Journal of Dermatology Research and Therapy
Curcumin-Induced Apoptosis in the Repair of Photodamaged Skin
Madalene CY Heng*
UCLA School of Medicine, USA
*Corresponding author: Madalene CY Heng MD, FRACP, FACD, FAAD, Professor of Medicine/Dermatology/David Geffen UCLA School of Medicine, 500 Paseo Camarillo, suite 100, Camarillo California 93010, USA, E-mail: madaleneheng@aol.com; madalene@madalenehengmd.com
J Dermatol Res Ther, JDRT-3-044, (Volume 3, Issue 1), Case Report; ISSN: 2469-5750
Received: October 24, 2016 | Accepted: March 18, 2017 | Published: March 20, 2017
Citation: Heng MCY (2017) Curcumin-Induced Apoptosis in the Repair of Photodamaged Skin. J Dermatol Res Ther 3:044. 10.23937/2469-5750/1510044
Copyright: © 2017 Heng MCY. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Ultraviolet light-induced damage to the DNA resulting in double-stranded triggers the DNA Damage Response (DDR). This results in Cell Cycle Arrest (CCA) and Nucleotide Excision and Repair (NER) of the photo-damaged DNA. These processes are regulated by a family of phosphatidylinositol-3-kinase family. Cell cycle arrest is inhibitory to rapid healing of tissues. When damage is severe, apoptosis is induced only after multiple failed attempts at repair. Moreover, the intrinsic repair processes are slow and laborious, and do not return the tissues to pre-injury levels. When large segments of the DNA are damaged, or if the damage involves both strands of the DNA, replication errors occur, producing a potentially premalignant cell. The activation of the DDR pathway is regulated by a family of phosphatidylinositol-3 kinase family, the phosphorylation of which may be enhanced by phosphorylase kinase. It is proposed that by avoiding activation of the DDR pathways through inhibition of phosphorylase kinase, curcumin may induce early apoptosis and avoid sequestration of the healing process in "Cell Cycle Arrest" The advantages of early apoptosis allows not only for removal of damaged cells, but prevents the survival of potentially premalignant cells in burns and premalignant lesions in photo-damaged skin. The replacement of damaged cells by new healthy cells allows for more complete repair process than would otherwise occur.
Introduction
Ultraviolet light-induced DNA damage results in helix distorting DNA lesions predominantly consisting of cyclobutane pyrimidine dimers (CPDs) and 6-4-photoproducts (6-4PP) [1-4]. The damaged DNA segments are removed by nucleotide excision repair (NER) that eliminates large segments of helix distorting lesions in a multistep "cut and patch" type reaction [5]. The damaged DNA is excised as a 25-30 oligonucleotide DNA fragment followed by gap filling through DNA repair synthesis and restoration of an intact DNA duplex by a final ligation step.
Cells activate distinct signaling pathways, in particular, the DNA damage response (DDR pathway, in response to various types of DNA damage, in particular to double stranded DNA breaks (DSBs). The recognition of these breaks is achieved by the multifunctional sensor complex, MRN [5] (MRE11-RAD50-NBS1) that consists of the structure-specific nuclease (MRE11; meiotic recombination 11 protein complex), the ATPase and adenylate kinase subunit (RAD50; radiation sensitive DNA repair protein 50), and the adaptor protein (NSB1; Nijmegan syndrome breakage 1). MRN complexes can bind to the exposed DNA ends directly and unwind the DNA ends in an ATP-dependent manner. One or the earliest events in DDR is the recruitment of the phosphatidylinositol 3-kinase-like kinase (PI-3KK), ATM kinase, by the MRN sensor complex [5-9]. This leads to the dissociation of the inactive dimer, and activation of ATM kinase, a key early enzyme in the DDR pathway.
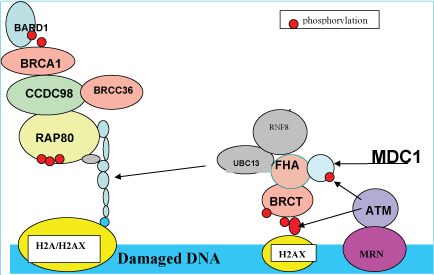
The presence of double stranded DNA breaks (DSB) following ultraviolet injury triggers the DNA damage response (DDR) signaling pathway (Figure 1), through activation of ATM kinase, resulting in nucleotide excision and repair (NER) through histone (H2A) ubiquitination [5,10]. ATM (ataxia telangiectasia mutated) kinase, a member of the phosphatidylinositol 3-kinase family, is responsible for phosphorylating H2AX at ser193 (γH2AX) in response to DSBs [10]. In chronic ultraviolet injury, the ultraviolet-induced replication stress activates ATR (ataxia telangiectasia rad 3 related) kinase, another member of the phosphatidylinositol 3-kinase family, resulting in the recruitment of ATR kinase by ATR-interacting protein to replication protein A (RPA)-coated single stranded DNA, which occurs at stalled replication forks [11]. Both ultraviolet damage and ultraviolet replicating stress triggers DNA damage signaling involving (1) RAD17, (2) the 9-1-1 complex, (3) ATM, (4) ATR, (5) CHK1 (checkpoint protein 1 and its mediator, MDC1 (mediator of DNA damage checkpoint protein 1) [12,13], and (6) CHK2 [14] (checkpoint protein 2) resulting in NER (nucleotide excision repair)-dependent histone H2A ubiquitination at sites of ultraviolet-induced DNA damage. Histone ubiquitination is achieved by the ubiquitin ligases, Ubc13 (E2 ubiquitin ligase) and RNF8 (E3 ubiquitin ligase). RNF8, which contains an ubiquitin interacting motif (UIM), is recruited to the sites of DNA damage through interaction of DSBs (DNA double-stranded breaks) and MDC1 (DDR scaffold protein) in a cell-cycle independent manner. The ATM-phosphorylated MDC1 scaffold protein, binds to the FHA (fork-head associated) domain of RNF8, which subsequently binds to the E2 ubiquitin ligase, Ubc13 [15]. This complex catalyzes K-63-linked polyubiquitination of histones H2A and γH2AX, which is recognized by RAP80 (receptor associated protein 80), a subunit of DNA-protein kinase (DNA-PK), thereby facilitating recruitment of the BRCA1/BARD1/CCDC98/RAP80 complex to DSB sites for DNA repair [15-17]. Figure 1 summarizes the recruitment of proteins to sites of DSB for DNA repair.
.
Figure 1: Recruitment of repair proteins to sites of DNA damage (double stranded breaks).
View Figure 1
Cells have developed checkpoint proteins to coordinate complex regulatory networks to sense DNA damage, coordinate DNA replication, cell-cycle arrest and DNA repair. The histone protein, H2AX, specifically controls recruitment of DNA repair proteins to sites of DNA damage. The MDC1 (mediator of CHK1) works with H2AX to promote recruitment of repair proteins to sites of DNA breaks, as well as induce cell-cycle arrest checkpoints [18]. Although it is known that these interactions are achieved in a phosphorylation-dependent manner, the exact protein kinase(s) responsible for these interactions are still uncertain. It has been proposed, however, that phosphorylation at multiple sites triggers ubiquitylation signals [19]. In addition, certain reactions can be bypassed. For example, DNA-PK is able to regulate H2AX phosphorylation in response to DNA damage by bypassing ATM-dependent phosphorylation [20].
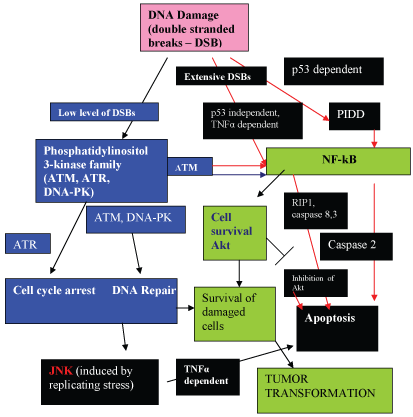
To ensure genomic integrity, cells have developed DDR pathways that sense DNA damage, and catalyze a multifaceted response that coordinates cell cycle arrest, apoptosis and DNA repair networks. The regulation of cell cycle checkpoints and DNA repair pathways are strictly coordinated and under tight control to achieve accurate DNA repair and prevent the introduction of errors. The genome is particularly vulnerable during DNA replication. If not properly repaired, cell can undergo apoptosis or senescence. Alternatively, dysregulation or impaired function of proteins involved in the improper repair during the DDR can result in chromosomal changes, genomic instability and increased cancer risk. The DDR pathways which result in apoptosis, cell cycle arrest, and DNA repair pathways are summarized in Figure 2.
Apoptosis and the Decision Process: To Die or Not to Die
In the case of severe irreparable DNA damage, apoptosis is induced. The intrinsic mechanism involves activation of caspases, and it is currently believed that pro- and anti-apoptotic pathways from environmental cues regulate the activation of caspases. The serine/threonine binding protein, 14-3-3, possesses a diverse array of client proteins and is thought to be responsible for coordinating survival and apoptotic pathways through this array of binding partner proteins [21,22]. However, the exact mechanisms for the decision making process of whether "to die" or "not to die" is still uncertain [21] and many reactions can result in both cell survival and apoptosis depending on the circumstances.
Clues to the decision-making mechanisms involved in the apoptotic and anti-apoptotic pathways are becoming apparent. The stress-induced serine/threonine kinase Jun-N-terminal kinase (JNK) is released by the AP-1 transcription regulator. JNK has be reported to antagonize the serine/threonine kinase, Akt (cell survival kinase)-mediated survival signals by phosphorylating 14-3-3 [23]. It is believed that JNK may disrupt 14-3-3 interactions by phosphorylating either the partner or the 14-3-3 proteins themselves, and phosphorylation with ligand release may be the primary mechanism by which activated JNK promotes apoptosis. Such may be the case with activated JNK causing sequestration of Bad (Bcl2 antagonist of death) in JNK-mediated apoptosis [23]. In the DNA Repair Response (DDR) pathway, JNK mediates replication stress-induced apoptosis (Figure 2).
Inhibition of the cell survival kinase (Akt) may also lead to apoptosis (Figure 2). The negative cell cycle regulator, 14-3-3sigma inhibits the activity of Akt [24]. By enhancing the degradation of the p27Kip1 cyclin-dependent inhibitor kinase. Akt has been shown to inhibit the AFX-Forkhead mediated transcription of p27 Kip1 [25]. The secretion of Akt occurs via NF-kB dependent pathways, and is activated by phosphorylase kinase and inhibited by curcumin. Inhibition of NF-kB activation through inhibition of phosphorylase kinase leads to depletion of Akt, which leads to apoptosis.
Additionally, genotoxic stress caused by double-stranded DNA breaks may also induce apoptosis via NF-kB dependent pathways [26]. The role of genotoxic stress (double stranded DNA breaks) in the induction of PIDD (p53 inducible death domain containing protein) has been studied by Janssens, et al. [27] with PIDD being responsible for activation of NF-kB-dependent apoptosis via caspase 2 activation (Figure 2).
It must also be pointed out that 14-3-3 proteins are better known for their promotion of cell proliferative or survival functions. Thus, members of the Raf family (MAP kinase, kinase, kinase), which are serine/threonine kinases, are key activators of the Ras GTPase that initiate the activation of the extracellular-regulated kinase (ERK) or MAP kinase, a tyrosine kinase, which promote cell proliferation and survival. These kinases, activated by NF-kB-dependent pathways, form a parallel pathway which are anti-apoptotic, and promote cell survival. The NF-kB activated pathways include activation of Akt, the cell-survival kinase, which promote the survival of damaged cells, leading to tumor transformation (Figure 2).
Curcumin: Advantages of Early Induction of Apoptosis
Curcumin (diferuloylmethane), the active ingredient from the spice, turmeric, has been recognized for its anti-oxidant and anti-inflammatory properties, and more recently for its ability to block phosphorylation events through its inhibition of phosphorylase kinase [28-31]. Protein kinases catalyze the transfer of high energy phosphate bonds from ATP to either serine/threonine or tyrosine residues [32], but usually not to both. This is because protein kinases usually allow only one configuration of its substrate binding site [32]. However, this is not the case with phosphorylase kinase, which is a protein kinase with multiple specificities attributable to its unique structure [33,34]. Phosphorylase kinase is a unique enzyme in which the spatial arrangements of its specificity determinants can be manipulated so that this enzyme can transfer high energy phosphate bonds to substrates of different specificities, including serine/threonine, tyrosine and even inositol. This is achieved by the presence of a hinge joint, which allows the manipulation of the size of the substrate binding site, and the ability to change the shape of the substrate binding site in two planes by either binding to Mn or Mg [34]. Curcumin is a well-known selective and non-competitive phosphorylase kinase inhibitor [35]. Its ability to inhibit the phosphorylation of a wide number of molecules with different substrate specificities is dependent on its ability to inhibit phosphorylase kinase [28-31].
Curcumin is hydrophobic and is poorly absorbed when taken orally, with negligible blood levels of the conjugated molecule even with high doses. However, the topical preparation appears to be effective on damaged skin [28-31]. The mechanism for the apparent rapidity of the observed repair in burns and acute skin damage is thought to be due to early curcumin-induced apoptosis, which allows new healthy (undamaged) cells to be formed, associated with rescue from cell-cycle arrest, which allows for rapid repair.
The damaged cell is programmed to always attempt to repair the DNA damage even if the healing process is prolonged, and even if the results are less than optimal, such as imperfect repair or excessive scarring. It is only when multiple attempts fail, that the cells undergoes "replication stress-induced apoptosis", which is achieved through JNK-dependent pathways. The salutary effects of curcumin gel on thermal and solar burns may be accomplished through curcumin-induced apoptosis, which has the advantage of the following:
(1) Early apoptosis, perhaps achieved by by-passing the DDR pathway through inhibition of ATM kinase, a phosphatidylinositol-3K-like kinase which is involved in sensing DNA damage and initiating the DDR response.
(2) Selective apoptosis of DNA damaged cells through selective cellular expression of phosphatidylinositol 3-kinase family. Since all three phosphatidylinositol-3 kinase-like kinases (ATM, ATR and DNA-PK) are induced by the presence of double stranded breaks (DSBs) in the DNA, only the damaged cells with DSBs secrete the inositol-containing kinases. These kinases are activated by phosphorylation, perhaps achieved though phosphorylase kinase and inhibited by curcumin (phosphorylase kinase inhibitor). The selectivity observed with curcumin in DNA damaged cells is achieved by selective production of these inositol-containing kinases in damaged cells with DSBs. This selectivity ensures that non-damaged cells are not killed by apoptosis. The removal of the damaged cells allows the space necessary for the replacement of the damaged cells by new healthy cells.
(3) Rapid repair and rescue the DNA damaged cells from cell cycle arrest may be achieved by bypassing the DDR pathway, including cell cycle arrest. This may be achieved blocking the activities of ATR kinase, a phosphatidylinositol-3kinase-like kinase in the DDR pathway. This allows for rapid repair of burns and acutely injured skin.
(4) Early removal of cells with DNA damage has the advantage of preventing the survival of potentially malignant cells with damaged DNA. This is particularly significant for repair of severe burns, including severe solar and radiation burns from ionizing radiation. Apoptosis with curcumin has also been reported to be achieved through other pathways, including depletion of Akt by blocking NF-kB activation [36,37]. It is possible that the action of curcumin through these additional pathways may synergize and amplify its apoptotic activity.
.
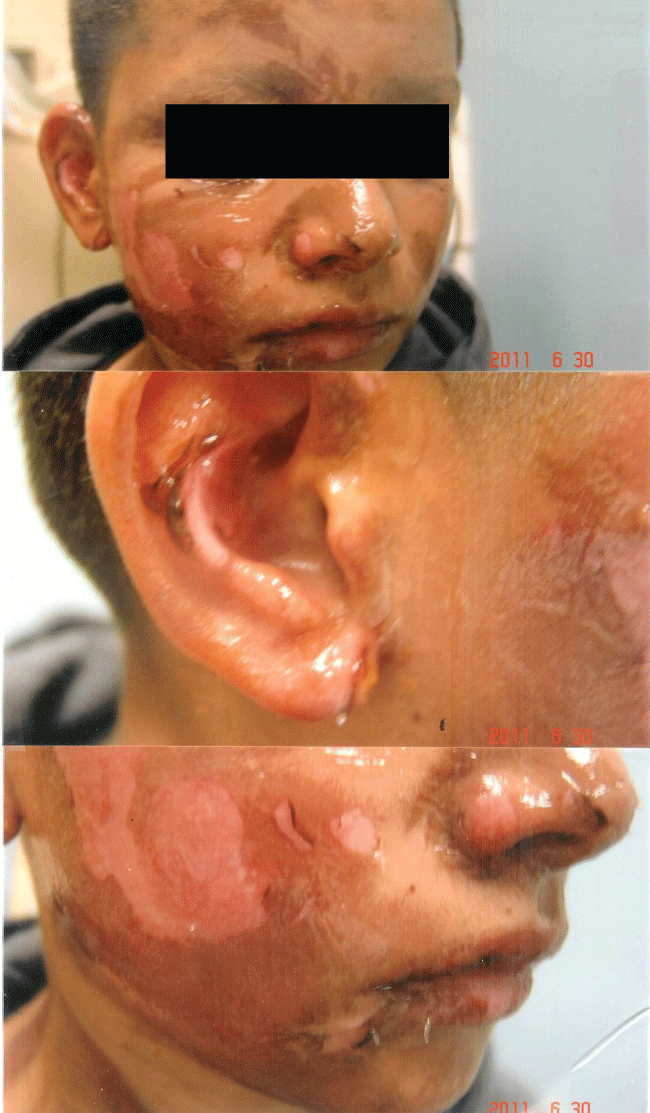
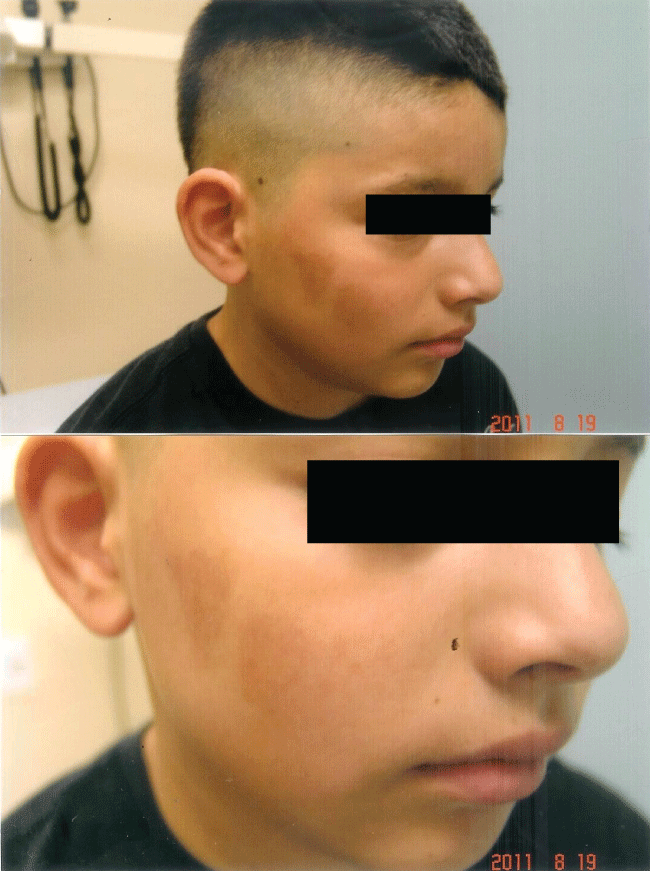
Figure 3a: Second degree flash burn from pouring lighter fluid on a burning barbeque: Patient sustained severe epidermal and dermal injury with sloughing of the epidermis.
View Figure 3a
.
Figure 3b: Significant improvement 5 days later following hourly application of curcumin gel: Re-epithelialization with post-inflammatory hypopigmentation.
View Figure 3b
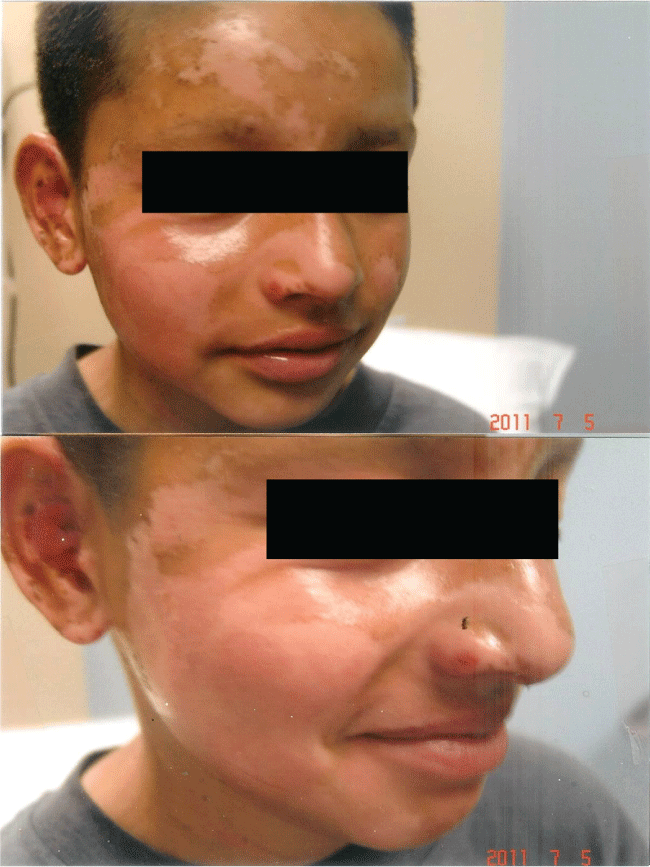
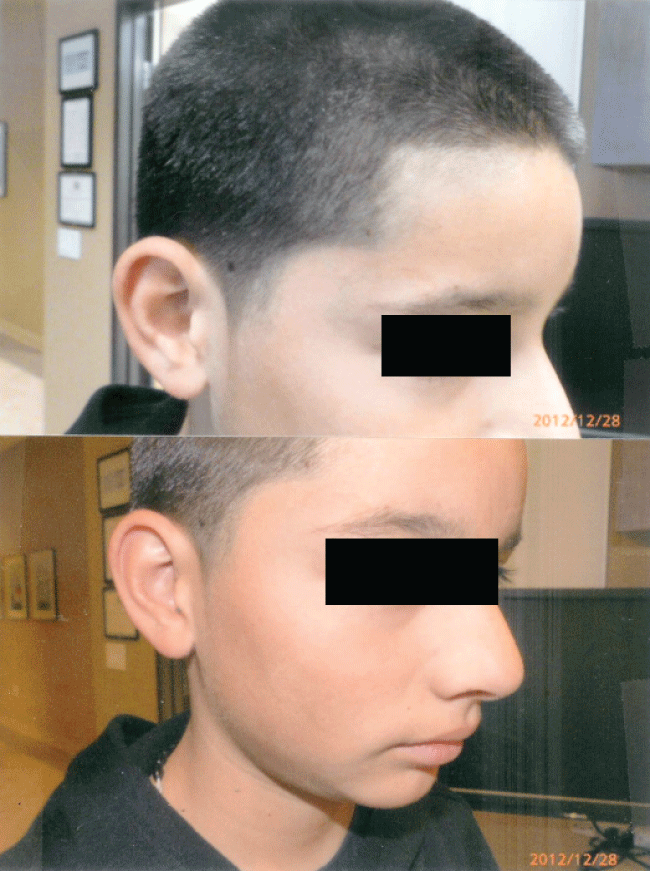
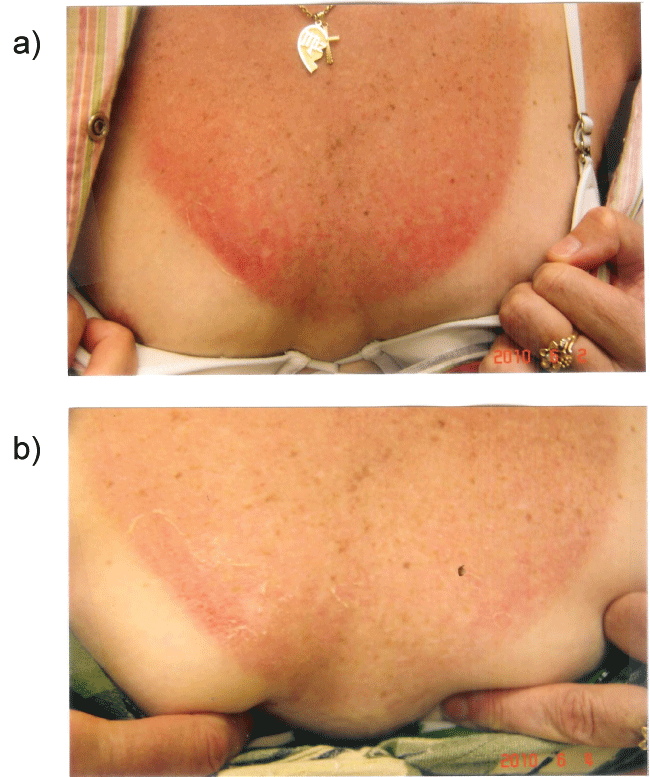
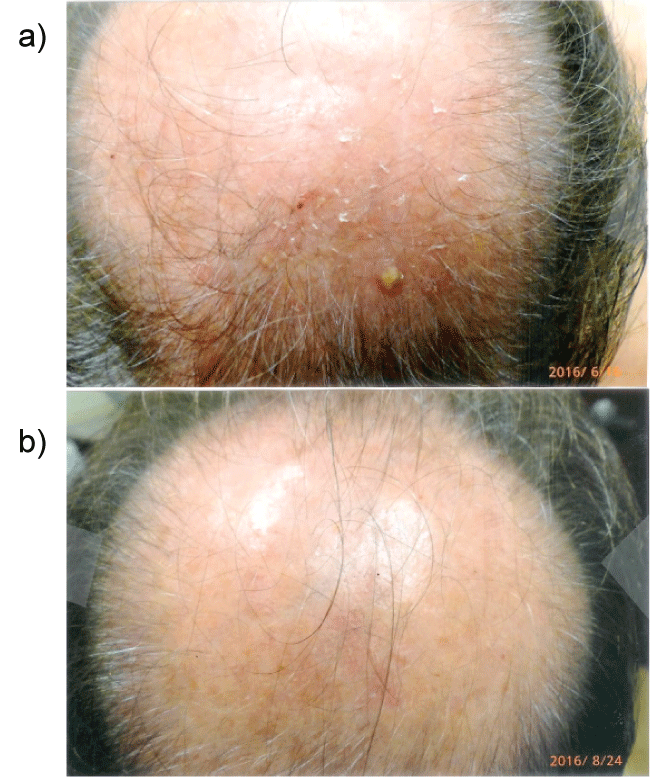
The rapidity of the healing observed after severe burns induced by curcumin gel (Figure 3a and Figure 3b) is thought to illustrate the early decision-making in favor of apoptosis, as well as by successful rescue from cell cycle arrest. Complete resolution without residual scarring suggests early replacement of the damaged cells by new healthy cells in burned skin treated with curcumin gel (Figure 3c and Figure 3d). The same rapidity of healing is also seen with solar burns (Figure 4a and Figure 4b), in which blister formation is aborted within 2 days of treatment with curcumin gel (Figure 4b). The healing of precancerous lesions (actinic keratoses) seen in Figures 5 and Figure 6 may also be achieved by killing and removal of the precancerous cells by curcumin-induced apoptosis.
.
Figure 3c: Complete healing 5 weeks later with curcumin gel treatment: Residual hyperpigmentation of his right cheek but no scarring.
View Figure 3c
.
Figure 3d: Shows the same patient many months later with complete resolution of his burns with the use of curcumin gel. There was no residual scarring or hyperpigmentation.
View Figure 3d
It is hypothesized that the salutary effects of curcumin gel may be achieved by inhibiting phosphatidylinositol 3-kinase-like kinases (ATM, ATR and DNA-PK) in the DDR pathway through inhibition of phosphorylase kinase (summarized in Figure 7). Rapid healing and reversal of burns and solar radiation burns and removal of precancerous lesions may prevent further malignant transformation in photo-damaged skin.
.
Figure 4a: Shows a patient with severe sunburn showing early blistering and hemorrhage before applying curcumin gel.
Figure 4b: Shows the same patient with rapid healing, with aborted blister formation and hemorrhage with frequent applications of curcumin gel.
View Figure 4 a&b
.
Figure 5a: Multiple actinic keratoses vertex scalp before the use of curcumin gel.
Figure 5b: Improvement of actinic keratoses after 3 months of extra strength curcumin gel. It is possible that improvement is achieved by curcumin induced apoptosis.
View Figure 5 a&b
The strategy of using an extraneous drug such as curcumin to selectively choose apoptosis versus repair in severely damaged cells, frees the cells from the decision-making process of whether or not to die. The concomitant ability of curcumin to rescue these cells from cell cycle arrest enables the injured tissue to rapidly replace the damaged cells by new uninjured cells, thus resulting in seamless and painless tissue regeneration.
.
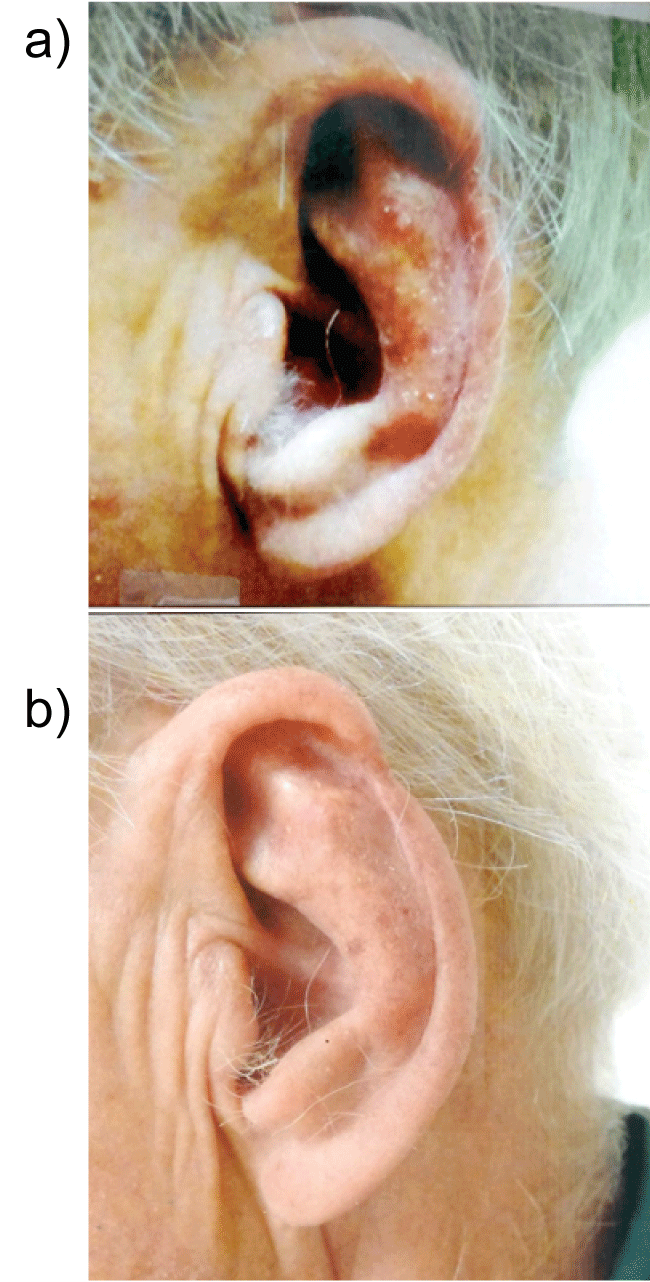
Figure 6a: Severely photodamaged skin with multiple confluent actinic keratoses left pinna ear before the use of curcumin gel.
Figure 6b: Marked improvement seen after application of extra strength curcumin gel bid to the left pinna ear after 3 months. It is believed that repair of photo-damaged skin is achieved by its anti-phosphorylase kinase activity through curcumin-induced apoptosis.
View Figure 6 a&b
.
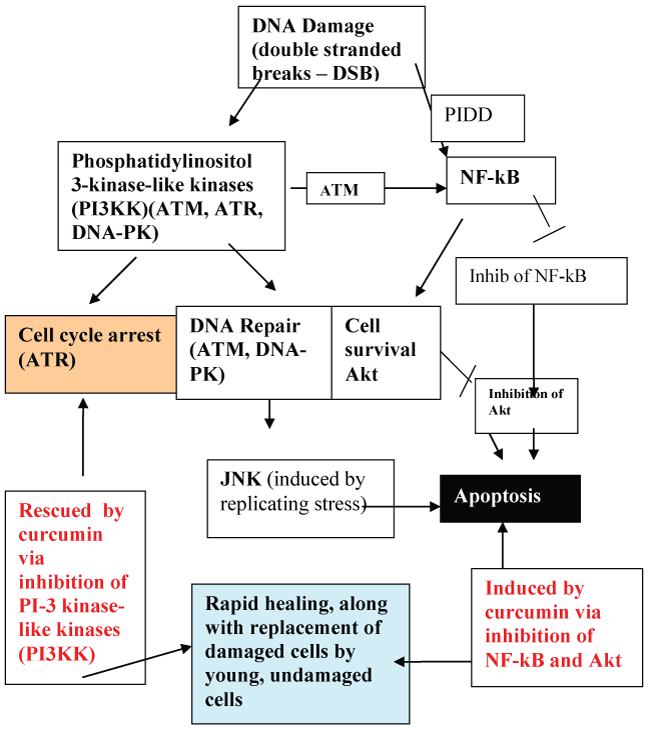
Figure 7: Curcumin-induced apoptosis and rescue from cell cycle arrest: Removal of damaged cells by curcumin-induced apoptosis allows the space for regeneration by new healthy cells. Rescue by curcumin from cell cycle arrest allows for rapid healing.
View Figure 7
Summary
We have detailed the DDR pathway triggered by DNA damage, which leads to cell cycle arrest, nucleotide excision and repair, and nucleotide replication. This is a laborious process resulting in recruitment of multiple proteins to the histones for nucleotide replication and repair. Mistakes are likely to be made if large segments of DNA are damaged or if both strands area damaged, leading to formation of premalignant and malignant cells. In addition, the need to channel all available resources to the repair processes, result in cell cycle arrest and cessation of healing.
Our clinical patients showing rapid healing with curcumin gel demonstrates the uniqueness of curcumin-induced apoptosis, which results in rapid healing from release or rescue from cell cycle arrest, and complete healing without scarring from apoptotic removal of damaged cells. The removal of damaged cells allows the space for regeneration of new healthy cells, which have no malignant potential.
We hypothesize that the mechanism whereby curcumin induces combined apoptosis and rescue from cell cycle arrest is achieved by targeting the early kinases in the DNA damage response (DDR) pathway (Figure 7). These kinases, namely ATM (ataxia telangiectasia mutated) kinase, ATR (ataxia telangiectasia related) kinase, and DNA-PK (protein kinase), all belong to the phosphatidylinositol 3-kinase family. Blocking ATR activation results in release from cell cycle arrest, while blocking ATM and DNA-PK releases the cell from entering into a prolonged unsatisfactory repair pathway with high risk for tumor transformation. Curcumin is believed to accomplish the inhibition of the above mentioned kinases by blocking phosphorylase kinase-dependent phosphorylation events. Apoptosis with curcumin has also been reported to be achieved through other pathways, including depletion of Akt by blocking NF-kB activation [36,37]. It is possible that the action of curcumin through these additional pathways may synergize and amplify its apoptotic activity. The capability of curcumin to inhibit phosphorylation of NF-kB and Akt-dependent pathways is also thought to be achieved through inhibition of phosphorylase kinase [1].
Phosphorylase kinase is a unique kinase in which the spatial arrangements of the specificity determinants can be manipulated to allow phosphorylase kinase to transfer high energy phosphate bonds from ATP to substrates of different specificities [32-34]. We have previously reported that phosphorylase kinase is capable of phosphorylating both serine kinases and tyrosine kinases in the NF-kB-dependent MAP kinase pathways. A previously unreported finding is that phosphorylase kinase may also be able to transfer high energy phosphate bonds to protein kinases of the phosphatidylinositol 3-kinase families including ATM (ataxia telangiectasia mutated) kinase, ATR (ataxia telangiectasia rad 3 related) kinase and DNA-PK (DNA-protein kinase). We propose that phosphorylase kinase may have an important role in the DDR (DNA damage response) pathways by activating the early response enzymes, such as ATM, ATR, and DNA-PK. This is achieved through phosphorylation events, which are necessary for removal of the inhibitory fragment which results in activation of the phosphatidylinositol 3-kinases (ATM kinase, ATR kinase and DNA-PK). This phosphorylation step has been previously attributed to autophosphorylation by other investigators. However, the efficacy of curcumin, a known phosphorylase kinase inhibitor, in blocking the DDR pathway suggests the possibility that phosphorylase kinase may also be involved in activation of phosphorylation events in this pathway (Figure 7). The previously reported induction of apoptosis by curcumin through NF-kB [36] and Akt [37] dependent pathways may also be achieved through inhibition phosphorylase kinase [28-30]. These additional pathways targeted by curcumin through phosphorylase kinase inhibition may synergize to enhance its apoptotic activity.
In conclusion, it is likely that the rapid healing achieved in burns and sun-burns may be achieved by two factors. First, the capability of curcumin to block phosphorylase kinase-dependent phosphorylation through NF-kB-dependent pathways allows the damaged cells to undergo rapid apoptosis using alternative pathways other than the usual DDR pathways. Second, the capability of curcumin to inhibit the phosphatidylinositol 3-kinase family also allows the damaged cell to undergo early apoptosis rather than be involved in a prolonged, unsatisfactory repair process, with increased risk of tumor transformation. This early decision-making achieves early induction of apoptosis in damaged cells, providing the space necessary for replacement by new healthy cells. The damaged tissues, "rescued" from cell cycle arrest" achieves rapid healing without residual scarring as shown our patients with solar and thermal injury treated with topical curcumin.
References
-
Heng MC (2010) Curcumin targeted signaling pathways: basis for anti-photoaging and anti-carcinogenic properties. Int J Dermatol 49: 608-622.
-
Jiang Y, Rabbi M, Kim M, Ke C, Lee W, et al. (2009) UVA generates pyrimidine dimers in DNA directly. Biophys J 96: 1151-1158.
-
Douki T, Raynaud-Angelin A, Cadet J, Sage E (2003) Bypyrimidine photoproducts rather than oxidative lesions are the main type of DNA damage involved in the genotoxic effect of solar UVA radiation. Biochemistry 42: 9221-9226.
-
Rochette PJ, Therrien JP, Drouin R, Perdiz D, Bastien N, et al. (2003) UVA-induced cyclobutane pyrimidine dimers form prevominantly at thymine-thymine dipyrimidines and correlate with the mutation spectum in rodent cells. Nucleic Acid Res 31: 2786-2794.
-
Marteijn JA, Bekker-Jensen S, Mailand N, Lans H, Schwertman P, et al. (2009) Nucleotide excision-repair induced H2A ubiquitination is dependent on MDC1 and RNF8 and reveals a universal DNA damage response. J Cell Biol 186: 835-847.
-
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM (1998) DNA double stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 273: 5858-5868.
-
Levin MF (2007) ATM and the Mre 11 complex combine to recognize and signal DNA double-strand breaks. Oncogene 26: 7749-7758.
-
Lee JH, Pauli TT (2007) Activation and regulation of ATM kinase activity in response to DNA double-strand breaks. Oncogene 26: 7741-7748.
-
Bali LG, Xiao W (2005) Molecular basis of ataxia telangiectasia nd related diseases. Acta Pharmacol Sin 26: 897-907.
-
Falck H, Coates J, Jackson SP (2005) Conserved modes of recruitment of ATM, ATR and DNA-PKcs to sites of DNA damage. Nature 434: 605-611.
-
Paulsen RD, Cimprich KA (2007) The ATR pathway: fine tuning the fork. DNA Repair 6: 953-966.
-
Solier S, Pommier Y (2011) MDC1 cleavage by caspase-3: a novel mechanism for inactivating the DNA damage response during apoptosis. Cancer Res 71: 906-913.
-
Goldberg M, Stucki M, Falck J, D'Amours D, Rahman D, et al. (2003) MDC1 is required for the intra-S-phase DNA damage checkpoint. Nature 421: 952-956.
-
Lou Z, Minter-Dykhouse K, Wu X, Chen J (2003) MDC1 is coupled to activated CHK2 in mammalian DNA damage response pathways. Nature 421: 957-961.
-
Wang B, Elledge S (2007) Ubc13/Rnf8 ubiquitin ligases control foci formation of the Rap80,/Abraxas/Brca1/Brcc36 complex in response to DNA damage. Proc Natl Acad Sci U S A 104: 20759-20763.
-
Kobayashi J (2004) Molecular mechanism of the recruitment of NBS1/hMRE11/hRAD50 complex to DNA double-strand breaks: NBS1 binds to γ-H2AX through FHA/BRCT domain. J Radiation Res 45: 473-478.
-
Yan J, Hetten AM (2008) RAP80 and RNF8, key players in the recruitment of repair proteins to DNA damage sites. Cancer Lett 271: 179-190.
-
Stewart GS, Wang B, Bignell CR, Taylor AM, Elledge SJ (2003) MDC1 is a mediator of the mammalian DNA damage checkpoint. Nature 421: 961-966.
-
Busino L, Chiesa M, Draetta GF, Donzelli M (2004) Cdc25a phosphatase: a combinatorial phosphorylation, ubiquitylation and proteolysis. Oncogene 23: 2050-2056.
-
An J, Huang YC, Xu QZ, Zhou LJ, Shang CF, et al. (2010) DNA-PKcs plays a dominant role in the regulation of H2AX phosphorylation in response to DNA damage and cell cycle progression. BMC Mol Biol 6: 11-18.
-
Porter GW, Khuri FR, Fu H (2006) Dynamic 14-33- client protein interactions integrate survival and apoptotic pathways. Sem Cancer Biol 16: 193-202.
-
Morrison DK (2009) The 14-3-3 proteins: integrators of diverse signaling cues that impact cell fate and cancer development. Trends Cell Biol 19: 16-23.
-
Sunayama J, Tsuruta F, Masuyama N, Gotoh Y (2005) JNK antagonizes Akt-mediated survival signals by phosphorylating 14-3-3. J Cell Biol 170: 295-304.
-
Yang H, Zhang Y, Zhao R, Wen Z, Fournier K, et al. (2006) Negative cell cycle regulator 14-3-3sigma stabilizes p27 Kip1 by inhibiting the activity of PKB/Akt. Oncogene 25: 4585-4594.
-
Graff JR, Konicek BW, McNulty AM, Wang Z, Houck K, et al. (2000) Increased AKT activity contributes to prostate cancer progression by dramatically accelerating prostate tumor growth and diminishing p27kip1 expression. J Biol Chem 275: 24500-24505.
-
Habraken Y, Piette J (2006) NF-kB activation by double-strand breaks. Biochem Pharmacol 72: 1132-1141.
-
Janssens S, Tinel A, Lippens S, Tschopp J (2005) PIDD mediated NF-kappaB activation in response to DNA damage. Cell 123: 1079-1092.
-
Heng MC, Song MK, Harker J, Heng MK (2000) Drug induced suppression of phosphorylase kinase activity correlates with resolution of psoriasis as assessed by clinical histological and immunohistochemical parameters. Br J Dermatol 143: 937-949.
-
Heng MC (2008) Signaling pathways tarted by curcumin: basis for anti-photoaging and anti-carcinogenic therapy. Anti-Aging Therapeutics 10: 151-160.
-
Heng MC (2013) Signaling pathways targeted by curcumin in acute and chronic injury: burns and photo-damaged skin. Int J Dermatol 52: 531-543.
-
Heng MC (2011) Wound healing in adult skin: aiming for perfect regeneration. Int J Dermatol 50: 1058-1066.
-
Johnson LN, Lowe ED, Noble ME, Owen DJ (1998) The Eleventh Datta Lecture. The structural basis for substrate recognition and control by protein kinases. FEBS Lett 430: 1-11.
-
Graves D, Bartleson C, Bjorn A, Pete M (1999) Substrate and inhibitor recognition of protein kinases: what is known about the catalytic subunit of phosphorylase kinase? Pharmacol Ther 82: 143-155.
-
Yuan CJ, Huang CY, Graves DJ (1993) Phosphorylase kinase: a metal ion-dependent and dual specificity kinase. J Biol Chem 268: 17683-17686.
-
Reddy S, Aggarwal BB (1994) Curcumin is a non-competitive and selective inhibitor of PhK. FEBS Lett 341: 19-22.
-
Bharti AC, Donato N, Singh S, Aggarwal BB (2003) Curcumin (diferuloylmethane) down-regulates the constitutive regulation of nuclear factor-kappa B and IkappaBalpha kinase in human myeloma cells, leading to suppression of proliferation and induction of apoptosis. Blood 101: 1053-1062.
-
Aggarwal S, Ichikawa H, Takada Y, Sandur SK, Shishodia S, et al. (2006) Curcumin (diferuloylmethane) down-regulates expression of cell proliferation and anti-apoptotic and metastatic gene products through suppression of IkappaBalpha kinase and Akt activation. Mol Pharmacol 69: 195-206.