Clinical Medical
Reviews and Case Reports
Ocular Lyme Disease Simulating Acute Posterior Multifocal Placoid Pigment Epitheliopathy
Mingwu Wang1*, Rahul N. Khurana2, Jill J. Hopkins3 and Narsing A. Rao4
1Department of Ophthalmology and Vision Science, University of Arizona College of Medicine, USA
2Northern California Retina-Vitreous Associates, USA
3Genentech, Burlingame, USA
4USC Eye Institute, Department of Ophthalmology, University of Southern California Keck School of Medicine, USA
*Corresponding author: Mingwu Wang, Department of Ophthalmology and Vision Science, University of Arizona College of Medicine, 655 N. Alvernon way, Suite 108, Tucson, Arizona 85711, USA, Tel: (520) 321-3677, Fax: (520) 321-3665, E-mail: mwang@eyes.arizona.edu
Clin Med Rev Case Rep, CMRCR-2-021, (Volume 2, Issue 3), Case Report; ISSN: 2378-3656
Received: January 26, 2015 | Accepted: March 10, 2015 | Published: March 12, 2015
Citation: Wang M, Khurana RN, Hopkins JJ, Rao NA (2015) Ocular Lyme Disease Simulating Acute Posterior Multifocal Placoid Pigment Epitheliopathy. Clin Med Rev Case Rep 2:021. 10.23937/2378-3656/1410021
Copyright: © 2015 Wang M. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A 27-year old man presented with ocular symptoms and signs of acute posterior multifocal placoid pigment epitheliopathy (APMPPE) who was also confirmed to have acute Lyme Disease (LD) by serology. The patient was treated timely for systemic LD and the APMPPE-like ocular findings regressed. Possible mechanism linking APMPPE and LD was discussed in conjunction with acute syphilitic posterior placoid choroiditis, another spirochete infection.
Keywords
Ocular Lyme disease, Acute posterior multifocal placoid pigment epitheliopathy
Introduction
Lyme Disease (LD) is a multisystem bacterial infection caused by the spirochete Borrelia burgdorferi [1]. A variety of ocular manifestations in LD have been described, including multifocal choroiditis [2-5]. There have been several case reports, suggesting an association between acute posterior multifocal placoid pigment epitheliopathy (APMPPE) and LD [6,7]. However, a study of 18 cases of APMPPE found that none had serum antibodies against Borrelia burgdorferi [8]. We herein describe a patient with confirmed LD who presented with ocular features resembling APMPPE.
Case Report
A 27-year old man presented with central scotomas and photopsia in the left eye. He reported fevers, generalized malaise and some joint discomfort for the past month. He is an avid hiker in the east coast although he denied any known incidence of tick bites or skin rash such as erythema chronicum migrans. On examination, his best-corrected visual acuities were 20/20 in right eye and 20/80 in left eye. There was no relative afferent pupillary defect. Slit lamp examination of the anterior segments of the eyes was unremarkable. Dilated fundoscopic examination disclosed multifocal yellowish white placoid lesions located in the posterior poles of both eyes at the level of retinal pigment epithelium (RPE) (Figure 1A). Intravenous fluorescein angiography (IVFA) revealed hypofluorescent lesions in the early phase (Figure 1B) with late phase staining of the lesions (Figure 1C), characteristic of APMPPE.
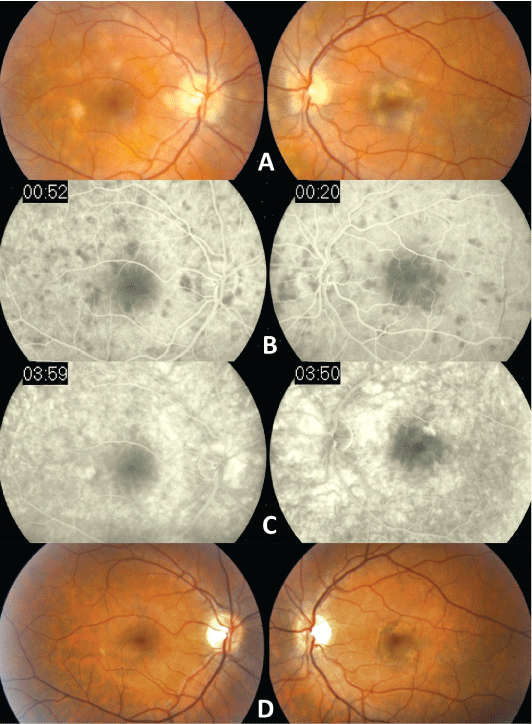
Figure 1: Fundoscopic photos showed multiofocal yellowish white placoid
lesions located in the posterior poles of both eyes, more in the left eye, at the
level of retinal pigment epithelium
(A). Fluorescein angiography revealed hypofluorescence of these lesions
in the early phase (B) with late phase staining of some of such lesions (C).
After 6 months, the previously yellowish white lesions regressed to faint
chorioretinal lesions with hyperpigmentation of the affected areas in both
eyes (D).
View Figure 1
The initial laboratory work up was significant for a positive ELISA Lyme titer of 3.4. The diagnosis of LD was then confirmed with Western blot showing 5 positive IgG bands (18KD, 39KD, 41KD, 58KD, and 93KD) which is diagnostic for LD [9]. Other laboratory results were normal, including negative reactive plasma regain, angiotensin-converting enzyme, Bartonella henslae, herpes simplex and varicella zoster titers, and normal levels of antinuclear antibody, C3 and C5. An MRI of the head and X-ray of the chest were unremarkable as well. A vitreous tap in search of Borrelia burgdorferi was declined by the patient, due to its invasiveness and uncertain benefit in guiding management in the presence of positive serology. The patient was immediately treated with intravenous ceftriaxone (2 gram per day) for 4 weeks and his left eye vision recovered to 20/20. At follow-up in 6 months, the yellowish white lesions regressed to faint chorioretinal lesions with hyperpigmentation of the affected areas (Figure 1D).
Comments
Ocular manifestations of the LD resemble that of ocular syphilis caused by another spirochete Treponema pallidum [10]. Although Borrelia burgdorferi and its DNA have been isolated from vitreous and iris specimens [10], the exact pathogenesis of ocular LD is still poorly understood. We believe ocular LD and syphilis may share similar pathogenic mechanisms. In describing acute syphilitic posterior placoid choroiditis (ASPPC), Gass et al. indicated striking similarities between ASPPC and APMPPE [11]. In addition to similar chorioretinal findings, IVFA of ASPPC often shows characteristic early phase hypofluorescence in areas of lesions which, in late phase, demonstrate staining at the RPE level [11]. The lesions of both disorders have a tendency to fade completely in 9 to 14 days. The ophthalmoscopic and angiographic findings of ocular LD may be due to active inflammatory reaction at the level of choriocapillaries and RPE. Such a reaction could be caused by deposition of soluble immune complexes, or by inflammation in response to disseminated spirochetes to the choriocapillaries, which in turn leads to focal nonperfusion � a mechanism believed to cause APMPPE [12]. Retinal vasculitis and chorioretinitis are indeed common findings in ocular LD [2,4,13].
Ocular manifestations of LD are similar to those of syphilis [10,14], and hence can readily mimic other ocular disorders. Although more evidence is needed to firmly link LD to APMPPE, in a patient with findings of APMPPE who has risk factors and presents clinical features suggesting Lyme infection, ocular LD should be considered. Due to high false positive rate, confirmatory tests [9] should be pursued and the patients treated timely. Anti-Borrelia burgdorferi antibody concentrations often decline, even to undetectable level, in patients with culture- or PCR-proven Lyme borreliosis [15]. This may explain why there were no antibodies detected against Borrelia burgdorferi in the 18 patients presented with APMPPE in one study [8]. Therefore, Western blot or detection of borrelial DNA by PCR would provide definitive diagnosis. Intraocular inflammation caused by LD is usually treated according to the regimen for neuroborreliosis [16]. To date, oral administration of drugs such as doxycyclin and intravenous treatment with beta lactam agents have been used in courses ranging from 10 to 28 days, without significant different outcomes [17].
References
-
Steere AC (2001) Lyme disease. N Engl J Med 345: 115-125.
-
Mikkil� HO, Sepp�l� IJ, Viljanen MK, Peltomaa MP, Karma A (2000) The expanding clinical spectrum of ocular lyme borreliosis. Ophthalmology 107: 581-587.
-
Howlett JM, Booth AP (2012) Ocular inflammation as a manifestation of Lyme borreliosis. BMJ 345: e4721.
-
Colucciello M (2001) Ocular Lyme borreliosis. N Engl J Med 345: 1350-1351.
-
Mora P, Carta A (2009) Ocular manifestations of Lyme borreliosis in Europe. Int J Med Sci 6: 124-125.
-
Bodine SR, Marino J, Camisa TJ, Salvate AJ (1992) Multifocal choroiditis with evidence of Lyme disease. Ann Ophthalmol 24: 169-173.
-
Framme C, Sachs HG, Gabler B, Roider J (2002) Fundus autofluorescence in APMPPE in association with lyme disease. Retina 22: 653-657.
-
Wolf MD, Folk JC, Nelson JA, Peeples ME (1992) Acute, posterior, multifocal, placoid, pigment epitheliopathy and Lyme disease. Arch Ophthalmol 110: 750.
-
Borchers AT, Keen CL, Huntley AC, Gershwin ME (2015) Lyme disease: A rigorous review of diagnostic criteria and treatment. J Autoimmun 57C: 82-115.
-
Karma A, Mikkil� H (1996) Ocular manifestations and treatment of Lyme disease. Curr Opin Ophthalmol 7: 7-12.
-
Gass JD, Braunstein RA, Chenoweth RG (1990) Acute syphilitic posterior placoid chorioretinitis. Ophthalmology 97: 1288-1297.
-
Dhaliwal RS, Maguire AM, Flower RW, Arribas NP (1993) Acute posterior multifocal placoid pigment epitheliopathy. An indocyanine green angiographic study. Retina 13: 317-325.
-
Mikkil� H, Sepp�l� I, Leirisalo-Repo M, Immonen I, Karma A (1997) The etiology of uveitis: the role of infections with special reference to Lyme borreliosis. Acta Ophthalmol Scand 75: 716-719.
-
Klig JE (2008) Ophthalmologic complications of systemic disease. Emerg Med Clin North Am 26: 217-231.
-
Oksi J, Uksila J, Marjam�ki M, Nikoskelainen J, Viljanen MK (1995) Antibodies against whole sonicated Borrelia burgdorferi spirochetes, 41-kilodalton flagellin, and P39 protein in patients with PCR- or culture-proven late Lyme borreliosis. J Clin Microbiol 33: 2260-2264.
-
Karma A, Sepp�l� I, Mikkil� H, Kaakkola S, Viljanen M, et al. (1995) Diagnosis and clinical characteristics of ocular Lyme borreliosis. Am J Ophthalmol 119: 127-135.
-
Halperin JJ, Shapiro ED, Logigian E, Belman AL, Dotevall L, et al. (2007) Quality Standards Subcommittee of the American Academy of N. Practice parameter: treatment of nervous system Lyme disease (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 69: 91-102.





