Journal of Dermatology Research and Therapy
A Fatal Infection due to Gordonia Terrae
Jeong Rae Yoo1, Miyeon Kim2, Keun Hwa Lee3, Seung Jin Yoo4, Chang lim Hyun5, Jonghwan Bae6, Sang Taek Heo1 and Yong-Geun Park6*
1Division of Infectious Disease, Jeju National University, Republic of Korea
2Division of Nephrology, Jeju National University, Republic of Korea
3Department of Microbiology, Jeju National University, Republic of Korea
4Chung-Ang University College of Medicine, Seoul, Republic of Korea
5Department of Pathology, Jeju National University, Republic of Korea
6Department of Orthopedic Surgery, Jeju National University, Republic of Korea
*Corresponding author:
Yong-Geun Park, M.D, Department of Orthopedic Surgery, Jeju National University, 15, Aran 13 gil, Jeju-si, Jeju Special-Governing Province 690-756, Republic of Korea, Tel: +82-64-717-2710, Fax: +82-64-717-1131, E-mail: pyk184@hanmail.net
J Dermatol Res Ther, JDRT-2-033, (Volume 2, Issue 5), Case Report; ISSN: 2469-5750
Received: August 04, 2016 | Accepted: October 03, 2016 | Published: October 05, 2016
Citation: Yoo JR, Kim M, Lee KH, Yoo SJ, Hyun CI, et al. (2016) A Fatal Infection due to Gordonia Terrae. J Dermatol Res Ther 2:033. 10.23937/2469-5750/1510033
Copyright: © 2016 Yoo JR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Mycetoma, Actinomycetoma, Gordonia terrae, Surgery, rRNA sequencing
Mycetoma is a chronic granulomatous, non-contagious disease of the subcutaneous tissues, this most often caused by Norcadia, Actinomycetes or fungi [1]. It is endemic in tropical and subtropical areas but not in temperate climates such as South Korea [2]. Among many causative pathogens for mycetoma, Gordonia species, rare nocardioform actinomycetes, are difficult to be identified by microbiologic and serologic tests [3,4], because the pathogen requires a precise identification using genomic sequencing [1]. In addition, Gordonia species have been reported in just a few numbers of case series, and its epidemiology and clinical importance have been underestimated [5]. Herein, we report a rare case of mycetoma caused by Gordonia terrae in an immunocompetent patient in a temperate climates area.
A 53-year-old man with a history of poliomyelitis underwent left total hip arthroplasty in December 2011. His past medical history was not significant except the medical conditions described disease. The patient had developed a sore on right perianal area since March 2013 (Figure 1A), and infected sore lesion was extended to abscess on a right buttock abscess one year later, it was managed with incision and drainage in right buttock abscess since November 2014. However, a few months later, a multifocal erosions and a sinus tract were presented in the same lesion (Figure 1B). The patient was again admitted for extensive surgical treatment. On admission, his body temperature remained within a normal range during hospitalization. Laboratory data showed white blood cell count 14,200 × 103/μl, hemoglobin 8.8 mg/dL, high sensitiviy C-reactive protein concentration 11.21 mg/dL, and erythrocyte sedimentation rate 120 mm/hr. Other laboratory results were normal. An initial computed tomography of the pelvis revealed an enhancement of skin and soft tissues on the right buttock area. Consequently, intravenous antibiotics treatment with daily 2 gram of ceftriaxone was initiated and continued for 7 days along with drainage. An initial blood culture result showed no pathogens, but a tissue culture of the wound site identified a penicillin susceptible Streptococcus anginosus and methicillin susceptible Staphylococcus aureus. All laboratory examinations for Mycobacterium tuberculosis, nontuberculous mycobacteria, and fungi were negative. Therefore, intravenous ceftriaxone was switched to intravenous Cefazolin with a daily dose of 6 grams (2 grams three times per day) according to antibiotic susceptibility test. However, the skin lesion continued to deteriorate despite adequate antibiotics trials. Following the result for gram stain of the wound site revealed a gram-positive bacilli, and the colonial morphology showed small and white-colored convex colonies on blood agar (Figure not shown). Furthermore, Corynebacterium species were identified in blood culture using the Vitek II system (bioMerieux Vitek Inc., Durham, NC). Consequently, vancomycin, ceftriaxone and clindamycin combination, and piperacillin/tazobactam were tried to target Corynebacterium species for a total of 4 months during hospitalization due to lack of response. In addition, the patient underwent incision and drainage precedures more than twenty times over the course of hospitalization. Despite medical and surgical treatments, his medical condition did not improve, and the extent of lesion became deeper and wider. Therefore, we decided to identify by 16S rRNA gene sequencing for gram-positive bacilli from blood and wound specimens at the another microbiology and immunology laboratory, the gram-positive bacilli was identified as Gordonia terrae by 16S rRNA gene sequencing (96% mach with type strain) using a pair 27FLP (5'-AGAGTTTGATCMTGGCTCAG-3') and 1492RPL (5'-GGTTACCTTGTTACGACTT-3') [5,6]. Unfortunately, an antibiotics susceptibility test was not performed due to lack of technological availability at our institution. As reported in previous articles, an antibiotic regimen was changed to a combination of intravenous amoxicillin/sulbactam and ciprofloxacin for 7 days. Despite changes in antibiotics regimen, his medical condition did not show any improvement, which made us consider a wider surgical resection rather than an incision and drainage procedure. However, the patient refused both blood transfusion for anemia of chronic disease and surgical treatment causing intraoperative blood loss, owing to the beliefs of Jehovah’s Witnesses. Then, we alternatively switched an antibiotic regimen to 500 mg of intravenous imipenem per 8 hours and 500 mg of levofloxacin per 24 hours. Despite multiple alterations in antibiotic regimens, the wound still continued to worsen for the following two weeks. At last, the patient was transferred to another hospital where surgical resection was possible without blood transfusion. However, he had refused the operation after all and was transferred back to our hospital three months later. While delaying in treatment, the wound developed to painless mycetoma in the subcutaneous tissue of right hip (Figure 1C and Figure 1D). Unfortunately, the patient died due to septic shock and multiple organ failure.
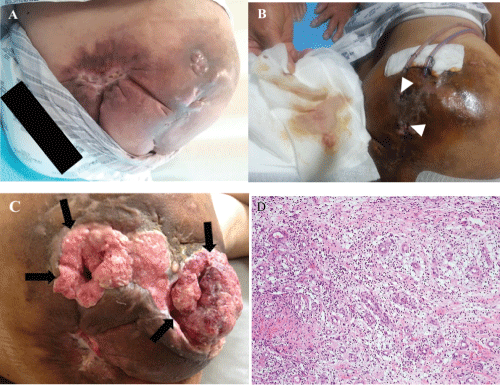
.
Figure 1:Clinical photo showing the changed morphologoies of a patient's lesion and histopathology of mycetoma.
(A) There are no signs of active inflammation until October 2014; (B) He had a pus discharge and multifocal sinus tract in the right hip after three months (white arrowhead; sinus tract); (C) The bulk tumorous lesion was showed in the subcutaneous tissue of right hip in July 2015 (after 7 months) (black arrow, granulation tissues); (D) Tissue section of a biopsy of the lesion showed a proliferations of vascular tissue with reactive endothelial cells, plump fibroblasts, and a mixed inflammatory infiltrate (granulation tissue). Focally, a dense cluster of neutrophils is seen in the vascular lumen. (H & E, ×100).
View Figure 1
Mycetoma which is reported 97% were actinomycetoma and 3.5% were eumycetoma is a major problem in tropical and subtropical areas [1]. The trial of a painless firm subcutaneous mass, multiple sinus formation, and a purulent discharge that contains grains is pathognomic of mycetoma [1]. Our case fulfilled the above signs. We identified Gordonia terrae from blood and gram positive bacilli from the wound by the 16S ribosomal RNA gene sequencing. To our knowledge, mycetoma caused by Gordonia terrae is a rare in temperate climate, although a case series has been just reported in worldwide.
Gordonia terrae has been reported in central venous catheter related infection [5,7-12], primary bacteremia [5], skin and soft tissue infection [5,13], brain abscess [11,14], traumatic wound infection [5], acute cholecystitis [15], suppurative mastitis [16], and abdominal wall abscess [5]. Previous cases were common in indwelling catheter-related blood stream infection and primary bacteremia in immunocompromised hosts, followed by skin and soft tissue infection. Among these reports, only few mycetoma related reports were found. And most of the cases were improved with antibiotic combination treatment with/without surgery. However, in the current case, the patient’s condition led to death despite antibiotic combination treatment and multiple procedures of incision and drainage because firstly antibiotic susceptibility test for Gordonia species was not available at our institution, secondly extensive surgical resection was not performed, and lastly Gordonia species are frequently misidentified as Rhodococcus, Corynebacterium, and Norcardia by conventional microbiological culture, which may lead to delay in accurate diagnosis [5,12]. Thus, mycetoma caused by Gordonia terrae would be consider as one of fatal pathogens causing skin and soft tissue infection.
As stated above, fatal infections by Gordonia species are rare disease in infected sore disease and in temperate climate areas. Because it is time consuming to identify Gordonia terrae in clinical settings, physicians would be consider a molecular sequencing methods for chronic tumorous granulation tissues and extensive surgical treatments for bulky mycetoma.
Acknowledgements
We thank Geun Hwa Lee and collagues for help with 16S rRNA gene sequencing at the department of microbiology and immunology, Jeju National University, Jeju, South Korea.
Conflict of Interest
No potential conflict of interest relevant to this article.
ORCID
Jeong Rae Yoo https://orcid.org/0000-0002-5488-7925.
References
-
Tsukamura M (1971) Proposal of a new genus, Gordona, for slightly acid-fast organisms occurring in sputa of patients with pulmonary disease and in soil. J Gen Microbiol 68: 15-26.
-
Arenskotter M, Broker D, Steinbuchel A (2004) Biology of the metabolically diverse genus Gordonia. Appl Environ Microbiol 70: 3195-3204.
-
Zijlstra EE, van de Sande WW, Welsh O, Mahgoub el S, Goodfellow M, et al. (2016) Mycetoma: A unique neglected tropical disease. Lancet Infect Dis 16: 100-112.
-
Lichon V, Khachemoune A (2006) Mycetoma: A review. Am J Clin Dermatol 7: 315-321.
-
Lai CC, Wang CY, Liu CY, Tan CK, Lin SH, et al. (2010) Infections caused by Gordonia species at a medical centre in Taiwan, 1997 to 2008. Clin Microbiol Infect 16: 1448-1453.
-
Suzuki MT, Giovannoni SJ (1996) Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes by PCR. Appl Environ Microbiol 62: 625-630.
-
Blaschke AJ, Bender J, Byington CL, Korgenski K, Daly J, et al. (2007) Gordonia species: emerging pathogens in pediatric patients that are identified by 16S ribosomal RNA gene sequencing. Clinical Infectious Diseases 45: 483-486.
-
Buchman AL, McNeil MM, Brown JM, Lasker BA, Ament ME (1992) Central venous catheter sepsis caused by unusual Gordona (Rhodococcus species: identification with a digoxigenin-labeled rDNA probe. Clin Infect Dis 15: 694-697.
-
Pham AS, Dé I, Rolston KV, Tarrand JJ, Han XY (2003) Catheter-related bacteremia caused by the nocardioform actinomycete Gordonia terrae. Clin Infect Dis 36: 524-527.
-
Grisold AJ, Roll P, Hoenigl M, Feierl G, Vicenzi-Moser R, et al. (2007) Isolation of Gordonia terrae from a patient with catheter-related bacteraemia. J Med Microbiol 56: 1687-1688.
-
Drancourt M, Pelletier J, Cherif AA, Raoult D(1997) Gordona terrae central nervous system infection in an immunocompetent patient. J Clin Microbiol 35: 379-382.
-
Ramanan P, Deziel PJ, Wengenack NL (2013) Gordonia bacteremia. J Clin Microbiol 51: 3443-3447.
-
Bakker XR, Spauwen PH, Dolmans WM (2004) Mycetoma of the hand caused by Gordona terrae: a case report. J Hand Surg Br 29: 188-190.
-
Drancourt M, McNeil MM, Brown JM, Lasker BA, Maurin M, et al. (1994) Brain abscess due to Gordona terrae in an immunocompromised child: case report and review of infections caused by G. terrae. Clin Infect Dis 19: 258-262.
-
Gil-Sande E, Brun-Otero M, Campo-Cerecedo F, Esteban E, Aguilar L, et al. (2006) Etiological misidentification by routine biochemical tests of bacteremia caused by Gordonia terrae infection in the course of an episode of acute cholecystitis. J Clin Microbiol 44: 2645-2647.
-
Zardawi IM, Jones F, Clark DA, Holland J (2004) Gordonia terrae-induced suppurative granulomatous mastitis following nipple piercing. Pathology 36: 275-278.





