International Journal of Anesthetics and Anesthesiology
A Comparative study on Efficacy of 10% and 20% Mannitol for Inducing Diuresis During Renal Transplant Surgery
Mohammed Husam Nafie*
Department of Anesthesia, Rizgary Teaching Hospital and Zheen International Hospital, Kurdistan, Iraq
*Corresponding author: Mohammed Husam Nafie, Department of Anesthesia, Rizgary Teaching Hospital and Zheen International Hospital, Kurdistan, Iraq, Tel: 00967504481926, E-mail: mhn.nafie@yahoo.com
Int J Anesthetic Anesthesiol, IJAA-2-036, (Volume 2, Issue 4), Original Research; ISSN: 2377-4630
Received: July 01, 2015 | Accepted: November 17, 2015 | Published: November 19, 2015
Citation: Nafie MH (2015) A Comparative study on Efficacy of 10% and 20% Mannitol for Inducing Diuresis During Renal Transplant Surgery. Int J Anesthetic Anesthesiol 2:036. 10.23937/2377-4630/2/4/1036
Copyright: © 2015 Nafie MH. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Mannitol is one of the most commonly used osmotic diuretics during renal transplant surgery. Two concentrations of mannitol, 10% and 20%, are available, however there is no consensus about which concentration is to be preferred. The purpose of this study was to compare between the efficacy of the two concentrations of mannitol for inducing diuresis during renal transplant surgery and the need for further diuretics (furosemide).
Methods: A prospective study was carried out on 60 patients of either sex undergoing living donor renal transplantation in Zheen International Hospital between April 2011 and May 2015 for the end-stage renal failure. Patients were assigned into two groups, group 1 received 10% mannitol and group 2 received 20% mannitol. All patients evaluated preoperatively by the anesthetist, immunosuppression was initiated the day before the surgery using combination of immunosuppressant drugs. General anesthesia induced with thiopentone and maintained with atracurium and isoflurane, vital parameters monitored, the donated kidney was placed retroperitoneally in the iliac fossa and anastomosis between renal and iliac vessels were established. Normal saline 0.9% used as intravenous fluid, once venous anastomosis started diuresis initiated with mannitol 10% or 20% (2-3 ml/kg). The urine output of the patients was monitored and furosemide was given as needed with the aim of at least 2 ml/kg/hr of urine output.
Results: The demographic characteristics between both study groups were comparable (p > 0.05). Patients treated with 10% mannitol (group 1) were found to require significantly less overall dose of mannitol as compared to patients received 20% mannitol (group 2) (p < 0.0001). Additionally, group 1 required significantly lower additional doses of furosemide as compared to group 2 (p < 0.0474). The duration of surgery, fluid requirement and urine output were comparable between both study groups (p > 0.05).
Conclusion: A lower dose of mannitol using lower concentration (10%) is more effective in terms of need of overall dose in grams for inducing diuresis with less need for furosemide compared to higher concentration (20%) of mannitol.
Keywords
Mannitol, Diuretics, Renal transplantation, Furosemide
Introduction
Osmotic diuretics are freely filterable at the glomerulus and undergo limited reabsorption or no reabsorption in the proximal tubules they limit the passive water reabsorption that normally follows active sodium reabsorption, in addition to their effect to increase water excretion, in large doses, they also increase electrolyte (sodium and potassium) excretion. The same process occurs in the loop of Henle [1]. Mannitol is a six-carbon non-metabolizable polyalcohol with a molecular weight of 182, is one of the oldest and most widely used osmotic diuretic. It is used orally and by intravenous route and supplied as sterile solutions of 10% and 20% concentrations for clinical use. It was introduced by Homer Smith in 1940 to estimate glomerular filtration rate (GFR) in humans and dogs and for the first time noted the osmotic diuretic effect of mannitol. In the half century that elapsed since then, mannitol is still in use for the prophylaxis against acute kidney injury [2].
Oliguria is considered a poor prognostic indicator in patients with acute kidney injury [3,4], frequently it leads to permanent damage to kidney and future fluid and electrolyte imbalance in patients. Diuretics used to improve urine output and facilitate fluid management in patients of acute kidney injury [5].
Mannitol has also been promoted as a renal protective agent especially in patients at high risk of developing acute kidney injury and renal failure, such as those undergoing cardiac and vascular surgeries, renal transplantation, and in patients with hepatic failure and rhabdomyolysis [6]. The overall evidences suggest that although mannitol increases urine output, it does not reduce the risk of acute kidney injury, but experiences and evidences for volume expansion in the setting of renal transplantation are encouraging and use of mannitol has shown to reduce the incidence of postoperative acute kidney injury in patient undergoing renal surgery [5].
The most effective drugs in producing diuresis in the patient with acute kidney injury are mannitol and the loop diuretics. A typical starting dose of mannitol (20%) is 1.25 to 2.5 g infused intravenously over 3 to 5 minutes. It has little non-renal clearance, so when given to anuric or oliguric patients, it remains in the circulation, potentially causing a hyperosmolar state. Mannitol is not metabolized and is excreted by glomerular filtration within 30-60 minutes, without any important tubular reabsorption or secretion. It should be used cautiously in patients with even mild renal insufficiency [7].
Many surgeons today use mannitol in a dose of 20% concentration with a target urine output of 2 ml/kg/hr. The 10% mannitol concentration is used less commonly and there are no universally accepted guidelines for preferential selection of any one. In addition to mannitol, many patients still require additional diuretics in the form of intravenous furosemide during renal surgery. The overall doses are selected as per requirement and there is no consensus among nephrologists about 10% versus 20% mannitol dose for selection and use for inducing diuresis and preventing acute kidney injury especially in transplanted kidney [2,8].
Still studies have shown that use of mannitol during donor kidney transplant can have positive effects in terms of prevention of delayed graph rejection. In contrast, it is a known fact that higher doses of mannitol can themselves causes acute kidney injury [9]. Therefore this prospective study was to compare between the efficacy and potency of 10% and 20% dose of mannitol in terms of diuresis and need for additional diuretics.
Materials and Methods
The study started after obtaining permission from the Institutional Ethics Committee of Zheen International Hospital, Kurdistan, Iraq. Written consent was taken from all the patients. The observed data was recorded in the patient's information sheet.
The proposed study was carried out at Department of Nephrology, Zheen International Hospital, Kurdistan, Iraq. The study population consisted of 60 patients with chronic kidney disease or end stage renal disease undergoing renal transplantation between April 2011 and May 2015. Patients of ages between 10-65 years of either sex were included. The sample size was selected based on the number of end stage renal disease patients admitted at our hospital per year and sample size for minimal statistical requirement.
All patients with other serious co-morbid cardiac or liver or any other systemic disease were excluded from the study. The surgical procedure followed was as per the protocol of Hospital. Patients were divided into two groups of 30 patients per group. All patients underwent investigations including complete blood count, bleeding and clotting time, prothrombine time and partial thromboplastin time, INR, liver function tests, renal function tests including electrolytes, thyroid function test, ECG, Echocardiogram and chest X-ray. They were assessed preoperatively by the anesthetist and nephrologist for surgical fitness. Then immune suppression was initiated the day before the surgery using combination of drugs like cyclosporine or tacrolimus, mycophenolate mofetil or mycophenolic acid, and corticosteroid, while Basiliximab in a dose of 10-20 mg, or antithymocyte globulin (ATG) in a dose of 1.5 mg/kg was started before the induction of anesthesia.
All patients were pre oxygenated for 5 minutes, and induction commenced using fentanyl 1 μg/kg and thiopentone 4-6 mg/kg, muscle relaxation was achieved with atracurium at a dose 0.5 mg/kg, patients were intubated and mechanically ventilated using volume controlled ventilation (tidal volume 6-8 ml/kg, respiratory rate 12/min), anesthesia was maintained using isoflurane with 50% air in oxygen. Patients were monitored with ECG, non-invasive blood pressure, pulse oximetry, capnograghy and central venous pressure (CVP). The CVP line was inserted preoperatively preferably into the right internal jagular vein under aseptic conditions.
The donated kidney was prepared and washed out by the surgeon using cold normal saline and placed retroperitoneally in the iliac fossa. The renal vein anastomosed to the external iliac vein (end to side) and renal artery to the internal iliac artery (end to end) or occasionally to external iliac artery (end to side), and ureters were anastomosis to the bladder.
All parenteral fluids were administered by anesthetist. Starting with 3-5 ml/kg/hour of 0.9% normal saline, once venous anastomosis started mannitol 10% or 20% was given at a dose of 2-3 ml/kg. At this stage intravenous fluid load was increased to 10-15 ml/kg. Methyl prednisolone and heparin were given before releasing vascular clamp. Following completion of arterial anastomosis, urine output was monitored and furosemide given according to the output starting from 10 mg up to 60 mg. The aim was at least 2 ml/kg/hr of urine.
At the end of the procedure, the patients were reversed with neostigmine and atropine (2.5:1 ratio), extubated and sent to recovery room with oxygen mask. Analgesia was given in the form of paracetamol and fentanyl. Patients were shifted to the isolation room when fully conscious, with adequate respiration and hemodynamic stability.
Statistical Analysis
All the data was expressed as Mean ± SD. The continuous variables were analyzed using unpaired t-test whereas categorical data was analyzed using Chi-Square test. The p value < 0.05 was considered significant. All statistical analysis done using GraphPad: InStat Version 3.06.
Results
The characteristics about patient demographics, including age, male to female ratio and body weight were compared between two study groups and results are summarized in table 1. All values are expressed as Mean ± SD or proportions.
![]()
Table 1: Baseline characteristics between study groups
View Table 1
The results of comparison of baseline demographic characteristics showed that both groups were comparable with respect to mean age (p = 0.5738), male to female ratio (p = 0.7945) and mean body weight of study population (p = 0.8509).
The efficacy was compared considering total urine output and total dose of furosemide required for additional diuresis.
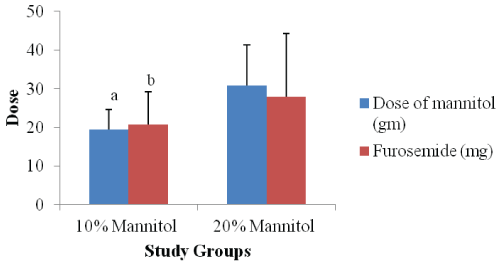
The results are presented in table 2 and figure 1.
.
Figure 1: Comparison of dose of mannitol and furosemide between study groups
aP value < 0.0001 & bP value 0.0474 as compared to 20% mannitol group
View Figure 1
![]()
Table 2: Comparison of efficacy between two study groups
View Table 2
The mean intravenous fluid given and mean urine output was comparable between both study groups. The duration of surgery remained comparable (p = 0.2177) between both study groups. The mean dose of mannitol required in 10% mannitol group was significantly lower (p < 0.0001) as compared to 20% mannitol group. Similarly the additional need of mean dose of furosemide was also significantly lower (p = 0.0474) in 10% mannitol group as compared to 20% mannitol group. One patient from each study groups did not require additional furosemide for diuresis. All the patients achieved desired urine output.
Discussion
Mannitol is commonly used as osmotic diuretic during renal transplant surgeries. It is administered immediately before opening the vascular clamp during the transplant. Mannitol, an inert sugar and offers protection against renal cortical ischemia through various mechanisms, it causes expansion of intravascular volume, diminishing the potential of tubular obstruction and increasing tubular flow rate through prevention of water reabsorption in the proximal tubule. Additional mechanisms include the release of vasodilatory prostaglandins in the kidney and action as a free radical scavenger [10]. Various clinical, single-center studies have found salutary effects of mannitol infusions for prevention of acute kidney injury in kidney transplantation [11]. Some of these studies either have a very small sample size or they have been done using retrospective analyses. With whatever evidence available, most commonly used dose of mannitol during renal transplantation is around 250 ml of 20% mannitol. It needs to be given immediately before vessel clamp removal to reduce the incidence of acute renal injury, which is indicated by adequate urinary output and a lower requirement of post-transplant dialysis [12].
There are several reasons why mannitol seems to be effective in preventing acute tubular necrosis and subsequent renal failure. Since mannitol induces osmotic diuresis, this 'flushing' effect within the tubules may help in reducing the accumulation of necrotic cell debris and casts. Osmotic diuresis even though used most commonly in such settings, it is not effective once complete tubular occlusion occurs. For this reason it is advised to administer mannitol before the ischemic insult becomes effective. Animal studies have demonstrated that mannitol can improves renal blood flow by changing the pressure-flow relationship within the kidneys leading to an increased flow at similar levels of perfusion pressure. Although this improvement in renal blood flow could be beneficial, but the overall effect is not so straightforward due to the increased oxygen consumption that occurs secondary to higher rates of energy demanding tubular solute reabsorption related to the parallel increase in GFR [6]. Mannitol also reduces post-ischaemic endothelial cell swelling and decreases ischaemic-reperfusion injuries by reducing the harmful effect of hydroxyl and other free radicals [6,13,14].
It is evident that the usage of mannitol also has some risks, because of the main mechanism of action which is to induce rapid intravascular volume expansion, which leads to systemic especially pulmonary edema. But concomitant hydration is proven to be a part of optimal prevention strategy for acute kidney injury after kidney transplantation [12].
Intravenous mannitol infusion before vascular clamp release and before the initiation of cyclosporin has been suggested for the prevention of post-transplant acute renal failure [10,15]. Such applications may require high doses of mannitol and may precipitate acute renal failure. Typically, mannitol-induced acute renal failure occurs in patients receiving larger cumulative doses of this agent than can be excreted [16]. In large doses, mannitol can also cause renal failure because of intra-renal vasoconstriction and intravascular volume depletion [5,16,17]. Additional mechanism which may contribute to reduced efficacy and acute kidney injury is that at higher concentrations mannitol diuresis can increase renal energy demands for Na+ reabsorption and so conceivably deplete ATP stores, thus rendering the kidney susceptible to acute kidney injury and more need for additional diuretic [2].
In our study it was found that patients in group 1 with 10% mannitol required overall lower dose of mannitol when used in grams. It was also observed that furosemide requirement as an additional diuretic is also significantly reduced which may signify the use of lower concentration over higher concentration and dose in renal transplant surgery. Additionally this may reduce necessity for several diagnostic and therapeutic interventions, with a concomitant decrease in the risks and advantage.
Small sample size and selection of fewer endpoints are few drawbacks of this study. This is due to limited number of end-stage renal disease patients available for renal transplant in our region.
Findings of this study needs to be verified with additional studies having larger sample size and more endpoints.
References
-
Butterworth JF, Mackey DC, Wasnick JD (2013) Morgan & Mikhail's Clinical Anesthesiology. 5th ed 29: 645-646.
-
Better OS, Rubinstein I, Winaver JM, Knochel JP (1997) Mannitol therapy revisited (1940-1997). Kidney Int 52: 886-894.
-
Guerin C, Girard R, Selli JM, Perdrix JP, Ayzac L (2000) Initial versus delayed acute renal failure in the intensive care unit: a multicenter prospective epidemiological study: Rhone-Alpes Area Study Group on Acute Renal Failure. Am J Respir Crit Care Med 161: 872-879.
-
de Mendonca A, Vincent JL, Suter PM, Moreno R, Dearden NM, et al. (2000) Acute renal failure in the ICU: risk factors and outcome evaluated by the SOFA score. Intensive Care Med 26: 915-921.
-
Bragadottir G, Redfors B, Ricksten SE (2012) Mannitol increases renal blood flow and maintains filtration fraction and oxygenation in postoperative acute kidney injury: a prospective interventional study. Crit Care 16: R159.
-
Shawkat H, Westwood M, Mortimer A (2012) Mannitol: a review of its clinical uses. Continuing Education in Anaesthesia, Critical Care & Pain 1: 1-4.
-
Dipiro JT, Talbert RL, Yee G, Matzke G, Wells B, et al. (2012) Pharmacotherapy: A Pathophysiologic Approach. 8th ed.
-
Product Information (2015) Osmitrol Intravenous Infusion (Mannitol).
-
Koning OH, Ploeg RJ, van Bockel JH, Groenewegen M, van der Woude FJ, et al. (1997) Risk factors for delayed graft function in cadaveric kidney transplantation: a prospective study of renal function and graft survival after preservation with University of Wisconsin solution in multi-organ donors. European Multicenter Study Group. Transplantation 63: 1620-1628.
-
Van Valenberg PL, Hoitsma AJ, Tiggeler RG, Berden JH, van Lier HJ, et al. (1987) Mannitol as an indispensable constituent of an intraoperative hydration protocol for the prevention of acute renal failure after renal cadaveric transplantation. Transplantation 44: 784.
-
Bugge JF, Hartmann A, Osnes S, Bentdal O, Stenstrom J (1999) Immediate and early renal function after living donor transplantation. Nephrol Dial Transplant 14: 389-393.
-
Schnuelle P, Johannes van der Woude F (2006) Perioperative fluid management in renal transplantation: a narrative review of the literature. Transpl Int 19: 947-959.
-
Luke RG, Briggs JD, Allison ME, Kennedy AC (1970) Factors determining response to mannitol in acute renal failure. Am J Med Sci 259: 168-174.
-
Sear JW (2005) Kidney dysfunction in the postoperative period. Br J Anaesth 95: 20-32.
-
Lauzurica R, Teixido J, Serra A, Torguet P, Bonet J, et al. (1992) Hydration and mannitol reduce the need for dialysis in cadaveric kidney transplant recipients treated with CyA. Transplant Proc 24: 46-47.
-
Dorman HR, Sondheimer JH, Cadnapaphornchai P (1990) Mannitol-induced acute renal failure. Medicine (Baltimore) 69: 153-159.
-
Veroux M, Veroux P (2012) Kidney Transplantation Challenging the Future 8: 91.





