Clinical Medical Reviews and Case Reports
A Case of Spontaneous Regression of High-Grade Hepatocellular Carcinoma
Ghufran A1, Agni R2, Winslow E3, Dalvie P4 and Agarwal PD5*
1Department of Medicine, University of Wisconsin School of Medicine and Public Health, USA
2Department of Pathology and Laboratory Medicine, University of Wisconsin School of Medicine and Public Health, USA
3Department of Surgery, University of Wisconsin School of Medicine and Public Health, USA
4Department of Radiology, University of Wisconsin School of Medicine and Public Health, USA
5Department of Gastronterology and Hepatology, University of Wisconsin School of Medicine and Public Health, USA
*Corresponding author: Parul D Agarwal, Department of Gastronterology and Hepatology, University of Wisconsin School of Medicine and Public Health, USA, E-mail: pagarwal@medicine.wisc.edu
Clin Med Rev Case Rep, CMRCR-3-128, (Volume 3, Issue 9), Case Report; ISSN: 2378-3656
Received: July 23, 2016 | Accepted: September 05, 2016 | Published: September 08, 2016
Citation: Ghufran A, Agni R, Winslow E, Dalvie P, Agarwal PD (2016) A Case of Spontaneous Regression of High-Grade Hepatocellular Carcinoma. Clin Med Rev Case Rep 3:128. 10.23937/2378-3656/1410128
Copyright: © 2016 Ghufran A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
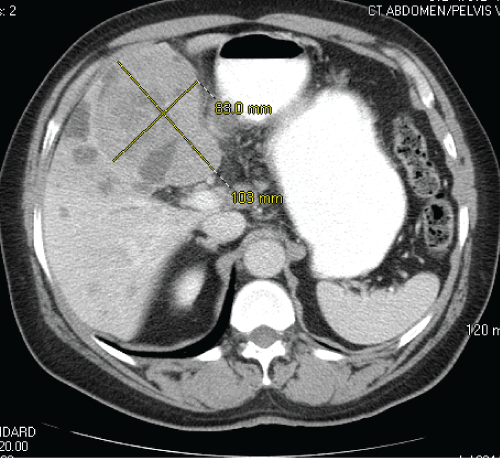
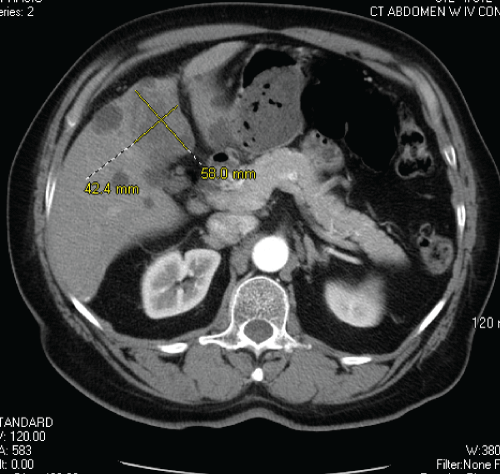
A 70-year-old South African male with a history of type 2 diabetes mellitus presented with vague abdominal discomfort of 2 months' duration. This was associated with anorexia and 15-pound weight loss over the preceding 6 months. A computerized tomogram (CT) of his abdomen and pelvis revealed a large heterogeneous mass, measuring 10.3 × 8.3 cm in liver segments 4A and B (Figure 1). His CT imaging did not show stigmata of cirrhosis or portal hypertension, although numerous simple hepatic cysts, consistent with polycystic liver disease, and a hemangioma were visualized. A biopsy of the hepatic mass revealed a poorly differentiated carcinoma, consistent with hepatocellular carcinoma (HCC), by morphology and immunohistochemistry (IHC) with HSA 1 (Hep Par 1) and glypican-3 (Figure 2a, Figure 2b, Figure 2c and Figure 2d). His serum alpha-fetoprotein (AFP) level was greater than 36300 ng/mL on initial presentation. His case was discussed at our institutional multidisciplinary primary liver tumor conference. He was not deemed to be a suitable candidate for surgical resection due to the large size of the liver mass. He was referred for liver-directed therapy with trans-arterial chemoembolization (TACE), which was performed 7 weeks following his initial presentation. However, repeat serologic and CT studies just prior to this procedure revealed that his serum AFP had fallen to 3619 ng/mL, and there was an interval decrease in the size of the tumor by approximately 30% since the initial CT, despite no prior treatment. He underwent chemoembolization (with doxorubicin, mitomycin, and cisplatin) of the segment IV hepatic arterial branch with subsequent placement of 300-500 μm bead blocks in the artery. CT imaging obtained 5 weeks after TACE revealed a further significant decrease in size of the left hepatic mass from 10.3 × 8.3 cm to 5.8 × 4.2 cm, although scant lipiodol uptake was observed within the tumor (Figure 3). In fact, there was reflux of lipiodol noted into the posterior right hepatic lobe and concentration within a hemangioma, without any apparent deposition within the tumor itself, indicating unsuccessful chemoembolization. At the same time, his serum AFP declined further to 145 ng/mL. Based on the significant interval decrease in size of the tumor, absence of extrahepatic disease and his performance status, he was referred for consideration of elective surgical resection of this hepatocellular carcinoma. The patient deferred surgery in favor of continued surveillance with cross-sectional imaging, which he received in 8-12 week intervals. Follow up imaging demonstrated continued decrease in the size of the segment 4 lesion, to 2.5 cm with ill-defined margins and without enhancement. No new arterially enhancing lesions were noted, and his serum AFP remained within the normal range, having normalized approximately 7 months since initial presentation. The patient finally elected to undergo surgical resection with left hepatectomy, which was performed approximately 22 months following his initial diagnosis. On gross examination, the cut surface of the liver was tan yellow and smooth, with innumerable cysts, 0.3-0.5 cm in diameter. A cystic mass, 3.5 × 3.5 × 1.3 cm is identified within this background as being the possible site of prior treatment. Yet on microscopy, a discrete nodule is not present, but rather, thrombosed vessels and hemorrhage are found in this area (Figure 4 and Figure 5).
.
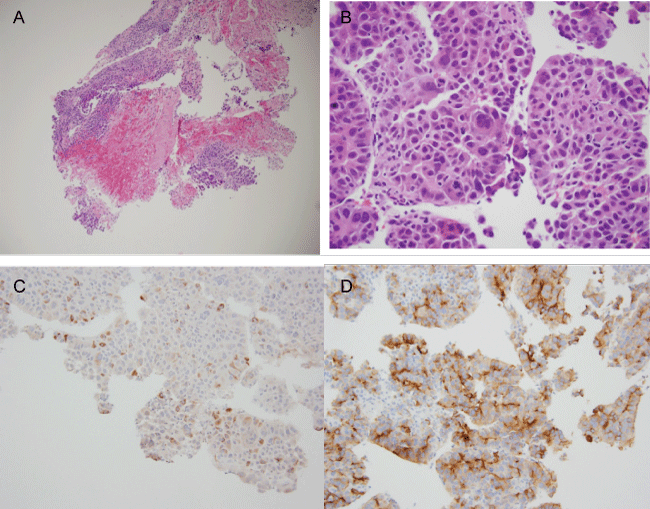
Figure 2a and b: Core biopsy of hepatic mass.
a) 100X (intermediate magnification) showing core of liver with cluster of hepatocellular carcinoma cells with central hemorrhage and infarction; b) 400X magnification of hepatocellular carcinoma: proliferation of atypical enlarged cells, resembling hepatocytes, growing in nests and trabeculae, with hyperchromatic nuclei with moderate nuclear pleomorphism, increased mitotic figures and abundant pink cytoplasm, indicative of poorly differentiated carcinoma.
Figure 2c and d: IHC of hepatic mass, with HSA1 (Figure 2c); Glypican-3 (Figure 2d).
By IHC, the neoplastic cells show rare focal positivity for Hepatocyte specific antigen /HSA 1(Hep Par 1) with granular cytoplasmic staining (not uncommon in poorly differentiated HCC) and strong positivity for glypican-3 supportive of the diagnosis of HCC (200x magnification).
View Figure 2
Serological studies confirmed that chronic hepatitis B infection was his risk factor for development of HCC. His aminotransferases were persistently elevated, in conjunction with positive hepatitis B surface antigen and circulating HBV DNA 85,100 IU/mL. He was initiated on anti-viral therapy with entecavir 0.5 mg daily, with subsequent normalization of his aminotransferases and undetectable HBV DNA.
.
Figure 3: CT abdomen showing significant decrease in size of hepatic mass despite scant lipiodol uptake within the tumor, owing to unsuccessful TACE.
View Figure 3
.
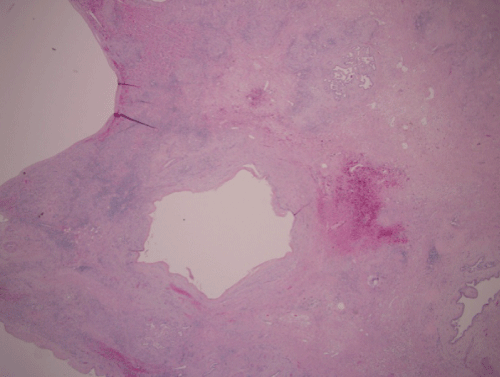
Figure 4: Liver lobectomy specimen.
20X (Scanning magnification): Cysts and infarct in area of prior hepatocellular carcinoma in segment 4. Residual carcinoma is no longer present. Pink hyalinized stroma between cysts (spaces in the above figure) represents an area of remote infarct with areas of hemorrhage.
View Figure 4
On follow-up, 3 years since elective left hepatectomy, and nearly 5 years since his initial diagnosis, he continued to show no evidence of residual neoplasm or recurrence on surveillance imaging, and had a normal serum AFP of 1.0 ng/mL.
.
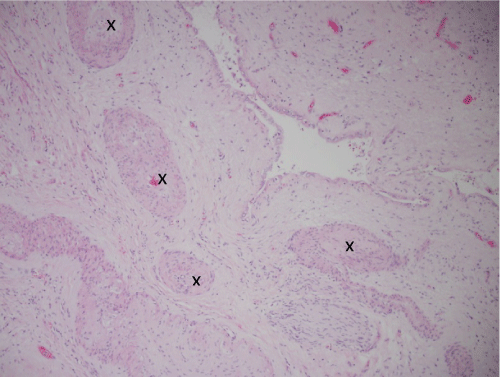
Figure 5: 100X thrombosed vessels (marked with an X) in area of infarction adjacent to area of prior hepatocellular carcinoma. The space in center is a collapsed cyst lined by low cuboidal epithelium.
View Figure 5
Discussion
Primary liver cancer, of which hepatocellular carcinoma (HCC) is the predominant type, is the fifth most common cancer and the third most common cause of cancer mortality worldwide. The vast majority of cases of HCC are associated with a known risk factor, such as chronic hepatitis B infection [1]. This century has witnessed a significant change in the management of HCC, and it is now a potentially curable cancer, if detected early. Unfortunately, most patients present at an advanced stage, with limited therapeutic options, and poor prognosis. Spontaneous regression of cancer, originally defined by Everson and Cole as ‘partial or complete disappearance of a malignant tumor in the absence of all treatment, or in the presence of therapy which is considered to be inadequate to exert a significant influence on neoplastic disease' has been well described in certain malignancies, such as renal cell carcinoma and neuroblastoma [2]. Spontaneous regression of HCC, although reported, is an extremely rare occurrence, with an estimated incidence ranging from 1 in 60,000 to 140,000 [3]. A detailed PubMed search from 1982 to 2011 yielded 87 reported cases of spontaneous regression of HCC described in the English literature. Patient demographics, etiology of liver disease and tumor burden vary widely in these cases, including patients with single or multiple lesions, portal vein invasion, with or without extra-hepatic spread. Most of the cases reported show partial regression of tumor [4].
The pathogenic mechanisms underlying spontaneous regression of HCC are unknown, however are hypothesized to be related to either tumor hypoxia or activation of a systemic inflammatory state [5,6]. Tumor ischemia may be triggered by occlusion of the portal vein or disruption of the hepatic artery, hemorrhagic shock or rapid tumor growth, which then induces necrosis of a highly-vascular malignancy. This mechanism for tumor regression appears plausible, especially as current treatment modalities, such as TACE, and anti-angiogenic therapies, such as Sorafenib, rely on affecting the arterial supply, thereby inducing hypoxia, to achieve their response [5]. This is the likely mechanism accounting for complete tumor regression in this case, although the inciting factor is unknown, but may be related to rapid tumor growth. This is suggested by the central hemorrhage and infarction adjacent to the malignancy on the index core biopsy (Figure 2b) as well as the site of the regressed HCC in the surgical specimen following resection (Figure 4 and Figure 5). Tumor hypoxia does not explain all of the reported cases of spontaneous HCC regression, particularly in those patients with metastases. Induction of immune mechanisms, involving intense systemic cytokine release, has also been implicated in tumor regression [7]. Activation of the systemic inflammatory response with cytokine surge may in turn induce production of interferon (IFN)-gamma and natural killer cells, thereby enhancing cytotoxicity towards cancer cells [7,8]. The inciting factor for the systemic inflammatory response has been variable in the literature, being related to systemic infection, following discontinuation of heavy alcohol use, as well as increased circulating levels of tumor necrosis factor alpha (TNF-α) following radiation therapy.
In conclusion, spontaneous regression of hepatocellular carcinoma, while reported in the literature, is a rare event. The pathogenic mechanisms underlying this process, which frequently involves tumor hypoxia or activation of a systemic inflammatory response, remain largely unknown and are likely multifactorial. Inducing tumor hypoxia to achieve regression of the tumor is the basis of current modalities, including chemoembolization and new anti-angiogenic agents. Targeted immune therapy is an area of active clinical research for a variety of cancers, including HCC. Further research into the pathogenic mechanisms of spontaneous regression may prove useful in management of patients with this increasingly prevalent cancer.
Authors' Disclosures of Potential Conflict of Interest
The authors indicated no potential conflicts of interest.
References
-
El-Serag HB (2012) Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 142: 1264-1273.
-
Everson TC, Cole WH (1959) Spontaneous regression of malignant disease. J Am Med Assoc 169: 1758-1759.
-
Chang WY (2000) Complete spontaneous regression of cancer: four case reports, review of literature, and discussion of possible mechanisms involved. Hawaii Med J 59: 379-387.
-
Randolph AC, Tharalson EM, Gilani N (2008) Sponteneous regression of hepatocellular carcinoma is possible and might have implications for future therapies. Eur J Gastroenterol Hepatol 20: 804-809.
-
Huz JI, Melis M, Sarpel U (2012) Spontaneous regression of hepatocellular carcinoma is most often associated with tumour hypoxia or a systemic inflammatory response. HPB 14: 500-505.
-
Lin TJ, Liao LY, Lin CL, Shih LS, Chang TA, et al. (2004) Spontaneous regression of hepatocellular carcinoma: a case report and literature review. Hepatogastroenterology 51: 579-582.
-
Abiru S, Kato Y, Hamasaki K, Nakao K, Nakata K, et al. (2002) Spontaneous regression of hepatocellular carcinoma associated with elevated levels of interleukin 18. Am J Gastroenterol 97: 774-775.
-
Jozuka H, Jozuka E, Suzuki M, Takeuchi S, Takatsu Y (2003) Psycho-neuro-immunological treatment of hepatocellular carcinoma with major depression-a single case report. Curr Med Res Opin 19: 59-63.