International Journal of Anesthetics and Anesthesiology
Use of Human Fibrinogen Concentrate in Pediatric Cardiac Surgery Patients
Christopher F. Tirotta*, Richard G. Lagueruela, Danielle Madril, Jorge Ojito, Chelsea Balli, Evelio Velis, Marilyn Torres, Francisco Alonso, Robert Hannan and Redmond P. Burke
The Heart Program, Nicklaus Children's Hospital, USA
*Corresponding author: Christopher F. Tirotta, Director, Cardiac Anesthesia, The Heart Program of Nicklaus Children's Hospital, 3100 S.W. 62nd Street, Miami, FL 33155, USA, Tel: 305-663-8456, Fax: 305-663-8673, E-mail: christirotta@att.net
Int J Anesthetic Anesthesiol, IJAA-2-037, (Volume 2, Issue 4), Original Research; ISSN: 2377-4630
Received: November 06, 2015 | Accepted: December 18, 2015 | Published: December 21, 2015
Citation: Tirotta CF, Lagueruela RG, Madril D, Ojito J, Balli C, et al. (2015) Use of Human Fibrinogen Concentrate in Pediatric Cardiac Surgery Patients. Int J Anesthetic Anesthesiol 2:037. 10.23937/2377-4630/2/4/1037
Copyright: © 2015 Tirotta CF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Bleeding after cardiac surgery can cause increased morbidity and mortality. This is a particularly serious problem in pediatric patients, especially neonates and infants, who may receive multiple units of blood products intra- and postoperatively. The aim of this study is to demonstrate that the use of human fibrinogen concentrate (HFC) during cardiopulmonary bypass (CPB) decreased operative blood loss and the need for perioperative blood component therapy in neonatal, infant and other high-risk bleeding patients.
Materials and methods:We have been using HFC in high-risk bleeding patients since April 2013.The drug is administered one time during the rewarming phase of CPB at a dose of 70 mg/kg, just prior to the termination of CPB. We conducted a retrospective cohort study of 100 pediatric cardiac surgery patients. The first 50 patients (HFC-treated group) that received the drug were compared to a group selected from the pre-HFC era (Pre-HFC group).We looked at total blood loss during the perioperative period, total transfusion requirements, including individual component therapy, duration of ventilation, length of stay (LOS) in the cardiac intensive care unit and LOS in the hospital. Thrombotic events were also compared between groups.
Results:There was no significant difference in the age, weight and type of surgical operation between groups. There was significantly less fresh frozen plasma (FFP) (61 cc/kg vs. 39 cc/kg) and cryoprecipitate (11 vs. 2.8 cc/kg) used in those patients receiving HFC. There was a significant reduction in plasma fibrinogen at the time of rewarming compared with the preoperative value (225 vs. 114 mg/dL). However, there was no difference in the incidence of thrombotic events.
Conclusion:HFC in a dose of 70 mg/kg significantly reduced the need for transfusion with cryoprecipitate and FFP in infants undergoing complex surgical repairs. Plasma fibrinogen concentration is significantly reduced during CPB.
Keywords
Blood loss, Surgical, Cardiac Surgery, Cardiopulmonary Bypass, Fibrinogen, Intensive Care, Pediatrics
Introduction
Bleeding after cardiac surgery is a dangerous complication that can cause increased morbidity and mortality [1,2]. This is an especially acute problem in pediatric patients, particularly neonates and infants [3,4]. Wolf et al. reported that early postoperative bleeding was independently associated with increased mortality in infants after cardiopulmonary bypass (CPB) [5]. Their study demonstrated that the quartile with the greatest bleeding in the first twelve hours had a 25-fold increased mortality risk (10% vs. 0.4%) compared to the quartile with the least bleeding. Iyangar et al. reported transfusion with packed red blood cells (PRBC), fresh frozen plasma(FFP) and cryoprecipitate increased pulmonary complications and hospital length of stay (LOS) in pediatric cardiac surgery patients [6]. Other authors have reported similar adverse events from transfusion therapy in pediatric cardiac surgery patients [7-9].
Fibrinogen (Factor I) is a plasma glycoprotein manufactured in the liver. Thrombin catalyzes the conversion of fibrinogen to fibrin, a step that is essential for clot formation, and fibrinogen also promotes platelet activation and agglomeration. The normal concentration of fibrinogen in the plasma is 1.5-4.0 g/L or 150-400 mg/dL [10-12]. Treatment options for augmenting fibrinogen include FFP, cryoprecipitate and/or human fibrinogen concentrate (HFC). FFP is issued as a single unit (200-250 mL), while cryoprecipitate is issued as a pooled sample of four to six units (10-20 mL). The concentration of fibrinogen in cryoprecipitate is typically 15 g/L, which is greater than the concentration of fibrinogen in FFP. FFP and cryoprecipitate have several limitations, including the need for ABO compatibility, the need for thawing, a variable fibrinogen content, the risk of transfusion-related complications (i.e., transfusion-related acute lung injury [TRALI] and transmission of infectious agents) and the presence of relatively high concentrations of citrate, which can acutely lower ionized calcium, leading to decreased myocardial contractility and hypotension [13,14].
The fibrinogen of children less than 12 months old with congenital heart disease is qualitatively dysfunctional [15]. Moreover, the coagulation systems of neonates and children undergoing CPB are profoundly affected by haemodilution [16] and by consumption [17]. Neonates and young infants undergoing complex cardiac repairs often receive multiple units of blood products, both in the operating room (OR) and intensive care unit (ICU) [18]. Reducing the total volume of transfused blood products and reducing the unit donor exposure can reduce transfusion-related morbidity and mortality.
The purpose of this retrospective analysis is to demonstrate that the use of HFC during CPB decreased operative blood loss and the need for perioperative blood component therapy in neonatal, infant and other high-risk bleeding patients.
Materials and Methods
This is a retrospective cohort study reviewed and approved by the Western Institutional Review Board.
Patient population
HFC has been used on over 50 neonates, infants and other high-risk bleeding patients scheduled for elective cardiac surgery at Miami Children's Hospital since April 23, 2013.
Human fibrinogen concentrate administration
The drug was administered prophylactically, one time, prior to the termination of CPB, during the rewarming phase, in a dose of 70 mg/kg; this is the dose recommended by the manufacturer in adult patients if the fibrinogen level is not known.
Endpoints
The first 50 patients (Group 2 or HFC-treated) that received the drug, during the time period from April 23, 2013 to February 26, 2014, were compared to a group of patients treated from October 1, 2012 through April 22, 2013 (Group 1 or Pre-HFC). The two cohorts were comparable in terms of age, diagnosis, procedure, and identity of the surgeon. We looked to determine if there was a statistical difference between the groups for the following primary parameters: postoperative blood loss (chest and mediastinal tube drainage), and the amount and type of blood components transfused during surgery, including during CPB, and for the first 24 hours after surgery. Secondary endpoints included duration of postoperative ventilation in hours, LOS in the cardiac intensive care unit (CICU), the LOS in the hospital, postoperative prothrombin time (PT), international normalized ratio (INR) and partial thromboplastin time (PTT), preoperative and rewarming fibrinogen and the incidence of clinical thrombosis. We also compared, between groups, the duration of: CPB, aortic cross clamp (AXC), regional low flow perfusion (RLFP), and deep hypothermic circulatory arrest (DHCA). The number of patients that experienced deep hypothermia (low temperature < 20°C) and the lowest intraoperative temperature achieved were also compared.
Anesthetic management
The anesthetic management and perfusion management were similar between groups. All patients were monitored with electrocardiogram (EKG), pulse oximeter (SPO2), non-invasive blood pressure (NIBP), capnography (ETCO2), near infrared spectroscopy (NIRS), arterial line (A-line), central venous pressure (CVP), and two temperatures (esophageal and bladder or rectal). The anesthetic induction was with sevoflurane when done by mask and with fentanyl or propofol when done via IV. Maintenance of anesthesia was done with fentanyl (5-20 mcg/kg), sevoflurane, rocuronium, and/or Precedex (0.5-1 mg/kg/hr). Inotropes used at the termination of CPB were at the discretion of the anesthesiologist. This included either milrinone (0.5-1 mcg/kg/min) alone or milrinone (0.5-1 mcg/kg/min) and epinephrine (0.05-0.2 mcg/kg/min).
Perfusion management
Patients under 10 kg in body weight had the CPB pump primed with one unit of PRBC and 200 cc FFP. In patients, over 10 kg a pure crystalloid prime was used. Target hemoglobin (Hg) while on CPB was 9 g/dL, but the transfusion trigger for additional units of PRBC was 7 g/dL. Heparin management for all patients was based on the following tests using the HEPCON® HMS PLUS system. First, a baseline activated clotting time (ACT) with a projected heparin dose-response is performed. The initial heparin bolus is determined based on a desired ACT target of 480 seconds and a projected patient heparin concentration of 3.0-4.5 mg/kg. Second, heparin levels and ACT tests were run every 30 minutes or more frequently as needed. Reversal is with 1.2 mg protamine for every 100 International Units (IU) of residual heparin activity as documented by the HEPCON. Third, a low range heparin assay to confirm heparin reversal or need for additional protamine is conducted after CPB.
Transfusion protocol
A ROTEM-based transfusion protocol was used in both groups (Figure 1); in Group 2 this was done after the administration of HFC. From that point forward the same transfusion protocol was used in both groups. It is our practice to employ rotational thromboelastometry (ROTEM) to guide component transfusion therapy, as described by Romlin et al. [19]. Therefore, nearing the conclusion of CPB, ROTEM is used to guide component transfusion therapy. It is our institutional practice to begin to correct the dilutional coagulopathy while still on CPB, but just prior to its termination. The goal is to commence correction of the CPB-induced coagulopathy and hypofibrinogenemia. The component therapy could include plateletpheresis, FFP and/or cryoprecipitate depending on the ROTEM results. Ongoing hypofibrinogenemia was corrected with cryoprecipitate in both groups. Further ROTEM tests were done as needed, both in the OR and in the CICU. PRBC were transfused based on the Hg level reported on the iStat blood gas machine; transfusion triggers were Hg less than 10 g/dL.
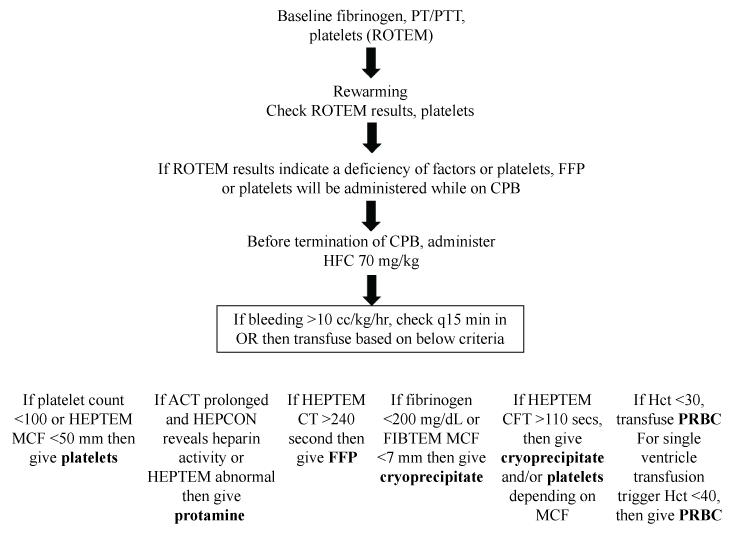
.
Figure 1: ROTEM-based transfusion protocol
ACT: Activated Clotting Time; CFT: Clot Formation Time; CPB: Cardiopulmonary Bypass; CT: Clotting Time; FFP: Fresh Frozen Plasma; Hct: Hematocrit; HFC: Human Fibrinogen Concentrate; MCF: Maximum Clot Firmness; OR: Operating Room; PRBC: Packed Red Blood Cells; PT: Prothrombin Time; PTT: Partial Prothrombin Time.
View Figure 1
Adverse event surveillance
The incidence of thrombotic events was also analyzed. A thrombotic event was defined as venous or arterial vessel occlusion secondary to placement of a vascular catheter documented by ultrasound (US), an aortic to pulmonary artery shunt occlusion, a Sano shunt occlusion, a thrombus in a major vessel or thrombosis while on cardiopulmonary support (CPS). Patients are routinely assessed for adequate peripheral circulation. Any patient demonstrating signs or symptoms of arterial or venous occlusion has an US done to confirm the diagnosis. Signs and symptoms of venous occlusion in the affected extremity include pain, edema, erythema, and distended peripheral veins. Signs and symptoms of arterial occlusion in the affected extremity include pain, pallor, loss of pulses, parathesia, paralysis and poikilothermia. Shunt occlusions and thrombosis in other vessels were confirmed by echocardiogram and/or cardiac catheterization.
Data analysis and data monitoring
Data was analyzed as mean ± standard deviation (SD), median and range, and proportions. Fifty charts were reviewed for each group.
The Statistical Package for Social Sciences (SPSS 21®) was used to organize, validate and analyze the collected data. Indicators of central tendency and dispersion were calculated. Group comparisons were made using both parametric (t-tests) and non-parametric (Mann-Whitney U) tests for continuous variables. Chi-Square or Fisher's exact test, when appropriate, were used for categorical variables. A determining level of 0.05 was selected for all tests of significance.
Results
Demographic characteristics
There was no significant difference in the age, weight and type of surgical operation between groups. One patient from Group 1 was eliminated due to being a statistical outlier. In Group 1, 46 out of 50 patients were less than 10 kg and in Group 2, 42 out of 50 patients were less than 10 kg. There was no variation in the duration of AXC, RLFP, and DHCA times. There was no difference in the number of patients that experienced deep hypothermia (χ2 = 0.83, p > 0.05) and no difference in the lowest temperature achieved between groups (U = 1091, p > 0.05, r = -0.09). There was a significantly longer duration of CPB in the HFC-treated patients (Group 2), (U = 938, p < 0.05, r = -0.20) (Table 1and Table 2).
![]()
Table 1: Demographic data
View Table 1
![]()
Table 2: Surgical procedures
View Table 2
Primary endpoints
The transfusion requirements in blood product type and amount are stratified into four groups: those given during CPB administered by the perfusionist; those products administered by anesthesia after termination of CPB; those products given by the ICU physicians once the patient leaves the OR; and then the total amount of all the blood products given during the three phases. With the above noted, there was a significant difference in the following parameters. There was significantly less FFP (U = 563, p < 0.01, r = 0.47) and cryoprecipitate (U = 411, p < 0.05, r = 0.68) given during CPB in the HFC-treated patients (Group 2). There was also significantly less FFP (U = 871, p < 0.05, r = 0.26) and cryoprecipitate (U = 722, p < 0.001, r = 0.49) given by anesthesia after termination of CPB in the HFC-treated patients (Group 2). Interestingly, the HFC-treated patients (Group 2) received significantly more Cell Saver than patients in Group 1 (U = 882, p < 0.05, r = 0.24). There was significantly less FFP (U = 730.5, p < 0.01, r = 0.36) and cryoprecipitate (U = 325.5, p < 0.01, r = 0.36) used in total in those patients receiving HFC (Group 2). Additionally, there was no difference in overall blood loss (U = 1039, p > 0.05, r = -0.13) or total transfusion requirements for other components (Table 3 and figure 2).
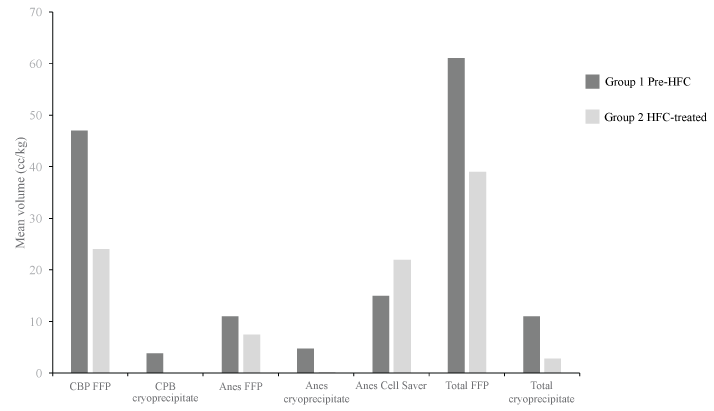
.
Figure 2: Statistically significant primary endpoints
P < 0.05 for all endpoints. CPB: Cardiopulmonary Bypass; FFP: Fresh Frozen Plasma; HFC: Human Fibrinogen Concentrate.
View Figure 2
![]()
Table 3: Quantitative results: primary endpoints
View Table 3
Secondary Endpoints
There was no difference in the duration of ventilation and LOS in both the CICU (U = 990, p > 0.05, r = 0.17) and hospital (U = 1013, p > 0.05, r = 0.15). No distinctions were obvious in Factor VII use or dose, nor the need for mediastinal exploration (χ2 = 1.69, p > 0.05). There was a significantly higher PT in the HFC-treated group (Group 2) compared with Group 1 (U = 770, p < 0.01, r = 0.27). There was no difference in postoperative INR (U = 10.5, p > 0.05, r = 0.14) and PTT (U = 14, p > 0.05, r = 0.11) between groups. On the contrary, there was a significant reduction in plasma fibrinogen at the time of rewarming compared with the preoperative value in the Group 2 patients. The rewarming fibrinogen level was below the normal range in 33 of the 34 patients that had the test performed. However, there was no difference in the incidence of thrombotic events (χ2 = 0.25, p > 0.05). See table 4 and table 5.
![]()
Table 4: Quantitative results: secondary endpoints
View Table 4
![]()
Table 5: Qualitative variables
View Table 5
Discussion
This is one of the first reports of the use of HFC in pediatric cardiac surgery patients. It demonstrated that using HFC one time at a dose of 70 mg/kg reduced the need for transfusion with FFP and cryoprecipitate during and after separation from CPB, while in the OR and overall, but not when in the ICU. This was despite the fact that the HFC-treated group had a significantly longer CPB time, which would normally predict a higher transfusion risk [20]. The reduced need for cryoprecipitate was certainly expected, since the HFC-treated patients were receiving a substantial dose of fibrinogen. The reduced need for FFP was not as intuitively obvious. It is noteworthy that in our analysis 82% (Group 1) and 81% (Group 2) of the blood products were administered in the OR, either by perfusion or anesthesia; less than 20% of the total was given by the ICU staff. This is a reflection of our programmatic philosophy of delivering a stable, non-bleeding patient to the ICU. This could also explain why there was no difference in the ICU component of the transfusion therapy.
This analysis corroborates previous studies demonstrating that the fibrinogen level at rewarming is typically 60% lower than the preoperative baseline value. Since the fibrinogen level can be used to dose HFC, quantifying the fibrinogen level at the time of administration can be potentially important. However, we had difficulty getting this lab value back in a timely enough fashion to utilize the fibrinogen level to dose the HFC. We believe this would be preferable to dosing based on weight. We also did not record unit donor exposure during this time period, so we did not include this in our analysis, but using HFC in lieu of cryoprecipitate necessarily must reduce unit donor exposure, since cryoprecipitate is issued as a pooled sample of 4-6 units, necessitating a 4-6 unit donor exposure from that one therapeutic intervention.
Our analysis partially corroborates the findings of studies in adult patients regarding a reduced transfusion requirement for FFP and cryoprecipitate, but did not demonstrate significantly less perioperative blood loss or a lower transfusion requirement for PRBC or platelets. We did not expect the administration of HFC to decrease the need for transfusion with platelets. Moreover, it is possible, given the profound dilutional coagulopathy that occurs in neonates and young infants undergoing CPB, that a dose of 70 mg/kg is not sufficient to produce similar results. Perhaps a greater dose, or multiple doses, is required. Further studies will need to be conducted.
Karlsson et al. showed in a prospective, observational study, using a multiple regression model, that preoperative fibrinogen concentration is an independent predictor of postoperative bleeding volume [21]. In this same study, the investigators also demonstrated that fibrinogen concentration, even when within the normal range, is a limiting factor for postoperative hemorrhage, i.e., fibrinogen values are inversely correlated with the risk of transfusion, even when within the normal range. The prophylactic infusion of HFC has been shown to reduce bleeding after adult coronary artery bypass graft (CABG) surgery without evidence of hypercoagulability [22]. Moreover, FIBTEM (fibrin-based extrinsically activated test with tissue factor and the platelet inhibitor cytochalasin D)-guided HFC administration was associated with reduced transfusion requirements and reduced postoperative bleeding within the first 24 hours in adult patients undergoing aortic valve operations and ascending aortic reconstructions [23]. In children, there is a paucity of data regarding the use of HFC in pediatric cardiac surgery patients. However, Galas et al. did report that in bleeding pediatric cardiac patients the use of HFC was "as efficient and safe as cryoprecipitate" [24]. Also, the American Society of Anesthesiologists (ASA) Practice Guidelines for Perioperative Blood Management, published in February 2015, recommends that "in patients with excessive bleeding, consider the use of fibrinogen concentrate" [25].
Thrombosis is a reported complication in HFC-treated patients, but the risk appears to be low [26]. However, Jakobsen et al. reported an increased risk of neurological thromboembolic complications and renal failure in their retrospective review of 1,876 adults undergoing CABG treated with fibrinogen concentrate [27]. Our analysis did not demonstrate an increased incidence of thrombotic events in HFC-treated patients, although the sample size was considerably smaller than that reported by Jakobsen et al. Virtually all of the thrombotic events were femoral arterial or femoral venous occlusions after percutaneous catheter insertion. There was one right atrial thrombus in Group 1.In Group 2, there was one shunt occlusion and one shunt occlusion and thrombosis on CPS in the same patient. All the other thrombotic events were related to percutaneous central venous catheters or arterial catheters. This incidence is high, but not inconsistent with other reports in the literature [28-30]. Furthermore, we did not examine any of the patients for end-organ damage due to thrombotic events by conducting brain MRIs or abdominal ultrasounds. This is certainly a limitation of this study.
The concentration of fibrinogen in HFC is standardized, in distinction to the variability that exists with FFP or cryoprecipitate.HFC is stored as a lyophilized powder at room temperature that permits fairly rapid reconstitution without unnecessary delays for cross matching or thawing [31]. Moreover, infusion volumes are low and the drug also does not contain any citrate. One unit of cryoprecipitate (10-20 cc of volume) contains 250-350 mg of fibrinogen [32]; the cost is $186 per unit whereas HFC costs $0.85 per mg. Thus, an equivalent dose of HFC at 250-350 mg would cost $212-$287.HFC is more expensive, but the reduced donor exposure might justify the increased cost.
One potential bias for this observational, retrospective analysis was patient selection in the pre-HFC group. We went back serially in time and, based on the patient demographic characteristics and planned surgery, included any patient we considered at high risk for bleeding and only then did we examine the record. Another limitation of this analysis is that we encountered numerous missing values for some of the variables of interest, particularly the preoperative coagulation studies. Therefore, the analysis had to be performed using only valid paired values. Furthermore, the postoperative PT was significantly longer in the HFC-treated group (Group 2), the only variable that was significantly worse for this cohort. We attribute this to the smaller amount of FFP this group received. Even though the PT was longer in Group 2, this did not lead to further bleeding. Since this is a retrospective observational study, results should be interpreted with caution. We are recommending a prospective, randomized controlled trial for more accurate comparison and generalizable conclusions.
This analysis has demonstrated an intriguing result, one deserving further study. Like recombinant coagulation factor VIIa (rFVIIa), which has been shown to be useful in preventing re-exploration for uncontrolled bleeding in children undergoing cardiac surgery with CPB [33-35], HFC can potentially be another useful drug in pediatric cardiac surgery patients. A prospective, randomized, controlled trial is planned.
In conclusion, HFC given during the rewarming phase of CPB in a dose of 70 mg/kg significantly reduced the need for transfusion with cryoprecipitate and FFP in infants undergoing complex surgical repairs placing them at risk for perioperative bleeding. There was also no increased risk of thrombotic complications in this series.
Disclosures
The retrospective analysis was submitted and approved by the Western Institutional Review Board. This study was funded by the Research Institute of Nicklaus Children's Hospital and a grant from CSL Behring.
References
-
Dacey LJ, Munoz JJ, Baribeau YR, Johnson ER, Lahey SJ, et al. (1998) Reexploration for hemorrhage following coronary artery bypass grafting: incidence and risk factors. Northern New England Cardiovascular Disease Study Group. Arch Surg 133: 442-447.
-
Moulton MJ, Creswell LL, Mackey ME, Cox JL, Rosenbloom M (1996) Reexploration for bleeding is a risk factor for adverse outcomes after cardiac operations. J Thorac Cardiovasc Surg 111: 1037-1046.
-
Williams GD, Bratton SL, Riley EC, Ramamoorthy C (1998) Association between age and blood loss in children undergoing open heart operations. Ann Thorac Surg 66: 870-875.
-
Williams GD, Bratton SL, Ramamoorthy C (1999) Factors associated with blood loss and blood product transfusions: a multivariate analysis in children after open-heart surgery. Anesth Analg 89: 57-64.
-
Wolf MJ, Maher KO, Kanter KR, Kogon BE, Guzzetta NA, et al. (2014) Early postoperative bleeding is independently associated with increased surgical mortality in infants after cardiopulmonary bypass. J Thorac Cardiovasc Surg 148: 631-636.e631.
-
Iyengar A, Scipione CN, Sheth P, Ohye RG, Riegger L, et al. (2013) Association of complications with blood transfusions in pediatric cardiac surgery patients. Ann Thorac Surg 96: 910-916.
-
Redlin M, Kukucka M, Boettcher W, Schoenfeld H, Huebler M, et al. (2013) Blood transfusion determines postoperative morbidity in pediatric cardiac surgery applying a comprehensive blood-sparing approach. J Thorac Cardiovasc Surg 146: 537-542.
-
Salvin JW, Scheurer MA, Laussen PC, Wypij D, Polito A, et al. (2011) Blood transfusion after pediatric cardiac surgery is associated with prolonged hospital stay. Ann Thorac Surg 91: 204-210.
-
Székely A, Cserép Z, Sápi E, Breuer T, Nagy CA, et al. (2009) Risks and predictors of blood transfusion in pediatric patients undergoing open heart operations. Ann Thorac Surg 87: 187-197.
-
Mosesson MW (2005) Fibrinogen and fibrin structure and functions. J Thromb Haemost 3: 1894-1904.
-
Standeven KF, Ariëns RA, Grant PJ (2005) The molecular physiology and pathology of fibrin structure/function. Blood Rev 19: 275-288.
-
Kreuz W, Meili E, Peter-Salonen K, Haertel S, Devay J, et al. (2005) Efficacy and tolerability of a pasteurised human fibrinogen concentrate in patients with congenital fibrinogen deficiency. Transfus Apher Sci 32: 247-253.
-
Weinkove R, Rangarajan S (2008) Fibrinogen concentrate for acquired hypofibrinogenaemic states. Transfus Med 18: 151-157.
-
Franchini M, Lippi G (2012) Fibrinogen replacement therapy: a critical review of the literature. Blood Transfus 10: 23-27.
-
Miller BE, Tosone SR, Guzzetta NA, Miller JL, Brosius KK (2004) Fibrinogen in children undergoing cardiac surgery: is it effective? Anesth Analg 99: 1341-1346.
-
Kern FH, Morana NJ, Sears JJ, Hickey PR (1992) Coagulation defects in neonates during cardiopulmonary bypass. Ann Thorac Surg 54: 541-546.
-
Chan AK, Leaker M, Burrows FA, Williams WG, Gruenwald CE, et al. (1997) Coagulation and fibrinolytic profile of paediatric patients undergoing cardiopulmonary bypass. Thromb Haemost 77: 270-277.
-
Kwiatkowski JL, Manno CS (1999) Blood transfusion support in pediatric cardiovascular surgery. Transfus Sci 21: 63-72.
-
Romlin BS, Wåhlander H, Berggren H, Synnergren M, Baghaei F, et al. (2011) Intraoperative thromboelastometry is associated with reduced transfusion prevalence in pediatric cardiac surgery. Anesth Analg 112: 30-36.
-
Schwartz JP, Bakhos M, Patel A, Botkin S, Neragi-Miandoab S (2008) Impact of pre-existing conditions, age and the length of cardiopulmonary bypass on postoperative outcome after repair of the ascending aorta and aortic arch for aortic aneurysms and dissections. Interact Cardiovasc Thorac Surg 7: 850-854.
-
Karlsson M, Ternström L, Hyllner M, Baghaei F, Nilsson S, et al. (2008) Plasma fibrinogen level, bleeding, and transfusion after on-pump coronary artery bypass grafting surgery: a prospective observational study. Transfusion 48: 2152-2158.
-
Karlsson M, Ternström L, Hyllner M, Baghaei F, Flinck A, et al. (2009) Prophylactic fibrinogen infusion reduces bleeding after coronary artery bypass surgery. A prospective randomised pilot study. Thromb Haemost 102: 137-144.
-
Rahe-Meyer N, Pichlmaier M, Haverich A, Solomon C, Winterhalter M, et al. (2009) Bleeding management with fibrinogen concentrate targeting a high-normal plasma fibrinogen level: a pilot study. Br J Anaesth 102: 785-792.
-
Galas FR, de Almeida JP, Fukushima JT, Vincent JL, Osawa EA, et al. (2014) Hemostatic effects of fibrinogen concentrate compared with cryoprecipitate in children after cardiac surgery: a randomized pilot trial. J Thorac Cardiovasc Surg 148: 1647-1655.
-
American Society of Anesthesiologists Task Force on Perioperative Blood Management (2015) Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Management*. Anesthesiology 122: 241-275.
-
Dickneite G, Pragst I, Joch C, Bergman GE (2009) Animal model and clinical evidence indicating low thrombogenic potential of fibrinogen concentrate (Haemocomplettan P). Blood Coagul Fibrinolysis 20: 535-540.
-
Jakobsen C-J, Tang M, Folkersen L (2011) Perioperative administration of fibrinogen is associated with increased risk of postoperative thromboembolic complications after cardiac surgery. J Blood Disord Transfus S1: 004.
-
Kusminsky RE (2007) Complications of central venous catheterization. J Am Coll Surg 204: 681-696.
-
Trottier SJ, Veremakis C, O'Brien J, Auer AI (1995) Femoral deep vein thrombosis associated with central venous catheterization: results from a prospective, randomized trial. Crit Care Med 23: 52-59.
-
Veldman A, Nold MF, Michel-Behnke I (2008) Thrombosis in the critically ill neonate: incidence, diagnosis, and management. Vasc Health Risk Manag 4: 1337-1348.
-
Fenger-Eriksen C, Ingerslev J, Sørensen B (2009) Fibrinogen concentrate--a potential universal hemostatic agent. Expert Opin Biol Ther 9: 1325-1333.
-
Shaz BH, Hillyer CD (2009) Cryoprecipitate. In: Transfusion Medicine and Hemostasis: Clinical and Laboratory Aspects. 1st ed. Elsevier BV, London pp. 175-178.
-
Yilmaz D, Karapinar B, Balkan C, Akisü M, Kavakli K (2008) Single-center experience: use of recombinant factor VIIa for acute life-threatening bleeding in children without congenital hemorrhagic disorder. Pediatr Hematol Oncol 25: 301-311.
-
Pychynska-Pokorska M, Moll JJ, Krajewski W, Jarosik P (2004) The use of recombinant coagulation factor VIIa in uncontrolled postoperative bleeding in children undergoing cardiac surgery with cardiopulmonary bypass. Pediatr Crit Care Med 5: 246-250.
-
von Heymann C, Spies C, Grubitzsch H, Schönfeld H, Sander M, et al. (2006) [Bleeding after cardiac surgery: the role of recombinant factor VIIa]. Hamostaseologie 26: S77-87.





