International Journal of Clinical Cardiology
Allograft Inflammatory Factor-1 Induction during Ischemia Reperfusion Injury: A Rat Model of LAD Occlusion
D. Olga McDaniel1,2*, Lance Majors2, Taolin Fang1, Feng Zhang1,2 and Larry S. McDaniel2,3
1Department of Surgery, University of Mississippi Medical Center, USA
2School of Medicine, University of Mississippi Medical Center, USA
3Department of Microbiology University of Mississippi Medical Center, USA
*Corresponding author: D. Olga McDaniel, Professor of Surgery and Pathology, University of Mississippi Medical Center, 2500 North State Street, Clinical Sciences Bldg. L-126, Jackson, MS, 39216-4505, Tel: 601-984-5081, Fax: 601-984-6614, E-mail: omcdaniel@umc.edu
Int J Clin Cardiol, IJCC-1-004, (Volume 1, Issue 1), Research Article; ISSN: 2378-2951
Received: September 24, 2014 | Accepted: October 27, 2014 | Published: October 29,
2014
Citation: McDaniel DO, Majors L, Fang T, Zhang F, McDaniel LS (2014) Allograft Inflammatory Factor-1 Induction during Ischemia Reperfusion Injury: A Rat Model of LAD Occlusion. Int J Clin Cardiol 1:004. 10.23937/2378-2951/1410004
Copyright: © 2014 McDaniel DO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The emerging role of the innate immune response in organ transplantation heightens the significance of molecular markers released from the donor organ due to ischemia/reperfusion injury (IRI) during organ procurement. These molecules are prime activators of innate immunity. Allograft inflammatory factor-1 (AIF-1) has been implicated in the regulation of inflammation and organ rejection. To verify expression level of AIF-1 in cardiac tissues, we developed a rat model of IRI, in which the left anterior descending artery (LAD) was occluded to generate ischemia in the left ventricle (LV) of the heart. Blood and cardiac tissues were tested for the presence of AIF-1 at different time intervals. AIF-1 expression was significantly increased in a time-dependent manner after IRI. AIF-1 was up regulated as early as 10 minutes after reperfusion, and further it was increased several-fold after 60 minutes of reperfusion in PBMCs and PMNCs as compared to the control group. Expression levels of AIF-1 in LV tissues were significantly increased after 60 minutes of ischemia or 30 minutes of ischemia followed by 60 minutes of reperfusion. In addition the level of AIF-1 was 2-fold greater in LV tissues after 60 minutes of reperfusion compared to LV after 30 or 60 minutes of ischemia, Thus, myocardial IRI activates AIF-1 up-regulation, which may result in allograft dysfunction in transplantation settings.
Keywords
Allograft inflammatory factor-1, Cardiac, Ischemia, LAD, Myocardium, Rat, Reperfusion
Introduction
Ischemia reperfusion during organ procurement and recovery is an unavoidable event that contributes significantly to tissue injury and may affect early organ dysfunction after transplantation. However, in clinical settings, the minimum ischemic time that results in a significant impact on cardiac transplantation is not clear. A preservation time >24 hour in kidney as well as lung transplantation has been linked to a decline of organ function during early post-transplantation [1-3]. It has been well documented that ischemia, (the disruption of the blood supply to the organ during procurement) followed by restoration of blood flow during reperfusion often creates an ultra-structural and metabolic/cellular injury (IRI). This leads to profound changes in the production of reactive oxygen species (ROS) causing myocardial apoptosis and cardiomyocyte death in ischemic regions [4-6]. The consequences of pathological obstruction or the experimental ligation of the coronary artery in the induction of ischemia and myocardial injury is well known. Inflammation is the hallmark of these consequences thus; targeting molecules initiating such inflammatory responses is an emerging therapeutic intervention with significance in the prevention of ischemia induced injury.
IRI induced inflammation typically occurs in a sterile environment but, shares many characteristics of the host immune response aimed at pathogenic microorganisms [7]. The immune response associated with cardiac IRI is initiated by activation of the myocardial resident dendritic cells and components of the innate immune system. This includes pattern recognition molecules such as Toll-like receptors (TLRs) [8-10], which results in the induction of proinflammatory cytokines and chemokines [7]. Cytokines are secreted by all resident cells in the myocardium and by the leukocytes that migrate into the heart [11]. Eventually, the innate response and the parameters involved in the activation of this early response bridges into adaptive immunity leading to allograft dysfunction and rejection [12]. Given the importance of innate immune responses in the recognition of pathogen or damaged tissue associated molecular patterns known as PAMPs or DAMPs respectively, we postulated that the molecules released from a donor organ during procurement may play a crucial role in the initial inflammatory response to the allograft.
A broad range of non-pathogenic motifs could trigger the induction of an inflammatory episode in the myocardium. It is possible that one of the contributing factors in the initial inflammatory response during IRI is the release of allograft inflammatory factor-1 (AIF-1). AIF-1 is a potential endogenous ligand that engages in activation of TLR-baring dendritic cells initiating the innate immune response in transplantation settings. An adapted hypothetical model of our study is summarized in Figure 1 [13]. AIF-1 has been implicated in the regulation of inflammation and thus far many investigators have shown an association between elevated levels of AIF-1 expression with cellular rejection and coronary vasculopathy after organ transplantation [14-16].
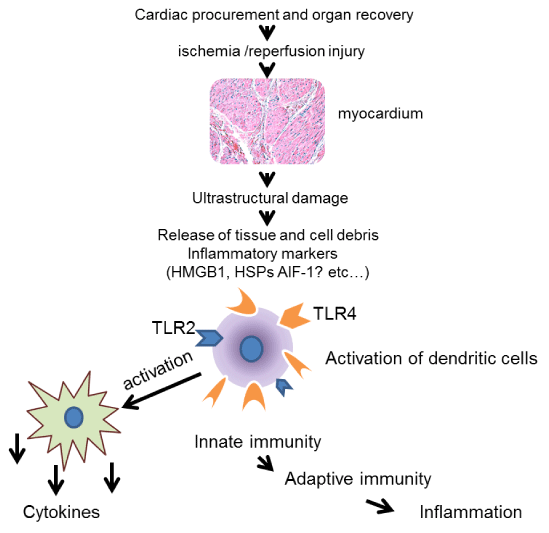
Figure 1: Graphic hypothesis of ischemia/reperfusion injury. A graphic
illustration of ischemia reperfusion injury (IRI) is described. A series of
molecular event occurs in which AIF-1 released from the tissue may serve
as an endogenous ligand to TLRs activating innate immune effector cells,
causing the release of proinflammatory cytokines, leading to inflammation.
View Figure 1
AIF-1 is a 143 amino acid, cytoplasmic, calcium binding protein, inducible by IL-1β, IFN-β, and IL-18, as well as T cell conditioned media [17-19]. In humans, the AIF-1 gene maps to the major histocompatibility complex class III region on chromosome 6p21.3. This is a region which is densely clustered with genes associated with the immune response, including TNF-α/β and NF-κB [20], suggesting a fundamental role of AIF-1 in the processing of the inflammatory responses. AIF-1 expression was first reported in 1995, in injured rat muscles in association with rat cardiac allograft rejection [21]. Subsequently AIF-1 was found in humans and is involved in many pathological processes including angiogenesis, cell proliferation, and differentiation [22,23]. It is expressed primarily in dendritic cells and macrophages [24]. In a blood vessel, AIF-1 was increased after balloon injury and angioplasty [25]. Over expression of AIF-1 is associated with cardiac allograft vasculopathy [14,15,26]. In renal allograft undergoing a rejection episode the infiltrating macrophages with increased AIF-1 signals may distinguish clinical rejection as opposed to subclinical rejection [27,28].
There have been limited reports associated with the early innate immune responses and the parameters involved in the activation of early inflammatory responses after transplantation. Recent evidence suggests that myocardial cell-death and associated inflammation are characteristics of cardiac IRI. Thus, strategies aimed at identification of molecular markers released during IRI could help in the design of blocking experiments to prevent or minimize cardiac injury induced by I/R. Direct evidence identifying the release of AIF-1 from cardiomyocytes during IR has not been established. In this study we investigated the release of AIF-1 during an experimental rat model of IRI, in which the left anterior descending artery (LAD) was occluded at different time intervals. We have compared the level of AIF-1 present in peripheral blood and cardiac tissue specimens before and after IRI. The goal was to provide evidence that AIF-1 is induced during cardiac IR in myocardial tissue and is a crucial biomarker in the injury induced immune response that indicates the magnitude of the inflammatory response initiated by IRI.
Materials and Methods
Surgical preparation of rats: Male Sprague-Dawley rats aged 2-3 months; 300-350 grams were tested in this study. Rats were housed in a temperature; humidity and 12:12 hour light-dark cycle controlled room in the animal facility at the University of Mississippi Medical center (UMMC) before surgery. Two to three rats were kept in one cage, providing free access to tap water and rat chow. All surgeries were performed during the light cycle. The study was conducted according to the guidelines required by Institutional Animal Care and Use Committee (IACUC) for the use of live rodents at UMMC.
Surgical approach: The procedure was performed under an operating microscope with 10X magnification. Surgical protocol for myocardial ischemia reperfusion was adapted from a procedure previously described [29]. Rats were anesthetized in an anesthesia chamber and isoflurane delivery system. Originally with inhalation rates of 3-5 % isoflurane and 2% oxygen via an isoflurane vaporizer (Isotech, Highland Medical Equipment, Tempecula, CA)followed by 1-3% as needed. The rat was placed in the supine position on a heated pad that was fixed on the operating table. The legs of the rat were secured to the side of body with tape, making sure the front legs were not over-stretched, which it may affect respiration. The body temperature was maintained at 37.5�C ± 0.5�C by the heating pad controller. A cotton umbilical surgical ribbon attached to the operating platform was placed horizontally under the front upper incisors to hold the upper jaw in place. The fur from surgical areas, which include: front of the neck, chest and the inner leg were shaved and decontaminated using Betadine, followed by a 70% ethanol swab.
For a successful oral-tracheal intubation, the trachea was exposed by making a1.5 cm cervical incision, then spreading the lobs of the thyroid gland and the sternohyoideus muscle facilitating viewing of the trachea. Holding the tongue with gauze slightly upwards, an 18- gauge, blunt end cannula with polyethylene (PE) tube attachment was gently inserted through the opening of throat and was viewed through the exposed trachea. The cannula was then secured in the place by a 2-0 suture while being mechanically ventilated using a rodent respirator (SAR-830/AP small animal ventilator). The ventilation rate was adjusted to 55-60 breaths/min, the tidal volume was set at 2-3 ml/100gram body weight. A left intercostal thoracotomy was performed by a 3-4 cm long incision at a site 0.5 cm away from the left sternal border. Before the ribs were cut, the 3rd- 5th ribs were clamped off with small curved hemostat to prevent the vessels underneath the ribs from bleeding. Then the ribs were spread with a small retractor to expose the heart. The pericardium was then opened with sharp forceps. The LAD, about 2-3 mm below the anterior-inferior edge of the left atrium was ligated with a tapered point needle and a 5-0 silk ligature. For a temporary ligature that can be removed during reperfusion, the ends of the tie was threaded through a 1 cm PE100 tube, which was placed parallel to the coronary artery to form a snare for reversible LAD occlusion. Thus, a local ischemic condition was achieved which was observed by a change in color of the anterior wall of the LV to paleness of myocardium (Figure 2).
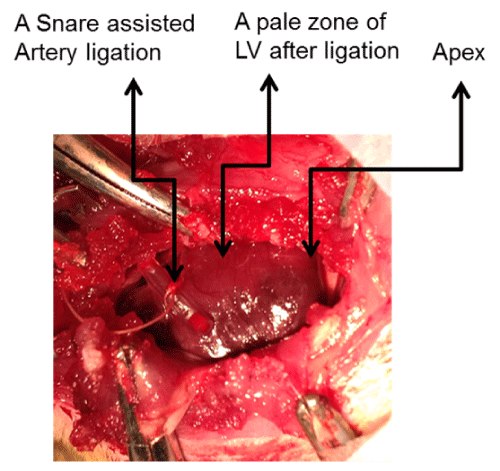
Figure 2: A model of rat LAD ligation and ischemia. A model of rat LAD
occlusion and ischemia development is presented. A, demonstrates ischemic
condition after 60 minutes, in which a change of color of the anterior wall of
the LV to paleness is observed. B, is a video presentation of ischemia. For
a temporary ischemia, a 1 cm PE tube was placed parallel to the coronary
artery, the ends of the suture was looped around the tube to form a snare for
reversible LAD occlusion.
View Figure 2
Blood was drawn from the femoral vein, after making an incision in the inguinal area, along the angle of the left hind leg. Using blunt-tipped scissors, the connective tissue was spread out until the femoral vein was exposed. A 26-gauge butterfly blood drawing needle attached to a 2 ml sterile syringe was used to collect blood. The syringe was flushed with heparin then filled with 0.1 ml heparin solution (20U/ml), to draw 1-2 ml of blood. At the completion of blood drawing, the needle was gently pulled. A 1-cm in diameter gauze-ball was placed over the vein, secured with a 5-0 silk suture into the muscle layer, and the incision was stitched with 4-0 suture.
Experimental protocol: In a preliminary study, 25 rats were assigned to five groups (5 in each group): control; 30 minutes LAD occlusion; 60 minutes LAD occlusion; 30 minutes LAD occlusion/10 minutes reperfusion and 30 minutes LAD occlusion/60 minutes reperfusion as summarized in Table 1. In all rats a left thoracotomy was performed, but in the control group no further procedure was applied. In groups 2, 4, and 5 the LAD was circled with suture and the ligation was tied around a small tube as described earlier for reversible LAD occlusion. Then 0.2ml of 1000U/ml heparin in physiological saline was injected through the penile vein to reduce blood coagulation during surgery. In group 2 and 3 at the end of 30 minutes or 60 minutes LAD ligation (ischemia), respectively, blood was drawn for testing of AIF-1 expression levels in the blood. Blood was drawn directly from left ventricle then the heart was excised while the rat was deeply anesthetized (5% isoflurane).In group 4 and 5, the LAD was ligated for 30 minutes followed by 10 minutes or 60 minutes reperfusion respectively. Blood was drawn at the completion of reperfusion for AIF-1 mRNA analysis. The heart was then excised as described earlier, flushed with physiologic-saline, wrapped in saran wrap and stored at -20oC for 30 minutes. Frozen heart was then sliced in two halves along the interventricular septum. Each half of the left and right ventricles (LV and RV) were sliced in 2x2square mm, some stored in formaldehyde for paraffin block preparation, for immunohistochemistry (IHC) staining, or at -80oC freezer to be used for RNA preparation for AIF-1 mRNA transcript.
![]()
Table 1: Study protocol for determination of AIF-1 release during ischemia vs
Reperfusion.
View Table 1
Measurement of aif-1 expression in rat peripheral blood cells and cardiac tissue: To determine AIF-1 mRNA transcript levels, RNA was isolated from rat peripheral blood and cardiac tissue specimen. In human, the AIF-1 expression level varies in mononuclear cells (MNCs) versus polymorphonuclear cells (PMNCs). Thus, both types of the cells were tested in this rat model of IRI.
Blood samples were subjected first to Ficoll-paque density gradient for isolation of PBMCs (the interphase layer between plasma and Ficoll-paque). The dense white layer above the red cells (buffy coat) was then suspended in a hypotonic solution prepared from 0.3 M NH4CL and 0.17 M Tris and layered over inactivated 100% Fetal Calf Serum, followed by centrifugation at 1,500rpm for 10 minutes at room temperature. The pink pellet, which contained PMNCs and residual amounts of red cells was washed 2X with 1X sterile phosphate buffered saline (PBS, pH=7.4), and adjusted to 2-5x106cells/ml. The pellets were immediately homogenized in TRIzol and were stored at -80oC for subsequent RNA extraction. The PBMCs and PMNCs were at ≥95% purity observed by differential cell count stain, and =95% viable with 0.4% Trypan blue stain.
RNA from PBMCs and PMNCs were extracted using �Pure Link� RNA kit (Life Technologies, Carlsbad, CA, USA), according to the manufacturer�s guidelines. RNA concentration and purity was determined using NanoDrop 2000C (Thermo-Fisher, GA, USA). For RNA preparation from cardiac tissues, frozen sections were homogenized in 800�l TRIzol and extracted using the same �Pure Link� kit. Afterward, the RNA was transcribed using the ImProm-II Reverse Transcription kit (Promega, Madison, WI), followed by semi-quantitative RT-PCR (95oC for 4 min, and a 25 cycles of amplification at 95�C/30sec, 60�C/30sec, 72�C/30sec, and 100nM of each forward and reverse primers) using primer sequences specific for rat AIF-1, and GAPDH shown in Table 2. The amplification products were analyzed in Alpha Innotech System using software v.6.0.14 (Proteinsimple, Santa Clara, CA USA).The mRNA transcript levels were normalized to the GAPDH gene expression levels. The normalized values (Unit) were presented as mean ± SEM.
![]()
Table 2: Rat primer sequences (5'-3') for Reverse Transcriptase �PCR.
View Table2
Immunohistochemistry (IHC): The formalin fixed paraffin embedded (FFPE) rat cardiac tissue sections were prepared for IHC stain as previously described [15]. The slides were stained with L5-B3662 primary antibody against AIF-1 (ID#1022-5, Ig2b, reactive in human and rat) (Life Span Bioscience Inc., Seattle, WA). The optimal working dilution for LS-B3662 was 1:50. The detail of the staining was previously reported [26]. The stained slides were evaluated by a pathologist to confirm the staining specificity
Data analysis: Both interstitial infiltrating MNCs and cardiac myocytes (CMCs) were evaluated by IHC staining. Sections were analyzed for the presence and absence of AIF-1 in relation with clinical condition (ischemia vs. reperfusion). The mRNA transcript values were expressed as the mean ± SEM. The group differences for mRNA transcript levels between tissue conditions were determined by one-way analysis of variance. The level of significance was set at p< 0.05 for comparison between the groups.
Results
Basic Measurements: A total of 25 rats were studied. The average body weight was 328 ± 21.5. The heart rates (HRs) and blood pressure were not recorded. The HR was stable after the initial anesthesia, and the follow up period (1-2% isoflurane and 1% oxygen). There were no drastic changes among the five groups. In the control group, in which the rats had undergone only a left thoracotomy, no significant HR changes were noticed throughout the observation. In other groups a significant increase of HR was observed during the first 1-5 minutes into ischemia condition, but then returned to a stable rate after oxygen was increased to a rate of 1.5-2%. During the reperfusion, there were no significant changes in HR among the group that had undergone reperfusion.
Rat model of LAD ligation and ischemia: The fate of an experimental coronary occlusion is highly dependent on the ideal ligation position. Thus, the procedure was performed under an operating microscope. In the groups with I/R some rats demonstrated a minimal ventricular fibrillation (VF), or ventricular tachycardia (VT), during the early period of both ischemia and reperfusion. The VF or VT did not have impact on the course of the study. A video of a LAD occlusion is shown in Figure 2, where myocardial color changes are visible on the left ventricular wall during ischemic condition.
Measurement of AIF-1 expression in peripheral blood cells: AIF-1 mRNA was tested in PBMCs and PMNCs. The normalized values (Unit) were presented as mean ± SEM. The levels of expression were increased after 30 minutes and 60 minutes of ischemia as compared with control group in both PBMCs and PMNCs (Controls: 1.03 ± 0.14 and 1.02 ± 0.54 respectively; 30 min LADI: 1.18 ± 0.4 and 1.46 ± 0.75; 60min LADI: 1.74 ± 0.45 and 2.1 ± 0.97). However, the data was not statistically significant (Figure 3). Compared to the control group, AIF-1 expression level was significantly increased in the group with 30 min ischemia followed by 10min or 60min reperfusion in both PBMCs and PMNCs (Controls: 1.03 ± 0.14 and 1.02 ± 0.54 respectively; 30 M LADI/10 M R: 2.73 ± 0.23 and 4.3 ± 1.35 (p< 0.05 and p< 0.03 respectively); 30 M LADI/60 M R: 9.1 ± 5.56 and 7.83 ± 3.1 (p< 0.01 and p< 0.01 respectively).
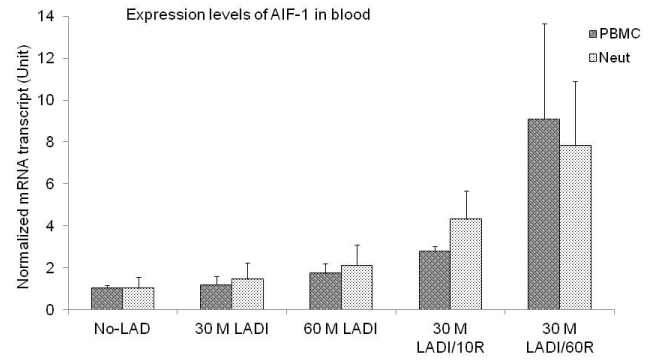
Figure 3: mRNA transcripts of AIF-1. Variations in mRNA transcripts levels
were expressed as the mean ± SEM. AIF-1 mRNA transcripts at different
time intervals are shown in PBMCs and Neutrophils/ or PMNCs. The levels
of expression were increased after ischemia compared with controls but was
not statistically significant. AIF-1 expression was significantly increased after
10 or 60 minutes of reperfusion observed in PBMCs and Neutrophils. For 30
minutes of ischemia (LAD)/10 minutes of reperfusion vs. control the p values
were (PBMCs, p< 0.05, Neutrophils, p< 0.03). For 30 minutes of ischemia
(LAD)/60 minutes of reperfusion vs. control the p values were (PBMCs,
p< 0.01, Neutrophils, p< 0.01).
View Figure 3
Measurement of AIF-1 expression in cardiac tissues: We examined mRNA expression levels in rat cardiac LV and RV tissues in all five groups. As shown in Figure 4, the mRNA expression levels were increased compared to control specimens. In group 2, the AIF-1 mRNA was increased 1.4-fold in LV tissues and 1.5-fold in RV tissues as compared with group 1. In group 3, the mean AIF-1 mRNA was 3.4-fold higher in LV as compared to 1.32-fold in RV tissues (p< 0.05). In group 4, after 10 min reperfusion the AIF-1 levels in LV tissues were 2.3-fold greater than the levels in tissues from group 1, and 2.2-fold greater than RV tissues (p< 0.05). In group 5, the AIF-1 mRNA was increased 6.24-fold in LV tissues as compared to the levels in group 1 (P< 0.01). In addition, the mean ± SEM levels in LV tissues were 5.13-fold greater than the levels in RV tissues (P< 0.01).
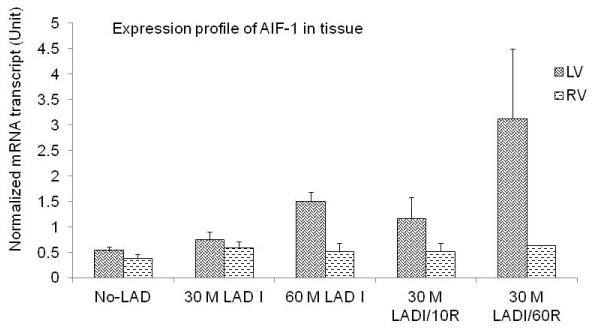
Figure 4: mRNA transcripts of AIF-1. Measurement of AIF-1 expression
in cardiac tissues expressed as the mean ± SEM. After 30 minutes of
ischemia AIF-1 mRNA was increased 1.4-fold in LV tissues and 1.5-fold in
RV tissues compared to control. After 60 minutes of ischemia AIF-1 mRNA
was increased 3.4-fold in LV tissues and 1.3-fold in RV tissues compared to
control (p< 0.05). After 10 minutes of reperfusion AIF-1 mRNA was reduced
but it was 2.3-fold greater than the levels in control and 2.2-fold greater than
in RV tissues (p< 0.5). After 60 minutes of reperfusion AIF-1 mRNA was
elevated 6.24-fold in LV tissues vs. control (p< 0.01), and 5.13-fold greater
than the levels in RV tissues (p< 0.01).
View Figure 4
Histopathologic examination and distribution of AIF-1: To localize the expression of AIF-1 after I/R, rat LV tissues were tested by histology and immunostaining. Figure 5, shows a representative hematoxylin and eosin (HE) staining of LV tissues from control and post IR groups. In control group LV myocardial fibers were arranged regularly with clear striation (Figure 5A,5B). No pathological changes were observed. In the post IR group, degenerative changes in myocardial fibers and inflammatory cells were seen in the perimeter of myocytes, which are an indication of myocyte injury (Figure 5C,5D). Immunohistochemistry staining for AIF-1 is shown in Figure 6, section A and section B. Anti-AIF-1 stain as present in both the nucleus of myocytes and in interstitial cells. AIF-1 positive cells were increased after reperfusion (Figure 6B).
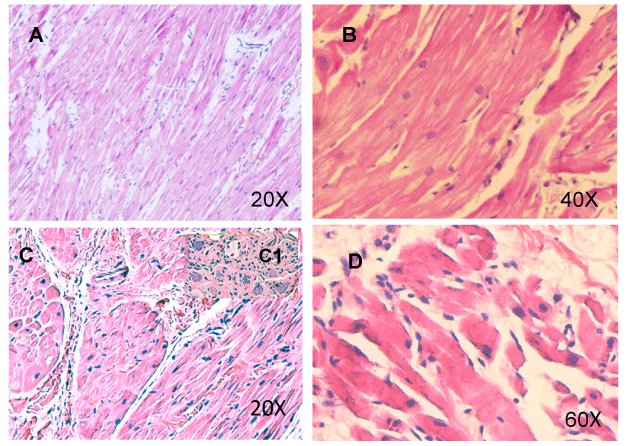
Figure 5: Cross-sections of cardiac LV after ischemia reperfusion. A
representative HE staining of LV tissues from control and post IR is shown. A
and B, a 20x and 40x illustration of LV from control group, in which myocardial
fibers are arranged regularly with clean striation and no pathological changes
are observed; C and D, presents after IR in which degenerative changes in
myocardial fibers and inflammatory cells in the perimeter of myocytes are
observed.
View Figure 5
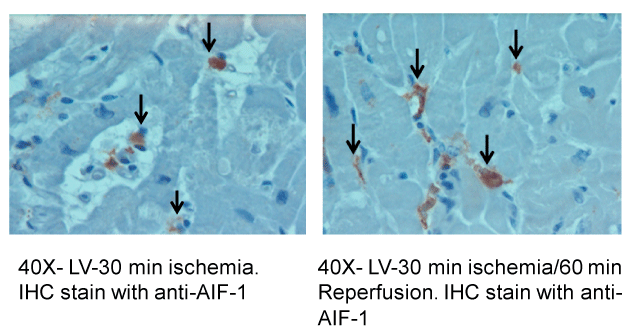
Figure 6: IHC stains of cardiac specimens after ischemia/reperfusion tested
with AIF-1 antibody. IHC stains of LV tissues after I/R is tested with AIF-1
antibody. A, after 30 minutes of ischemia AIF-1 positive stain indicated by
brown color are observed in both nucleus of myocytes and in interstitial cells;
B, after 60 minutes of reperfusion AIF-1 positive cells were 2-fold increased.
View Figure 6
Discussion
The scope of this study was to verify the release of AIF-1 after experimental LAD occlusion followed by reperfusion. It has been documented that a series of pathophysiologic and immunologic events occurs when tissue or organ has undergone IRI [30,31]. IRI increases the systemic availability of inflammatory mediators, leading to the activation of myocardial endothelial cells, as well as infiltrating MNCs and PMNCs initiating vascular and organ dysfunction. In addition molecular markers associated with innate immunity have been implicated in the regulation of inflammatory responses during IRI [32,33]. In this study we have generated ischemic condition followed by reperfusion in a rat in vivo model of LAD occlusion. The practical value of this study was the findings that the AIF-1 was variably expressed in cardiac tissues after IRI. Our data revealed that AIF-1 was up regulated as early as 10 minutes after reperfusion, and further it was increased several-fold after 60 minutes in PBMCs and PMNCs as compared to the control group. Similarly, expression levels of AIF-1 in LV tissues were significantly increased after 60 minutes ischemia or 30 minutes ischemia followed by 60 minutes reperfusion (Figure 4). This indicates that AIF-1 was up regulated during both ischemia and reperfusion. In addition AIF-1 level was 2-fold greater in LV tissues after 60 minutes reperfusion compared to LV after 30 or 60 minutes ischemia, suggesting that AIF-1 upon induction is actively expressed during IRI. This powers the impact of AIF-1 during IRI and thereafter on organ dysfunction.
Recent studies have reported increased levels of AIF-1 expression in IRI [34,35]. In the latter study using human cardiac atrium tissues in vitro, it was shown that AIF-1 levels increased in association with IL-18, TLR2, and TLR4 at different time intervals. IL-18 and TLRs are essential receptor molecules on macrophages. They are known for their association with early innate immune responses and several studies have reported the presence of these inflammatory markers in association with allograft rejection [35,36]. On the other hand, AIF-1 is known for its participation in inflammatory responses and regulation of inflammatory pathways of macrophages [37,38]. This was supported by the studies in which AIF-1 cDNA was transfected into a macrophage cell line, Raw264.7. The transfectant cells with over expressed AIF-1 had characteristics of the dendritic cells, with significant production of IL-6, IL-10 and IL-12 upon stimulation with LPS [37]. Moreover, blocking the expression of AIF-1 by siRNA was associated with apoptosis of AIF-1 baring macrophages [38,39]. Macrophages play a significant role in linking injury, inflammation, and organ dysfunction [40]. The level of AIF-1 expression in rat macrophages has been shown to be augmented by IFN-γ and IL-1β [17,18]. Furthermore, it was shown in a rat model of IRI AIF-1 expression was parallel with the expression levels of IFN-γ after reperfusion, thus suggesting the immunologic involvement of AIF-1 in proinflammatory processes of macrophages. In our unpublished studies of a rat model of IRI, as well as in in vitro studies of human cardiac tissue IR [34], we were unable to demonstrate increased expression levels of IFN-γ. However, we observed a significant increase in the levels of TLR-2 and TLR-4 mRNA transcripts in cardiac tissues after IR. The TLRs particularly TLR-2,-3, -4, and -9 have been shown to be involved in the pathophysiology of IRI [41-43]. Although we have not tested expression levels of TLRs in rat cardiac tissues after IRI, one study reported the impact of TLR-3 in myocardial injury through the recognition of DAMPs during myocardial IRI [44].
We do not know whether AIF-1 is just a marker of immune effector cells associated with inflammation, or directly engaged in regulation of inflammatory processes in cardiomyocytes and resident dendritic cells/macrophages during IR. It has been shown that increased levels of AIF-1 in liver-allograft after cold and warm IR could be reduced with low dose of FK-506 pretreatment [45]. In addition, studies have shown that reducing inflammatory responses during IR significantly protects organ function [46-48].
The functional role of AIF-1 remains to be determined. Hypothetically there is growing evidence suggesting that allograft injury during organ procurement induces innate immune response in which donor-derived damaged molecules may serve as endogenous ligands for TLRs in the activation of an initial inflammatory event. This is an immunologic concept that was originally proposed by Janeway, 1989 in which he described how the innate immune system discriminates noninfectious self from infectious non-self [49]. Later the concept was retackled by Polly Matzinger [50]. These self-derived molecules are known as �danger signals� and are associated with non-pathogenic, host originated damaged tissues causing sterile inflammation. In a similar context we are considering that a segment of AIF-1 may represent a potential candidate ligand and may initiate a danger signal, leading to inflammatory responses. Such a model of cardiac allograft IRI is currently under investigation.
This study was the first to investigate expression levels of AIF-1 in a time-dependent manner from myocardial tissues and peripheral distribution after IRI. Considering the involvement of AIF-1 in the initial inflammatory processes, the specific inhibition or minimizing the expression levels during ischemia may have important clinical implications in transplantation. However, whether such inhibition can protect the cardiac tissue from injury remains to be established. Emerging studies have reported the significance of post-ischemia reconditioning in combination with inflammatory agonist that may attenuate myocardial IRI by inhibiting reperfusion induced pro-inflammatory cytokines [48,51]. Thus, identification of signals and/or activators of myocardial IRI and signaling pathways, which leads to early inflammatory response, suggests that AIF-1 and TLRs may prove efficient therapeutic targets for pharmacologic agents. While the inhibition of such markers will remain crucial for treatment of IRI and organ protection, many other aspects of allograft rejection and allograft tolerance induction need further investigation.
Acknowledgement
The authors thank the Microsurgery Laboratory in the Department of Surgery at University of Mississippi Medical Center (UMMC) for providing the facility; Dr. Merry L. Lindsey, Professor of Physiology and Medicine, and the Director of Jackson Center for Heart Research at UMMC for her technical guidance; Dr. Richard J. Roman, Professor and Chair of Pharmacology and Toxicology at UMMC providing us small rodent ventilator. This work was supported in part by a grant from Alcon Research to L.S.M and by the Transplantation Immunogenetics Laboratory (PI: D.O.M) in the Department of Surgery at UMMC.
Ethical Statement
The study was conducted according to the guidelines required by Institutional Animal Care and Use Committee (IACUC) for the use of live rodents at the University of Mississippi Medical Center.
References
-
Tozzi M, Franchin M, Soldini G, Ietto G, Chiappa C, et al. (2013) Impact of static cold storage VS hypothermic machine preservation on ischemic kidney graft: inflammatory cytokines and adhesion molecules as marker of ischemia/reperfusion tissue damage. Our preliminary results. Int J Surgery 11: S110-S114.
-
Sert I, Colak H, Tugmen C, Dogan SM, Karaca C (2014) The effect of cold ischemia time on delayed graft function and acute rejection in kidney transplantation. Saudi J Kidney Dis Transpl 25: 960-966.
-
Rodr�guez NS, Barrasa JL, Gonz�lez MA, Garc�a AL, Caballero JA, et al. (2007) [Assessment of ischemia-reperfusion injury and early acute rejection in experimental lung transplantation after prolonged ischemia]. Arch Bronconeumol 43: 373-377.
-
Carden DL, Granger DN (2000) Pathophysiology of ischaemia-reperfusion injury. J Pathol 190: 255-266.
-
Eltzschig HK, Eckle T (2011) Ischemia and reperfusion--from mechanism to translation. Nat Med 17: 1391-1401.
-
Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357: 1121-1135.
-
Chen GY, Nu�ez G (2010) Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol 10: 826-837.
-
Beutler B (2004) Inferences, questions and possibilities in Toll-like receptor signalling. Nature 430: 257-263.
-
Frantz S, Ertl G, Bauersachs J (2008) Toll-like receptor signaling in the ischemic heart. Front Biosci 13: 5772-5779.
-
Yu L, Wang L, Chen S (2010) Endogenous toll-like receptor ligands and their biological significance. J Cell Mol Med 14: 2592-2603.
-
Prabhu SD (2004) Cytokine-induced modulation of cardiac function. Circ Res 95: 1140-1153.
-
Land WG (2005) The role of post ischemia reperfusion injury and other non-antigen-dependent inflammatory pathways in transplantation. Transplantation 79: 505-514.
-
Land WG (2012) Emerging role of innate immunity in organ transplantation part II: Potential of damage-associated molecular patterns to generate immune-stimulatory dendritic cells. Transplantation Reviews 26: 73-87.
-
Autieri MV, Kelemen S, Thomas BA, Feller ED, Goldman BI, et al. (2002) Allograft inflammatory factor-1 expression correlates with cardiac rejection and development of cardiac allograft vasculopathy. Circulation 106: 2218-2223.
-
Zhou X, He Z, Henegar J, Allen B, Bigler S (2011) Expression of allograft inflammatory factor-1 (AIF-1) in acute cellular rejection of cardiac allografts. Cardiovasc Pathol 20: e177-e184.
-
McDaniel DO, Piazza LK, Barker A, Robertson H, Moore CK et al. (2012) AIF-1 gene expression and polymorphism in association with cardiac allograft rejection. J Clin Exp Cardiology S11: 002.
-
Autieri MV (1996) cDNA cloning of human allograft inflammatory factor-1: tissue distribution, cytokine induction, and mRNA expression in injured rat carotid arteries. Biochem Biophys Res Commun 228: 29-37.
-
Liu G, Ma H, Jiang L, Zhao Y (2007) Allograft inflammatory factor-1 and its immune regulation. Autoimmunity 40: 95-102.
-
Del Galdo F, Jim�nez SA (2007) T cells expressing allograft inflammatory factor 1 display increased chemotaxis and induce a profibrotic phenotype in normal fibroblasts in vitro. Arthritis Rheum 56: 3478-3488.
-
Iris FJ, Bougueleret L, Prieur S, Caterina D, Primas G, et al. (1993) Dense Alu clustering and a potential new member of the NF kappa B family within a 90 kilobase HLA class III segment. Nat Genet 3: 137-145.
-
Utans U, Arceci RJ, Yamashita Y, Russell ME (1995) Cloning and characterization of allograft inflammatory factor-1: a novel macrophage factor identified in rat cardiac allografts with chronic rejection. J Clin Invest 95: 2954-2962.
-
Tian Y, Jain S, Kelemen SE, Autieri MV (2009) AIF-1 expression regulates endothelial cell activation, signal transduction, and vasculogenesis. Am J Physiol Cell Physiol 296: C256-266.
-
Kuschel R, Deininger MH, Meyermann R, Bornemann A, Yablonka-Reuveni Z, et al. (2000) Allograft inflammatory factor-1 is expressed by macrophages in injured skeletal muscle and abrogates proliferation and differentiation of satellite cells. J Neuropathol Exp Neurol 59: 323-332.
-
Utans U, Quist WC, McManus BM, Wilson JE, Arceci RJ (1996) Allograft inflammatory factor-1. A cytokine responsive macrophage molecule expressed in transplanted human hearts. Transplantation 61: 1387-1392.
-
Autieri MV, Carbone C, Mu A (2000) Expression of allograft inflammatory factor-1 is a marker of activated human vascular smooth muscle cells and arterial injury. Arterioscler Thromb Vasc Biol 20: 1737-1744.
-
Barker AK, McDaniel DO, Zhou X, He Z, Aru G, et al. (2010) Combined analysis of allograft inflammatory factor-1, interleukin-18, and Toll-like receptor expression and association with allograft rejection and coronary vasculopathy. Am Surg 76: 872-878.
-
Grimm PC, McKenna R, Nickerson P, Russell ME, Gough J, et al. (1999) Clinical rejection is distinguished from subclinical rejection by increased infiltration by a population of activated macrophages. J Am Soc Nephrol 10: 1582-1589.
-
McDaniel DO, Rigney DA, McDaniel KY, Windham WJ, Redmond P, et al. (2013) Early expression profile of inflammatory markers and kidney allograft status. Transplantation Proceedings 45: 1520-1523.
-
Xiong J, Wang Q, Xue FS, Yuan YJ, Li S, et al. (2011) Comparison of cardioprotective and anti-inflammatory effects of ischemia pre- and postconditioning in rats with myocardial ischemia-reperfusion injury. Inflamm Res 60: 547-554.
-
Land WG (2007) Innate immunity-mediated allograft rejection and strategies to prevent it. Transplant Proc 39: 667-672.
-
Parlakpinar H, Orum MH, Sagir M (2013) Pathophysiology of myocardial ischemia reperfusion injury: A review. Med Science 2: 934-954.
-
Kaczorowski DJ, Tsung A, Billiar TR (2009) Innate immune mechanisms in ischemia/reperfusion. Front Biosci (Elite Ed) 1: 91-98.
-
Takahashi M (2011) Role of the inflammasome in myocardial infarction. Trends Cardiovasc Med 21: 37-41.
-
McDaniel DO, Zhou X, Rigney DA, McDaniel LS, Aru G et al. (2013) Allograft inflammatory factor-1 in cardiac ischemia reperfusion injury: release of molecular markers in an in vitro setting. OJOTS 3: 5-12.
-
McDaniel DO, Zhou X, Moore CK, Aru G (2010) Cardiac allograft rejection correlates with increased expressions of Toll-like receptors 2 and 4 and allograft inflammatory factor 1. Transplant Proc 42: 4235-4237.
-
Leventhal JS, Schr�ppel B (2012) Toll-like receptors in transplantation: sensing and reacting to injury. Kidney Int 81: 826-832.
-
Watano K, Iwabuchi K, Fujii S, Ishimori N, Mitsuhashi S, et al. (2001) Allograft inflammatory factor-1 augments production of interleukin-6, -10 and -12 by a mouse macrophage line. Immunology 104: 307-316.
-
Yang ZF, Ho DW, Lau CK, Lam CT, Lum CT, et al. (2005) Allograft inflammatory factor-1 (AIF-1) is crucial for the survival and pro-inflammatory activity of macrophages. Int Immunol 17: 1391-1397.
-
Tian Y, Kelemen SE, Autieri MV (2006) Inhibition of AIF-1 expression by constitutive siRNA expression reduces macrophage migration, proliferation, and signal transduction initiated by atherogenic stimuli. Am J Physiol Cell Physiol 290: C1083-C1091.
-
Anselmi A, Abbate A, Girola F, Nasso G, Biondi-Zoccai GG, et al. (2004) Myocardial ischemia, stunning, inflammation, and apoptosis during cardiac surgery: a review of evidence. Eur J Cardiothorac Surg 25: 304-311.
-
Oyama J, Blais C Jr, Liu X, Pu M, Kobzik L, et al. (2004) Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 109: 784-789.
-
Cha J, Wang Z, Ao L, Zou N, Dinarello CA, et al. (2008) Cytokines link Toll-like receptor 4 signaling to cardiac dysfunction after global myocardial ischemia. Ann Thorac Surg 85: 1678-1685.
-
Arslan F, Keogh B, McGuirk P, Parker AE (2010) TLR2 and TLR4 in ischemia reperfusion injury. Mediators Inflamm 2010: 704202.
-
Lu C, Ren D, Wang X, Ha T, Liu L, et al. (2014) Toll-like receptor 3 plays a role in myocardial infarction and ischemia/reperfusion injury. Biochim Biophys Acta 1842: 22-31.
-
Jiang W, Kong L, Wu X, Pu L, Wang X (2011) Allograft inflammatory factor-1 is up-regulated in warm and cold ischemia-reperfusion injury in rat liver and may be inhibited by FK506. J Surg Res 165: 158-164.
-
Reutzel-Selke A, Zschockelt T, Denecke C, Bachmann U, Jurisch A, et al. (2003) Short-term immunosuppressive treatment of the donor ameliorates consequences of ischemia/reperfusion injury and long-term graft function in renal allografts from older donors. Transplantation 75:1786-1792.
-
Lutz J, Th�rmel K, Heemann U (2010) Anti-inflammatory treatment strategies for ischemia/reperfusion injury in transplantation. J Inflamm (Lond) 7: 27.
-
Xiong J, Yuan YJ, Xue FS, Wang Q, Li S, et al. (2012) Combined postconditioning with ischemia and a7nAChR agonist produces an enhanced protection against rat myocardial ischemia reperfusion injury. Chin Med J (Engl) 125: 326-331.
-
Janeway CA Jr (1992) The immune system evolved to discriminate infectious nonself from noninfectious self. Immunol Today 13: 11-16.
-
Matzinger P (1994) Tolerance, danger, and the extended family. Annu Rev Immunol 12: 991-1045.
-
Guo J, Wang SB, Yuan TY, Wu YJ, Yan Y, et al. (2013) Coptisine protects rat heart against myocardial ischemia/reperfusion injury by suppressing myocardial apoptosis and inflammation. Atherosclerosis 231: 384-391.





