Clinical Medical
Reviews and Case Reports
Aortoenteric Fistula in Patient with Infected Aortic Endovascular Stent
Giuseppe Grande*, Helga Bertani, Angelo Caruso and Rita Conigliaro
Gastroenterology and digestive endoscopy unit, NOCSAE Hospital, Modena, Italy
*Corresponding author: Giuseppe Grande, Gastroenterology and digestive endoscopy unit, NOCSAE hospital, via Pietro Giardini 1355, ZIP Code 41126, Baggiovara di Modena, Italy, Tel: +39-0593961269/+39-0593961216, Fax: +39 0593961216, E-mail: giuseppegrande1984@gmail.com
Clin Med Rev Case Rep, CMRCR-3-135, (Volume 3, Issue 10), Case Report; ISSN: 2378-3656
Received: August 29, 2016 | Accepted: October 10, 2016 | Published: October 12, 2016
Citation: Grande G, Bertani H, Caruso A, Conigliaro R (2016) Aortoenteric Fistula in Patient with Infected Aortic Endovascular Stent. Clin Med Rev Case Rep 3:135. 10.23937/2378-3656/1410135
Copyright: © 2016 Grande G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Text
A 70-years-old man was admitted at the emergency room for onset of fever, malaise and discharge of melena. At the admission, his vital signs were: blood pressure 135/70 mmHg, heart rate 92 bpm, O2 saturation 95%. Blood sample showed mild normocyitc anaemia (Hb 12.1 g/dL), leucocytosis (White blood cells count 12.300/mmc, Neutrophil 77%), rise of C-reactive protein (7.3 mg/dL, normal value < 0.5 mg/L), normal kidney function (creatinine 1.0 mg/dL). Clinical examination was remarkable for mild epigastric pain.
Eight months before, the patient underwent to endovascular aneurism repair (EVAR) of abdominal aorta, placing prosthetic aortic endovascular stent (Zilver stent 518, Cook Inc., USA). For what concern pharmacologic anamnesis, at the moment of the admission the patient took aspirin, antihypertensive and hypocholesterolemic agents.
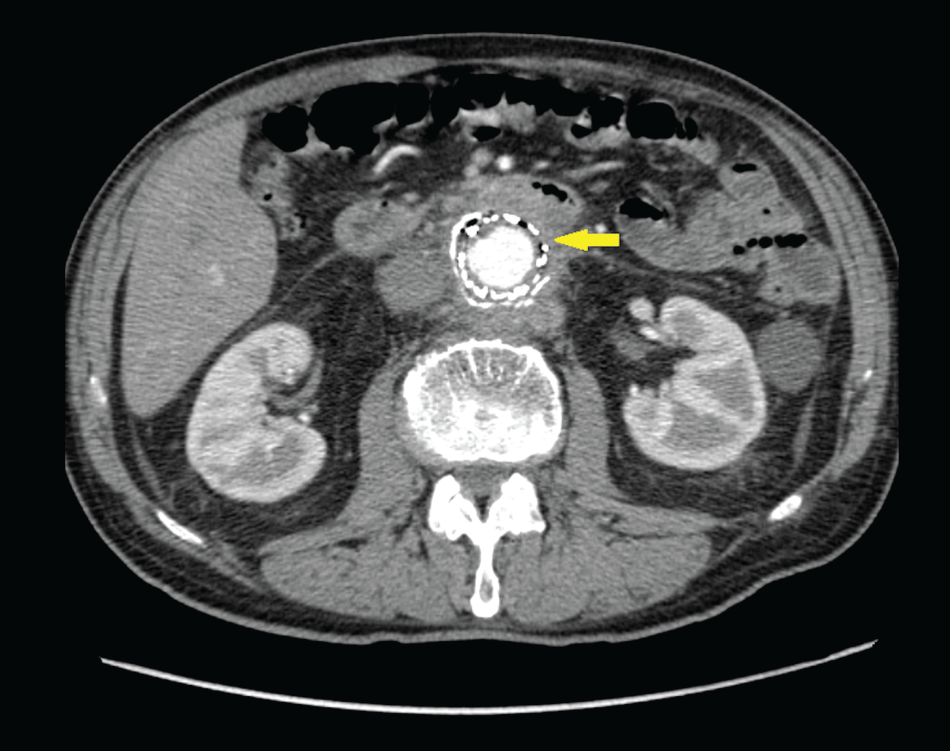
After first medical evaluation, a contrast-enhanced abdominal CT was performed, showing air microbubbles around aortic endovascular prosthesis without clear communication between aortic and duodenal wall (Figure 1).
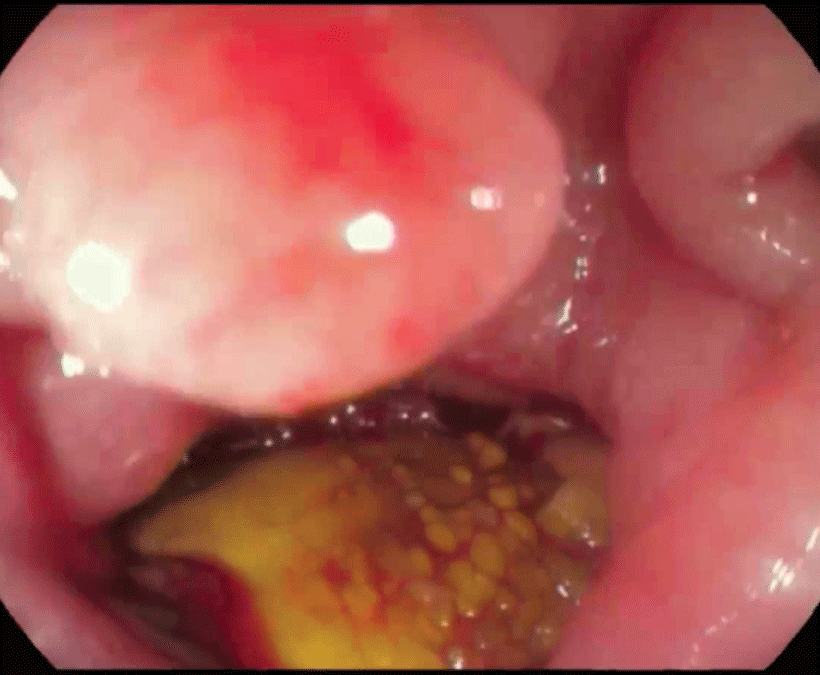
Given the suspicious of aorto-enteric fistula (AEF), an urgent upper endoscopy was performed, showed ulcerated, actively bleeding lesion within metal mesh of endovascular stent outcropping in the distal duodenal wall (Figure 2) (Video).
.
Figure 2: Upper endoscopy showed metal mesh of endovascular stent outcropping in the distal duodenal wall, confirming secondary aortoenteric fistula.
View Figure 2
On this basis, the patient was referred to open laparotomy, performing initial extra-antomical axillo-bifemoral bypass, followed by aortic endarterectomy, removing the infected aortic stent and positioning a Dacron polyester tube graft. Even more, the duodenal orifice of AEF was resected and the leak sutured.
In the days after, the patient didn't show melena anymore. However, the renal function became to fail and 15 days later, the patient was moved to intensive care unit (ICU) and he was subjected to haemodialysis. The most likely cause of renal failure was the partial or complete exclusion of both renal arteries from the blood flow. Two months after abdominal the patient died for septic shock due to antibiotic-resistant Pseudomonas aeruginosa infection.
Secondary AEF is an infrequent but potentially lethal complication of patients with endovascular stent [1,2]. The pathogenesis of the AEF is not clear and different factors could play a role. In the case of open surgery, the most important are local infection, direct bowel injury during the surgical dissection, bowel injury by anastomotic pseudoaneurysm or sutures, and fibrous contact between the graft body and bowel leading to pulse synchronous repetitive trauma [3]. Among them, the prosthesis infection seems to be the stronger. In fact, during open abdominal aortic aneurysm (AAA) repair, even a minimal bacterial translocation, attachment and biofilm formation on the vascular prosthesis (difficult to reach by host defence and antibiotic therapy), sustains bacterial colonization and local spreading of the infection that can result in intestinal necrosis and the formation of a fistula between the aneurysm and the intestinal wall [4,5]. The pathogenetic mechanism of AEF development after EVAR may be related to mechanical failure of the stent-graft such as rupture or migration with kinking of the device in the aneurysmal sack, which leads to aortic graft erosion and pressure necrosis of the bowel wall [1]. Another mechanism witch can lead to AEF appeared to be related to transient bacteremia with endograft seeding and local spreading of the infection [6].
The exact incidence of secondary AEF is not known, but it seems to be around 1% [3], occurring from several weeks or months to several years following first EVAR procedure [5]. The third or fourth duodenal segment is the most frequently involved site [5]. Once AEF was developed, the communication permits the passage of the blood flow into the bowel lumen and at the same time it allows the spreading of intestinal microorganism into the systemic circulation. Therefore, in the patients carrying aortic endovascular stent with clinical signs of sepsis and gastrointestinal bleeding, the presence of AEF should be kept a high suspicion [7], even if the classic triad of gastrointestinal bleeding, pulsatile abdominal mass, and pain is present in only a quarter of cases. The main symptom is “herald” haemorrhage from the upper gastrointestinal tract, whereas bleeding recur over a period of hours, days, or weeks and sometimes culminating in massive haemorrhage and hypovolemic shock [3,8].
Unfortunately, despite early diagnosis with aggressive antibiotic and surgical treatment, overall AEF mortality rate remains between 10% and 50% [4].
Conflicts of Interest
The authors disclose no conflicts.
Video
Urgent upper endoscopy was performed in the suspicious of AEF. No others sources of bleeding were found in the esophagus, stomach or proximal duodenal tract. A 10 mm, slightly bleeding orifice was found in the third duodenal portion and the prosthetic mesh were visible through the orifice.
References
-
Tagowski M, Vieweg H, Wissgott C, Andresen R (2014) Aortoenteric Fistula as a Complication of Open Reconstruction and Endovascular Repair of Abdominal Aorta. Radiology Research and Practice 2014.
-
Geraci G, Pisello F, Volsi FL, Facella T, Platia L, et al. (2008) Secondary aortoduodenal fistula. World J Gastroenterol 14: 484-486.
-
Bergqvist D, Björck M (2009) Secondary arterioenteric fistulation: a systematic literature analysis. Eur J Vasc Endovasc Surg 37: 31-42.
-
de Donato G, Setacci F, Galzerano G, Ruzzi U, Setacci C, et al. (2014) Prosthesis infection: prevention and treatment. J Cardiovasc Surg (Torino) 55: 779-792.
-
Saratzis N, Saratzis A, Melas N, Ktenidis K, Kiskinis D (2008) Aortoduodenal fistulas after endovascular stent-graft repair of abdominal aortic aneurysm: single-center experience and review of the literature. J Endovasc Ther 15: 441-448.
-
Murphy EH, Szeto WY, Herdrich BJ, Jackson BM, Wang GJ, et al. (2013) The management of endograft infections following endovascular thoracic and abdominal aneurysm repair. J Vasc Surg 58: 1179-1185.
-
Hye RJ, Inui TS, Anthony FF, Kiley ML, Chang RW, et al. (2015) A multiregional registry experience using an electronic medical record to optimize data capture for longitudinal outcomes in endovascular abdominal aortic aneurysm repair. J Vasc Surg 61: 1160-1166.
-
Simon T, Feller E (2011) Diverse Presentation of Secondary Aortoenteric Fistulae. Case Reports in Medicine 2011.