International Journal of Clinical Cardiology
Herpes Simplex Encephalitis, an Unusual Cause of Sinus Node Dysfunction: Case Report and Literature Review
Pierre-Philippe Nicol1*, Jean-Philippe Talarmin2 and Thierry Joseph1
1Department of Cardiology, Hôpital Cavale Blanche, France
2Department of Internal Medicine and Infectious Diseases, Hôpital Cavale Blanche, France
*Corresponding author: Pierre-Philippe Nicol, Department of Cardiology, Hôpital Cavale Blanche, Boulevard Tanguy Prigent, 29609 Brest Cedex, France, Tel: +33298347505, E-mail : pph.ncl@gmail.com
Int J Clin Cardiol, IJCC-2-042, (Volume 2, Issue 4), Case Report; ISSN: 2378-2951
Received: May 30, 2015 | Accepted: July 23, 2015 | Published: July 25, 2015
Citation: Nicol PP, Talarmin JP, Joseph T. (2015) Herpes Simplex Encephalitis, an Unusual Cause of Sinus Node Dysfunction: Case Report and Literature Review. Int J Clin Cardiol 2:042. 10.23937/2378-2951/1410042
Copyright: © 2015 Nicol PP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Herpes simplex encephalitis is responsible for progressive alteration of behaviour, epilepsy, focal neurological signs, and cognitive problems, but is rarely involved in sinus node dysfunction. We describe this association in a 66-year-old woman with a complete cardiac and neurologic recovery after acyclovir treatment. Consequently, this aetiology of sinoatrial arrest should be recognized in order to systematically assess cardiac monitoring and avoid unnecessary pacemaker implantation. Moreover the literature shows that sinus node dysfunction may also result from several other neurological diseases.
Keywords
Sinus node dysfunction, Syncope, Herpes simplex encephalitis
Introduction
Herpes simplex encephalitis is responsible for progressive alteration of behaviour, epilepsy, focal neurological signs, and cognitive problems, but is rarely involved in sinus node dysfunction. We describe this association in a 66-year-old woman. Available data of literature is also reviewed and discussed.
Case Report
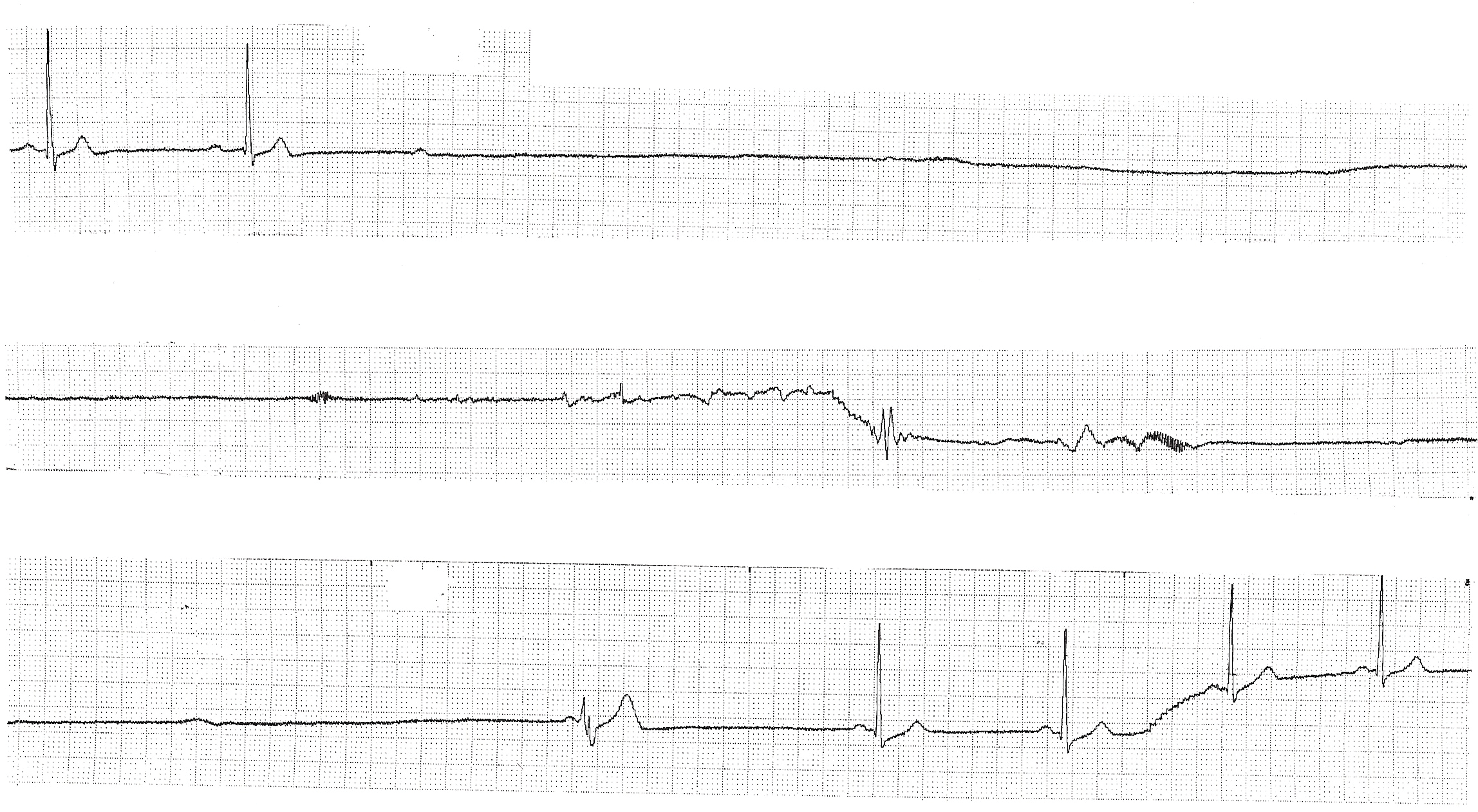
A 66-year-old woman was admitted to the emergency department of a primary hospital following an abrupt transient loss of consciousness. Her past medical history included a mitral valve prolapse with moderate regurgitation. Shortly after arrival, she presented repeated syncopal episodes with documented sinus pauses on cardiac monitoring (Figure 1), efficiently treated with intravenous isoprenalin.
The patient was referred to our institution for a pacemaker implantation, which was delayed because of a fever with episodes of acute confusional state, including troubles in spatial and temporal orientation with a Glasgow coma score of 14, without speech or memory impairment. Clinical examination revealed severe systolic murmur of mitral regurgitation (MR) without heart failure. The neurological exam revealed no evidence of focal neurological signs, neck stiffness, epilepsy.
Transthoracic and transoesophageal echocardiography showed moderate to severe MR with ruptured chordae and prolapse of the posterior leaflet without vegetation or abscess, ruling out the diagnosis of infectious endocarditis that had been initially suspected. Moreover, blood cultures remained sterile, and procalcitonin and C reactive protein were normal. The cerebral computed tomography (CT) scan was also normal. A lumbar puncture was performed and examination of the cerebrospinal fluid (CSF) showed a mildly raised protein level of 0.68g/L, a normal glucose level of 3.87mmol/L, and an elevated white blood cell count with lymphocytes predominance (69/mm3). Polymerase Chain Reaction (PCR) documented herpes simplex virus (HSV) type 1 DNA in the CSF, and intravenous acyclovir (10mg/kg/8h) was initiated for a total duration of 3 weeks.
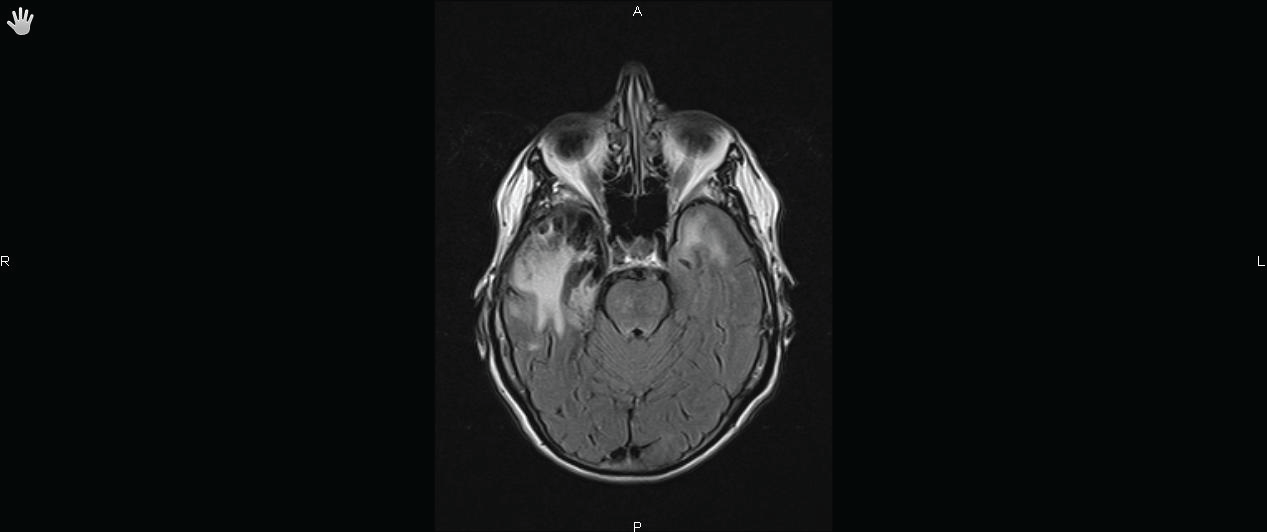
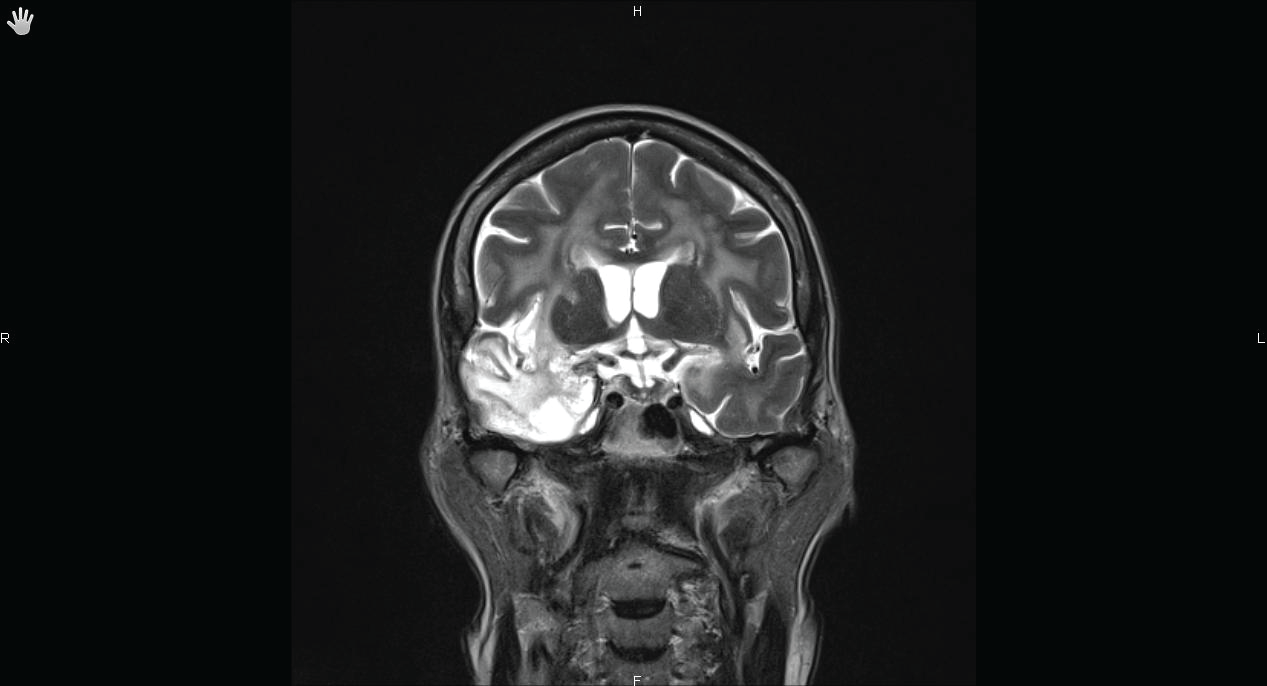
Unfortunately, the patient experienced refractory seizures requiring prolonged sedation and mechanical ventilation in the intensive care unit (ICU). The evolution was marked by the recovery of sinus node dysfunction allowing the discontinuation of isoprenalin, and disappearance of epileptic figures on electroencephalogram (EEG). After a 3-months follow-up, there was no sinus pause recurrence and a complete neurologic recovery was achieved. A cerebral Magnetic Resonance Imaging (MRI) performed seven months after the onset of the illness showed persistent signal abnormalities in the right temporal lobe and more particularly in the right insula (Figures 2A,2B).
Discussion
Encephalitis is defined by the presence of an inflammatory process of the brain in association with clinical evidence of neurologic dysfunction [1]. Diagnostic criteria and algorithm for the etiological diagnosis of encephalitis have been recently described by the International Encephalitis Consortium [2]. Major and minor criteria required for the diagnosis of possible, probable or confirmed encephalitis are summarized in table 1.
![]()
Table 1: Diagnostic Criteria for Encephalitis and Encephalopathy of Presumed Infectious or Autoimmune Aetiology (extracted from Venkatesan et al. [2])
View Table 1
Etiological diagnosis of encephalitis remains a medical challenge. Despite extensive testing 32 to 75% of cases of presumed infectious encephalitis remain unexplained [1]. Many pathogens have been involved, mainly viruses and primarily HSV1 in adults [3,4]. In a French prospective study including 253 patients with acute infectious encephalitis, an etiologic diagnosis could be established in only 52% of cases, HSV representing the most frequent isolated pathogen (42%) [5].
Common symptoms of herpes simplex encephalitis include fever, headache, consciousness impairment, focal neurological signs, and seizures. Definite diagnosis is based on PCR detection of HSV DNA in the CSF, with a sensitivity of 98%, a specificity of 94%, a positive predictive value of 95% and a negative predictive value of 98% in adults [1,3,6]. In more than 90% of patients, MRI reveals temporal and/or inferior frontal lobe oedema with high signal intensity on FLAIR and T2-weighted images [1]. Recent data suggested temporal trends of brain involvement with a relative superiority of DWI sequences over FLAIR's ones to detect involved areas (especially cortical) in acute and subacute phase of HSV encephalitis except for thalamic attempt early identified by FLAIR imaging [7]. In the late phase of disease, FLAIR is superior, mainly to detect white matter involvement.
Treatment of herpes simplex encephalitis is based on intravenous acyclovir (10 mg/kg/8h), administered as soon as possible [1]. Indeed, despite antiviral therapy 18-month mortality remains high reaching 28% [1], but is considerably decreased by early initiation of treatment. However, survivors frequently suffer from neurological sequelae that greatly impair their quality of life [8]. Interestingly, our patient presented complete neurological recovery, despite a diagnostic delay of four days due to the atypical presentation with inaugural cardiac manifestations.
Sinus node dysfunction secondary to HSV encephalitis has rarely been reported in the literature. To the best of our knowledge, only two confirmed and two probable cases have been published, another one remaining more controversial, according to the diagnostic criteria of the International Encephalitis Consortium [2]. The main characteristics of these five cases are summarized in table 2.
![]()
Table 2: Main characteristics of the five reported cases
View Table 2
Smith et al. [9] reported the case of a 59-year-old woman suffering from infectious encephalitis complicated by periods of sinus arrest, requiring temporary cardiac pacing. HSV encephalitis was confirmed, as PCR detection of HSV DNA in the CSF was positive. Acyclovir therapy resulted in complete cardiac recovery, with no need for a permanent pacemaker implantation.
Pollock et al. [10] published a case of infectious encephalitis, presenting as a sick sinus syndrome in a 60-year-old man admitted for multiple fainting attacks. A permanent cardiac pacemaker was implanted on the first day of hospitalization. In the next three days, the patient experienced neurological deterioration without abnormalities on two consecutive cerebral CT scans and two lumbar punctures. Twelve days after admission, acyclovir therapy was initiated because of sharp activity in the left temporal lobe on EEG, and appearance of low attenuation in the left frontal and temporal lobes on CT scan. Unfortunately, the patient died the same day. HSV 1 encephalitis was confirmed by necropsy.
Saffran et al. [11] reported the case of a 39-year-old man presenting with several episodes of asystole requiring temporary cardiac pacing, associated with seizures, lethargy, confusion, and fever. Acyclovir therapy was initiated because of an elevated white blood cell count in the CSF, and presence of an extensive area of lucency in the right temporal lobe on the CT scan. Interestingly, an electrophysiological heart study revealed no abnormalities. After completion of antiviral therapy and cessation of cardiac pacing, there was no recurrence of syncope. According to the International Encephalitis Consortium guidelines [2], infectious encephalitis is probable. Unfortunately HSV aetiology could not be confirmed as no PCR was performed, but remains likely.
Alsolaiman et al. [12] also reported a case of infectious encephalitis in a 48-year-old woman presenting with multiple periods of asystole due to sinoatrial arrest, requiring temporary cardiac pacing. According to the International Encephalitis Consortium guidelines [2], infectious encephalitis is probable. Unfortunately, PCR testing for HSV was not performed on the CSF. Nevertheless, herpetic aetiology remains likely, as MRI features are typical, and treatment with intravenous acyclovir produced a complete recovery, with a follow-up of more than 4 years [12].
In 2009, Gooch [13] reported the case of a 33-year-old man presenting with seizures and asystole requiring permanent pacemaker implantation. Infectious encephalitis was suspected, and acyclovir was initiated. According to the International Encephalitis Consortium guidelines [2], this case seems more controversial, as the patient presented no altered mental status, no fever, no focal neurological sign. Two lumbar punctures were normal with negative PCR for HSV. MRI could not be performed because of the pacemaker implantation, but two CT scans of the head were normal. EEG only showed nonspecific abnormality of the right temporal lobe [13]. Therefore, in our opinion, despite a favourable outcome with acyclovir therapy, the diagnosis of HSV encephalitis remains doubtful.
Interestingly, in all of these patients, diagnosis of encephalitis was delayed because of first sinus node dysfunction, resulting in cardiology units admission for temporary or permanent cardiac pacing. In our case pacemaker implantation was postponed only because of fever and initial suspicion of infectious endocarditis.
In the literature cases, antiviral therapy was initiated between 2 and 13 days after symptoms beginning, with a complete cardiac recovery for all the patients except one, suggesting that cardiac manifestations were reversed by antiviral treatment. Similarly, our patient presented no additional episode of sinus node dysfunction on cardiac monitoring after the beginning of acyclovir.
Alongside this cardiac improvement, it is noteworthy that all surviving patients including our case presented almost complete neurological recovery.
It is hypothesized that the sinus node dysfunction occurring during encephalitis results from lesions of the central nervous system and not from primary cardiac attempt. Indeed, in the fatal case, necropsy revealed no cardiac structure abnormalities especially in conduction pathways [10]. Likewise, polymorphic Ventricular Tachycardia (VT) secondary to acquired long QT syndrome has been described in a 26-year old woman hospitalized for confirmed herpes encephalitis. VT was never detected again after antiviral therapy. As electrocardiogram, cardiac MRI and genetic studies were negative, the authors suggested that these cardiac manifestations were triggered by cerebral infection [14].
Moreover, asystole or others cardiac arrhythmias have been also reported in case of seizures and ischaemic strokes, especially originating from temporal lobes, and could possibly be related to some unexplained sudden death observed in these situations [15,16].
Neurological lesions (cortical and subcortical abnormalities including insular region) due to autoimmune mechanisms, such as anti-N-methyl-D-aspartate receptors (NMDAR) encephalitis, associated in many cases with tumors such as ovarian teratoma, have been also found to be associated with sinus node dysfunction [17,18], generally reversed by tumor extraction and/or immunosuppressive therapy. It's noteworthy that recent investigations indicate that herpes simplex encephalitis can trigger autoimmunity and then NMDAR encephalitis [19]. But in our patient, presence of N-methyl-D-aspartate receptors (NMDA-R) antibodies was unfortunately not tested in serum or CSF.
All these data highlight the fact that injury of insular cortex especially in its right side, that is thought to be involved in the control of autonomic system, can result in central sinus node dysfunction [20], whatever the aetiology [15]. Anterior cingulated cortex seems to play a major role in the central autonomic network [21], but to our knowledge, lesions of this area have not been proved to be associated with cardiac disturbance.
HSV encephalitis is a rare and possibly underestimated cause of reversible sinus node dysfunction. Hence, this association should be recognized when encephalitis is suspected, in order to systematically monitor cardiac rhythm, and to avoid unnecessary permanent cardiac pacing. Antiviral treatment should be promptly initiated, resulting in cardiac recovery and preventing severe neurological complications. Heart specialists should be aware that sinus node dysfunction can be related to a neurological disease and can be successfully treated by an aetiology-related and not specific cardiac therapy.
References
-
Tunkel AR, Glaser CA, Bloch KC, Sejvar JJ, Marra CM, et al. (2008) The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 47: 303-327.
-
Venkatesan A, Tunkel AR, Bloch KC, Lauring AS, Sejvar J, et al. (2013) Case definitions, diagnostic algorithms, and priorities in encephalitis: consensus statement of the international encephalitis consortium. Clin Infect Dis 57: 1114-1128.
-
Stahl JP, Mailles A, Dacheux L, Morand P (2011) Epidemiology of viral encephalitis in 2011. Med Mal Infect 41: 453-464.
-
Jmor F, Emsley HC, Fischer M, Solomon T, Lewthwaite P (2008) The incidence of acute encephalitis syndrome in Western industrialised and tropical countries. Virol J 5: 134.
-
Mailles A, Stahl JP; Steering Committee and Investigators Group (2009) Infectious encephalitis in france in 2007: a national prospective study. Clin Infect Dis 49: 1838-1847.
-
Debiasi RL, Tyler KL (2004) Molecular methods for diagnosis of viral encephalitis. Clin Microbiol Rev 17: 903-925, table of contents.
-
Renard D, Nerrant E, Lechiche C (2015) DWI and FLAIR imaging in herpes simplex encephalitis: a comparative and topographical analysis. J Neurol .
-
Raschilas F, Wolff M, Delatour F, Chaffaut C, De Broucker T, et al. (2002) Outcome of and prognostic factors for herpes simplex encephalitis in adult patients: results of a multicenter study. Clin Infect Dis 35: 254-260.
-
Smith BK, Cook MJ, Prior DL (2008) Sinus node arrest secondary to HSV encephalitis. J Clin Neurosci 15: 1053-1056.
-
Pollock S, Reid H, Klapper P, Metcalfe RA, Ahmed N (1986) Herpes simplex encephalitis presenting as the sick sinus syndrome. J Neurol Neurosurg Psychiatry 49: 331-332.
-
Saffran L, Goldner BG, Adler H, Feingold BD, Feingold RM, et al. (1995) Asystole associated with herpes simplex encephalitis. J Invasive Cardiol 7: 152-155.
-
Alsolaiman MM, Alsolaiman F, Bassas S, Amin DK (2001) Viral encephalitis associated with reversible asystole due to sinoatrial arrest. South Med J 94: 540-541.
-
Gooch R (2009) Ictal asystole secondary to suspected herpes simplex encephalitis: a case report. Cases J 2: 9378.
-
Huemer M, Boldt LH, Wutzler A, Parwani A, Rolf S, et al. (2012) Polymorphic ventricular tachycardia in a patient with herpes encephalitis. J Clin Neurosci 19: 483-484.
-
Sörös P, Hachinski V (2012) Cardiovascular and neurological causes of sudden death after ischaemic stroke. Lancet Neurol 11: 179-188.
-
Duncan S, Brodie MJ (2011) Sudden unexpected death in epilepsy. Epilepsy Behav 21: 344-351.
-
Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, et al. (2008) Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7: 1091-1098.
-
Nazif TM, Vázquez J, Honig LS, Dizon JM (2012) Anti-N-methyl-D-aspartate receptor encephalitis: an emerging cause of centrally mediated sinus node dysfunction. Europace 14: 1188-1194.
-
Armangue T, Leypoldt F, Málaga I, Raspall-Chaure M, Marti I, et al. (2014) Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann Neurol 75: 317-323.
-
Oppenheimer SM, Wilson JX, Guiraudon C, Cechetto DF (1991) Insular cortex stimulation produces lethal cardiac arrhythmias: a mechanism of sudden death? Brain Res 550: 115-121.
-
Critchley HD, Mathias CJ, Josephs O, O'Doherty J, Zanini S, et al. (2003) Human cingulate cortex and autonomic control: converging neuroimaging and clinical evidence. Brain 126: 2139-2152.