International Journal of Neurology and Neurotherapy
Neuromyelitis Optica Spectrum Disorder in A Ninety-year-old Woman. A Case Report
Fernando Romero-Delgado1*, Juan-Pablo Cuello1, Jose-Manuel García-Domínguez1, Yolanda Higueras-Hernández1, Haydee Goicochea-Briceño1, Juan-Adán Guzmán-de-Villoria2 and Maria-Luisa Martínez-Ginés1
1Department of Neurology, Hospital General Universitario Gregorio Marañón, Madrid, Spain
2Department of Radiology, Hospital General Universitario Gregorio Marañón, Madrid, Spain
*Corresponding author:
Romero-Delgado Fernando, Department of Neurology, Hospital General Universitario Gregorio Marañón, Dr. Esquerdo 46, 28007, Madrid, Spain, Tel: 0034 914269610, E-mail: fernandoromerodelgado@gmail.com
Int J Neurol Neurother, IJNN-3-043, (Volume 3, Issue 2), Case Report; ISSN: 2378-3001
Received: February 01, 2016 | Accepted: March 25, 2016 | Published: March 28, 2016
Citation: Romero-Delgado F, Cuello JP, García-Domínguez JM, Higueras-Hernández Y, Goicochea-Briceño H, et al. (2016) Neuromyelitis Optica Spectrum Disorder in A Ninety-yearold Woman. A Case Report. Int J Neurol Neurother 3:043. 10.23937/2378-3001/3/2/1043
Copyright: © 2016 Romero-Delgado F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Neuromyelitis optica spectrum disorder (NMOSD), Optic neuritis, Longitudinally extensive myelitis lesion (LETM), Elderly, Nonagenarian, Anti-AQP4 antibody
Introduction
Neuromyelitis Optica (NMO) is an inflammatory demyelinating, necrotizing disease of the central nervous system, with a predilection for the optic nerves and spinal cord. Clinical, radiological and immunopathological characteristics distinguish it from multiple sclerosis (MS). Recently, new diagnostic criteria have been published, and the terms NMO and neuromyelitis optica spectrum disorder (NMOSD) have been unified, allowing a diagnosis of NMOSD on clinical grounds or on anti-AQP4 antibody status [1]. The onset age of NMO is commonly around the fourth decade of life, but the first attack may occur at any age from early childhood to elderly patients [2,3].
Case Report
We report the case of a 90-year-old woman with medical history of atrial fibrillation, anticoagulated with acenocoumarol, valvular pathology resulting from rheumatic heart disease with severe mitral and tricuspid insufficiency, cardioembolic ischemic stroke of the left middle cerebral artery in 2006 with residual aphasia and right hemiparesis, pulmonary hypertension, and chronic renal failure.
She was admitted to an emergency department due to subacute, painless loss of sight on her left eye. Fundus examination was normal. Brain Computerized Tomography (CT) scan showed a known residual ischemic lesion on the left temporal region. Laboratory exams were normal, including sedimentation rate. Her symptoms were interpreted as a non-Arteritic Anterior Ischemic Optic Neuropathy and she was discharged.
A week later the patient returned to the emergency department with progressive weakness in her lower limbs and upwards hypoesthesia. On physical examination, she was afebrile, had left amaurosis with relative afferent pupillary defect, decreased pain and thermal sensation below dorsal four spinal cord level, moderate tetraparesis, urinary retention, generalized hyperreflexia and bilateral babinski sign. Spinal MRI revealed myelitis with a longitudinally extensive spinal cord lesion, extending from the medulla to C6-C7 level, consistent with myelitis of inflammatory origin (Figure 1).
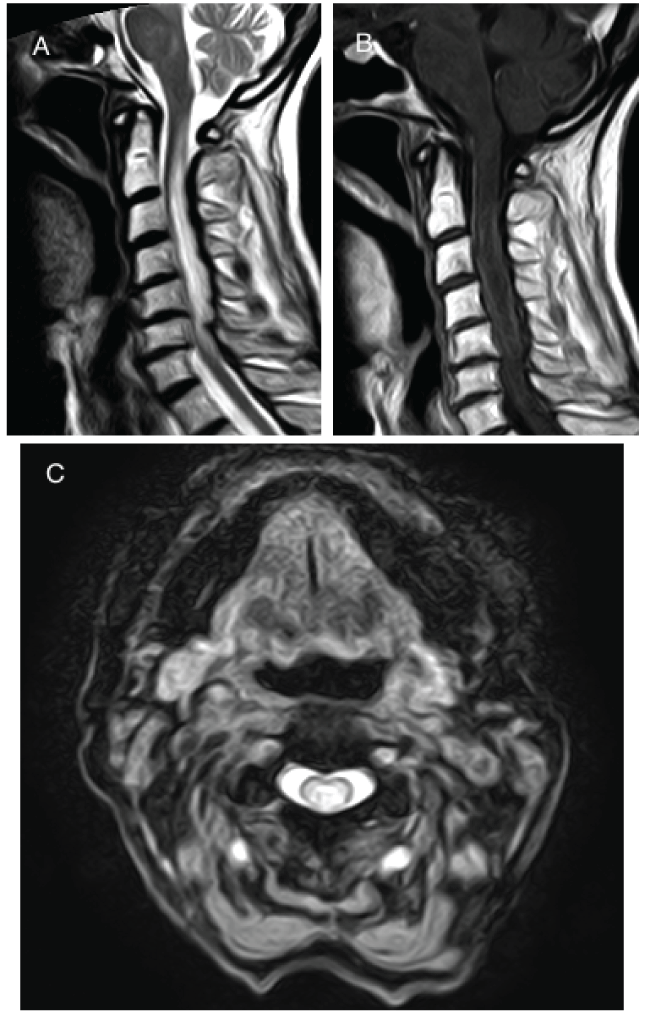
.
Figure 1: A) Sagittal T2-weighted MRI of cervical spinal cord demonstrates a longitudinally extensive myelitis lesion (LETM); B) Sagittal T1-weighted MRI with gadolinium shows tenuous irregular peripheral enhancement; C) Axial T2-weighted MRI of cervical spinal cord demonstrates a lesion involving the central cord.
View Figure 1
Cerebrospinal Fluid (CSF) analysis showed a protein count of 89mg/dL, 2/μL white cells, and a normal glucose. CSF cultures were all negative. No oligoclonal bands were found. Laboratory examination showed positive anti-AQP4 in the serum. Anti-AQP4 determination was performed by cell-based assay using transfected human embryonic kidney cells. Routine blood test, serological studies for viruses and a comprehensive autoimmunity panel (including tests for antinuclear, anti-double-stranded DNA, anti-neutrophil cytoplasmic, anti-smooth muscle antibodies and cryoglobulines) were negative. A whole body CT scan, mammography and neoplastic serological markers were normal. Onconeural antigens Anti-amphiphysin, anti-CV2, anti-Hu, anti-Ma2/Ta, anti-Ri and anti-Yo were all negative. Brain Magnetic Resonance (MRI) revealed ischemic injury on the left middle cerebral artery and signs of mild supratentorial microangiopathy. A follow up CSF analysis showed a 57 mg/dL protein concentration; negative cell counts, negative immunophenotypic study and no oligoclonal bands.
Our patient fulfilled classical Wingerchuk criteria for NMO, presenting with a Devic's syndrome. A diagnosis of NMOSD was made on clinical and AQP4 seropositivity grounds. She was treated with two courses of methylprednisolone therapy (1 g/day during 5 days/course), but no clinical improvement was observed. Plasmapheresis and IV immunoglobulin treatment were considered, but they were rejected because of patient comorbidity. Two months after hospital discharge, the patient died due to decompensated heart failure complicated with bronchopneumonia.
Discussion
Since the discovery of anti-AQP4-IgG as the pathogenic marker for NMO, the spectrum of NMOSD has widened to include limited and unexpected forms of the disease [1,4]. Optic neuritis is more common in paediatric patients, while myelitis is more common in older patients [3]. The rate of onset above 80 years of age is around 1% for NMOSD [5].
Screening for cancer is mandatory even in the anti-AQP4 antibody positive cases, for anti-AQP-4 antibodies have been reported in paraneoplastic context. Breast carcinoma is the most common neoplasm reported with paraneoplastic NMOSD. Lung, uterus, thymus, cervix, bladder, and thyroid carcinomas; seminoma; ovarian teratoma; pituitary adenoma; lymphoma and monoclonal gammopathy have also been reported. In a minority of cases of paraneoplastic NMOSD, no cancer is found [6,7]. Patients with paraneoplastic NMOSD are generally older than 55 years of age.
Only a few cases of NMOSD in nonagenarian patients have been described so far [5,8-10]. The presence of anti-AQP-4 antibodies, and the previous optic neuritis, prompted us to consider a diagnosis of NMOSD. The possibility of a paraneoplastic NMOSD could not be completely ruled out.
Our report suggest that even in the very elderly, NMOSD must be considered in patients with classical features of NMO such as LETM or suspicion of Non-Arteritic Anterior Ischemic Optic Neuropathy without typical features. Anti-AQP4 antibodies should be determined in such cases.
Ethical Statement
The authors state that they have no Conflict of Interest (COI).
References
-
Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, et al. (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85: 177-189.
-
Jacob A, McKeon A, Nakashima I, Sato DK, Elsone L, et al. (2013) Current concept of neuromyelitis optica (NMO) and NMO spectrum disorders. J Neurol Neurosurg Psychiatry 84: 922-930.
-
Nagaishi A, Takagi M, Umemura A, Tanaka M, Kitagawa Y, et al. (2011) Clinical features of neuromyelitis optica in a large Japanese cohort: comparison between phenotypes. J Neurol Neurosurg Psychiatry 82: 1360-1364.
-
Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG (2007) The spectrum of neuromyelitis optica. Lancet Neurol 6: 805-815.
-
Niino M, Uesugi H, Takahashi T, Fukazawa T, Minami N, et al. (2012) Recurrent Brainstem Lesions Mimicking Infarctions in an Elderly Patient with Neuromyelitis Optica Spectrum Disorder. Intern Med 51: 809-812.
-
Figueroa M, Guo Y, Tselis A, Pittock SJ, Lennon VA, et al. (2014) Paraneoplastic neuromyelitis optica spectrum disorder associated with metastatic carcinoid expressing aquaporin-4. JAMA Neurol 71: 495-498.
-
De Santis G, Caniatti L, De Vito A, De Gennaro R, Granieri E, et al. (2009) A possible paraneoplastic neuromyelitis optica associated with lung cancer. Neurol Sci 30: 397-400.
-
Quek AM, McKeon A, Lennon VA, Mandrekar JN, Iorio R, et al. (2012) Effects of age and sex on aquaporin-4 autoimmunity. Arch Neurol 69: 1039-1043.
-
Ohe Y, Fukuoka T, Deguchi I, Hayashi T, Tanahashi N (2014) Case of elderly-onset neuromyelitis optica spectrum disorder mimicking cervical spondylotic myelopathy. Neurology and Clinical Neuroscience 2: 18-19.
-
Kitazawa Y, Warabi Y, Bandoh M, Takahashi T, Matsubara S (2012) Elderly-Onset Neuromyelitis Optica which Developed after the Diagnosis of Prostate Adenocarcinoma and Relapsed after a 23-Valent Pneumococcal Polysaccharide Vaccination. Intern Med 51: 103-107.





