Clinical Medical
Reviews and Case Reports
Paediatric Hodgkin Lymphoma and Radiation Therapy: 3DCRT vs VMAT. A Case Report
Annalisa Nardone1, Leonarda Rella2*, Anna Milella1, Antonio Pascali1, Pasquale Tamborra3, Michele Telegrafo2 and Marco Lioce3
1Department of Oncologic Radiotherapy, IRCCS Istituto Tumori Giovanni Paolo II, Italy
2DIM - Section of Diagnostic Imaging and Radiotherapy, University of Bari Medical School, Italy
3Department of Medical Phisics, IRCCS Istituto Tumori Giovanni Paolo II, Italy
*Corresponding author: Leonarda Rella, DIM - Section of Diagnostic Imaging and Radiotherapy, University of Bari Medical School, Bari, Italy, Viale Orazio Flacco 65, 70124 Bari, Italy, Tel: 00390805555211, Fax: 00390805555239, E-mail: lea.rella@gmail.com
Clin Med Rev Case Rep, CMRCR-2-026, (Volume 2, Issue 4), Case Report; ISSN: 2378-3656
Received: December 16, 2014 | Accepted: March 27, 2015 | Published: April 02, 2015
Citation: Nardone A, Rella L, Milella A, Pascali A, Tamborra P, et al. (2015) Paediatric Hodgkin Lymphoma and Radiation Therapy: 3DCRT vs VMAT. A Case Report. Clin Med Rev Case Rep 2:026. 10.23937/2378-3656/1410026
Copyright: © 2015 Nardone A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Hodgkin Lymphoma (HL) is one of the most common cancers in young adult. Early stage Hodgkin disease has a good prognosis and high survival rates with modern therapeutic approaches. Clinical management of early stage HL involves two therapeutic options: one is combined modality therapy with abbreviated chemotherapy plus involved field radiation therapy (IFRT) and the other is chemotherapy alone. Combined modality ensures high cure rates but exposes young HL survivors to short-term and long-term complications such as cardiovascular and pulmonary disease and second malignancies especially in case of mediastinal involvement. Radiation therapy is crucial in the management of HL and has changed over the last decades. The introduction of new irradiation techniques such as Intensity modulated Radiation Therapy (IMRT) and Volumetric Modulated Arc Therapy (VMAT), the delivery of lower doses regimens and the reduction of field sizes from extended field radiotherapy (EFRT ) to involved field RT (IFRT) and involved �nodal RT (INRT) allow to choose the most effective and safest technique. We present a case report of a paediatric HL, stage II bulky mediastinal, already treated with different chemotherapy regimens containing bleomicyn and candidate to autologous transplantation in order to demonstrate which radiation technique is more effective and safer for lungs between 3D conformational therapy (3DCRT) and VMAT. VMAT seems safer for better lung sparing.
Keywords
Hodgkin disease, Combined modality, Radiation therapy, Long-term toxicity
Introduction
Hodgkin Lymphoma (HL) is one of the most common cancers in young adults with an annual incidence of approximately 3 of 100.000 in US and 5-7 of 100.000 in Italy [1,2]. Classical Hodgkin lymphoma includes 4 different subtypes: nodular sclerosis (NSHL), Mixed Cellularity (MCHL), lymphocyte depleted (LDHL) and lymphocyte rich (LRHL). Clinical presentation, therapeutic management and prognosis of HL allow identifying two subgroups of Hodgkin disease (HD): early stage defined as IA to IIA stage with no bulky or B symptoms (favourable disease) and advanced stage disease defined as stage I to II unfavourable with bulky or B symptoms and stage III to IV [3]. Early stage HD has an excellent prognosis with survival rates approaching 95% with modern therapeutic options [4].
Clinical management of early stage HD (favourable disease) is variable consisting mainly in two therapeutic approaches: one is the combined modality therapy with abbreviated chemotherapy with 2 or 4 ABVD cycles (doxorubicin, bleomycin, vinblastine, and dacarbazine) or Stanford V protocol for 8 weeks plus 20-30 Gy of involved field radiation therapy (IFRT) and the other is chemotherapy alone.
Patients with stage I/II with bulky mediastinal disease are typically treated with six to eight cycles of ABVD plus FRT [3] or Stanford V protocol for 12 weeks or according to the German Hodgkin Study Group (GHSG) HD14 study two cycles of escalated doses of bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone (Esc BEACOPP) plus two cycles of ABVD followed by IFRT [5].
HD is a curable disease and many patients are less than 40 years old at diagnosis so it is very important to evaluate all potential complications of each therapeutic option. Radiation therapy of mediastinal HD has acute and late effects. Approximately 95% of children and young adults with this stage and 85% of patients with advanced-stage disease are treated with chemotherapy and radiation therapy [6,7]. A proper evaluation of treatment-induced short term effects like interstitial pneumonitis is mandatory especially for children with mediastinal HD receiving combined-modality therapy and who are candidate to preparative chemotherapy to peripheral blood stem cell (PBSC) transplantation in case of refractory or relapsed HD. Late complications (3-40 years) include cardiovascular and pulmonary disease, secondary leukemia and solid tumors arising in radiated fields, soft tissue effects [8-10] and the risk for radiation-related cancer and for cardio-pulmonary complications is proportional to the irradiated volume [11].
Radiation therapy for HD has changed over the last few decades in terms of irradiated volumes, doses and techniques [12,13] becoming increasingly central in the management of this disease. The introduction of new irradiation techniques such as Intensity modulated Radiation Therapy (IMRT) and Volumetric Modulated Arc Therapy (VMAT), the delivery of lower doses regimen with 30 Gy or less obtaining similar results in combined modality than previous 35-45 Gy schedule [10,14,16] and the reduction of field sizes from extended field radiotherapy (EFRT ) to involved field RT (IFRT) and involved �nodal RT (INRT) [17-19] allow to the radiation oncologist to choose the most effective technique with less side effects.
Case Report
A 12�year old girl presented to us in July 2011 with 5 months before hyperpyrexia not responding to antibiotic therapies, left painful laterocervical adenopathy measuring 7 cm and left supraclavicular and axillary adenopathies, sweats, asthenia and arthralgias at lower limbs. Cervical lymph nodes biopsy was diagnostic of Hodgkin�s lymphoma, nodular slerosis type, CD 15 and CD 30 positive.
Contrast chest CT revealed multiple mediastinal lymphadenopathies: bilateral laterocervical, supraclavicular, especially on left side with compression of the ipsilateral jugular vein, left axillary adenopathies. Global mediastinal lymph node involvement had a transverse diameter of 10 cm in the area of the thoracic outlet. PET scan revealed increased uptake in the above mentioned nodes mostly in the laterocervical regions (SUV max 8.3). Her chest X-ray examination showed mediastinal widening: mediastinal to thoracic ratio (M/T ratio) of 0.47.
She was diagnosed with Hodgkin�s lymphoma, nodular sclerosis type, unfavourable stage II and was planned to start COPP/ABV chemotherapy (cyclophosphamide, vincristine, procarbazine, prednisone, doxorubicin, bleomycin and vinblastine) according to the Italian Association of Pediatric Hematology and Oncology Protocol (AIEOP-LH 2004), second therapeutic group . After 2 cycles of COPP/ABV she repeated chest CT revealing a reduction of global mediastinal involvement (4.5 cm of diameter) and PET scan showing less sites of increased uptake with a lower intensity of uptake (SUV max 6 for supraclavicular bilateral regions). Chest X Rays documented a M/T ratio of 0.34.
She continued with other two cycles of COPP/ABV and then she had again chest CT and PET-CT revealing no significant change. In consideration of these data, the haematologist decided to start on new chemotherapy according to IEP protocol (ifosfamide, etoposide, prednisone) and to make subsequent PBSC transplantation. She received two cycles of IEP and subsequent PET TC revealed persistence of previously detected lymphadenopathies (SUV max 6.7) and CT pointed out a volumetric increase of the mediastinal adenopathic mass with a transverse diameter of 5 cm and enlargement of bilateral laterocervical lymphadenopathies and left supraclavicular nodes. In consideration of disease progression, the patient was addressed to DHAP (dexamethasone, high dose cytarabine and cisplatin) chemotherapy. After two cycle HLA typing was performed and subsequent chest CT and PET-CT showed persistence of laterocervical, subclavicular and upper mediastinal adenopathies.
She presented to us to perform mediastinal RT. We had two available techniques: 3DCRT and VMAT. We decided for VMAT in order to preserve the largest lung volume as possible because the patient had already been subjected to drugs potentially toxic for lungs, such as bleomycin, cyclophosphamide, vinblastine, contained in COPP-ABV regimen and she was candidate to haematopoietic stem cell transplant with further risk of pulmonary infectious and non infectious complications [20] and radiation-induced lung toxicity.
The young patient was simulated supine in an individualized thermoplastic mask with the arms close to the hips. All of the CT images were acquired with 16-row multislice CT, Big Bore, Siemens and transferred to and registered in the treatment planning system (TPS).
The RT treatment volume CTV was contoured according to the AIEOP-LH 2004 Protocol and encompassed both pre-chemotherapy involved lymph nodes showed with chest CT and PET TC and post-chemotherapy CT and PET-TC residual lymph nodes.
An additional margin of 2 cm was added to CTV in all directions for physiological movement and set-up error to form the planning target volume.
The RT dose was 25.2 Gy to PTV including upper mediastinum, bilateral supraclavicular regions and left laterocervical region (II-III-IV levels) and 36 Gy (25.2 + boost of 10.8 Gy) to residual masses > 50 cc to be delivered in 20 fractions (1.8 Gy per fraction).
The organs at risk (OARs) included: lungs (ipsilateral and contralateral), spinal cord, esophagus, heart, breasts, thyroid. The dose-volume constraints for the lungs were set as follows: V20< 20%, V15< 33% and mean lung dose < 20 Gy. The objective of planning was to deliver the 95% of the prescribed dose to at least 98% of the PTV with a maximum dose (Dmax) to the target not exceeding 105%. We obtained that the 95% of the dose was given to99 % of the target and D1% (maximum dose given to 1% of the PTV) has been 104% of the prescribed dose. The plan was optimized plans with Rapid Arc Optimizer PRO 8.6.15 version and calculated with Algoritm AAA version 8.6.15. Follow up CT examinations showed a remission of the disease and the patient is now disease free.
Discussion
The choice of the radiation therapy optimal technique for HL bases on some aspects including the reduction of treatment volume to only involved node station, the total delivered dose, the reduction of the amount of healthy tissue exposed to radiation, the use of highly conformal RT technique such as Varian Rapid Arc, treatment time duration, dose-volume constraints for OARs.
Regarding to the irradiated volumes, we used the current approach for radiation therapy involving node radiotherapy (INRT) instead of involved field radiotherapy (IFRT). This modality introduced by Girinsky et al. [18] is associated with less risk of late effects because the irradiated volume is limited to initially involved lymph nodes and excludes the adjacent uninvolved nodal groups in order to reduce the volumes of OAR exposed to radiation [4]. Some studies reported that INRT show no statistical difference in progression-free survival and overall survival at 10 years compared with IFRT [19] and it has been adopted in randomized trials of favourable risk limited stage HD by EORTC and GHGS. A new concept, involved site radiation therapy (ISRT), has been introduced, although not yet validated in a formal study; it is more conservative than INRT, accounting for suboptimal information wherein optimal imaging is not available [21].
The choice between Varian Rapid Arc and 3D CRT plans was not so simple and immediate as it could be in an adult patient case. Rapid Arc (RA) technique allows higher conformity dose and homogeneity than 3DCRT technique, smearing low doses on larger volumes and lowering the probabilities for OARs complications. Unfortunately, larger irradiated volumes imply always a higher risk of radio-induced tumors. This risk is more important in pediatric patients who have a longer life expectance.
Moreover, as VMAT plans deliver higher dose gradients than 3DCRT plans, it is increased the risk of target missing especially for treatments of thoracic district. The obvious consequent need for shorter treatment times, induced us to prefer only VMAT plans to IMRT ones. In fact the latter have a similar quality of the former, but remarkable longer treatment times associated to larger Monitor Units (MU).
Evaluating the advantages and limitations of Rapid Arc plan adoption, we considered the dose constraints for OARs , the risk of target missing, the risk of secondary malignancies .
Although the risk for secondary malignancies is calculated always with theoretical model [22] and the risk for target missing could be minimized with gating techniques such as deep-inspiration breath-hold (DI) [23] that need an implementation in our institute, the only reliable data are on heart and lung toxicity which we considered as the most important to reduce late effects for thoracic structures. In fact, Filippi et al. found no difference between 3DCRT and VMAT regarding the organ equivalent dose in female HL patients [22] while Schneider et al. calculated even higher risks for secondary malignancies with VMAT in their series; in our case, however, only Rapid Arc plan guaranteed acceptable values of V20, V15, mean dose for lungs and for heart.
Analyzed parameters for PTV 25 included DMax (D1%) and DMin (95%), homogeneity range (D5%-95%), Conformity Index (C.I.) and Conformation Number (C.N.) for a 3DCRT plan and a RA one (Table 1).
![]()
Table 1: Parameters analyzed for PTV 25 Gy
View Table 1
DMax was lower in RA plan than in 3DCRT one, DMin was higher in RA plan than 3DCRT one. Homogeneity range for RA plan was 1.42, lower than 3DCRT plan where we found 1.85 as homogeneity range. C.I. and C.N. were better with RA plan than 3DCRT one. Dose distribution for 20 Gy isodose for RA plan is shown in Figure 1a, for 3DCRT plan is shown in Figure 1b.
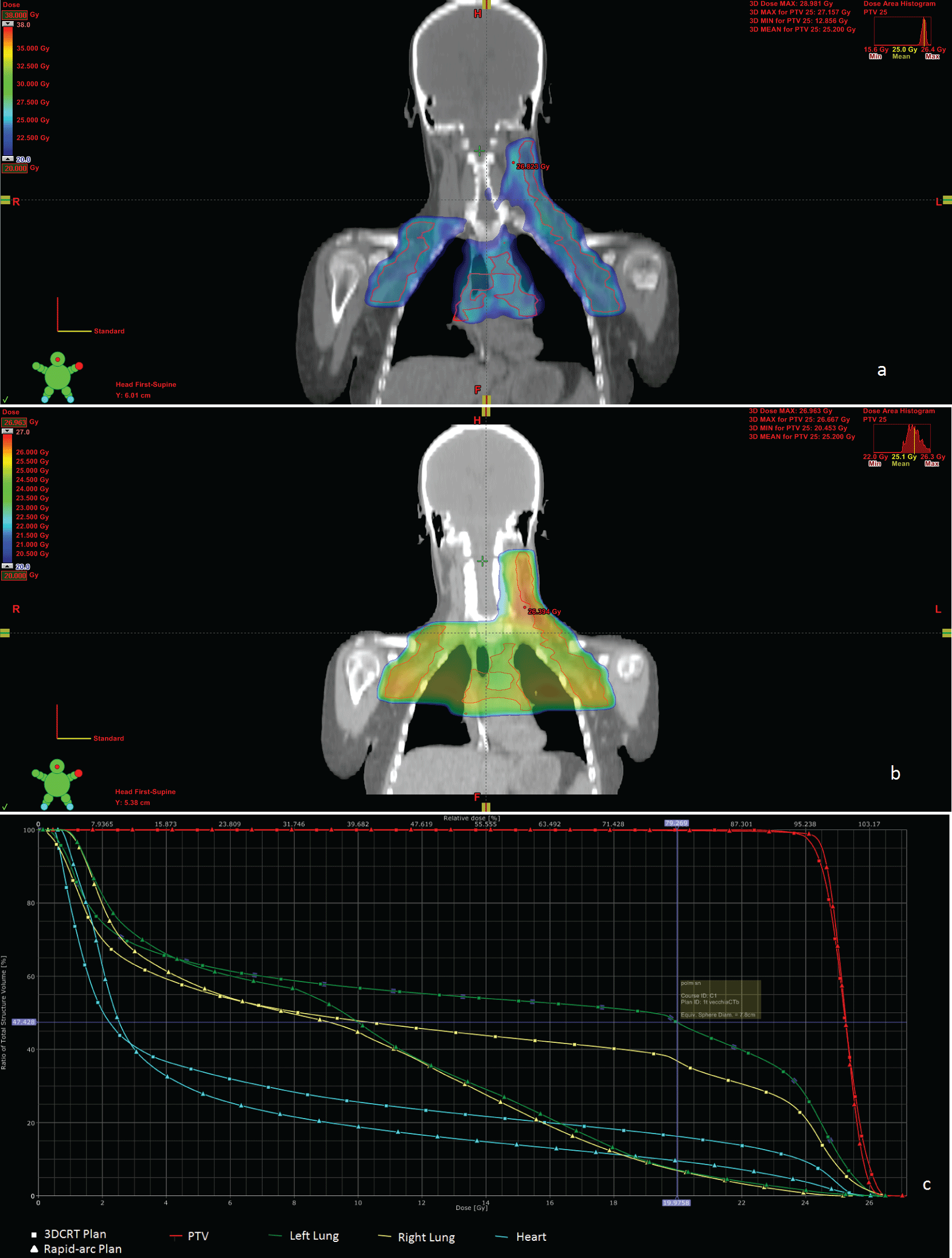
Figures 1: Dose distribution for 20 Gy isodose for Rapid Arc plan (a). and for 3DCRT plan (b). Cumulative dose-volume histograms (DVHs) for both plans (c).
DVH for lungs (20 Gy isodose) showed larger volume receiving this dose with 3DCRT compared to RA plan both for the left and the right lung. DVH for the heart
showed larger volume receiving 15 Gy with 3DCRT plan than RA plan.
View Figure 1
Quantitative evaluations were performed by means of cumulative dose-volume histograms (DVHs) for both plans (Figure 1c). DVH for lungs (20 Gy isodose) showed larger volume receiving this dose with 3DCRT compared to RA plan both for left and right lung. DVH for heart showed larger volume receiving 15 Gy with 3DCRT plan than RA plan.
About PTV coverage we found that 95% of prescribed dose covers 99% of target in RA plan and 98,3% in 3DCRT plan; the 98% of prescribed dose covers 88,3% with RA plan vs 82,5% with 3DCRT plan.
In conclusion, VMAT showed better target coverage, superior conformity around the target and better lung sparing in the presented case.
References
-
Bauer K, Herbst C, Brillant C, Monsef I, Kluge S, et al. (2010) Eleventh biannual report of the Cochrane Haematological Malignancies Group: Focus on Hodgkin lymphoma. J Natl Cancer Inst 102: E1.
-
Wethal T, Lund MB, Edvardsen T, Foss� SD, Pripp AH, et al. (2009) Valvular dysfunction and left ventricular changes in Hodgkin's lymphoma survivors. A longitudinal study. Br J Cancer 101: 575-581.
-
Hoppe RT, Advani RH, Ai WZ, Ambinder RF, Aoun P, et al. (2012) Hodgkin lymphoma, version 2.2012 featured updates to the NCCN guidelines. J Natl Compr Canc Netw 10: 589-597.
-
Campbell BA, Voss N, Pickles T, Morris J, Gascoyne RD, et al. (2008) Involved-nodal radiation therapy as a component of combination therapy for limited-stage Hodgkin's lymphoma: a question of field size. J Clin Oncol 26: 5170-5174.
-
von Tresckow B, Pl�tschow A, Fuchs M, Klimm B, Markova J, et al. (2012) Dose-intensification in early unfavorable Hodgkin's lymphoma: final analysis of the German Hodgkin Study Group HD14 trial. J Clin Oncol 30: 907-913.
-
Schwartz CL, Constine LS, Villaluna D, London WB, Hutchison RE, et al. (2009) A risk-adapted, response-based approach using ABVE-PC for children and adolescents with intermediate- and high-risk Hodgkin lymphoma: the results of P9425. Blood 114: 2051-2059.
-
Herbst C, Rehan FA, Skoetz N, Bohlius J, Brillant C, et al. (2011) Chemotherapy alone versus chemotherapy plus radiotherapy for early stage Hodgkin lymphoma. Cochrane Database Syst Rev : CD007110.
-
Ng AK, Bernardo MV, Weller E, Backstrand K, Silver B, et al. (2002) Second malignancy after Hodgkin disease treated with radiation therapy with or without chemotherapy: long-term risks and risk factors. Blood 100: 1989-1996.
-
Khimani N, Chen YH, Mauch PM, Recklitis C, Diller L, et al. (2013) Influence of new late effects on quality of life over time in Hodgkin lymphoma Survivors: a longitudinal survey study. Ann Oncol 24: 226-230.
-
Eich HT, Diehl V, G�rgen H, Pabst T, Markova J, et al. (2010) Intensified chemotherapy and dose-reduced involved-field radiotherapy in patients with early unfavorable Hodgkin's lymphoma: final analysis of the German Hodgkin Study Group HD11 trial. J Clin Oncol 28: 4199-4206.
-
De Bruin ML, Sparidans J, van't Veer MB, Noordijk EM, Louwman MW, et al. (2009) Breast cancer risk in female survivors of Hodgkin's lymphoma: lower risk after smaller radiation volumes. J Clin Oncol 27: 4239-4246.
-
Meyer RM, Hoppe RT (2012) Point/counterpoint: early-stage Hodgkin lymphoma and the role of radiation therapy. Blood 120: 4488-4495.
-
Engert A, Pl�tschow A, Eich HT, Lohri A, D�rken B, et al. (2010) Reduced treatment intensity in patients with early-stage Hodgkin's lymphoma. N Engl J Med 363: 640-652.
-
Yeoh KW, Mikhaeel NG (2011) Role of Radiotherapy in Modern Treatment of Hodgkin's Lymphoma. Adv Hematol 2011: 258797.
-
Eichenauer DA, Engert A, Andr� M, Federico M, Illidge T (2011) Hodgkin's lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 22:55-58.
-
Ferme C, Eghbali H, Meerwaldt JH, Rieux C, Bosq J, et al. (2007) Chemotherapy plus involved-field radiation in early-stage Hodgkin's disease. N Engl J Med 357: 1916-1927.
-
(1999) NCCN practice guidelines for Hodgkin's disease. National Comprehensive Cancer Network. Oncology (Williston Park) 13: 78-110.
-
Girinsky T, van der Maazen R, Specht L, Aleman B, Poortmans P, et al. (2006) Involved-node radiotherapy (INRT) in patients with early Hodgkin lymphoma: concepts and guidelines. Radiother Oncol 79: 270-277.
-
Campbell BA, Hornby C, Cunninghame J, Burns M, MacManus M, et al. (2012) Minimising critical organ irradiation in limited stage Hodgkin lymphoma: a dosimetric study of the benefit of involved node radiotherapy. Ann Oncol 23: 1259-1266.
-
Gower WA, Collaco JM, Mogayzel PJ Jr (2010) Lung function and late pulmonary complications among survivors of hematopoietic stem cell transplantation during childhood. Paediatr Respir Rev 11: 115-122.
-
Specht L, Yahalom J, Illidge T, Berthelsen AK, Constine LS, et al. (2014) Modern radiation therapy for Hodgkin lymphoma: field and dose guidelines from the international lymphoma radiation oncology group (ILROG). Int J Radiat Oncol Biol Phys 89: 854-862.
-
Filippi AR, Ragona R, Fusella M, Botticella A, Fiandra C, et al. (2013) Changes in breast cancer risk associated with different volumes, doses, and techniques in female Hodgkin lymphoma patients treated with supra-diaphragmatic radiation therapy. Pract Radiat Oncol 3: 216-222.
-
Schneider U, Sumila M, Robotka J, Weber D, Gruber G, et al. (2014) Radiation-induced second malignancies after involved-node radiotherapy with deep inspiration breath-hold technique for early stage Hodgkin lymphoma : a dosimetric study. Radiation Oncology 9:58.





