International Journal of Neurology and Neurotherapy
Neurology and Cardiac Arrhythmias
Zhengqin Zhai, Min Tang* and Shu Zhang
Department of Arrhythmia, State Key Laboratory of Cardiovascular Disease, National Clinical Research Center of Cardiovascular Diseases, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China
*Corresponding author:
Min Tang, Department of Arrhythmia, State Key Laboratory of Cardiovascular Disease, National Clinical Research Center of Cardiovascular Diseases, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, NO.167, Beilishi Road, Xicheng District, Beijing, China, Tel: +86-138-1066-5178, Fax: 6833-4688, E-mail: Doctortangmin@hotmail.com
Int J Neurol Neurother, IJNN-3-051, (Volume 3, Issue 3), Short Review; ISSN: 2378-3001
Received: May 09, 2016 | Accepted: June 23, 2016 | Published: June 25, 2016
Citation: Zhai Z, Tang M, Zhang S (2016) Neurology and Cardiac Arrhythmias. Int J Neurol Neurother 3:051. 10.23937/2378-3001/3/3/1051
Copyright: © 2016 Zhai Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The imbalance of cardiac autonomic nervous system (CANS) is one of the major causes of cardiac arrhythmias and several novel therapies of arrhythmias through modulating the activity of CANS have emerged. This review is aimed to address the relationship between the CANS and cardiac arrhythmias, and the present situation of novel therapies.
Keywords
Cardiac autonomic nervous system, Cardiac arrhythmias, Atrial fibrillation, Ventricular tachyarrhythmias, Denervation, Nerve stimulation, Neuromodulation
Abbreviations
AF: Atrial Fibrillation; BRS: Baroreceptor Stimulation; CANS: Cardiac Autonomic Nervous System; CPVT: Catecholaminergic Polymorphic Ventricular Tachycardia; CSD: Cardiac Sympathetic Denervation; ECNS: Extrinsic Cardiac Nervous System; GP: Ganglionated Plexi; ICNS: Intrinsic Cardiac Nervous System; IVT: Idiopathic Ventricular Tachycardia; LL-TS: Low-Level Tragus Stimulation; LL-VNS: Low-Level Vagal Nerve Stimulation; LQTS: Long QT Syndrome; PAF: Paroxysmal AF; PV: Pulmonary Vein; PVI: Pulmonary Vein Isolation; RDN: Renal Denervation; SCS: Spinal Cord Stimulation; VT: Ventricular Tachyarrhythmias; VNS: Vagal Nerve Stimulation; VF: Ventricular Fibrillation
Introduction
A host of studies on the cardiac innervation have found a complex link known as cardiac autonomic nervous system (CANS), connecting extracardiac nerves, intracardiac ganglia, and myocardial cells. Now it is well known that CANS plays a critical role in regulating the functions of the heart and its imbalance is regarded as one of the major causes of cardiac arrhythmias [1]. With the development of medical technology and the advancement of ideas, several novel therapies of arrhythmias through modulating the activity of CANS have emerged. This review is aimed to address the relationship between CANS and cardiac arrhythmias, and the present situation of novel treatment approaches.
Cardiac autonomic nervous system
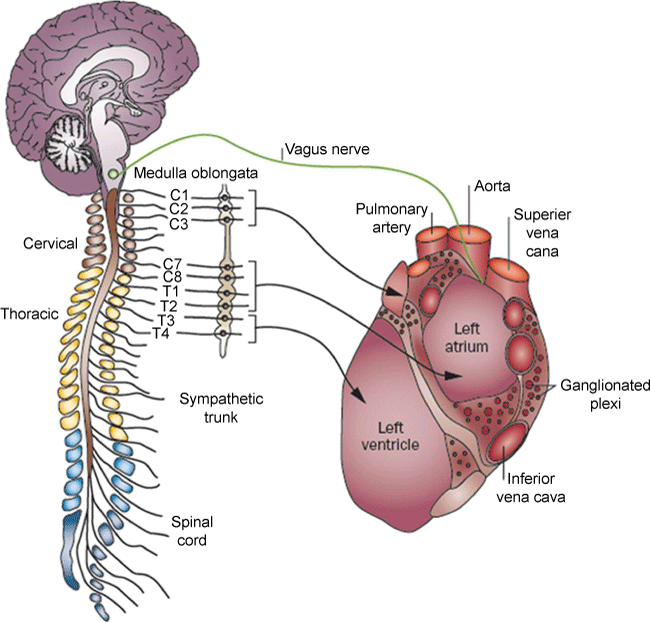
Heart is innervated by CANS, which mediates signals from physiologic "sensors" in the heart and great vessels, and can be divided into two components, extrinsic and intrinsic cardiac nervous system (Figure 1) [1,2].
.
Figure 1: Scheme of autonomic innervation of the heart. The cardiac sympathetic ganglia consist of cervical ganglia, stellate (cervicothoracic) ganglia, and thoracic ganglia. Parasympathetic innervation comes from the vagus nerves. Reprinted from Shen et al. [2] with permission of the publisher. Copyright © American Heart Association, Inc.
View Figure 1
Extrinsic cardiac nervous system
Extrinsic cardiac nervous system (ECNS) is composed of sympathetic cardiac nerves and parasympathetic cardiac branches. The former arises from the stellate ganglia and the caudal halves of the cervical sympathetic trunks, and is subdivided into 4 parts: superior, middle and inferior cervical ganglia and vertebral ganglia, communicating with the spinal nerves C1-4, C3-6, C5-T4, and C4-7 respectively [3-5]. The cardiac nerves from these ganglia innervate the heart by following different courses [6,7]. Parasympathetic cardiac branches are derived from vagus nerves. Its efferent component to the heart includes preganglionic fibers and makes synaptic connections with ganglion cells in the cardiac ganglia [3-5,8].
Intrinsic cardiac nervous system
Intrinsic cardiac nervous system (ICNS) is a complex neural network formed by the nerves and ganglia located around the large vessels and on the heart itself all within the pericardium. Autonomic inputs to the heart converge at several locations and these convergence points form ganglionated plexi (GP) that contain interconnecting ganglia and nerves. The positions of GP are asymmetrical and extensive [3,9]. GPs contain afferent neurons from myocardium and from the extrinsic system [10] and function as the "integration centers" that modulate the interaction between ECNS and ICNS [11].
CNS has yin-yang nature in its physiological function. The activation of its sympathetic component is related to the increased heart rate and ventricular contraction, and the enhanced atrioventricular conductivity. However, its parasympathetic component functions in contrary to its sympathetic component [12].
Physiological Function of CNS
It is well known that the characteristic of physiological function of CNS is its ying-yang nature. Its sympathetic influences on cardiac electrophysiology are similar on both atrial and ventricular myocytes. Electrophysiologically, sympathetic stimulation could shorten action potential duration and reduce transmural dispersion of repolarization [13,14]. Besides, sympathetic activation can also increase heart rate and ventricular contraction, and enhance atrioventricular conductivity [12,15]. In contrast, parasympathetic stimulation decreases heart rate and ventricular contraction, and slows atrioventricular conductivity [12]. In addition, parasympathetic stimulation prolongs action potential duration and effective refractory period in ventricles, while in atrium, reduces the effective refractory period, increases spatial electrophysiological heterogeneity, and promotes early after depolarization toward the end of phases 3 in the action potential [13,16-19], which is different with sympathetic activation.
Interaction between CANS and tachyarrhythmias
Atrial fibrillation: Although the mechanism responsible for atrial fibrillation (AF) has not been completely understood, it is well known that CANS plays an important role in its initiation and maintenance [20].
ECNS and AF: The interaction between ECNS and AF has been approved by several studies. Patients with idiopathic paroxysmal atrial fibrillation (PAF), most appear to be vagally dependent, while in most patients with structural heart diseases, PAF episodes appear more sympathetically mediated [21]. Besides, the incidence of AF in patients with increased sympathetic activity increased as well [22] and in some cases, variations of the autonomic tone were observed before the occurrence of PAF [23,24]. Several studies have shown that beta-adrenergic agonists and the combination of sympathetic activation and acetylcholine infusion could facilitate the induction of AF [25-28] and beta-receptor blockade and atropine were effective in preventing recurrence of AF or decreasing AF inducibility [29-31]. Beside, it was also observed that in rapid atrial pacing induced AF models, simultaneous sympathovagal discharges were common triggers for AF and cryoablation of extrinsic sympathovagal nerves eliminated PAF, which further supported the interrelationship between ECNS and the initiation and maintenance of AF [32].
ICNS and AF: Previous studies provided substantial evidences of the interrelationship between ICNS and AF. The abnormal focal firing in PV is regarded as the major trigger of AF and the four of the left atrial GP each innervates one of the four PVs [9,33,34]. Related to this, a study demonstrated that stimuli applied to PVs would not induce AF unless there was simultaneous simulation of the adjacent GP [35]. Similarly, Po and colleagues showed that focal firing originating from the PV and AF could be induced by injection of acetylcholine into the adjacent GP [36]. It has also been shown that focal AF could be induced or eliminated by stimulating or interrupting the ICNS [37]. Besides, in animal models of AF, a significant increase of sympathetic and/or parasympathetic neurons was present in atrial intrinsic cardiac ganglia [22,38,39]. All these data approve that ICNS is a critical element in the genesis and maintenance of AF.
CANS and non-inherited ventricular tachyarrhythmias: Participation of CANS in the genesis of non-inherited ventricular tachyarrhythmias (VT) has been observed in several studies, most with elevated sympathetic activity that could reduce the ventricular fibrillation (VF) threshold and provoke VT [40-46]. The increased vagal activity seems to be protective in most cases, especially in the presence of elevated sympathetic tone [47-49]. Sympathetic hyperactivity may partly due to sympathetic nerve sprouting in heart. An association between a history of ventricular arrhythmias and an increase of sympathetic nerves in the heart of patients was discovered by Cao et al. [50], and was further approved by a experiment, which found the increased sympathetic nerve sprouting was along with a high-yield model of spontaneous VT [50,51]. Besides, an alteration in neurotransmitters also contributes to sympathetic hyperactivity. The impairment of catecholamine reuptake contributes to myocardial catecholamine overexposure and thus VT after experimental myocardial infarction [52]. As to the protective effect of increased vagal activity against VT, its evidences are mainly obtained by vagal nerve stimulation [53]. We will discuss it later.
CANS and inherited ventricular tachyarrhythmias: For the patients with catecholaminergic polymorphic ventricular tachycardia (CPVT) or long QT syndrome (LQTS), the occurrences of VT are often precipitated by increased sympathetic activity and could be prevented by beta-blockage. CPVT is associated with sympathetic hyperactivity [54-56], and beta-blockage has great efficacy in preventing CPVT related cardiac events [57,58]. As for patients with LQTS, early after depolarization-induced triggered activity is thought to be the primary arrhythmogenic trigger of VT and sympathetic hyperactivity could create the substrate for this trigger and prolong the QT interval in LQTS type 1 and 2 [14,59-63]. However, LQTS type 3 has VT triggered by increased vagal tone [64]. Administration of beta-blockage and left cervicothoracic sympathetic denervation are effective in preventing VT in patients with LQTS type 1 and 2, but have no evident effect in type 3 [65].
Besides LQTS type 3, Brugada syndrome and idiopathic ventricular tachycardia (IVT) have also been reported to be associated with vagal hyperactivity. Most VT episodes in patients are observed during periods of high vagal tone [66-70]. A sudden increase of vagal activity before the onsets of VF was reported in patients with Brugada syndrome, and the ST-segment elevation could be augmented by parasympathomimetic agents, while be reduced by sympathomimetic agents [71,72]. For patients with IVT, J-wave elevation is associated with VF onsets and sudden cardiac death [73-75], and bradycardia could result in the augmentation of J-wave amplitude [76,77], while isoproterenol infusion may eliminate J-wave and suppress VF [78]. All these data demonstrate a critical role of vagal hyperactivity in the occurrence of VT in patients with Brugada syndrome or IVT.
Therapeutic Neuromodulation for Tachyarrhythmias
GP ablation
GP ablation is the major way to modulate intrinsic cardiac nervous system to treat cardiac tachyarrhythmias. For patients with AF, GP ablation alone could significantly decrease the occurrence of PV firing and inducibility of AF [10,79,80]. Its effect has been further supported by a meta-analysis and other clinical trials. In the treatment of AF, addition of GP ablation to PV isolation (PVI) confers better outcomes than PVI alone no matter during a short or long-term follow-up period, and GP ablation alone is inferior to PVI alone [79-82]. Of note, the identification of GP sites is very important. There are two main approaches to the GP sites: selective and anatomic location. The selective approach is performed by high-frequency stimulation, and GP sites were identified as sites showing a vagal response [79,83]. The latter was first reported by Katrisis and colleagues [84], based on the autonomic innervation of the heart, experiences of selective location, and then ablations were delivered to the presumed GP sites [84]. However, there is no available data of GP ablation in the treatment of patients with ventricular arrhythmias.
Vagal nerve stimulation (VNS)
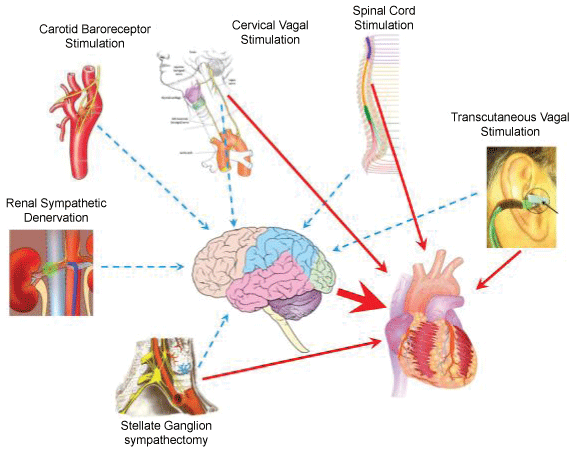
Low-level vagal nerve stimulation (LL-VNS), defined as combination of intensity and frequency without effect on heart rate or atrioventricular conduction, has been found to be protective against cardiac tachyarrhythmias (Figure 2) [1].
.
Figure 2: Schematic representation of various approach of neuromodulation. Solid lines indicate direct effects on cardiac autonomic nervous system, while dashed lines indicate effects on cardiac autonomic nervous system, possibly through neuroal reflexes involving the brain or spinal cord. The thick red arrow denotes that many of the beneficial effects of neuromodulation depend on the brain to process the neural inputs. Reprinted from Hou et al. [1] with permission of the publisher, Copyright © Elsevier.
View Figure 2
For patients with AF, several studies have found that LL-VNS can prevent and reverse atrial remodeling, shorten AF duration and suppress the occurrence or inducibility [85-90]. The mechanism of LL-VNS may be very complex. It is reported that LL-VNS have both anticholinergic and antiadrenergic effects and its inhibition of the GP may be responsible for protective effect on AF [85,86,88,90].
For the treatment of VT, experimental studies showed that VNS could increase VF threshold, and protect against ventricular arrhythmias [53,91-93]. However, there is no available data on its performance in patients with ventricular arrhythmias, and the clinical trials designed to assess the efficacy of VNS among patients with heart failure didn't provide positive results involving ventricular arrhythmias [94-96].
LL-VNS with the electrodes located in vagosympathetic trunk or cervical vagus nerve, is invasive and needs surgery. In 2013, a novel noninvasive approach of VNS by simulating the auricular branch of the vagus nerve at the anterior protuberance of the outer ear was reported by Yu and colleagues [97]. In several studies on animals or humans, LL-TS has been shown to be effective in the prevention or treatment of AF [85,97-105], and may be a promising alternative to VNS.
Spinal cord stimulation (SCS)
SCS of T1-T5 appears to have an antiarrhythmic effect on cardiac tachyarrhythmias [106,107]. For AF, SCS could prolong atrial effective refractory periods and reduce AF burden and inducibility in animal models, suggesting that SCS may represent a treatment option [108,109]. Furthermore, a study indicates that long-term SCS shows sustained protection against AF and that the efficacy of arrhythmia stabilization increases with duration of treatment [110].
For the treatment of VT, it is reported that SCS could reduce the episodes of VT in animal model of post-infarction heart failure, acute myocardial infarction or ischemia-reperfusion [47,106,107,111]. However, there are limited clinical data on SCS. Grimaldi et al. demonstrated its effect on the occurrence of VT episodes in 2 patients [112]. More clinical studies on the protection of SCS against cardiac arrhythmias are needed to further confirm these findings mentioned above.
Cardiac sympathetic denervation (CSD)
CSD is mainly used to prevent the cardiac events in patients with inherited VT [113,114]. Several clinical studies have shown that CSD is associated with a significant reduction in the incidence of VT symptoms and the episodes of VT, especially in patients with LQTS or CPVT [57,115-119]. Besides, CSD has also shown benefits in treatment of patients with structural heart diseases. In a clinical study involving patients with VT storm and structural heart diseases, CSD could also reduce the burden of implantable cardioverter-defibrillator shocks [120]. However, although CSD has been approved to be highly effective in prevention of cardiac events of patients with VT, especially LQTS and CPVT, it is rarely used in clinical practice. The complex surgery of CSD or its high rate of complications, such as Horner syndrome may be the main reasons. Collura and colleagues reported a safe and effective video-assisted thoracoscopic surgery of CSD, with several important advantages, including a more accurate sympathetic chain resection and a lower risk of Horner syndrome [113,116]. Nagels et al. has also reported a percutaneous approach to CSD, and it may be an alternative to surgical intervention [116].
Renal denervation (RDN)
Originally used to manage the blood pressure in patients with hypertension, while in a large multicenter clinical trial, RDN doesn't show benefit on systolic blood pressure in patients with refractory hypertension compared to control [121]. However, RDN may have protective effect on cardiac arrhythmias. In animal experiments, RDN could suppress the atrial remodeling after rapid atrial pacing and reduce the occurrence of VT during left ventricular ischemia and reperfusion [122,123]. Besides, there is also evidence indicating that RND reduces atrial sympathetic nerve sprouting, structural alterations in goats with persistent AF [124]. In patients with AF and refractory hypertension, RND has been reported to provide incremental AF suppressing after PVI or improve the outcomes of PVI [125,126]. These results indicate that an addition of RDN to PVI may be beneficial in patients with AF and/or refractory hypertension.
Baroreceptor stimulation (BRS)
Although BRS is regarded as a new promising approach to control blood pressure and manage heart failure [127,128], there are interests in its impact on cardiac arrhythmias. Liao and colleagues have shown that low-level BRS could reduce the occurrence of ventricular arrhythmias during acute ischemia in dogs [129] and it has also been reported that low-level BRS prolonged the effective refractory period and attenuated rapid atrial pacing induced atrial remodeling in rabbits and could inhibit atrial fibrillation [130,131]. These data appear to indicate that low-level BRS may have protective effect on arrhythmias, while the related studies are rare and there are no clinical studies.
Conclusion
Comprehensive studies on the characteristics and mechanism of cardiac autonomic nervous system in initiating and maintaining arrhythmias have brought out several novel therapeutic approaches to arrhythmias. However, the related clinical trials are limited, and the mechanisms of these therapies are also not completely elucidated. More larger-scale clinical studies and animal experiments are necessary in the future.
Acknowledgement
The work was supported by the National Natural Science Foundation of China (Grant 81441018 to Min Tang and 61527811 to Cong Wang).
References
-
Hou Y, Zhou Q, Po SS (2016) Neuromodulation for cardiac arrhythmia. Heart Rhythm 13: 584-592.
-
Shen MJ, Zipes DP (2014) Role of the autonomic nervous system in modulating cardiac arrhythmias. Circ Res 114: 1004-1021.
-
Kawashima T (2005) The autonomic nervous system of the human heart with special reference to its origin, course, and peripheral distribution. Anat Embryol (Berl) 209: 425-438.
-
Kawashima T (2011) Anatomy of the cardiac nervous system with clinical and comparative morphological implications. Anat Sci Int 86: 30-49.
-
Janes RD, Brandys JC, Hopkins DA, Johnstone DE, Murphy DA, et al. (1986) Anatomy of human extrinsic cardiac nerves and ganglia. Am J Cardiol 57: 299-309.
-
Randall WC, Szentivanyi M, Pace JB, Wechsler JS, Kaye MP (1968) Patterns of sympathetic nerve projections onto the canine heart. Circ Res 22: 315-323.
-
Zipes DP (1990) Influence of myocardial ischemia and infarction on autonomic innervation of heart. Circulation 82: 1095-1105.
-
Guth L (1956) Regeneration in the mammalian peripheral nervous system. Physiol Rev 36: 441-478.
-
Armour JA, Murphy DA, Yuan BX, Macdonald S, Hopkins DA (1997) Gross and microscopic anatomy of the human intrinsic cardiac nervous system. Anat Rec 247: 289-298.
-
Nakagawa H, Scherlag BJ, Patterson E, Ikeda A, Lockwood D, et al. (2009) Pathophysiologic basis of autonomic ganglionated plexus ablation in patients with atrial fibrillation. Heart rhythm 6: S26-34.
-
Hou Y, Scherlag BJ, Lin J, Zhang Y, Lu Z, et al. (2007) Ganglionated plexi modulate extrinsic cardiac autonomic nerve input: effects on sinus rate, atrioventricular conduction, refractoriness, and inducibility of atrial fibrillation. J Am Coll Cardiol 50: 61-68.
-
Chatterjee NA, Singh JP (2015) Novel Interventional Therapies to Modulate the Autonomic Tone in Heart Failure. JACC Heart Fail 3: 786-802.
-
Ng GA, Brack KE, Coote JH (2001) Effects of direct sympathetic and vagus nerve stimulation on the physiology of the whole heart-a novel model of isolated Langendorff perfused rabbit heart with intact dual autonomic innervation. Exp Physiol 86: 319-329.
-
Shimizu W, Antzelevitch C (1998) Cellular basis for the ECG features of the LQT1 form of the long-QT syndrome: effects of beta-adrenergic agonists and antagonists and sodium channel blockers on transmural dispersion of repolarization and torsade de pointes. Circulation 98: 2314-2322.
-
Dukes ID, Vaughan Williams EM (1984) Effects of selective alpha 1-, alpha 2-, beta 1-and beta 2-adrenoceptor stimulation on potentials and contractions in the rabbit heart. J Physiol 355: 523-546.
-
Zipes DP, Mihalick MJ, Robbins GT (1974) Effects of selective vagal and stellate ganglion stimulation of atrial refractoriness. Cardiovasc Res 8: 647-655.
-
Wijffels MC, Kirchhof CJ, Dorland R, Allessie MA (1995) Atrial fibrillation begets atrial fibrillation. A study in awake chronically instrumented goats. Circulation 92: 1954-1968.
-
Fareh S, Villemaire C, Nattel S (1998) Importance of refractoriness hetero-geneity in the enhanced vulnerability to atrial fibrillation induction caused by tachycardia-induced atrial electrical remodeling. Circualtion 98: 2202-2209.
-
Burashnikov A, Antzelevitch C (2003) Reinduction of atrial fibrillation immediately after termination of the arrhythmia is mediated by late phase 3 early afterdepolarization-induced triggered activity. Circulation 107: 2355-2360.
-
He B, Scherlag BJ, Nakagawa H, Lazzara R, Po SS (2012) The intrinsic autonomic nervous system in atrial fibrillation: a review. ISRN Cardiol 2012: 490674.
-
Huang JL, Wen ZC, Lee WL, Chang MS, Chen SA (1998) Changes of autonomic tone before the onset of paroxysmal atrial fibrillation. Int J Cardiol 66: 275-283.
-
Olshansky B (1996) Management of atrial fibrillation after coronary artery bypass graft. Am J Cardiol 78: 27-34.
-
Bettoni M, Zimmermann M (2002) Autonomic tone variations before the onset of paroxysmal atrial fibrillation. Circulation 105: 2753-2759.
-
Klingenheben T, Gerian G, Li YG (1999) Heart rate variability to assess changes in cardiac vagal modulation before the onset of par- oxysmal atrial fibrillation in patients with and without structural heart disease. Ann Noninvas Electrocardiol 4: 19-26.
-
Burashnikov A, Antzelevitch C (2003) Reinduction of atrial fibrillation immediately after termination of the arrhythmia is mediated by late phase 3 early afterdepolarization-induced triggered activity. Circulation 107: 2355-2360.
-
Patterson E, Po SS, Scherlag BJ, Lazzara R (2005) Triggered firing in pulmonary veins initiated by in vitro autonomic nerve stimulation. Heart Rhythm 2: 624-631.
-
Sharifov OF, Fedorov VV, Beloshapko GG, Glukhov AV, Yushmanova AV, et al. (2004) Roles of adrenergic and cholinergic stimulation in spontaneous atrial fibrillation in dogs. J Am Coll Cardiol 43: 483-490.
-
Jayachandran JV, Sih HJ, Winkle W, Zipes DP, Hutchins GD, et al. (2000) Atrial Fibrillation Produced by Prolonged Rapid Atrial Pacing Is Associated With Heterogeneous Changes in Atrial Sympathetic Innervation. Circulation 101: 1185-1191.
-
Schauerte P, Scherlag BJ, Patterson E, Scherlag MA, Matsudaria K, et al. (2001) Focal atrial fibrillation: Experimental evidence for a pathophysiologic role of the autonomic nervous system. J Cardiovasc Electrophysiol 12: 592-529.
-
Kuhlkamp V, Schirdewan A, Stangl K, Homberg M, Ploch M, et al. (2000) Use of metoprolol CR/XL to maintain sinus rhythm after conversion from persistent atrial fibrillation: a randomized, double-blind, placebo-controlled study. J Am Coll Cardiol 36: 139-446.
-
Tan AY, Zhou S, Ogawa M, Song J, Chu M, et al. (2008) Neural mechanisms of paroxysmal atrial fibrillation and paroxysmal atrial tachycardia in ambulatory canines. Circulation 118: 916-925.
-
Pauza DH, Skripka V, Pauziene N, Stropus R (2000) Morphology, distribution, and variability of the epicardiac neural ganglionated subplexuses in the human heart. Anat Rec 259: 353-382.
-
January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, et al. (2014) 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol 64: e1-76.
-
Scherlag BJ, Yamanashi W, Patel U, Lazzara R, Jackman WM (2005) Autonomically induced conversion of pulmonary vein focal firing into atrial fibrillation. J Am Coll Cardiol 45: 1878-1886.
-
Po SS, Scherlag BJ, Yamanashi WS, Edwards J, Zhou J, et al. (2006) Experimental model for paroxysmal atrial fibrillation arising at the pulmonary vein-atrial junctions. Heart 3: 201-208.
-
Lu Z, Scherlag BJ, Lin J, Niu G, Fung KM, et al. (2008) Atrial fibrillation begets atrial fibrillation: autonomic mechanism for atrial electrical remodeling induced by short-term rapid atrial pacing. Circ Arrhythm Electrophysiol 1: 184-192.
-
Yu Y, Wei C, Liu L, Lian AL, Qu XF, et al. (2014) Atrial fibrillation increases sympathetic and parasympathetic neurons in the intrinsic cardiac nervous system. Pacing Clin Electrophysiol 37: 1462-1469.
-
Chang CM, Wu TJ, Zhou S, Doshi RN, Lee MH, et al. (2001) Nerve sprouting and sympathetic hyperinnervation in a canine model of atrial fibrillation produced by prolonged right atrial pacing. Circualtion 103: 22-25.
-
Vladimir Shusterman M, Benhur Aysin, Venkateshwar Gottipaty M, Raul Weiss, Susan Brode, et al. (1998) Autonomic Nervous System Activity and the Spontaneous Initiation of Ventricular Tachycardia. ESVEM Investigators. Electrophysiologic Study Versus Electrocardiographic Monitoring Trial. J Am Coll Cardiol 32: 1891-1899.
-
Leclercq JF, Maisonblanche P, Cauchemez B, Coumel P (1988) Respective role of sympathetic tone and of cardiac pauses in the genesis of 62 cases of ventricular fibrillation recorded during Holter monitoring. Eur Heart J 9: 1276-1283.
-
Huikuri HV, Valkama JO, Airaksinen J, Seppanen T, Kessler KM, et al. (1993) Frequency domain measures of heart rate variability before the onset of nonsustained and sustained ventricular tachycardia in patients with coronary artery disease. Circulation 87: 1220-1228.
-
Pratt CM, Francis MJ, Luck JC, Wyndham CR, Miller RR, et al. (1983) Analysis of ambulatory electrocardiograms in 15 patients during spontaneous ventricular fibrillation with special reference to preceding arrhythmic events. J Am Coll Cardiol 2: 789-797.
-
Valkama JO, Huikuri HV, Koistinen MJ, Yli-Mayry S, Airaksinen KE, et al. (1995) Relation between heart rate variability and spontaneous and induced ventricular arrhythmias in patients with coronary artery disease. J Am Coll Cardiol 25: 437-443.
-
Huang B, Yu L, Scherlag BJ, Wang S, He B, et al. (2014) Left renal nerves stimulation facilitates ischemia-induced ventricular arrhythmia by increasing nerve activity of left stellate ganglion. J Cardiovasc Electrophysiol 25: 1249-1256.
-
Noordzij W, Elvan A, Demirel F, Jager P, Tio R, et al. (2016) Sympathetic denervation in patients with ischemic cardiomyopathy and risk on ventricular tachy-arrhythmias. A pilot study. Q J Nucl Med Mol Imaging.
-
Issa ZF, Zhou X, Ujhelyi MR, Rosenberger J, Bhakta D, et al. (2005) Thoracic spinal cord stimulation reduces the risk of ischemic ventricular arrhythmias in a postinfarction heart failure canine model. Circulation 111: 3217-3220.
-
Liu YB, Wu CC, Lu LS, Su MJ, Lin CW, et al. (2003) Sympathetic nerve sprouting, electrical re- modeling, and increased vulnerability to ventricular fibrillation in hypercholesterolemic rabbits. Circ Res 92: 1145-1152.
-
B S Kolman, R L Verrier, B Lown (1975) The effect of vagus nerve stimulation upon vulnerability of the canine ventricle: role of sympathetic-parasympathetic interactions. Circulation 52: 578-585.
-
Kolman BS, Verrier RL, Lown B (1975) The effect of vagus nerve stimulation upon vulnerability of the canine ventricle: role of sympathetic-parasympathetic interactions. Circulation 52: 578-585.
-
Cao JM, Fishbein MC, Han JB, Lai WW, Lai AC, Wu, et al. (2000) Relationship Between Regional Cardiac Hyperinnervation and Ventricular Arrhythmia. Circulation 101: 1960-1969.
-
Chen PS, Chen LS, Cao JM, Sharifi B, Karagueuzian HS, et al. (2001) Symapathetic nerve sprouting, electrical remodeling and the mechanism of sudden cardiac death. Cardiovasc Res 50: 409-416.
-
Sasano T, Abraham MR, Chang KC, Ashikaga H, Mills KJ, et al. (2008) Abnormal sympathetic innervation of viable myocardium and the substrate of ventricular tachycardia after myocardial infarction. J Am Coll Cardiol 51: 2266-2275.
-
Brack KE, Winter J, Ng GA (2013) Mechanisms underlying the autonomic modulation of ventricular fibrillation initiation--tentative prophylactic properties of vagus nerve stimulation on malignant arrhythmias in heart failure. Heart Fail Rev 18: 389-408.
-
Leenhardt A, Lucet V, Denjoy I, Grau F, Ngoc DD, et al. (1995) Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients. Circulation 91: 1512-1519.
-
Knollmann BC, Chopra N, Hlaing T, Akin B, Yang T, et al. (2006) Casq2 deletion causes sarcoplasmic reticulum volume increase, premature Ca2+ release, and catecholaminergic polymorphic ventricular tachycardia. J Clin Invest 116: 2510-2520.
-
Cerrone M, Noujaim SF, Tolkacheva EG, Talkachou A, O'Connell R, et al. (2007) Arrhythmogenic mechanisms in a mouse model of catecholaminergic polymorphic ventricular tachycardia. Circ Res 101: 1039-1048.
-
Wilde AA, Zahurul A Bhuiyan, Crotti L, Facchini M, De Ferrari GM, et al. (2008) Left cardiac sympathetic denervation for catecholaminergic polymorphic ventricular tachycardia. N Engl J Med 358: 2024-2029.
-
Napolitano C, Priori SG (2007) Diagnosis and treatment of catecholaminergic polymorphic ventricular tachycardia. Heart Rhythm 4: 675-678.
-
el-Sherif N, Caref EB, Yin H, Restivo M (1996) The electrophysiological mechanism of ventricular arrhythmias in the long qt syndrome: tri- dimensional mapping of activation and recovery patterns. Circ Res 79: 474-492.
-
Antzelevitch C, Sun ZQ, Zhang ZQ, Yan GX (1996) Cellular and ionic mechanisms underlying erythromycin-induced long QT intervals and torsade de pointes. J Am Coll Cardiol 28: 1836-1848.
-
Dukes ID, Vaughan Williams EM (1984) Effects of selective alpha 1-, alpha 2-, beta 1-and beta 2-adrenoceptor stimulation on potentials and contractions in the rabbit heart. J Physiol 355: 523-546.
-
Huffaker R, Lamp ST, Weiss JN, Kogan B (2004) Intracellular calcium cycling, early afterdepolarizations, and reentry in simulated long QT syndrome. Heart rhythm 1: 441-448.
-
Noda T, Takaki H, Kurita T, Suyama K, Nagaya N, et al. (2002) Gene-specific response of dynamic ventricular repolarization to sympathetic stimulation in LQT1, LQT2 and LQT3 forms of congenital long QT syndrome. Eur Heart J 23: 975-983.
-
Antzelevitch C (2002) Sympathetic modulation of the long QT syndrome. Eur Heart J 23: 1246-1252.
-
Goldenberg I, Moss AJ (2008) Long QT syndrome. J Am Coll Cardiol 51: 2291-2300.
-
Matsuo K, Kurita T, Inagaki M, Kakishita M, Aihara N, et al. (1999) The circadian pattern of the development of ventricular fibrillation in patients with Brugada syndrome. Eur Heart J 20: 465-470.
-
Takigawa M, Noda T, Shimizu W, Miyamoto K, Okamura H, et al. (2008) Seasonal and circadian distributions of ventricular fibrillation in patients with Brugada syndrome. Heart Rhythm 5: 1523-1527.
-
Kim SH, Nam GB, Baek S, Choi HO, Kim KH, et al. (2012) Circadian and seasonal variations of ventricular tachyarrhythmias in patients with early repolarization syndrome and Brugada syndrome: analysis of patients with implantable cardioverter defibrillator. J Cardiovasc Electrophysiol 23: 757-763.
-
Maeda S, Takahashi Y, Nogami A, Yamauchi Y, Osaka Y, et al. (2015) Seasonal, weekly, and circadian distribution of ventricular fibrillation in patients with J-wave syndrome from the J-PREVENT registry. J Arrhythm 31: 268-273.
-
Abe A, Ikeda T, Tsukada T, Ishiguro H, Miwa Y, et al. (2010) Circadian variation of late potentials in idiopathic ventricular fibrillation associated with J waves: insights into alternative pathophysiology and risk stratification. Heart Rhythm 7: 675-682.
-
Miyazaki T, Mitamura H, Miyoshi S, Soejima K, Aizawa Y, et al. (1996) Autonomic and antiarrhythmic drug modulation of ST segment elevation in patients with Brugada syndrome. J Am Coll Cardiol 27: 1061-1070.
-
Antzelevitch C, Patocskai B (2016) Brugada Syndrome: Clinical, Genetic, Molecular, Cellular, and Ionic Aspects. Curr Probl Cardiol 41: 7-57.
-
Haissaguerre M, Derval N, Sacher F, Jesel L, Deisenhofer I, et al. (2008) Sudden cardiac arrest associated with early repolarization. N Engl J Med 358: 2016-2023.
-
Rosso R, Kogan E, Belhassen B, Rozovski U, Scheinman MM, et al. (2008) J-point elevation in survivors of primary ventricular fibrillation and matched control subjects: incidence and clinical significance. J Am Coll Cardiol 52: 1231-1238.
-
Shinohara T, Takahashi N, Saikawa T, Yoshimatsu H (2006) Characterization of J wave in a patient with idiopathic ventricular fibrillation. Heart Rhythm 3: 1082-1084.
-
Antzelevitch C, Yan GX (2010) J wave syndromes. Heart Rhythm 7: 549-558.
-
Mizumaki K, Nishida K, Iwamoto J, Nakatani Y, Yamaguchi Y, et al. (2012) Vagal activity modulates spontaneous augmentation of J-wave elevation in patients with idiopathic ventricular fibrillation. Heart Rhythm 9: 249-55.
-
Nam GB, Ko KH, Kim J, Park KM, Rhee KS, et al. (2010) Mode of onset of ventricular fibrillation in patients with early repolarization pattern vs. Brugada syndrome. Eur Heart J 31: 330-339.
-
Zhou Q, Hou Y, Yang S (2011) A meta-analysis of the comparative efficacy of ablation for atrial fibrillation with and without ablation of the ganglionated plexi. Pacing Clin Electrophysiol 34: 1687-1694.
-
Katritsis DG, Pokushalov E, Romanov A, Giazitzoglou E, Siontis GC, et al. (2013) Autonomic denervation added to pulmonary vein isolation for paroxysmal atrial fibrillation: a randomized clinical trial. J Am Coll Cardiol 62: 2318-2325.
-
Pokushalov E, Romanov A, Katritsis DG, Artyomenko S, Shirokova N, et al. (2013) Ganglionated plexus ablation vs linear ablation in patients undergoing pulmonary vein isolation for persistent/long-standing persistent atrial fibrillation: a randomized comparison. Heart Rhythm 10: 1280-1286.
-
Katritsis D, Giazitzoglou E, Sougiannis D, Goumas N, Paxinos G, et al. (2008) Anatomic approach for ganglionic plexi ablation in patients with paroxysmal atrial fibrillation. Am J Cardiol 102: 330-334.
-
Mitsuru Takami M, Kohei Yamashiro, Yuichiro Sakamoto, Koyo Satoh, Takahiko Suzuki (2014) Impact of ganglionated plexi ablation on high-frequency stimulation-induced changes in atrial fibrillation cycle length in the pulmonary vein. Journal of arrhythmia 30: 356-361.
-
Krul SPJ, Berger WR, Veldkamp MW, Driessen AHG, Wilde AAM, et al. (2015) Treatment of Atrial and Ventricular Arrhythmias Through Autonomic Modulation. JACC: Clinical Electrophysiology 1: 496-508.
-
Stavrakis S, Humphrey MB, Scherlag BJ, Hu Y, Jackman WM, et al. (2015) Low-level transcutaneous electrical vagus nerve stimulation suppresses atrial fibrillation. J Am Coll Cardiol 65: 867-875.
-
Sheng X, Scherlag BJ, Yu L, Li S, Ali R, et al. (2011) Prevention and reversal of atrial fibrillation inducibility and autonomic remodeling by low-level vagosympathetic nerve stimulation. J Am Coll Cardiol 57: 563-571.
-
Li S, Scherlag BJ, Yu L, Sheng X, Zhang Y, et al. (2009) Low-level vagosympathetic stimulation: a paradox and potential new modality for the treatment of focal atrial fibrillation. Circ Arrhythm Electrophysiol 2: 645-651.
-
Sha Y, Scherlag BJ, Yu L, Sheng X, Jackman WM, et al. (2011) Low-level right vagal stimulation: anticholinergic and antiadrenergic effects. J Cardiovasc Electrophysiol 22: 1147-1153.
-
Shen MJ, Shinohara T, Park HW, Frick K, Ice DS, et al. (2011) Continuous low-level vagus nerve stimulation reduces stellate ganglion nerve activity and paroxysmal atrial tachyarrhythmias in ambulatory canines. Circulation 123: 2204-2212.
-
Yu L, Scherlag BJ, Li S, Sheng X, Lu Z, et al. (2011) Low-level vagosympathetic nerve stimulation inhibits atrial fibrillation inducibility: direct evidence by neural recordings from intrinsic cardiac ganglia. J Cardiovasc Electrophysiol 22: 455-463.
-
Brack KE, Coote JH, Ng GA (2011) Vagus nerve stimulation protects against ventricular fibrillation independent of muscarinic receptor activation. Cardiovasc Res 91: 437-446.
-
Ando M, Katare RG, Kakinuma Y, Zhang D, Yamasaki F, et al. (2005) Efferent vagal nerve stimulation protects heart against ischemia-induced arrhythmias by preserving connexin43 protein. Circulation 112: 164-170.
-
Zhang L, Lu Y, Sun J, Zhou X, Tang B (2016) Subthreshold vagal stimulation suppresses ventricular arrhythmias and inflammatory response in a canine model of acute cardiac ischemia and reperfusion. Exp Physiol 101: 41-49.
-
Zannad F, De Ferrari GM, Tuinenburg AE, Wright D, Brugada J, et al. (2015) Chronic vagal stimulation for the treatment of low ejection fraction heart failure: results of the NEural Cardiac TherApy foR Heart Failure (NECTAR-HF) randomized controlled trial. Eur Heart J 36: 425-433.
-
Premchand RK, Sharma K, Mittal S, Monteiro R, Dixit S, et al. (2014) Autonomic regulation therapy via left or right cervical vagus nerve stimulation in patients with chronic heart failure: results of the ANTHEM-HF trial. J Card Fail 20: 808-816.
-
Gold MR, Van Veldhuisen DJ, Hauptman PJ, Borggrefe M, Kubo SH, et al. (2016) Vagus nerve stimulation for the treatment of heart failure: The INOVATE-HF Trial. J Am Coll Cardiol.
-
Yu L, Scherlag BJ, Li S, Fan Y, Dyer J, et al. (2013) Low-level transcutaneous electrical stimulation of the auricular branch of the vagus nerve: a noninvasive approach to treat the initial phase of atrial fibrillation. Heart Rhythm 10: 428-435.
-
Clancy JA, Mary DA, Witte KK, Greenwood JP, Deuchars SA, et al. (2014) Non-invasive vagus nerve stimulation in healthy humans reduces sympathetic nerve activity. Brain Stimul 7: 871-877.
-
Chen M, Yu L, Liu Q, Wang Z, Wang S, et al. (2015) Noninvasive vagus nerve stimulation: A novel promising modulator for cardiac autonomic nerve system dysfunction. Int J Cardiol 187: 338-339.
-
Chen M, Yu L, Ouyang F, Liu Q, Wang Z, et al. (2015) The right side or left side of noninvasive transcutaneous vagus nerve stimulation: Based on conventional wisdom or scientific evidence? Int J Cardiol 187: 44-45.
-
Ajijola OA, Hamon D (2016) Noninvasive Neuromodulation Via Tragal Stimulation. JACC: Clinical Electrophysiology.
-
Li S, Zhou X, Yu L, Jiang H (2015) Low level non-invasive vagus nerve stimulation: a novel feasible therapeutic approach for atrial fibrillation. Int J Cardiol 182: 189-190.
-
Chinda K, Tsai WC, Chan YH, Lin AY, Patel J, et al. (2016) Intermittent left cervical vagal nerve stimulation damages the stellate ganglia and reduces the ventricular rate during sustained atrial fibrillation in ambulatory dogs. Heart Rhythm 13: 771-780.
-
Madhavan M, Venkatachalam KL, Swale MJ, Desimone CV, Gard JJ, et al. (2016) Novel percutaneous epicardial autonomic modulation in the canine for atrial fibrillation: results of an efficacy and safety study. Pacing and Clinical Electrophysiology 39: 407-417.
-
Yu L, Wang S, Zhou X, Wang Z, Huang B, et al. (2015) Chronic Intermittent Low-Level Stimulation of Tragus Reduces Cardiac Autonomic Remodeling and Ventricular Arrhythmia Inducibility in a Post-Infarction Canine Model. JACC: Clinical Electrophysiology.
-
Lopshire JC, Zhou X, Dusa C, Ueyama T, Rosenberger J, et al. (2009) Spinal cord stimulation improves ventricular function and reduces ventricular arrhythmias in a canine postinfarction heart failure model. Circulation 120: 286-94.
-
Odenstedt J, Linderoth B, Bergfeldt L, Ekre O, Grip L, et al. (2011) Spinal cord stimulation effects on myocardial ischemia, infarct size, ventricular arrhythmia, and noninvasive electrophysiology in a porcine ischemia-reperfusion model. Heart Rhythm 8: 892-898.
-
Bernstein SA, Wong B, Vasquez C, Rosenberg SP, Rooke R, et al. (2012) Spinal cord stimulation protects against atrial fibrillation induced by tachypacing. Heart Rhythm 9: 1426-1433.
-
Wang S, Zhou X, Huang B, Wang Z, Zhou L, et al. (2016) Spinal cord stimulation suppresses atrial fibrillation by inhibiting autonomic remodeling. Heart Rhythm 13: 274-821.
-
Ardell JL, Cardinal R, Beaumont E, Vermeulen M, Smith FM, et al. (2014) Chronic spinal cord stimulation modifies intrinsic cardiac synaptic efficacy in the suppression of atrial fibrillation. Auton Neurosci 186: 38-44.
-
Wang S, Zhou X, Huang B, Wang Z, Liao K, et al. (2015) Spinal cord stimulation protects against ventricular arrhythmias by suppressing left stellate ganglion neural activity in an acute myocardial infarction canine model. Heart Rhythm 12: 1628-1635.
-
Grimaldi R, de Luca A, Kornet L, Castagno D, Gaita F (2012) Can spinal cord stimulation reduce ventricular arrhythmias? Heart Rhythm 9: 1884-1887.
-
Yongkeun Cho (2015) Left cardiac sympathetic denervation: An important treatment option for patients with hereditary ventricular arrhythmias. Journal of arrhythmia.
-
Schwartz PJ (2014) Cardiac sympathetic denervation to prevent life-threatening arrhythmias. Nat Rev Cardiol 11: 346-353.
-
Schwartz PJ, Priori SG, Cerrone M, Spazzolini C, Odero A, et al. (2004) Left cardiac sympathetic denervation in the management of high-risk patients affected by the long-QT syndrome. Circulation 109: 1826-1833.
-
Collura CA, Johnson JN, Moir C, Ackerman MJ (2009) Left cardiac sympathetic denervation for the treatment of long QT syndrome and catecholaminergic polymorphic ventricular tachycardia using video-assisted thoracic surgery. Heart Rhythm 6: 752-759.
-
Miller MA, Bhasin K, Reddy VY, d'Avila A (2011) Left cardiac sympathetic denervation for the treatment of methadone-induced long QT syndrome. Heart Rhythm 8: 1955-1957.
-
Nagels W, Brabant S, Van Aelst L, Pollet P, Anne W, et al. (2014) Percutaneous left cardiac sympathetic denervation in a patient with long-QT syndrome. J Cardiothorac Vasc Anesth 28: 1580-1582.
-
De Ferrari GM, Dusi V, Spazzolini C, Bos JM, Abrams DJ, et al. (2015) Clinical Management of Catecholaminergic Polymorphic Ventricular Tachycardia: The Role of Left Cardiac Sympathetic Denervation. Circulation 131: 2185-2193.
-
Vaseghi M, Gima J, Kanaan C, Ajijola OA, Marmureanu A, et al. (2014) Cardiac sympathetic denervation in patients with refractory ventricular arrhythmias or electrical storm: intermediate and long-term follow-up. Heart Rhythm 11: 360-366.
-
Bhatt DL, Kandzari DE, O'Neill WW, D'Agostino R, Flack JM, et al. (2014) A controlled trial of renal denervation for resistant hypertension. N Engl J Med 370: 1393-1401.
-
Wang X, Zhao Q, Huang H, Tang Y, Xiao J, et al. (2013) Effect of renal sympathetic denervation on atrial substrate remodeling in ambulatory canines with prolonged atrial pacing. PLoS One 8: e64611.
-
Linz D, Wirth K, Ukena C, Mahfoud F, Poss J, et al. (2013) Renal denervation suppresses ventricular arrhythmias during acute ventricular ischemia in pigs. Heart Rhythm 10: 1525-1530.
-
Linz D, van Hunnik A, Hohl M, Mahfoud F, Wolf M, et al. (2015) Catheter-based renal denervation reduces atrial nerve sprouting and complexity of atrial fibrillation in goats. Circ Arrhythm Electrophysiol 8: 466-474.
-
Pokushalov E, Romanov A, Corbucci G, Artyomenko S, Baranova V, et al. (2012) A randomized comparison of pulmonary vein isolation with versus without concomitant renal artery denervation in patients with refractory symptomatic atrial fibrillation and resistant hypertension. J Am Coll Cardiol 60: 1163-1170.
-
Pokushalov E, Romanov A, Katritsis DG, Artyomenko S, Bayramova S, et al. (2014) Renal denervation for improving outcomes of catheter ablation in patients with atrial fibrillation and hypertension: early experience. Heart Rhythm 11: 1131-1138.
-
Abraham WT, Zile MR, Weaver FA, Butter C, Ducharme A, et al. (2015) Baroreflex Activation Therapy for the Treatment of Heart Failure With a Reduced Ejection Fraction. JACC Heart Fail 3: 487-496.
-
Heusser K, Tank J, Engeli S, Diedrich A, Menne J, et al. (2010) Carotid baroreceptor stimulation, sympathetic activity, baroreflex function, and blood pressure in hypertensive patients. Hypertension 55: 619-626.
-
Liao K, Yu L, Yang K, Saren G, Wang S, et al. (2014) Low-level carotid baroreceptor stimulation suppresses ventricular arrhythmias during acute ischemia. PLoS One 9: e109313.
-
Dai M, Bao M, Liao J, Yu L, Tang Y, et al. (2015) Effects of low-level carotid baroreflex stimulation on atrial electrophysiology. J Interv Card Electrophysiol 43: 111-119.
-
Linz D, Hohl M, Khoshkish S, Mahfoud F, Ukena C, et al. (2016) Low-level but Not High-Level Baroreceptor Stimulation Inhibits Atrial Fibrillation in a Pig Model of Sleep Apnea. J Cardiovasc Electrophysiol.





