International Journal of Ophthalmology and Clinical Research
Generation of Retinal Progenitor Cell Sheets which Differentiate into Rhodopsin Positive Photoreceptors from Mouse iPS Cell Derived Retinal Progenitor Cell Clones
Noboru Suzuki1,2*, Jun Shimizu1, Chieko Hirotsu1, Erika Takada1, Nagisa Arimitsu1, Yuji Ueda1, Naruyoshi Fujiwara1, Tomoko Suzuki1 and Kenji Takai1
1Departments of Immunology and Medicine, St. Marianna University School of Medicine, Japan
2Department of Regenerative Medicine, Institute of Advanced Medical Science, St. Marianna University Graduate School of Medicine, Japan
*Corresponding author: Dr. Noboru Suzuki, Departments of Immunology and Medicine, St. Marianna University School of Medicine, 2-16-1 Sugao, Miyamae-ku, Kawasaki, Kanagawa 216-8511, Japan, Tel: 81-44-977-8111, Fax: 81-44-975-3315, E-mail: n3suzuki@marianna-u.ac.jp
Int J Ophthalmol Clin Res, IJOCR-2-011, (Volume 2, Issue 1), Research Article; ISSN: 2378-346X
Received: January 05, 2015 | Accepted: February 03, 2015 | Published: February 05, 2015
Citation: Suzuki N, Shimizu J, Hirotsu C, Takada E, Arimitsu N, et al. (2015) Generation of Retinal Progenitor Cell Sheets which Differentiate into Rhodopsin Positive Photoreceptors from Mouse iPS Cell Derived Retinal Progenitor Cell Clones. Int J Ophthalmol Clin Res 2:011. 10.23937/2378-346X/1410011
Copyright: © 2015 Suzuki N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We previously reported successful establishment of retinal progenitor cell clones. Here, we cultured them on temperature-reversible gelation polymer, with supplementation of several chemokines. The retinal progenitor cell clones formed a cell sheet with extension of their axon like processes. RT-PCR of the cell sheets after two week culture, they were positive for retinal ganglion cell markers, bipolar cell markers, and photoreceptor cell markers. Immunohistochemistry suggested that the cell sheets contained photoreceptors and synaptic connection was observed within the sheet.
We found that in vitro culture of cloned retinal progenitor cells led to formation of the cell sheet being rhodopsin positive. The cell sheet made of photoreceptor cells may have the potential to benefit various retinal diseases.
Keywords
Rhodopsin, Photoreceptor, iPS cells, Retinal progenitor, Retinal degeneration, Retinal transplantation, and Cell sheet
Introduction
Retinal diseases often provoke severe visual disturbance. Currently, there are several approaches to rescue or restore vision in humans with retinal degeneration. One major treatment strategy is cell replacement therapy including transplantation of retinal sheets [1-4] which aims at replacing photoreceptors and/or retinal pigment epithelium (RPE) (and other retinal cells) lost in the retinal diseases. Intact sheets of fet al and neonatal retina have also been transplanted into degenerated eyes with the aim of restoring visual function [5-7].
However, donor tissues used in the reports mainly came from fet al eyes from humans and rats. Alternative donor tissue for retinal transplantation has long been awaited.
We previously reported establishment of retinal neuron progenitors from mouse embryonic stem (ES) cells and mouse induced pluripotent stem (iPS) cells by pax6 gene transfection and subsequent limiting dilution culture [8,9]. Because the cell clones derived from mouse iPS cells expressed CD73 predominantly, and further differentiated into cells with rhodopsin expression, we considered them as a candidate cell source of photoreceptor precursor cells. Then, we tried to make a cell sheet composed of retinal progenitors which were able to differentiate into rhodopsin positive photoreceptor cells, aiming to experimental and clinical application in the near future, and the histological and immune-histological characteristics of the retinal progenitor cell sheet were analyzed.
Materials and Methods
Establishing retinal progenitor cell clones from mouse iPS cells
A culture protocol for establishing retinal progenitor cell clones has been reported [9] (Figure 1A). In brief, we used a mouse iPS cell line, iPS-MEF-Ng-20D-17 (Figure 1B) [10]. The culture (growth) medium consisted of DMEM, supplemented with 2 mM glutamine, 0.1 mM beta-mercaptoethanol, 1 mM pyruvate, and 15% FCS [8-10]. Mouse pax6 cDNA was kindly donated by Dr. Barbara Norman (National Eye Institute, Bethesda, MD), and preparation of the pax6 cDNA expression vectors and transfection of them were conducted as previously reported [8]. Although cytomegalovirus promoter allows for a strong short-term expression of transduced genes, it becomes silent within a few weeks after gene transfer [11]. Fourteen days after initiation of selection culture with G418, the resistant cells were harvested. And then, we conducted limiting dilution culture and resultant cloned cell lines were characterized as previously described [8,9].
Generation of retinal progenitor cell sheets on Upcell� (CellSeed, Tokyo, Japan) and their detachment from it for the characterization
After expansion cultures of the cloned retinal progenitor cell lines in growth media for 5 days, we cultured the cells in 6-well and 12-well plates precoated with thermo-reversible gelation polymer (Upcell�) [12]. We changed the culture medium (designated as differentiation medium) composed of Dulbecco's modified Eagle's medium, Nutrient mixture F-12 (DMEM/F12) with N2 supplement (Invitrogen, Oslo, Norway), and 0.05% fibronectin (FN) (Sigma-Aldrich, St. Louis, MO), and cultured them until they became confluent, leading to formation of a cell sheet.
To release confluent cells as a cell sheet, culture dishes were incubated in another CO2 incubator set at 22�C [12]. The cell sheets detached spontaneously within 1 hour and floated into the aqueous media. Immediately after detachment, the entire cell sheet was processed for RT-PCR and immunohistological staining.
RT-PCR
Total RNA extraction, cDNA synthesis and PCR amplification including their primer sequences have been reported [8,9]. Beta-actin was used to detect housekeeping gene expression in all RT-PCR.
HE staining and confocal microscopic examinations
The cell sheets were fixed in 4% paraformaldehyde solution [8,9]. They were incubated with one of the following primary antibodies: anti-Nestin (Millipore, Billerica, MA), anti-Beta III tubulin (Promega, Madison, WI), anti-CD73 (BD Bioscience, Tokyo, Japan), anti-Rhodopsin (Cosmo Bio, Tokyo, Japan), anti-S opsin (millipore), anti-M opsin (millipore), and anti-SynapsinI (millipore). An Alexa Fluor 594 second antibody (Invitrogen) was used for the development of immunofluorescence. A confocal laser scanning microscope (LSM510, Carl Zeiss, and Jena, Germany) was used to detect signals. In some experiments, two color immunofluorescence staining was conducted as previously reported [8].
Results
Cell culture for making retinal progenitor cell sheets
We recently reported that transfection with Pax6 gene of mouse iPS cells (Figure 1B) induced their differentiation and brought about emergence of retinal progenitors [9]. In brief, we conducted transfection of mouse iPS cells with the Pax6 expression vector. After selection and limiting dilution culture (Figure 1A), we picked up the cloned cell lines expressing nestin mRNA and musashi1 mRNA by RT-PCR. Then we conducted the second screening where cell clones expressing Chx10 mRNA and Six3 mRNA simultaneously were selected. After expansion culture in the growth medium, the cloned cells were transferred to the differentiation medium. When the cells were cultured for a longer period of time, they gradually extended axon like processes (Figure. 1CD) that eventually led to the formation of neural network like structures in vitro [9]. The cloned pax6 transfected cells were highly purified as for retinal progenitors and they showed high cell yield after expansion culture in vitro.
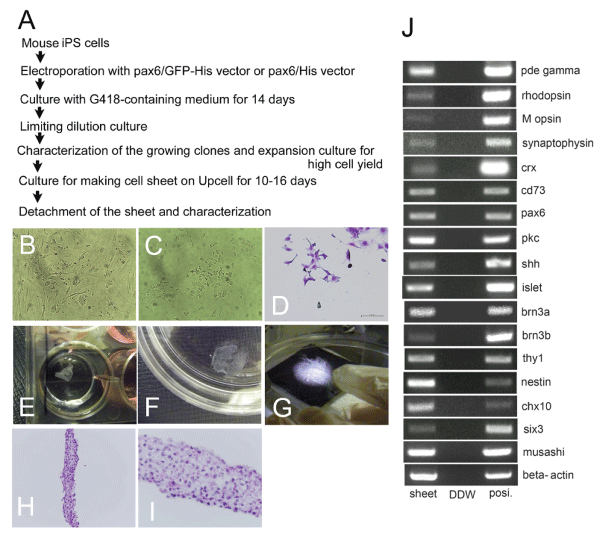
Figure 1: Characterization of cell sheets composed of the cloned retinal
progenitors derived from Pax6-transfected mouse iPS cells.
View Figure 1

A). An experimental protocol of cell sheet formation by culturing retinal
progenitors derived from Pax6-transfected mouse iPS cells.
We conducted transfection of mouse iPS cells with the Pax6 expression
vectors. After selection and limiting dilution culture, we picked up the cloned
cells expressing nestin mRNA and musashi1 mRNA. Then we conducted
second screening where cell clones expressing chx10 mRNA and six3
mRNA simultaneously were selected. After expansion culture in the growth
medium, the cloned cells were harvested. Then the cells were cultured on
temperature-responsive gelation polymer coated plate (Upcell�) for 10 to 16
days depending on the neuronal maturation, where they extended axon like
processes, leading to the formation of a cell sheet.
In some experiments, we introduced low concentrations of SDF1 and MCP1,
in order to facilitate cell sheet formation. Nonetheless, both chemokines
exhibited marginal effects on cell sheet formation (data not shown). The cell
sheet was recovered by lowering temperature of culture plate below 22�C, and
was used for subsequent characterization.
B). Microscopic view of parental mouse iPS cells which were used for
transfection on the mouse fet al fibroblasts at day 1. The iPS cells have already
started to form colonies at day 1.
C). Microscopic view of cloned Pax6-transfected cells in growth media at day
1. Some of the cell had elongated axon like processes, suggesting that Pax6-
transfected cells have different morphology from the undifferentiated iPS cells.
D). Diff-quick staining (a modification of the Wright Giemsa stain) of the cloned
Pax6-transfected cells at day 1.
E). Stereomicroscopic view of the retinal progenitor cell sheet cultured in a
12- well culture plate.
F). Stereomicroscopic view of the sheet transferred into a 10 cm culture dish.
G). Close-up photography of the cell sheet on a black board.
HI. Hematoxylin and eosin (HE) staining of the cell sheet (E, lower
magnification, x 200 and F, higher magnification, x 400).
J). mRNA expressions of the retinal progenitor cell sheet. "sheet" indicated
cDNA of the retinal progenitor cell sheet. "DDW" meant the same PCR
reactions without addition of cDNA. "posi." indicated positive control reactions
using appropriate cDNA.
.
After full characterization of the cells as retinal progenitors, the aliquot of the harvested cells were further cultured in 6-well and 12-well plate coated with FN and temperature-responsive gelation polymers for 10�16 days (average 13 days, depending on confluence and axon elongation). In some experiments, we introduced low concentrations of stromal cell derived factor (SDF) 1 and monocyte chemotactic factor (MCP) 1, in order to facilitate cell sheet formation. Nonetheless, both chemokines exhibited marginal effects on cell sheet formation (data not shown). After termination of cell culture periods, the plates were transferred to another incubator, set at 22�C, for 1 hour to release the cultured cells as an intact cell sheet. Under this protocol, a confluent retinal progenitor cell sheet was spontaneously detached from the plate (Figure 1E-1G). Hematoxylin and eosin (HE) staining (Figure 1H,1I) disclosed that the sheet consisted of cells of rather small nuclei with pale cytoplasm. The thickness of the sheet varied considerably. Nonetheless, we have estimated thickness of the sheet with the scale bars (Figure 2), and found that it was usually from 100 ?m to 300 ?m.
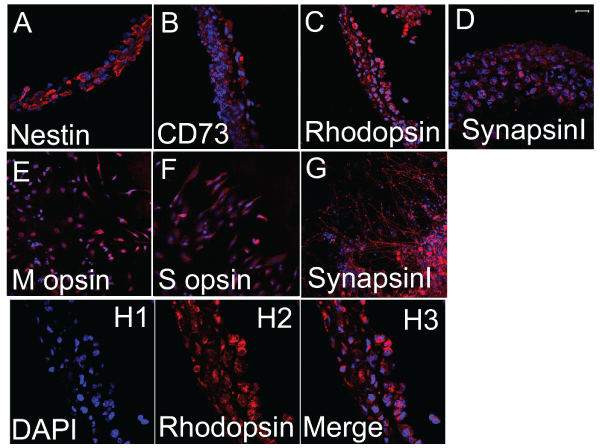 Figure 2: Single color immunostaining of retinal progenitor cell sheets
composed of the cloned Pax6-transfected cells of mouse iPS cell origin.
Figure 2: Single color immunostaining of retinal progenitor cell sheets
composed of the cloned Pax6-transfected cells of mouse iPS cell origin.
Horizontal view (panels E, F, and G) of the sheet and view of vertical section
(panels A, B, C, D and H) of the sheet were presented. The sheets were
positive for Nestin, CD73, Rhodopsin and SynapsinI. The sheets were partly
positive for S opsin and M opsin. The scale bar represents 20�m (panels D
and H) and 40�m (panels A, B, C, E, F and G).
View Figure 2
RT-PCR analyses of the cell sheet disclosed that cells in the sheet expressed nestin mRNA. As for the retinal ganglion cell associated genes, they were positive for shh, islet1, thy1, brn3a and brn3b. They were positive for a bipolar cell marker, pkc and for photoreceptor-related markers, crx, cd73, rod cGMP phosphodiesterase (pde) gamma, rhodopsin, and M opsin (Figure 1J; data not shown) [1,10]. The mRNA expression pattern of the cell sheets was quite similar to the parental cloned pax6 transfected cells. Thus, formation of cell sheets by culturing on the thermo-reversible gelation polymer marginally affected the differentiation status of the cloned pax6 transfected cells and they formed the cell sheet.
Immunostaining of the retinal progenitor cell sheets
With immunostaining (Figure 2), the cloned Pax6-transfected cells 10�16 days after culture in the differentiation medium on Upcell� culture plates expressed Nestin and CD73. The cells in the sheet became expressing Rhodopsin extensively. Some cells of the sheet expressed M opsin and S opsin. SynapsinI was positively stained suggesting synaptic connections.
With two color staining, the Nestin negative cells expressed M opsin in the sheet. Rhodopsin positive cells and M opsin positive cells had some connection with Beta III tubulin positive cells (Figure 3).
 Figure 3: Characterization of retinal progenitor cell sheets derived from the
cloned Pax6-transfected cells by two color immunostaining.
Figure 3: Characterization of retinal progenitor cell sheets derived from the
cloned Pax6-transfected cells by two color immunostaining.
A). The sheets were stained with anti-M opsin (red color) and anti-Nestin
(green color,) mAbs. The Nestin negative cells expressed M opsin in the sheet.
B). The sheets were stained with anti-Rhodopsin (red color) and anti-Beta III
tubulin (green color) mAbs.
C). The sheets were stained with anti- M opsin (red color) and anti-Beta III
tubulin (green color) mAbs.
D). Higher magnification of the panel C.
The scale bar in panel A represents 20�m (for panels A and B). The scale bar
in panel C represents 50 �m for panel C and 20�m for panel D.
View Figure 3
Discussion
Diseases affecting the outer retina are incurable once photoreceptors are lost. We here showed that cloned pax6 transfected mouse iPS cells we had established formed retinal progenitor cell sheets, where the cells were able to differentiate into Rhodopsin and M opsin positive. Our RT-PCR study suggested that our retinal progenitor cell sheets contained various cell types including retinal ganglion cells, bipolar cells and photoreceptor cells. Nonetheless, the cell sheets we made were totally white as can be seen in Figure 1, EFG and completely lacked pigmented cells. We conducted RT-PCR of RPE65, and found that the cell sheets were RPE65 negative (data not shown).
It is possible that the dead cells were included in the sheets after long term cell culture. We need to shorten our cell culture periods to reduce dead cells in the cell sheets.
iPS cells were originally generated via genetic reprogramming of dermal fibroblasts to pluripotency using retroviral transduction with the four transcription factors Oct4, Sox2, KLF4 and c-Myc [10]. Production of iPS cell derived retinal cells expressing a variety of retinal proteins, including those specific to photoreceptors, have been achieved [13-15]. In different animal models of retinal degeneration, retinal transplants can morphologically reconstruct a damaged retina, and restore visual sensitivity [16].
Photoreceptor cell populations generated from mouse iPS cells suggested applications of autologous iPS cells for the effective treatment of retinal degenerative diseases [15]. It is not easy to calculate percentage of each particular cell type in the sheets precisely. We found that Rhodopsin positive cells were most prevalent as could be seen in Figure 2.
Clinically relevant derivation of patient specific iPS cells from accessible adult tissue has already become possible.
This cell sheet showed clearly that Beta III tubulin positive axons connected with the photoreceptor cells. Because both cell types formed the sheet, we did not need to used trypsin or other dispases for recovering the cells, suggesting that cellular connection be retained after recovering cells for transplantation. Unlike retinal sheet transplantation, retinal cell transplantation cannot reconstruct the retinal layer [17- 20], and integrated cell survival decreased significantly in the long term [21]. In contrast, long-term cell survival has been reported via retinal sheet transplantation [22- 24].
Initial encouraging results obtained from animal experiments persuaded researchers to perform retinal transplantation surgeries in human patients [25, 26] and some studies yielded promising results [25]. They conducted transplantation of sheets of fet al retina into the subretinal space using a special device that allows gentle placement of the fragile donor tissue as a flat sheet with the proper orientation/polarity [25]. These sheets were shown to develop a normal lamination pattern in a variety of rat models of retinal degeneration and restored visual responses in the superior colliculus [14, 27, 28]. We need to test whether our retinal progenitor cell sheets are comparable to those reported previously with regard to regenerative potential of the retinal functions [18, 27, 28].
Collectively, our present study supports that retinal progenitor cell sheets derived from patient specific iPS cells may become applicable for treating severe retinal degeneration diseases.
References
-
Pearson RA, Barber AC, Rizzi M, Hippert C, Xue T, et al. (2012) Restoration of vision after transplantation of photoreceptors. Nature 485: 99-103.
-
Barber AC, Hippert C, Duran Y, West EL, Bainbridge JW, et al. (2013) Repair of the degenerate retina by photoreceptor transplantation. Proc Natl Acad Sci U S A 110: 354-359.
-
Mandai M, Homma K, Okamoto S, Yamada C, Nomori A, et al. (2012) Adequate time window and environmental factors supporting retinal graft cell survival in rd mice. Cell Med 4: 45�54.
-
Singh MS, Charbel Issa P, Butler R, Martin C, Lipinski DM, et al. (2013) Reversal of end-stage retinal degeneration and restoration of visual function by photoreceptor transplantation. Proc Natl Acad Sci U S A 110: 1101-1106.
-
Hasan SM, Vugler AA, Miljan EA, Sinden JD, Moss SE, et al. (2010) Immortalized human fet al retinal cells retain progenitor characteristics and represent a potential source for the treatment of retinal degenerative disease. Cell Transplant 19: 1291�1306.
-
Luo J, Baranov P, Patel S, Ouyang H, Quach J, et al. (2014) Human retinal progenitor cell transplantation preserves vision. J Biol Chem 2289: 6362�6371.
-
Zhao T, Li Y, Tang L, Li Y, Fan F, et al. (2011) Protective effects of human umbilical cord blood stem cell intravitreal transplantation against optic nerve injury in rats. Graefes Arch Clin Exp Ophthalmol 249: 1021-1028.
-
Kayama M, Kurokawa MS, Ueda Y, Ueno H, Kumagai Y, et al. (2010) Transfection with pax6 gene of mouse embryonic stem cells and subsequent cell cloning induced retinal neuron progenitors, including retinal ganglion cell-like cells, in vitro. Ophthalmic Res 43: 79�91.
-
Suzuki N, Shimizu J, Takai K, Arimitsu N, Ueda Y, et al. (2012) Establishment of retinal progenitor cell clones by transfection with Pax6 gene of mouse induced pluripotent stem (iPS) cells. Neurosci Lett 509: 116-120.
-
Okita K, Ichisaka T, Yamanaka S (2007) Generation of germline-competent induced pluripotent stem cells. Nature 448: 313-317.
-
Smith JR, Maguire S, Davis LA, Alexander M, Yang F, et al. (2008) Robust, persistent transgene expression in human embryonic stem cells is achieved with AAVS1-targeted integration. Stem Cells 26: 496-504.
-
Yamato M, Utsumi M, Kushida A, Konno C, Kikuchi A, (2001) Thermo-responsive culture dishes allow the intact harvest of multilayered keratinocyte sheets without dispase by reducing temperature. Tissue Eng 7: 473�480.
-
Meyer JS, Shearer RL, Capowski EE, Wright LS, Wallace KA, et al. (2009) Modeling early retinal development with human embryonic and induced pluripotent stem cells. Proc Natl Acad Sci U S A 106: 16698-16703.
-
Lamba DA, McUsic A, Hirata RK, Wang PR, Russell D, et al. (2010) Generation, purification and transplantation of photoreceptors derived from human induced pluripotent stem cells. PLoS One 5: e8763.
-
Tucker BA, Park IH, Qi SD, Klassen HJ, Jiang C, et al. (2011) Transplantation of adult mouse iPS cell-derived photoreceptor precursors restores retinal structure and function in degenerative mice. PLoS One 6: e18992.
-
Zhong X, Gutierrez C1, Xue T2, Hampton C1, Vergara MN1, et al. (2014) Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nat Commun 5: 4047.
-
Gouras P, Du J, Kjeldbye H, Yamamoto S, Zack DJ (1992) Reconstruction of degenerate rd mouse retina by transplantation of transgenic photoreceptors. Invest Ophthalmol Vis Sci 33: 2579-2586.
-
Seiler MJ, Aramant RB (2012) Cell replacement and visual restoration by retinal sheet transplants. Prog Retin Eye Res 31: 661-687.
-
Peng Q, Thomas BB, Aramant RB, Chen Z, Sadda SR, et al. (2007) Structure and function of embryonic rat retinal sheet transplants. Curr Eye Res 32: 781-789.
-
Eberle D, Kurth T, Santos-Ferreira T, Wilson J, Corbeil D, et al. (2012) Outer segment formation of transplanted photoreceptor precursor cells. PLoS One 7: e46305.
-
West EL, Pearson RA, Barker SE, Luhmann UF, Maclaren RE, et al. (2010) Long-term survival of photoreceptors transplanted into the adult murine neural retina requires immune modulation. Stem Cells 28: 1997-2007.
-
Ghosh F, Johansson K, Ehinger B (1999) Long-term full-thickness embryonic rabbit retinal transplants. Invest Ophthalmol Vis Sci 40: 133-142.
-
Gouras P, Du J, Kjeldbye H, Yamamoto S, Zack DJ (1994) Long-term photoreceptor transplants in dystrophic and normal mouse retina. Invest Ophthalmol Vis Sci 35: 3145-3153.
-
Hambright D, Park KY, Brooks M, McKay R, Swaroop A, et al. (2012) Long-term survival and differentiation of retinal neurons derived from human embryonic stem cell lines in un-immunosuppressed mouse retina. Mol Vis 18: 920-936.
-
Radtke ND, Aramant RB, Seiler MJ, Petry HM, Pidwell D (2004) Vision change after sheet transplant of fet al retina with retinal pigment epithelium to a patient with retinitis pigmentosa. Arch Ophthalmol 122: 1159-1165.
-
Radtke ND, Aramant RB, Petry HM, Green PT, Pidwell DJ, et al. (2008) Vision improvement in retinal degeneration patients by implantation of retina together with retinal pigment epithelium. Am J Ophthalmol 146: 172-182.
-
Qiu G, Seiler MJ, Mui C, Arai S, Aramant RB, et al. (2005) Photoreceptor differentiation and integration of retinal progenitor cells transplanted into transgenic rats. Exp Eye Res 80: 515-525.
-
Thomas BB, Aramant RB, Sadda SR, Seiler MJ (2006) Retinal transplantation. A treatment strategy for retinal degenerative diseases. Adv Exp Med Biol 572: 367-376.





