Clinical Medical
Reviews and Case Reports
Three-Vessel Spontaneous Coronary Artery Dissection in an Old Woman
Huseyin Bozbas1*, Tugba Kayhan Altuner1, Ezgi Polat Ocakli1, Mehmet Sanser Ates2, Murat Kurtoglu2 and Mehmet Emin Korkmaz1
1Department of Cardiology, Guven Hospital, Ankara, Turkey
2Cardiovascular Surgery, Guven Hospital, Ankara, Turkey
*Corresponding author: Huseyin Bozbas, MD, FESC, Department of Cardiology, Güven Hospital, Paris Cad No: 58, Kavaklıdere, Ankara, Turkey, Tel: +90-532-7480151, Fax: +90-312-4572873, E-mail: hbozbas@gmail.com
Clin Med Rev Case Rep, CMRCR-3-113, (Volume 3, Issue 6), Case Report; ISSN: 2378-3656
Received: May 07, 2016 | Accepted: June 24, 2016 | Published: June 27, 2016
Citation: Bozbas H, Altuner TK, Ocakli EP, Ates MS, Kurtoglu M, et al. (2016) Three-Vessel Spontaneous Coronary Artery Dissection in an Old Woman. Clin Med Rev Case Rep 3:113. 10.23937/2378-3656/1410113
Copyright: © 2016 Bozbas H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Spontaneous coronary artery dissection (SCAD) is one of the rare causes of acute coronary syndrome. Regarding the etiology, it is classified broadly into 2 groups: atherosclerotic and non-atherosclerotic. It is more common in females and left anterior descending coronary artery is the most commonly involved artery. Optimal therapy for SCAD is not well-defined. Treatment options include a conservative approach, percutaneous intervention, and bypass grafting surgery. We present an old woman with three-vessel SCAD. She was admitted with acute coronary syndrome. By angiography, SCAD was identified in all three coronary arteries. Her case was discussed by our heart team and she underwent bypass surgery. Her postoperative course was uneventful and she was discharged from the hospital in good clinical condition.
Keywords
Spontaneous coronary artery dissection, Acute coronary syndrome, Three vessel
Introduction
Spontaneous coronary artery dissection (SCAD) is a very rare cause of acute coronary syndrome. Its etiology and pathogenesis are not well-defined. The prevalence of SCAD is reported to be approximately 0.1% in patients undergoing coronary angiography [1]. Saw et al. showed that 24% of young women who presented with a myocardial infarction had SCAD [2]. Endovascular imaging modalities, intravascular ultrasound (IVUS) technology, and optical coherence tomography (OCT) improved the recognition and characterization of SCAD [3,4]. It is much more prevalent in patients with acute coronary syndrome (ACS) than in those with stable coronary artery disease CAD.
There is a clear female preponderance; 70-80% of affected patients are women. It is most commonly seen during pregnancy and the peripartum period. The mean age of women suffering SCAD is approximately 35-40 years of age and the left anterior descending (LAD) coronary artery is the most commonly involved vessel [5]. We present an old woman who presented with ACS and had three-vessel SCAD.
Case Report
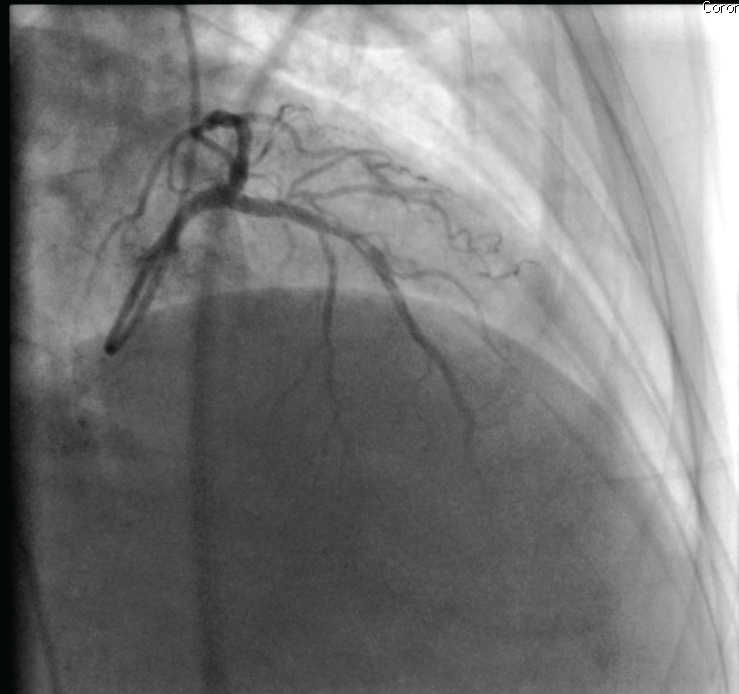
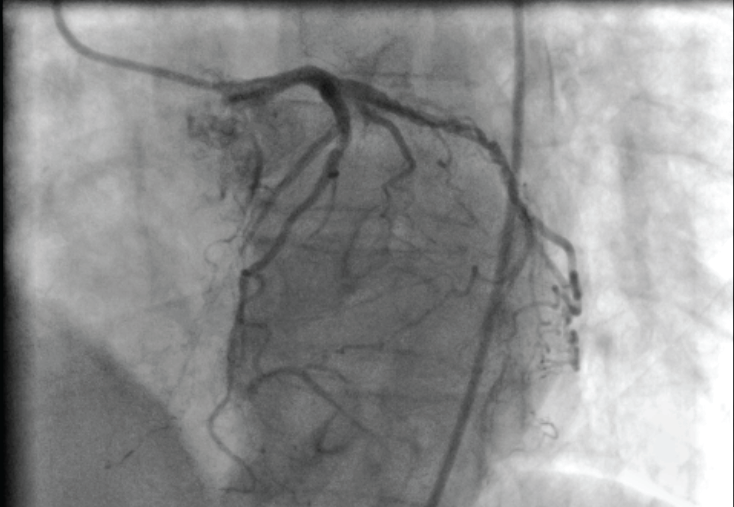
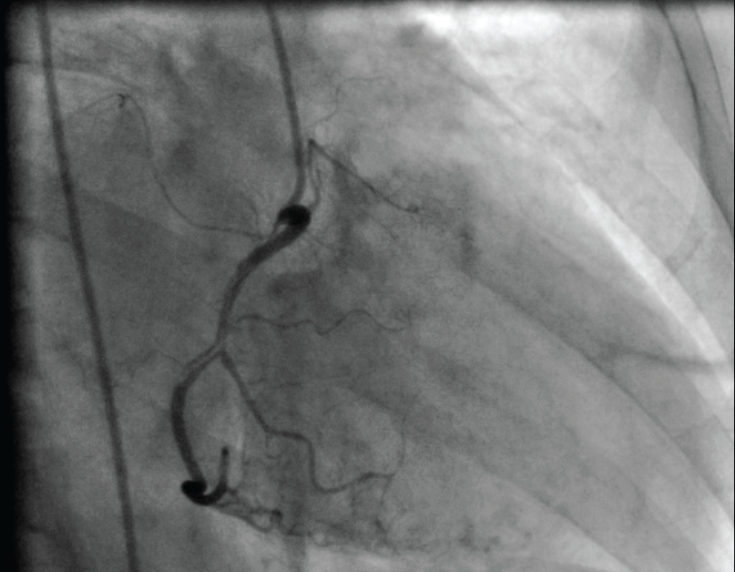
A 66-year-old female patient was admitted into our hospital with chest pain. In her past medical history, hypertension and hyperlipidemia were noted. She was suffering chest pain for four days. She described the chest pain as a feeling of severe pressure on her chest. Upon physical examination, her blood pressure was 120/70 mmHg and her pulse was 74 beats/minute. No murmur was heard by auscultation. Her peripheral pulses were palpable and no pulse deficit was noted. There was no murmur in her carotid arteries. Her troponin level was high. By ECG, anterolateral ischemic changes were identified. Upon Doppler echocardiographic examination, her left ventricular ejection fraction was 45% with anterolateral and apical wall motion abnormalities. Further, mild mitral regurgitation was noted. Doppler examination of the carotid arteries suggested luminal irregularities and plaque formation. Coronary angiography was performed and dissection was visualized in the LAD, circumflex, and right coronary arteries (Figure 1, Figure 2 and Figure 3). Her case was discussed by our heart team; because of the multi-vessel involvement, they advised surgery. A coronary artery bypass grafting operation was performed successfully. Her postoperative course was uneventful and she was discharged from the hospital in good condition.
Discussion
SCAD is a rare and complex disease with multiple factors contributing to the etiology. Clinical presentation may vary from unstable angina to sudden cardiac death. Chest pain is the most common presenting symptom of affected patients. During the pathogenesis of SCAD, two events are proposed to result in disease. First, there is an intimal tear through which blood enters into the intimal space creating a false lumen. The second event is the rupture of the vasa vasorum.
Regarding the etiology, SCAD is classified broadly into two groups: atherosclerotic and non-atherosclerotic SCAD. For the non-atherosclerotic type, predisposing and precipitating factors for the development of SCAD include pregnancy, fibromuscular dysplasia, connective tissue diseases (Marfan’s syndrome, systemic lupus erythematosus, etc.), hormonal therapy, systemic inflammatory conditions, spasms in the coronaries, vigorous exercise, emotional stress, labor/delivery, cocaine use, severely elevated blood pressure, and chest trauma [6-8]. Recent data show a strong association between fibromuscular dysplasia and SCAD, suggesting a causal relationship [9]. Based on this information, special care must be given to identify those predisposed to arteriopathy, for example women with SCAD.
Peripartum SCAD is usually seen during the last trimester or early post-puerperal period. The exact role of the peripartum state in the pathogenesis of SCAD is not well-defined yet. Hormonal changes such as high estrogen levels are proposed to play role. Additionally, increased cardiac output and heart rate during pregnancy and labor are psychological factors that seem to increase shear stress. Thus, both hormonal and hemodynamic changes seem to be responsible for the development of SCAD.
In a small percentage of SCAD cases, no specific cause can be identified despite detailed evaluation. These patients are usually young, lack identified atherosclerosis, and have no conventional risk factors.
Atherosclerosis is another cause of SCAD. Studies have shown that this type of SCAD is more common than previously assumed [10]. The mechanism of this type of SCAD is different than that of the non-atherosclerotic type. An increased density of the vasa vasorum due to the presence of an atherosclerotic plaque may lead to dissection.
Vanzetto et al. examined the prevalence and course of SCAD in 11,605 patients who underwent coronary angiography during a five year period [11]. Twenty three of these patients had SCAD (0.2%). Of these cases 17 (74%) were female and the mean age was 46 years. The clinical presentation was acute coronary syndrome in 91% of the cases. LAD was the most common coronary artery affected and atheromatous CAD was identified in only five patients. Regarding disease management, 43% were treated medically and 57% underwent coronary revascularization [48% percutaneous coronary intervention (PCI), 9% coronary artery bypass grafting CABG) ]. Of note, in this large cohort, no cases of SCAD that affected three vessels were detected.
Based on the age of the patient in our case study and her associated risk factors, we thought that atherosclerosis was the underlying cause of SCAD. To our knowledge, this is one of the oldest patients with three-vessel SCAD reported in the literature.
For SCAD diagnosis, angiography is not the gold standard method. We cannot examine the arterial wall using the angiogram of the involved vessel. IVUS and OCT help us to better visualize the structure of the arterial wall [3,4]. Each method has limitations and advantages. For example, IVUS has a lower spatial resolution but better penetration capability. OCT, on the other hand, provides higher resolution but has lower penetration ability. Discrimination of false and true lumens can be achieved precisely using OCT. Even small tears in the vessel wall can be detected.
Optimal therapy for SCAD is not well-defined. Treatment options include a conservative approach, PCI, and CABG [12-14]. The severity of disease, extent of dissection, and the quantity of myocardium at risk are important factors to consider when choosing a therapy. In the acute phase, when the patient is unstable, PCI seems to be the best reperfusion strategy. In stable patients with an established and normal flow, conservative treatment may be an appropriate option. Recent data show that spontaneous healing is common, making a conservative approach a valid option [4,6]. Saw et al. reported a high angiographic recovery rate and a good outcome using this approach [6]. However, because detrimental cardiovascular events are still common, these patients require close follow up. Medical therapy, such as the anti-coagulant heparin, is recommended during a patient’s hospital stay. Further, aspirin should be given. Thrombolytic therapy is not recommended because of the potential harms and exacerbation of the dissection. Beta blockers seem beneficial because they can reduce the arterial wall tension. Nitroglycerin helps to resolve spasms and ischemic symptoms. In symptomatic patients with multi-vessel disease, a CABG operation seems to be a valid treatment strategy. Because our patient had SCAD in three vessels and had chest pains, our heart team decided that CABG would be a better treatment choice. She did well during the peri- and post-operative periods.
In our case report, the diagnosis of SCAD was confirmed using angiography. Lack of intravascular imaging is a weakness of our case report. If it had been done, we would have learned the exact site of the intimal tear.
Conflict of Interest
None.
References
-
Maeder M, Ammann P, Angehrn W, Rickli H (2005) Idiopathic spontaneous coronary artery dissection: incidence, diagnosis and treatment. Int J Cardiol 101: 363-369.
-
Saw J, Aymong E, Mancini GB, Sedlak T, Starovoytov A, et al. (2014) Nonatherosclerotic coronary artery disease in young women. Can J Cardiol 30: 814-819.
-
Maehara A, Mintz GS, Castagna MT, Pichard AD, Satler LF, et al. (2002) Intravascular ultrasound assessment of spontaneous coronary artery dissection. Am J Cardiol 89: 466-468.
-
Alfonso F, Paulo M, Gonzalo N, Dutary J, Jimenez-Quevedo P, et al. (2012) Diagnosis of spontaneous coronary artery dissection by optical coherence tomography. J Am Coll Cardiol 59: 1073-1079.
-
DeMaio SJ Jr, Kinsella SH, Silverman ME (1989) Clinical course and long-term prognosis of spontaneous coronary artery dissection. Am J Cardiol 64: 471-474.
-
Saw J, Aymong E, Sedlak T, Buller CE, Starovoytov A, et al. (2014) Spontaneous coronary artery dissection: association with predisposing arteriopathies and precipitating stressors and cardiovascular outcomes. Circ Cardiovasc Interv 7: 645-655.
-
Tanis W, Stella PR, Kirkels JH, Pijlman AH, Peters RH, et al. (2008) Spontaneous coronary artery dissection: current insights and therapy. Neth Heart J 16: 344-349.
-
Aldoboni AH, Hamza EA, Majdi K, Ngibzadhe M, Palasaidi S, et al. (2002) Spontaneous dissection of coronary artery treated by primary stenting as the first presentation of systemic lupus erythematosus. J Invasive Cardiol 14: 694-696.
-
Saw J, Poulter R, Fung A, Wood D, Hamburger J, et al. (2012) Spontaneous coronary artery dissection in patients with fibromuscular dysplasia: a case series. Circ Cardiovasc Interv 5: 134-137.
-
Celik SK, Sagcan A, Altintig A, Yuksel M, Akin M, et al. (2001) Primary spontaneous coronary artery dissections in atherosclerotic patients. Report of nine cases with review of the pertinent literature. Eur J Cardiothorac Surg 20: 573-576.
-
Vanzetto G, Berger-Coz E, Barone-Rochette G, Chavanon O, Bouvaist H, et al. (2009) Prevalence, therapeutic management and medium-term prognosis of spontaneous coronary artery dissection: results from a database of 11,605 patients. Eur J Cardiothorac Surg 35: 250-254.
-
Ozturk C, Yildirim AO, Demir M, Haqmal H, Balta S, et al. (2016) The spontaneous coronary artery dissection may need intervention in the proximal segment of the arteries. Int J Cardiol 202: 943-944.
-
Ozturk C, Celik T, Demirkol S, Demir M, Balta S, et al. (2015) The healing of spontaneous coronary artery dissection with conservative treatment: When to stop. Int J Cardiol 189: 249-251.
-
Iyisoy A, Kursaklioglu H, Kose S, Ozturk C, Amasyali B, et al. (2003) Spontaneous intimal dissection in a patient with post-infarct angina: identification with intravascular ultrasound and treatment with coronary stenting. Jpn Heart J 44: 557-564.