International Journal of Ophthalmology and Clinical Research
Biweekly Bevacizumab for Refractory Neovascular Age-related Macular Degeneration
Ryan B. Rush1-3* and Sloan W. Rush2,3
1Southwest Retina Specialists, USA
2Panhandle Eye Group, USA
3Texas Tech University Health Science Center, USA
*Corresponding author: Ryan Rush, MD, Southwest Retina Specialists, 7411 Wallace Blvd., marillo, TX, 79106, USA, Tel: (806) 351-1870, Fax: (806) 351-1690, E-mail: Ryanbradfordrush21@hotmail.com
Int J Ophthalmol Clin Res, IJOCR-2-006, (Volume 2, Issue 1), Research Article; ISSN: 2378-346X
Received: September 04, 2014 | Accepted: January 10, 2015 | Published: January 13, 2015
Citation: Rush RB, Rush SW (2015) Biweekly Bevacizumab for Refractory Neovascular Age-related Macular Degeneration. Int J Ophthalmol Clin Res 2:006. 10.23937/2378-346X/1410006
Copyright: © 2015 Rush RB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: To evaluate the effectiveness of biweekly (twice-per-month) bevacizumab for treatment-resistant neovascular age-related macular degeneration (nAMD).
Methods: The charts of nAMD subjects treated with biweekly bevacizumab injections because of persistent retinal edema on optical coherence tomography (OCT) despite consecutive monthly anti-vascular endothelial factor (VEGF) therapy were retrospectively reviewed.
Results: There were 17 subjects included in the analysis. An average of 10.8 (+/- 2.5) anti-VEGF injections were given prior to biweekly bevacizumab initiation. There were 64.7 % (11/17) of subjects that received both bevacizumab and aflibercept prior to initiation of biweekly bevacizumab therapy. At the 6-month follow-up evaluation after initiating biweekly bevacizumab, the mean best-corrected visual acuity improved from0.53 logMAR (+/- 0.19) (Snellen 20/68) to 0.36 logMAR (+/- 0.16) (Snellen 20/46) (p=0.0088), central macular thickness on OCT decreased from 373.6 (+/- 42.8) microns to 308.8 microns (+/- 37.5) (p< 0.0001), and choroidal neovascularization surface area on indocyanine green angiography decreased from 2.74 mm2 (+/- 1.9) to 1.55mm2 (+/- 0.96) (p=0.0306). Subjects received an average of 6.6 (+/- 2.8) biweekly bevacizumab injections during the study interval. At the study�s end, 64.7 % (11/17) of subjects had a macula without retinal edema on OCT, and 76.4 % (13/17) were able to receive bevacizumab at an interval of 4 weeks or beyond.
Conclusion: Biweekly bevacizumab can improve the visual and anatomic outcomes in nAMD subject�s recalcitrant to consecutive monthly anti-VEGF therapy; this strategy may especially provide a feasible treatment option in subjects with persistent retinal edema despite a switch in anti-VEGF medication.
Introduction
The Comparison of Age-related Macular Degeneration Treatments Trials reported persistent retinal edema on optical coherence tomography (OCT) in more than half of the patients treated with both ranibizumab (Lucentis; Genetech, Inc, South San Francisco, CA) and bevacizumab (Avastin; Genetech, Inc) after 1 and 2 years[1,2]. Persistent retinal edema despite consecutive monthly anti-vascular endothelial growth factor (VEGF) therapy may be designated as �refractory� and the following strategies have been described to address this clinical dilemma: injections more frequently than every 4 weeks [3], higher anti-VEGF dosages [4], switching anti-VEGF medications [5-10], and anti-VEGF combined with photodynamic therapy [11]. In this study, the authors explore the effectiveness of biweekly (twice-per-month) bevacizumab for the management of refractory nAMD.
Methods
This retrospective medical record review of nAMD patients treated from November 2011 to May 2014 at a single private-practice institution was approved by the Southwest Retina Specialists Institutional Review Board (IORG0007600/IRB00009122). All research components were conducted according to the tenets of the Declaration of Helsinki and in accordance with human research regulations and standards.
The inclusion criteria were as follows: 1) age = 55 years, 2) subfoveal nAMD was demonstrated by clinical examination, fluorescein angiography (FA), spectral domain OCT, and indocyanine green (ICG) angiography at the subject�s baseline evaluation, 3) Snellen best-corrected visual acuity (BCVA) was between 20/25 and 20/200 prior to anti-VEGF therapy and at the time of the initiation of biweekly bevacizumab, 4) at least 6 consecutive monthly anti-VEGF injections were given before the initiation of biweekly bevacizumab, 5) persistent retinal edema on OCT was present at all monthly follow-up evaluations prior to initiation of biweekly bevacizumab, and 6) a 6 month follow-up period after initiation of biweekly bevacizumab was fully documented with interval BCVA, examination details, OCT, FA, and ICG angiography.
The exclusion criteria were as follows: 1) ocular media opacity (corneal scar, cataract, etc), which in the opinion of the examiner, was significantly affecting BCVA or hindering retinal examination and imaging acquisition, 2) intraocular surgery for any indication was performed during the study period or within 3 months prior to enrollment, 3) a visually-significant retinal disease other than nAMD (epiretinal membrane, diabetic retinopathy, polypoidal choroidal vasculopathy, etc.), active uveitis, or uncontrolled glaucoma was present at enrollment or developed during the study period, 4) a previous vitrectomy (pars plana or anterior) was performed at any time to the study eye (before or during the study period).
Baseline examinations for treatment-na�ve subjects included BCVA, slit-lamp assessment of the anterior and posterior segments, FA, OCT, and ICG angiography. Subjects were considered �refractory� to anti-VEGF therapy only after failing to achieve a dry macula (without intraretinal or subretinal fluid) on OCT following 6 or more consecutive monthly anti-VEGF injections. Subjects were permitted to switch anti-VEGF medications during the study interval prior to the initiation of biweekly bevacizumab so long as they continued receiving consecutive monthly injections without ever achieving a dry macula on OCT. Once biweekly bevacizumab was initiated, subjects underwent examinations with OCT every 4 weeks, and FA and ICG angiography testing approximately every 8 weeks. Subjects were extended back to a monthly injection interval with bevacizumab once a dry macula on OCT was achieved with biweekly injections. Subjects were then managed according to a standard treat-and-extend protocol [12]. If retinal exudation recurred at a monthly interval following extension from the biweekly schedule, the decision to return to biweekly injections was made at the discretion of the clinician.
OCT, FA, and ICG angiography procedures were performed on the Heidelberg Spectralis (Heidelberg Engineering, Heidelberg, Germany) system. OCT was used to determine the presence or absence of intraretinal and subretinal fluid and to assess central macular thickness. The central macular thickness was acquired from the center subfield of the macular thickness map and only well-centered scans were designated for data analysis. Choroidal neovascularization (CNV) surface areas were measured using centered early to mid-frame (within the first 3 minutes) ICG angiography images. The �Inlay� function was selected on the ICG angiography image menu and the CNV margins were manually encircled, followed by surface area computation with the Heidelberg Spectralis software. This technique of CNV size determination on ICG angiography in the setting of nAMD has been described by a previous study [13].
Outcome Measures and Statistical Analysis
The study�s primary outcome was the percentage of subjects that achieved a dry macula on OCT at 6 months after initiating biweekly bevacizumab. Secondary outcome measures were change in BCVA, change in central macular thickness on OCT, and change in CNV surface area on ICG angiography during the study interval. Statistical calculations were performed using the JMP 11 software package (SAS Institute, Cary, NC, USA). For statistical analysis, Snellen BCVA was converted into a logarithm of the minimal angle of resolution (logMAR). Numerical means were compared using one-way analysis of the variance (ANOVA). Nominal variables were compared using Chi-Square and Likelihood Ratios where appropriate. A probability of < 0.05 was considered statistically significant.
Results
There were 17 cases that met the inclusion/exclusion criteria. At enrollment, the study population�s mean age was 75.1 (+/-7.4) years, 64.7 % (11/17) were female and 35.3 % (6/17) male, and 58.8 % (10/17) were pseudophakic and 41.2 % (7/17) were phakic. The mean pretreatment baseline BCVA was 0.51 (+/-0.22) logMAR (Snellen 20/65), central macular thickness on OCT was 443.9 (+/- 95) microns, and CNV surface area on ICG was 2.05 (+/- 1.3) mm2. All 17 subjects were initially started on monthly bevacizumab injections upon presentation as treatment na�ve subjects, and 64.7 % (11/17) of subjects were switched from monthly bevacizumab to monthly aflibercept (VEGF-Trap Eye/Eylea; Regeneron, Tarrytown, NY) prior to receiving biweekly bevacizumab. Of the subjects that received aflibercept during the study interval, the mean number aflibercept injections prior to receiving biweekly bevacizumab were 5.2 (+/- 1.9). None of the subjects received combination treatment with photodynamic therapy or macular photocoagulation during the study interval. None of the subjects received ranibizumab during the study interval. An average of 10.8 (+/- 2.5) anti-VEGF injections were given prior to biweekly bevacizumab initiation (Median=12 injections; Range=6 to 14 injections).
At the evaluation in which biweekly bevacizumab therapy was decided upon, the BCVA was 0.53 (+/- 0.19) logMAR (Snellen 20/68), central macular thickness on OCT was 373.6 (+/- 42.8) microns, and CNV surface area on ICG angiography was 2.74 (+/- 1.9) mm2. During the treatment interval from anti-VEGF treatment naivety to the evaluation in which biweekly bevacizumab therapy was decided upon, the study population did not demonstrate a change in BCVA (p=0.7499), although central macular thickness on OCT improved (p=0.0278), but CNV surface area on ICG angiography trended toward enlargement but was not significantly different (p=0.2426). At the 6-month follow-up evaluation after initiating biweekly bevacizumab, the mean BCVA was 0.36 logMAR (+/- 0.16) (Snellen 20/46), central macular thickness on OCT was 308.8 microns (+/- 37.5), and CNV surface area on ICG angiography was 1.55mm2 (+/- 0.96). From the evaluation in which biweekly bevacizumab therapy was decided upon until 6-month follow-up after initiating biweekly bevacizumab, the BCVA improved (p=0.0088), central macular thickness on OCT decreased (p< 0.0001), and CNV surface area on ICG angiography decreased (p=0.0306).
Subjects received an average of 6.6 (+/- 2.8) biweekly bevacizumab injections during the study interval (Median=6; Range=2 to 12). There were 11.7 % (2/17) of subjects still receiving biweekly bevacizumab injections despite 12 consecutive biweekly injections without ever attaining a dry macula on OCT. There were 17.6 % (3/17) of subjects that achieved a dry macula on OCT while receiving biweekly bevacizumab injections and then were extended back to a monthly injection interval only to experience recurrent exudation; two of these three subjects returned back to biweekly bevacizumab injections. There were 29.4 % (5/17) of subjects successfully extended beyond monthly bevacizumab therapy without recurrent exudation by study�s end. Overall, 64.7 % (11/17) of subjects had a macula without retinal edema on OCT, and 76.4 % (13/17) were able to receive bevacizumab at an interval of 4 weeks or beyond by the close of the study. There were 29.4 % (5/17) of subjects that experienced 3 or more lines of improved BCVA at the 6-month follow-up evaluation after initiating biweekly bevacizumab; there were no subjects that lost 3 or more lines of BCVA.
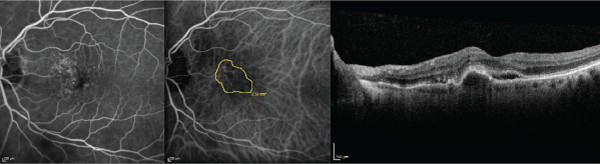
Figure 1: The baseline fluorescein angiography (left), indocyanine green angiography (middle), and optical coherence tomography (right) images are displayed in
a 72 year-old treatment-na�ve subject with a Snellen visual acuity of 20/50.
View Figure 1
There were 88.2 % (15/17) of subjects that had a reduction in CNV size at the 6-month follow-up evaluation after initiating biweekly bevacizumab. CNV size on ICG angiography enlarged by 33 % or more in 29.4 % (5/17) from treatment naivety until the evaluation in which biweekly bevacizumab therapy was decided upon. CNV size on ICG angiography decreased by 33 % or more in 47.0 % (8/17) at the 6-month follow-up evaluation after initiating biweekly bevacizumab; CNV size on ICG angiography decreased by 33 % or more in 0 % from treatment naivety until the evaluation in which biweekly bevacizumab therapy was decided upon (Figures 1-3).
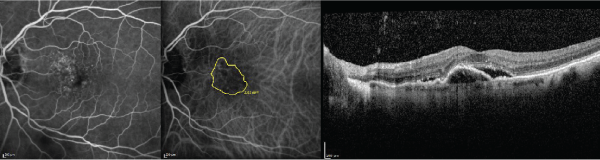 Figure 2: The fluorescein angiography (left), indocyanine green angiography (middle), and optical coherence tomography (right) images are displayed in the same
subject as Figure 1 after 8 consecutive monthly bevacizumab injections and 4 aflibercept injections. The Snellen visual acuity has decreased to 20/60. Note the
enlargement in choroidal neovascularization surface area from baseline.
View Figure 2
Figure 2: The fluorescein angiography (left), indocyanine green angiography (middle), and optical coherence tomography (right) images are displayed in the same
subject as Figure 1 after 8 consecutive monthly bevacizumab injections and 4 aflibercept injections. The Snellen visual acuity has decreased to 20/60. Note the
enlargement in choroidal neovascularization surface area from baseline.
View Figure 2
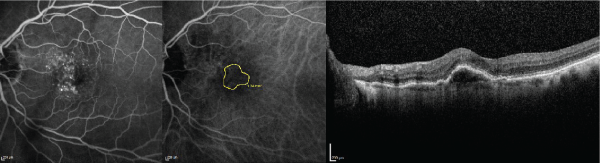 Figure 3: The fluorescein angiography (left), indocyanine green angiography (middle), and optical coherence tomography (right) images are displayed in the same
subject as Figures 1 and 2 after 4 biweekly bevacizumab injections followed by 3 monthly bevacizumab injections. The Snellen visual acuity has improved to 20/30.
Note the reduction in choroidal neovascularization surface area from Figure 2.
View Figure 3
Figure 3: The fluorescein angiography (left), indocyanine green angiography (middle), and optical coherence tomography (right) images are displayed in the same
subject as Figures 1 and 2 after 4 biweekly bevacizumab injections followed by 3 monthly bevacizumab injections. The Snellen visual acuity has improved to 20/30.
Note the reduction in choroidal neovascularization surface area from Figure 2.
View Figure 3
Discussion
Persistent retinal edema despite repetitive monthly anti-VEGF injections presents a significant challenge for clinicians, and there exists an unmet need for management strategies to effectively tackle this problem. Many recent reports suggest that treatment-resistant nAMD subjects receiving either bevacizumab or ranibizumab may benefit from a medication switch to aflibercept [5-10]. However, a significant portion of those subjects continued to have retinal edema even after switching medications. Sparse clinical information exists regarding the management of treatment-resistant nAMD with biweekly anti-VEGF therapy. Stewart et al. [3] performed pharmacokinetic modeling to determine if more frequent treatment with anti-VEGF medications could theoretically be useful; they concluded that biweekly bevacizumab resulted in trough binding levels superior to monthly 0.5mg ranibizumab and potentially superior to the levels reached with monthly 2.0mg ranibizumab. Stewart et al. [3] also described two case reports in the same study demonstrating the clinical effectiveness of biweekly anti-VEGF therapy in these nAMD subjects refractory to monthly dosing. At the present time, these case reports by Stewart et al. [3] are the only ones found on pubmed.org describing biweekly anti-VEGF therapy for refractory nAMD. Thus, we are presenting the first case series of treatment-resistant nAMD subjects managed with biweekly anti-VEGF therapy.
Our results demonstrate that biweekly bevacizumab can improve visual acuity, lower central macular thickness, and reduce CNV surface area in nAMD subjects with persistent retinal edema despite repetitive monthly anti-VEGF therapy, even after a prolonged time period has elapsed since treatment naivety. Of particular note, subjects switched from bevacizumab to aflibercept prior to receiving biweekly bevacizumab still demonstrated clinical improvement at the end of the study. This subgroup (refractory to multiple anti-VEGF drugs) within the overall nAMD population may be one of the most difficult to manage, and biweekly bevacizumab may offer an effective treatment option for these patients. There were 35.2 % (6/17) of subjects that were not switched from monthly bevacizumab to monthly aflibercept prior to biweekly bevacizumab initiation; the reasons for this were the lack of availability of aflibercept at the time of treatment in 2 subjects (treatment occurred prior to the commercial release of the drug) and excessive financial burden in 4 subjects (unable to afford the cost of aflibercept). Although biweekly bevacizumab therapy results in more frequent injections, it is still significantly less expensive than a switch to monthly aflibercept for underinsured nAMD subjects� refractory to monthly bevacizumab. In sufficiently insured refractory nAMD subjects, the authors suggest that switching anti-VEGF medications would be a reasonable and appropriate first move, followed by biweekly bevacizumab if retinal edema persists after the switch. However, for underinsured refractory nAMD subjects, a move to biweekly bevacizumab therapy first may provide the necessary means of obtaining the goal of a dry macula on OCT, which in our study resulted in better vision,while at the same time lowering cost. As the results of this study demonstrate, most subjects do not require biweekly bevacizumab therapy for a period longer than three months before attaining a dry macula on OCT, after which time most are able to then return to monthly therapy without recurrent exudation.
Weaknesses of this study include the relatively small number of cases, limited follow-up, use of logMAR visual acuity, the retrospective collection of data, and the observer-dependent determination of CNV size with the Heidelberg Spectralis surface area measurement software. Recently, the authors described the clinical utility of ICG angiography in nAMD subjects undergoing bevacizumab therapy [13]. Although CNV size on ICG angiography decreased for nearly all of the subjects after initiating biweekly bevacizumab (15/17), none of the subjects had complete resolution of CNV on ICG angiography. More subjects with longer follow-up are necessary to determine the precise role of ICG angiography in the management of this specific patient population. In summary, biweekly bevacizumab may provide an effective treatment option for nAMD subjects that continue to have retinal edema despite monthly anti-VEGF therapy. More research is needed to further establish the role of biweekly bevacizumab therapy in nAMD subject�s refractory to monthly anti-VEGF, and it remains unknown how this treatment strategy compares to other potential treatment options such as combined anti-VEGF with photodynamic therapy or higher dose ranibizumab.
References
-
CATT Research Group, Martin DF, Maguire MG, Ying GS, Grunwald JE, et al. (2011) Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med 364: 1897-1908.
-
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group1, Martin DF, Maguire MG, Fine SL, Ying GS, et al. (2012) Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 119: 1388-1398.
-
Stewart MW, Rosenfeld PJ, Penha FM, Wang F, Yehoshua Z, et al. (2012) Pharmacokinetic rationale for dosing every 2 weeks versus 4 weeks with intravitrealranibizumab, bevacizumab, and aflibercept (vascular endothelial growth factor Trap-eye). Retina 32: 434-457.
-
Brown DM, Chen E, Mariani A, Major JC Jr; SAVE Study Group (2013) Super-dose anti-VEGF (SAVE) trial: 2.0 mg intravitrealranibizumab for recalcitrant neovascular macular degeneration-primary end point. Ophthalmology 120: 349-354.
-
Bakall B, Folk JC, Boldt HC, Sohn EH, Stone EM, et al. (2013) Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol 156: 15-22.
-
Cho H, Shah CP, Weber M, Heier JS (2013) Aflibercept for exudative AMD with persistent fluid on ranibizumab and/or bevacizumab. Br J Ophthalmol 97: 1032-1035.
-
Yonekawa Y, Andreoli C, Miller JB, Loewenstein JI, Sobrin L, et al. (2013) Conversion to aflibercept for chronic refractory or recurrent neovascular age-related macular degeneration. Am J Ophthalmol 156: 29-35.
-
Ho VY, Yeh S, Olsen TW, Bergstrom CS, Yan J, et al. (2013) Short-term outcomes of aflibercept for neovascular age-related macular degeneration in eyes previously treated with other vascular endothelial growth factor inhibitors. Am J Ophthalmol 156: 23-28.
-
Kumar N, Marsiglia M, Mrejen S, Fung AT, Slakter J, et al. (2013) Visual and anatomical outcomes of intravitrealaflibercept in eyes with persistent subfoveal fluid despite previous treatments with ranibizumab in patients with neovascular age-related macular degeneration. Retina33:1605-1612.
-
Chang AA, Li H, Broadhead GK, Hong T, Schlub TE, et al. (2014) Intravitrealaflibercept for treatment-resistant neovascular age-related macular degeneration. Ophthalmology 121: 188-192.
-
Tozer K, Roller AB, Chong LP, Sadda S, Folk JC, et al. (2013) Combination therapy for neovascular age-related macular degeneration refractory to anti-vascular endothelial growth factor agents. Ophthalmology 120: 2029-2034.
-
Rush RB, Simunovic MP, Vandiver L, Aragon AV 2nd, Ysasaga JE (2014) Treat-and-extend bevacizumab for neovascular age-related macular degeneration: the importance of baseline characteristics. Retina 34: 846-852.
-
Rush RB, Rush SW, Aragon AV 2nd, Ysasaga JE (2014) Evaluation of Choroidal Neovascularization with Indocyanine Green Angiography in Neovascular Age-related Macular Degeneration Subjects Undergoing IntravitrealBevacizumab Therapy. Am J Ophthalmol158:337-344.





