 2013 demonstrated overall improvement and response to radiation, with smaller parietal (1.1 cm), basal ganglia (0.9 cm), and cerebellar (0.7 cm) lesions. PET scan on October 30th showed mixed extracranial response without any uptake in the mediastinum but with new FDG-avidity in a level IV left cervical lymph node (max SUV 2.9 g/mL), as well as increased radiotracer uptake in the distal left femur. She had associated pain in her left femur, and received empiric palliative radiation to this area (18 Gy). Brain MRI on December 27th showed a stable parietal lesion (1.1 cm); however, the basal ganglia (1.2 cm) and cerebellar (1.1 cm) lesions had enlarged, and there were multiple new smaller lesions (Figure 1A). Her lesions were still asymptomatic. CT imaging on December 30th also showed progression of disease, with bony metastases in the left 10th rib and left iliac wing.
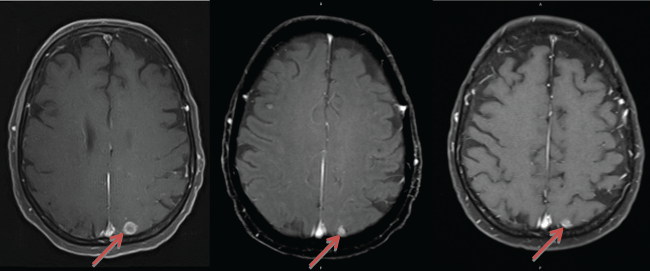
.
Figure 1: Brain MRI images showing regression and stabilization of parietal lesion with targeted chemotherapy. A) Lesion in December 2013, after WBRT (11.4 mm × 10.8 mm). B) Lesion after 3 months of targeted chemotherapy (March 2014), now measuring 6.1 mm × 7.8 mm. C) Continued stabilization and regression of the lesion after 7 months of targeted chemotherapy (July 2014), measuring 5.8 mm × 7.1 mm.
View Figure 1
Given her recurrent brain metastases, which at this point represented the most serious problem facing this patient, we selected drugs that had a reasonable probability to penetrate into the CNS and also to be effective with her lung cancer. Since sequential therapy was unlikely to be feasible with this patient, we elected to use a simultaneous combination of agents. We selected targeted therapies with a low toxicity profile because if these drugs were effective, we did not want to discontinue them due to toxicity, which often occurs with the majority of other lung cancer drugs. Hence, she was treated with a simultaneous combination of three drugs: erlotinib, bevacizumab, and eribulin. While eribulin is not a targeted agent per se, we used it weekly at a lower dose (1.0 mg/m2 every 1-2 weeks for 16 doses) to minimize toxicity. Erlotinib was started at 150 mg PO daily (continued for 9 months) and the dose titrated for development of minimal rash on the cheeks and chest. Bevacizumab was given at 15 mg/kg every 3 weeks for 8 doses.
A MRI brain scan in March 2014 (three months into targeted treatment) showed decreased metastatic burden, including smaller parietal (0.8 cm) (Figure 1B), basal ganglia (1.1 cm), and cerebellar (0.6 cm) lesions. In addition to T1 post-contrast, FLAIR and T2 sequences confirmed the treatment response, reducing the possibility of a pseudo-response, which has been reported with bevacizumab in T1 sequences with other brain tumors. Intracranial disease continued to be controlled without progression or development of new lesions, including up until her last brain MRI in July 2014 (7 months into treatment). At this time, the parietal (0.7 cm) (Figure 1C), basal ganglia (0.8 cm), and cerebellar (0.5 cm) lesions continued to decrease in size. Nonetheless, PET scan showed progression with multiple new FDG-avid bony metastases. She ultimately developed a pathological fracture of her left femoral neck and underwent hemi-arthroplasty in June 2014, which was complicated by a septic left hip joint in October 2014 and resulted in therapy being stopped. She was referred to hospice, and expired in November 2014. She never developed any new neurologic symptoms that would suggest clinical progression of BM.
Discussion and Review of the Literature
Brain metastases are associated with a poor prognosis, with a median survival time measured in months. The management of BM has thus far been largely limited to surgical resection, WBRT, and SRS. Nevertheless, the role of chemotherapy has become increasingly promising, with advances in drug delivery and many new targeted agents that demonstrate better CNS penetration due to their small size and molecular weight. We will discuss the chemotherapeutic agents for NSCLC (both currently approved and under investigation) focusing on their documented efficacy, if any, on BM.
The Blood Brain Barrier and Advances in Drug Delivery
The BBB and its associated cellular junctions, lipophilic properties, and membrane transporters with efflux pumps have been proposed as the mechanisms responsible for limiting the entry of chemotherapeutic agents [13]. Studies have demonstrated that brain metastases are able to physically disrupt the BBB, suggesting that systemic chemotherapy is able to reach intracranial tissues, at least in the macroscopic or relapsed setting [6]. It is likely through these mechanisms that standard chemotherapy has been able to achieve marginal BM response rates (RR). However, perhaps due to alternative mechanisms such as efflux pumps and others that are poorly understood, drug concentrations within the metastatic lesions have been limited, and the RR to systemic chemotherapy have thus far not been considered significant [6,14]. Some of the novel agents (small molecular inhibitors) are smaller in size, which may help explain their markedly higher RR.
Our enhanced understanding of the properties of the BBB and the mechanism of tumor cell invasion into the CNS provides additional insight for advances in drug delivery as well as potential targets for chemotherapy. Numerous modifications in the approach to drug delivery are under current investigation with varying results. These include changes in the route of drug delivery (intra-arterial, intrathecal, intraventricular, intratumoral, intranasal), modification to the drug itself (smaller, more lipophilic), the use of agents to disrupt the BBB, inhibition of drug efflux, receptor-mediated transport (endocytosis and exocytosis of the drug), and nanosystem delivery (liposomal or nanoparticle carriers) [13]. Preclinical studies on platinum drugs, the back-bone of standard chemotherapy for NSCLC, have recently shown enhanced platinum delivery to the CNS via nanoparticles and liposomes [15,16].
Documented Efficacy of Systemic Therapy on Brain Metastases
Standard systemic chemotherapy
Prior to targeted agents, studies on NSCLC BM showed limited efficacy of platinum-based combinations, with BM response rates mostly ranging between 15-30%, and rarely higher, if using triple-agent combinations [17,18]. Nevertheless, these studies demonstrated no effect on survival, and standard chemotherapy has thus not been recommended for treatment of BM [5,6,17]. Moreover, the duration of therapy is an important consideration. While platinum chemotherapy is the standard for first-line therapy in stage 4 lung cancer patients, its toxicity limits its duration to 4-6 cycles. In contrast, the use of weekly chemotherapy agents (at lower doses, such as eribulin mesylate) or targeted therapies with low toxicity profiles allows the use of these drugs for longer periods of time. Even if tumors are not decreased in size, the stabilization of disease would likely be palliative due to the prolongation of life with minimal treatment side effects.
A few of the standard systemic chemotherapy drugs known to cross the BBB that have been studied in NSCLC include temozolamide, topotecan, irinotecan, and pemetrexed. Temozolamide has been extensively studied due to its activity in primary brain tumors; however results in NSCLC BM have been mixed, without significant survival advantage [18]. Topotecan has been demonstrated to have an advantage over WBRT alone (in a study merging both NSCLC and small cell lung cancer patients into a single cohort); however, it also did not affect survival in NSCLC [19]. Irinotecan has been studied in combination with cisplatin and ifosfamide with a BM RR of 50% as well as a mild benefit in overall survival [20]. Pemetrexed, FDA-approved for NSCLC in 2008, has been an agent with more promising CNS activity: Observational studies have demonstrated that pemetrexed (either as single-agent or combined with platinum-based therapy) results in an intracranial RR of 33-82% [21-23].
Tyrosine kinase inhibitors
Tyrosine kinase inhibitors (TKIs) against the epidermal growth factor receptor (EGFR) tyrosine kinase and the anaplastic lymphoma kinase (ALK) are now approved for the treatment of NSCLC cases with these activating mutations. EGFR-TKIs and ALK-TKIs have generally demonstrated improved progression-free survival (PFS) with insignificant changes in overall survival (OS) [24].
EGFR-TKIs: EGFR-activating mutations account for 10-15% of NSCLC cases in westerners and 25-55% in Asians [4,24]. They are highly associated with adenocarcinomas, female gender, non-smokers, and Asian populations. Erlotinib and gefitinib, both FDA- approved for EGFR-mutated NSCLC, have a markedly improved BM RR of 27-100%, based on analyses of multiple small studies [4,18]. However, robust and adequate powered trials are still lacking. Afatinib is a second generation EGFR-TKI that overcomes the resistance associated with erlotinib and gefitinib [24]. Based on one study of 32 patients, afatinib had a comparable BM RR of 35%, with 66% of patients showing BM stability [25]. Osimertinib (formerly AZD9291) is a third-generation EGFR-TKI that recently received accelerated FDA approval for refractory EGFR-positive NSCLC, showing clinical response in leptomeningeal disease [26]. AZD3759 is another EGFR-TKI, currently in a phase I clinical trial for NSCLC (NCT02228369), and preliminary data showed BBB penetration in animal models as well as promising activity in BM of a number of patients in the BLOOM trial [27]. Dacomitinib is a second- generation EGFR-TKI currently in a phase III clinical trial for NSCLC (NCT01774721); its activity in BM is being studied in a phase II trial (NCT02047747). Another agent currently under clinical trials for NSCLC is rociletinib (phase III; NCT02322281), but it has not yet been studied in BM.
ALK-TKIs: ALK-activating mutations represent 3-6% of NSCLC cases [24]. They are associated with adenocarcinoma, non-smokers, and younger patients. Crizotinib, FDA-approved for the treatment of ALK-mutated NSCLC, demonstrated significant intracranial activity with a disease control rate of 56% for radiation-naïve BM and 62% for post-radiation BM [28]. However, as is the case with systemic disease, a portion of the cases showed eventual progression, indicating resistance to crizotinib. Ceritinib, a second-generation ALK-TKI, is FDA-approved for the treatment of crizotinib-resistant NSCLC. A phase I trial studying its activity on BM showed a significant BM RR of 50% in crizotinib-treated patients and 69.2% in crizotinib-naïve patients [29]. Alectinib is another second generation ALK-TKI currently in a phase III trial; it has demonstrated significant results: it showed a greater than 90% overall RR (including resistant mutations), with CNS-specific disease showing a 52% RR [24,30]. Similarly, another ALK-TKI with great promise, AP26113, has demonstrated 60% RR in CNS in a phase 2 study [31]. Other ALK-TKIs currently in phase I/II trials include X-396 (phase I, NCT01625234), PF-06463922 (phase II, NCT01970865), and ASP3026 (phase I, NCT01401504); to date, no published studies have evaluated their effectiveness in BM.
Other TKIs: BRAF, ROS1, HER2: Gene rearrangements in the v-RAF murine sarcoma viral oncogene homolog B (BRAF) have a 1-3% incidence in NSCLC, often occurring in current or former smokers [24]. Dabrafenib and vemurafenib are both BRAF-TKIs being studied in NSCLC and are currently FDA-approved for melanoma; they are commonly used along with the MEK inhibitor trametinib to prevent resistance. Dabrafenib is currently in a phase II clinical trial for NSCLC (NCT01336634), with preliminary results showing an overall RR of 63% [32]. Though dabrafenib has not yet been studied in NSCLC BM, it has shown activity in melanoma BM [5]. Vemurafenib will be studied in a phase II clinical trial for NSCLC (NCT02314481); it has one reported case with documented activity on NSCLC BM [33], as well as good evidence of working on melanoma BM [5]. Mutations of the c-ros oncogene 1, receptor tyrosine kinase (ROS1), are rare with, an incidence of 0.7-1.7% in NSCLC, and are associated with young age, non-smokers, and adenocarcinoma (being similar to ALK-activating mutations) [34]. Crizotinib, AP26113, and PF-06463922 are all TKIs that work on NSCLC with either ALK or ROS1 rearrangements, and their activity on BM has been discussed above under ALK-TKIs. Mutations encoding the human epidermal growth factor receptor 2 (HER2) are present in 1-2% of NSCLC cases [24]. Afatinib and neratinib, both with TKI activity against HER2, have documented success in HER2-mutant NSCLC [24]. Afatinib, as discussed earlier in this review as an EGFR-TKI, has demonstrated activity in BM. Neratinib has not yet been studied in BM.
Angiogenesis inhibitors
Lung cancer BM is characterized by early angiogenesis upon invasion into the brain [4] and angiogenesis inhibitors are associated with improved PFS and OS, especially when used in combination with standard regimens in NSCLC [35]. Increased risk of cerebral hemorrhage in patients with BM has been a concern regarding these agents; however, multiple large trials and meta-analyses have not demonstrated an increased risk [4]. Vascular endothelial growth factor (VEGF) and its receptor are new emerging targets in NSCLC, with both monoclonal antibodies and TKIs being used [4]. Bevacizumab is a monoclonal antibody against VEGF, currently FDA-approved for NSCLC in combination with carboplatin and paclitaxel. In the BRAIN trial, it demonstrated significant activity on BM in combination with carboplatin and paclitaxel, with a BM RR of 61.2% [36]. The BRAIN trial also showed that the combination of bevacizumab and erlotinib achieves a BM RR of 20.8%. Rapidly emerging data including retrospective trials and analyses have continued to show that bevacizumab, when added to standard chemotherapy, has favorable results in NSCLC BM [37,38].
A major past concern within the medical community was the safety of bevacizumab on patients with treated and untreated brain metastases. Because the initial phase II trial studying bevacizumab reported 6 cases of severe hemoptysis that were associated with large central tumors located near major blood vessels [39], the drug has only been FDA-approved in non-squamous lung cancer. Subsequently, with increasingly stringent patient selection such as that in the AVAiL trial, which excluded patients with squamous tumors invading or against blood vessels, the risk of pulmonary hemorrhage has been reduced to less than 1.5% [40]. Recently, bevacizumab has been proven to be relatively safe when used with patients with both treated and untreated lung cancer brain metastases [36].
Ramucirumab, a monoclonal antibody against VEGFR2, was recently FDA-approved for pre-treated NSCLC in combination with docetaxel. Currently no studies evaluated its activity in BM. Cilengitide, with a unique mechanism as an integrin receptor antagonist, inhibits the integrin-assisted angiogenesis and tumor invasion through the BBB. Cilengitide demonstrated potential activity in NSCLC when added to cetuximab and platinum-based chemotherapy [41], but no studies in NSCLC BM have been done to date.
Nintedanib, vandetanib, motesanib, and cediranib are all VEGF-TKIs under investigation in clinical trials for the treatment of NSCLC with positive results [35,42,43]; however, these agents have not yet been studied in BM. Sunitinib and sorafenib, both approved for other solid tumors, have not shown to significantly improve survival in NSCLC [35].
Other novel agents
Other monoclonal antibodies: Nivolumab, an immune-modulatory monoclonal antibody targeting the programmed cell death protein 1 (PD-1), was recently FDA-approved for the treatment of pre-treated squamous NSCLC. In melanoma patients, nivolumab improves BM control and overall survival when combined with CNS radiation, without any major side effects [44]. In contrast, a recent study has shown an association between nivolumab and worsening neurologic events in patients with NSCLC and concurrent BM, leading to premature discontinuation of the drug in 58% of these patients; therefore, nivolumab may not be tolerated in NSCLC BM [45]. Cetuximab, a monoclonal antibody against EGFR, has shown improved survival when combined with standard chemotherapy in several large trials [46]; however, due to its higher profile of toxicity, the FDA has required identification of a biomarker prior to approving the drug for NSCLC. Based on one case report, cetuximab combined with WBRT does cross the BBB [47].
PARP enzyme inhibitors: Agents targeting the poly ADP ribose polymerase (PARP) are currently under investigation in clinical trials, with preliminary activity in NSCLC. Veliparib, when combined with carboplatin and paclitaxel, has shown marginally improved overall survival in a phase II trial for NSCLC [48], while another phase II trial is investigating the effect of veliparib and WBRT on NSCLC BM (NCT01657799). Olaparib is also currently in trials for the treatment of NSCLC (NCT01788332); no studies on BM have been done.
Eribulin mesylate: Eribulin mesylate has demonstrated activity in a phase II trial for NSCLC [11]; it is FDA-approved as a third-line treatment for metastatic breast cancer. Though preclinical studies initially demonstrated a limited ability of eribulin to cross the BBB, a subsequent preclinical murine study found that novel second generation analogs of the drug were able to cross by avoiding transporter efflux [49]. Matsuoka et al. described the first case of eribulin mesylate after WBRT with a significant BM response in a patient with advanced breast cancer [12]. A clinical trial is currently recruiting participants to study the concentration of eribulin in the brain of patients with metastatic breast, bladder, and lung cancer (NCT02338037). Prior to our patient, no case has been reported of eribulin on NSCLC BM.
Concluding Remarks
The current standard of care for the treatment of brain metastases is still limited to surgery and radiation. However, ongoing data about the use of systemic agents, especially targeted agents and the use of novel pathways for drug delivery, are paving way for an exciting new era of BM treatment. Our patient, with partial response and continued stabilization of BM, demonstrates how effective systemic treatment can be for intracranial metastases even after they have become refractory to WBRT. In this case, systemic therapy was more effective in controlling intracranial disease than extracranial disease. Though the reason for this is poorly understood, the same phenomenon was noted in the BRAIN trial, the only trial to date to study the effect of combined bevacizumab + erlotinib on NSCLC [36]. A follow-up question naturally arises: What is the best primary endpoint to assess efficacy of systemic treatment in brain metastases? OS and PFS are widely used as primary endpoints in BM trials. However, up to 60% of patients with BM ultimately die from extracranial disease (as our patient did), making OS or PFS not truly reflective of the agent's activity on BM [5]. This remains a challenge in many BM trials, though some have designated endpoints such as neurocognitive outcomes or radiological progression in the brain. Even with progression of extracranial disease and unchanged overall survival, the control of BM has the potential to significantly improve morbidity and quality of life.
After treatment with combined bevacizumab, erlotinib, and eribulin mesylate, our patient had a BM RR that lasted longer than 7 months (based on radiological response alone), and possibly up to 11 months as she remained neurologically asymptomatic until her death. This response continued to improve on each sequential MRI. Although eribulin mesylate has been FDA-approved for advanced breast cancer with one reported case of activity in breast cancer BM [12], it is important to note that its current role in NSCLC is only experimental, with moderately positive results in a phase II trial [11]. There is an ongoing clinical trial with eribulin in NSCLC BM, but otherwise our patient is, to our knowledge, the first reported case of eribulin being used in NSCLC BM.
It is important to highlight that the additional benefit of eribulin mesylate to the combination of erlotinib and bevacizumab would be difficult to assess with a single case report, and a clinical trial would be needed. However, we note that erlotinib + bevacizumab alone only had a BM RR of 20.8% in the BRAIN trial [36], suggesting that the addition of erlotinib was likely beneficial in our patient. As such, this case primarily highlights how a combination of agents with low toxicity profiles has the potential to achieve noteworthy results.
While platinum-based chemotherapy is the standard first-line therapy for NSCLC, it is important to consider that its toxicity limits its duration of treatment to typically only 4-6 cycles. In contrast, the use of chemotherapy agents at lower doses or with lower toxicity profiles allows for longer duration of treatment, potentially keeping BM controlled even in otherwise terminal cases. Thus, the stabilization of disease may be used in the palliative setting as well, due to the improved quality of life and control of neurological symptoms with minimal treatment side effects. Given the remarkable intracranial response in our patient, the aforementioned activity of eribulin mesylate on BM in other solid tumors, and the agent's overall low profile of toxicity, we propose that the activity of eribulin mesylate in NSCLC BM be studied in further clinical trials.
Acknowledgements/Funding
We received no outside assistance in the preparation of this manuscript. This research received no grants from any funding agency in the public, commercial, private, or not-for-profit sectors.
Declaration of Conflict of Interests
The authors declare that there is no conflict of interest.
Authors' Contribution
All authors contributed, read, and approved the final manuscript.
References
-
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, et al. (2015) Global cancer statistics, 2012. CA Cancer J Clin 65: 87-108.
-
American Cancer Society (2015) Society AC: Cancer Facts and Figures 2015.
-
Ali A, Goffin JR, Arnold A, Ellis PM (2013) Survival of patients with non-small-cell lung cancer after a diagnosis of brain metastases. Curr Oncol 20: e300-306.
-
Preusser M, Capper D, Ilhan-Mutlu A, Berghoff AS, Birner P, et al. (2012) Brain metastases: pathobiology and emerging targeted therapies. Acta Neuropathol 123: 205-222.
-
Ahluwalia MS, Vogelbaum MV, Chao ST, Mehta MM (2014) Brain metastasis and treatment. F1000 Prime Rep 6: 114.
-
Mehta MP, Paleologos NA, Mikkelsen T, Robinson PD, Ammirati M, et al. (2010) The role of chemotherapy in the management of newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol 96: 71-83.
-
Marks LB, Yorke ED, Jackson A, Ten Haken RK, Constine LS, et al. (2010) Use of normal tissue complication probability models in the clinic. Int J Radiat Oncol Biol Phys 76: S10-S19.
-
Yamada MK (2016) A link between vascular damage and cognitive deficits after whole-brain radiation therapy for cancer: A clue to other types of dementia? Drug Discov Ther 10: 79-81.
-
DeAngelis LM, Delattre JY, Posner JB (1989) Radiation-induced dementia in patients cured of brain metastases. Neurology 39: 789-796.
-
Tallet AV, Azria D, Barlesi F, Spano JP, Carpentier AF, et al. (2012) Neurocognitive function impairment after whole brain radiotherapy for brain metastases: actual assessment. Radiat Oncol 7: 77.
-
Gitlitz BJ, Tsao-Wei DD, Groshen S, Davies A, Koczywas M, et al. (2012) A phase II study of halichondrin B analog eribulin mesylate (E7389) in patients with advanced non-small cell lung cancer previously treated with a taxane: a California cancer consortium trial. J Thorac Oncol 7: 574-578.
-
Matsuoka H, Tsurutani J, Tanizaki J, Iwasa T, Komoike Y, et al. (2013) Regression of brain metastases from breast cancer with eribulin: a case report. BMC Res Notes 6: 541.
-
Bhowmik A, Khan R, Ghosh MK (2015) Blood brain barrier: a challenge for effectual therapy of brain tumors. Biomed Res Int 2015: 320941.
-
Gerstner ER, Fine RL (2007) Increased permeability of the blood-brain barrier to chemotherapy in metastatic brain tumors: establishing a treatment paradigm. J Clin Oncol 25: 2306-2312.
-
Charest G, Sanche L, Fortin D, Mathieu D, Paquette B (2013) Optimization of the route of platinum drugs administration to optimize the concomitant treatment with radiotherapy for glioblastoma implanted in the Fischer rat brain. J Neurooncol 115: 365-373.
-
Dhami NK, Pandey RS, Jain UK, Chandra R, Madan J (2014) Non-aggregated protamine-coated poly(lactide-co-glycolide) nanoparticles of cisplatin crossed blood-brain barrier, enhanced drug delivery and improved therapeutic index in glioblastoma cells: in vitro studies. J Microencapsul 31: 685-693.
-
Bartolotti M, Franceschi E, Brandes AA (2012) EGF receptor tyrosine kinase inhibitors in the treatment of brain metastases from non-small-cell lung cancer. Expert Rev Anticancer Ther 12: 1429-1435.
-
Zimmermann S, Dziadziuszko R, Peters S (2014) Indications and limitations of chemotherapy and targeted agents in non-small cell lung cancer brain metastases. Cancer Treat Rev 40: 716-722
-
Ge XH, Lin Q, Ren XC, Liu YE, Chen XJ, et al. (2013) Phase II clinical trial of whole-brain irradiation plus three-dimensional conformal boost with concurrent topotecan for brain metastases from lung cancer. Radiat Oncol 8: 238.
-
Fujita A, Fukuoka S, Takabatake H, Tagaki S, Sekine K (2000) Combination chemotherapy of cisplatin, ifosfamide, and irinotecan with rhG-CSF support in patients with brain metastases from non-small cell lung cancer. Oncology 59: 291-295.
-
Bailon O, Chouahnia K, Augier A, Bouillet T, Billot S, et al. (2012) Upfront association of carboplatin plus pemetrexed in patients with brain metastases of lung adenocarcinoma. Neuro Oncol 14: 491-495.
-
Bearz A, Garassino I, Tiseo M, Caffo O, Soto-Parra H, et al. (2010) Activity of Pemetrexed on brain metastases from Non-Small Cell Lung Cancer. Lung Cancer 68: 264-268.
-
Zhu W, Roe OD, Wu C, Li W, Guo R, et al. (2015) Activity of pemetrexed-based regimen as first-line chemotherapy for advanced non-small cell lung cancer with asymptomatic inoperable brain metastasis: a retrospective study. J Chemother 27: 221-226.
-
Nguyen KS, Neal JW, Wakelee H (2014) Review of the current targeted therapies for non-small-cell lung cancer. World J Clin Oncol 5: 576-587.
-
Hoffknecht P, Tufman A, Wehler T, Pelzer T, Wiewrodt R, et al. (2015) Efficacy of the irreversible ErbB family blocker afatinib in epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI)-pretreated non-small-cell lung cancer patients with brain metastases or leptomeningeal disease. J Thorac Oncol 10: 156-163.
-
Nanjo S, Ebi H, Arai S, Takeuchi S, Yamada T, et al. (2016) High efficacy of third generation EGFR inhibitor AZD9291 in a leptomeningeal carcinomatosis model with EGFR-mutant lung cancer cells. Oncotarget 7: 3847-3856.
-
Kim D-W, Chih-Hsin Yang J, Chen K, Cheng Z, Yin L, et al. (2015) AZD3759, an EGFR inhibitor with blood brain barrier (BBB) penetration for the treatment of non-small cell lung cancer (NSCLC) with brain metastasis (BM): Preclinical evidence and clinical cases. Presented at the ASCO Annual Meeting.
-
Costa DB, Shaw AT, Ou SH, Solomon BJ, Riely GJ, et al. (2015) Clinical Experience with Crizotinib in Patients With Advanced ALK-Rearranged Non-Small-Cell Lung Cancer and Brain Metastases. J Clin Oncol 33:1881-1888.
-
Shaw AT, Kim DW, Mehra R, Tan DS, Felip E, et al. (2014) Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med 370: 1189-1197.
-
Gadgeel SM, Gandhi L, Riely GJ, Chiappori AA, West HL, et al. (2014) Safety and activity of alectinib against systemic disease and brain metastases in patients with crizotinib-resistant ALK-rearranged non-small-cell lung cancer (AF-002JG): results from the dose-finding portion of a phase 1/2 study. Lancet Oncol 15:1119-1128.
-
Gettinger SN, Bazhenova L, Salgia R, Langer CJ, Gold KA, et al. (2014) Updated Efficacy and Safety of the ALK Inhibitor AP26113 in Patients with Advanced Malignancies, Including ALK+ Non-Small-Cell Lung Cancer (NSCLC). J Clin Oncol suppl; abstr 8047.
-
Planchard D, Groen HJM, Kim TM, Rigas JR, Souquet PJ, et al. (2015) Interim results of a phase II study of the BRAF inhibitor (BRAFi) dabrafenib (D) in combination with the MEK inhibitor trametinib (T) in patients (pts) with BRAF V600E mutated (mut) metastatic non-small cell lung cancer (NSCLC). J Clin Oncol 33, 2015 (suppl; abstr 8006), ASCO Annual Meeting.
-
Robinson SD, O'Shaughnessy JA, Cowey CL, Konduri K (2014) BRAF V600E-mutated lung adenocarcinoma with metastases to the brain responding to treatment with vemurafenib. Lung Cancer 85: 326-330.
-
Chin LP, Soo RA, Soong R, Ou SH (2012) Targeting ROS1 with anaplastic lymphoma kinase inhibitors: a promising therapeutic strategy for a newly defined molecular subset of non-small-cell lung cancer. J Thorac Oncol 7:1625-1630.
-
Hong S, Tan M, Wang S, Luo S, Chen Y, et al. (2015) Efficacy and safety of angiogenesis inhibitors in advanced non-small cell lung cancer: a systematic review and meta-analysis. J Cancer Res Clin Oncol 141: 909-921.
-
Besse B, Le Moulec S, Mazières J, Senellart H, Barlesi F, et al. (2015) Bevacizumab in Patients with Nonsquamous Non-Small Cell Lung Cancer and Asymptomatic, Untreated Brain Metastases (BRAIN): A Nonrandomized, Phase II Study. Clin Cancer Res 21: 1896-1903.
-
Ilhan-Mutlu A, Osswald M, Liao Y, Gömmel M, Reck M, et al. (2016) Bevacizumab Prevents Brain Metastases Formation in Lung Adenocarcinoma. Mol Cancer Ther 15: 702-710.
-
Tang N, Guo J, Zhang Q, Wang Y, Wang Z (2016) Greater efficacy of chemotherapy plus bevacizumab compared to chemo- and targeted therapy alone on non-small cell lung cancer patients with brain metastasis. Oncotarget 7: 3635-3644.
-
Johnson DH, Fehrenbacher L, Novotny WF, Herbst RS, Nemunaitis JJ, et al. (2004) Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol 22: 2184-2191.
-
Gadgeel SM (2012) Safety profile and tolerability of antiangiogenic agents in non-small-cell lung cancer. Clin Lung Cancer 13: 96-106.
-
Vansteenkiste J, Barlesi F, Waller CF, Bennouna J, Gridelli C, et al. (2015) Cilengitide combined with cetuximab and platinum-based chemotherapy as first-line treatment in advanced non-small-cell lung cancer (NSCLC) patients: results of an open-label, randomized, controlled phase II study (CERTO). Ann Oncol 26: 1734-1740.
-
Kubota K, Ichinose Y, Scagliotti G, Spigel D, Kim JH, et al. (2014) Phase III study (MONET1) of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous nonsmall-cell lung cancer (NSCLC): Asian subgroup analysis. Ann Oncol 25: 529-536.
-
Reck M, Kaiser R, Mellemgaard A, Douillard JY, Orlov S, et al. (2014) Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): a phase 3, double-blind, randomised controlled trial. Lancet Oncol 15: 143-155.
-
Ahmed KA, Stallworth DG, Kim Y, Johnstone PA, Harrison LB, et al. (2016) Clinical outcomes of melanoma brain metastases treated with stereotactic radiation and anti-PD-1 therapy. Ann Oncol 27: 434-441.
-
Kanai O, Fujita K, Okamura M, Nakatani K, Mio T (2016) Severe exacerbation or manifestation of primary disease related to nivolumab in non-small-cell lung cancer patients with poor performance status or brain metastases. Ann Oncol.
-
Yang ZY, Liu L, Mao C, Wu XY, Huang YF, et al. (2014) Chemotherapy with cetuximab versus chemotherapy alone for chemotherapy-naive advanced non-small cell lung cancer. Cochrane Database Syst Rev 11: CD009948.
-
Rades D, Nadrowitz R, Buchmann I, Hunold P, Noack F, et al. (2010) Radiolabeled cetuximab plus whole-brain irradiation (WBI) for the treatment of brain metastases from non-small cell lung cancer (NSCLC). Strahlenther Onkol 186: 458-462.
-
Rizvi NA, Mazieres J, Planchard D, Stinchcombe TE, Dy GK, et al. (2015) Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2, single-arm trial. Lancet Oncol 16: 257-265.
-
Narayan S, Carlson EM, Cheng H, Condon K, Du H, et al. (2011) Novel second generation analogs of eribulin. Part III: Blood-brain barrier permeability and in vivo activity in a brain tumor model. Bioorg Med Chem Lett 21:1639-1643.





