International Journal of Ophthalmology and Clinical Research
Nasal Mucosa Graft and Lamellar Keratoplasty as Treatment in Recurrent Pterygium
Ricardo Romero1*, Ricardo Romero-Fontenlos1, Almudena del Hierro1, Alvaro Arbizu1, Margarita Sanchez-Orgaz1 and Ana Romero2
1Department of Oculoplasty, University Hospital La Paz, Spain
2Department of Oculoplasty, Centro de Oftalmolog�a Castilla Eye Clinic, Spain
*Corresponding author: Ricardo Romero, Department of Oculoplasty, University Hospital La Paz, Madrid, Calle Ana Tutor 1, portal 3,2 B, Las Rozas 28232, Madrid, Spain, Tel: + 34 616726182, Fax: + 34 915625266, E-mail: romeromartinricardo@hotmail.com
Int J Ophthalmol Clin Res, IJOCR-2-016, (Volume 2, Issue 2), Case Report; ISSN: 2378-346X
Received: January 31, 2015 | Accepted: February 25, 2015 | Published: March 02, 2015
Citation: Romero R, Romero-Fontenlos R, del Hierro A, Arbizu A, Sanchez-Orgaz M, et al. (2015) Nasal Mucosa Graft and Lamellar Keratoplasty as Treatment in Recurrent Pterygium. Int J Ophthalmol Clin Res 2:016. 10.23937/2378-346X/1410016
Copyright: © 2015 Romero R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aim: We propose the use of lamellar keratoplasty associated with nasal mucosal graft for the reconstruction of the ocular surface in multiple recurrent pterygium.
Methods: Our patient had undergone four operations of nasal pterygium excision on the right eye, using different surgical procedures: simple excision, excision plus conjunctival autograft, removal plus amniotic membrane graft, excision plus mucosa oral graft plus mitomycin C. All the procedures failed.
Given the need for sizeable graft to reconstruct the lower fornix symblepharon, we decided to take a nasal mucosa graft. We also proposed an eccentric lamellar keratoplasty in order to restore corneal thickness and transparency.
Results: Twenty months after surgery, the fornices were preserved, donor cornea was transparent and there were no signs of recurrence of pterygium.
Discussion: Traditionally, the conjunctival autograft has been the elected treatment for recurrent pterygium. Its main limitation is the inability to obtain large grafts. For this reason we decided to extract nasal mucosa for the reconstruction of the ocular surface. One of the main advantages of the nasal mucosa is that it contains goblet cells, critical for a proper function and production of the film tear. We considered an eccentric lamellar keratoplasty because it provides the Bowman's membrane, which acts as a barrier to impede the growth of conjunctival tissue, decreasing the risk of recurrence.
Keywords
Pterygium, Nasal mucosal graft, Goblet cells, Lamellar keratoplasty
Introduction
Pterygium is a fibrovascular proliferation [1] of conjunctival tissue that grows from the bulbar conjunctiva into the cornea. The term derives from the Greek word pterygium that means wing, because pterygium is a winged shaped tissue with its base located on the periphery and the apex invading the cornea. It is usually placed on interpalpebral conjunctiva, with an axis of 180 degrees. It generally affects the nasal sector, although it may also affect the temporal field.
The pterygium is a relatively common condition that occurs more frequently in countries close to the Equator with warm weather and working populations exposed to the sunlight. The common denominator seems to be the exposure to solar radiation. Other factors involved in its pathogenesis are: rapid evaporation of the tear film, irritation and chronic inflammation of the ocular surface and familial trend.
Clinical manifestations depend on the degree of activity and size, and can range from gritty feeling to significant loss of visual acuity, due to astigmatism or invasion of the visual axis.
The treatment of pterygium is surgical. Many techniques have been employed, from simple resection to more complex ones, such as conjunctival autograft [2] or lamellar keratoplasty [3], associated or not to the use of adjuvant antimetabolites such as mitomycin C. One of the main problems of pterygium surgery is the recurrence rate. Therapeutic management of recurrent pterygium is a challenge for ophthalmologists. We propose the use of lamellar keratoplasty associated with nasal mucosal graft for the reconstruction of the ocular surface in multiple recurrent pterygium.
Case Presentation
Male patient, 68 years old, caucasian, without relevant medical history. He had been operated four times for nasal pterygium on his right eye, having a new relapse affecting the visual axis and causing inferior symblepharon. Since 1996, the patient had undergone four operations of nasal pterygium excision on the right eye, using different surgical procedures: simple excision, excision plus conjunctival autograft, removal plus amniotic membrane graft, excision plus mucosa oral graft plus mitomycin C. All the procedures failed because the pterygium recurred after each intervention.
On the ophthalmic examination, the corrected visual acuity was 20/200 in the right eye and 20/20 in the left eye. Biomicroscopy of the right eye revealed a recurrent nasal pterygium that extends beyond 6 mm on the corneal surface affecting the visual axis, and with inferomedial symblepharon (Figure 1). The patient suffered from binocular diplopia due to the restriction of abduction and supraduction of that eye caused by the pterygium. The rest of the ophthalmologic examination was normal.
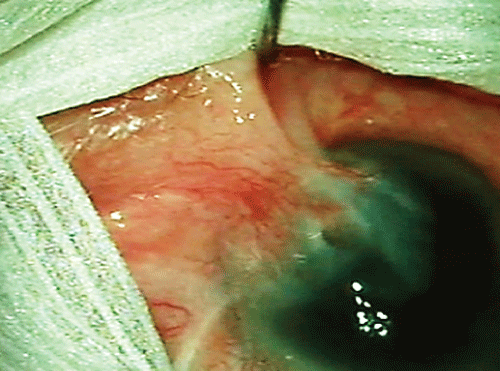
Figure 1: Recurrent nasal pterygium invading visual axis and causing symblepharon.
View Figure 1
Given the need for sizeable graft to reconstruct the lower fornix, we decided to take a nasal mucosa graft. We also proposed an eccentric lamellar keratoplasty in order to restore corneal thickness and transparency. The patient was operated under general anesthesia. The surgery was carried out in three phases. In the first phase, we performed a resection of the middle turbinate of the left nostril, dissecting the mucosa of the turbinate on the operating table. The nasal mucosa obtained was thinned with a No 15 blade before placing it on the ocular surface. In a second phase the pterygium was excised with an extensive removal of Tenon�s layer (13mm by 13 mm), preserving intact the medial rectus muscle, and the symplepharon was dissected and moved into the inferior pouch. To reconstruct the fornix and cover the denuded sclera area, we used the nasal mucosa graft which was sutured with 5-0 Vicryl to the scar tissue dissected, placing it in the pouch; and to the inferonasal sclera using 9-0 nylon. To deepen the fornix, a silicone tube was anchored to it through the total thickness of the lower lid (Figure 2) with 5-0 Vicryl suture. The silicon tube helps to maintain the nasal mucosa graft immobile which is basic for the graft attachment. The silicone tube was removed two weeks after the surgery. Finally, the eccentric lamellar keratoplasty was carried out (Figure 3). An anterior lamella was carved on the donor cornea by Moria artificial chamber, with a 100 micron knife, after corneal disepithelialization. A manual superficial lamellar keratectomy of 7 mm was performed on the recipient cornea, removing the total leucomatous area. The anterior lamella was sutured with 10-0 nylon. To finish the surgery an amniotic membrane layer was placed covering the nasal mucosa and the lamellar keratoplasty, and fixed with 10-0 nylon and fibrin glue (Tissucolc, Baxter, Madrid, Spain). Postoperatively, topical dexamethasone was used every hour during the day, for the first ten days. The topical corticosteroids were slowly tapered over three months.
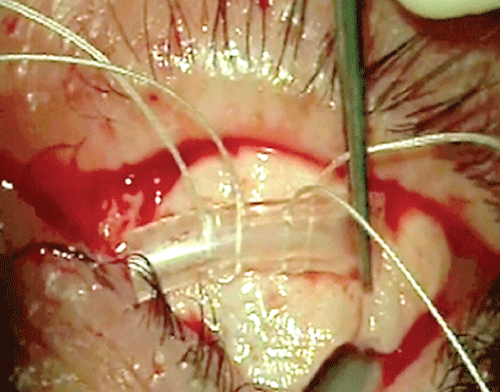
Figure 2: Placement of a silicone tube on the nasal mucosa anchored to the fornix.
View Figure 2
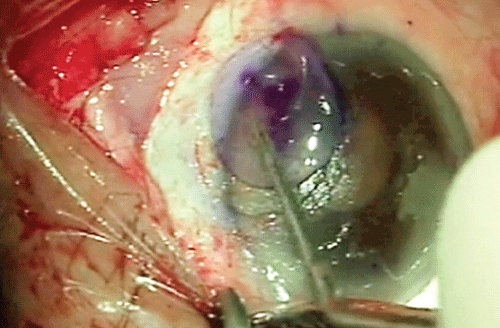
Figure 3: Anterior corneal lamella being placed on the recipient bed. Note the nasal mucosa in the inferior fornix.
View Figure 3
Results
Ten months after surgery, the fornices were preserved, donor cornea was transparent, the corrected visual acuity improved to 20/70, the restriction of abduction and supraduction was minimal and the patient only suffered from binocular diplopia in extreme gazes. There were no clear signs of recurrence of pterygium, but the existence of a neovessel on the inferonasal donor cornea (Figure 4) was treated with two intrastromal bevacizumab injections, spaced for fifteen days.
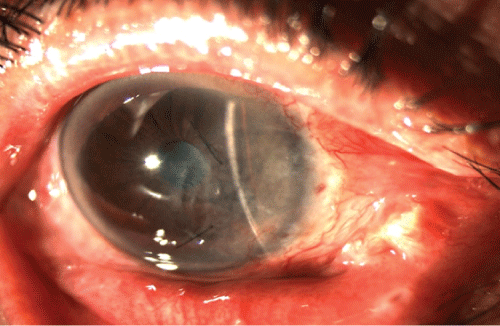
Figure 4: Right eye twenty months after surgery. It has recovered the lower fornix and the cornea remains transparent.
View Figure 4
Discussion
The clinical behaviour of primary and recurrent pterygium differs considerably. The recurrent pterygium presents a more aggressive behaviour, with higher fibrovascular growth and inflammation, with the consequent tendency to symblepharon, ocular motility restrictions and corneal scarring. Pathological studies show that as in primary pterygium predominates the elastotic degeneration of the conjunctiva, in case of recurrences, the fibrotic and vascular proliferation is more prominent1. This is one of the reasons why the degree of failure after recurrent pterygium surgery is higher.
Currently the gold standard treatment for recurrent pterygium is excision with conjunctival autograft, followed or not by supportive treatment, such as mitomycin C. In a randomized clinical trial, Luanratanakorn et al. [2] compared the treatment of primary and recurrent pterygium by excision and conjunctival autograft versus excision and amniotic membrane transplantation. They concluded that when using amniotic membrane graft the recurrence rate was higher than when using the conjunctival autograft.
As mentioned, the conjunctival autograft has been the elected treatment for recurrent pterygium. Its main limitation is the inability to obtain large grafts. Our patient was treated with a conjunctival autograft obtaining poor results including the formation of symblepharon. Therefore, we needed a graft large enough to cover the denuded sclera after pterygium excision, and to restore the fornices. For this reason we decided to extract nasal mucosa for the reconstruction of the ocular surface. The nasal mucosa can be obtained from turbinates or nasal septum, although removing it from turbinates is advisable. The nasal septum mucosa is not recommended because it is usually stiffer due to its content of cartilage which makes difficult the submucosal dissection.
There are published works using buccal mucosa [4] for the surgical treatment of symblepharon with good results. However, based on our experience and the published literature, we consider that nasal mucosa is the best therapeutic option. Pathological studies [5] have shown that the nasal mucosa, as well as the conjunctiva, has many goblet cells, whereas the buccal mucosa lacks of them. As it is known, goblet cells are critical for a proper function and production of the film tear, which is of utmost importance in patients with ocular surface disease. Kim et al. [6] have shown by immunohistochemistry, the persistence of goblet cells in the nasal mucosa grafts placed on the ocular surface, one year after surgery. The aforementioned work by Kim et al. [6] also collected a series of six patients with symblepharon secondary to chemical burns, three of them treated with buccal mucosa graft and the other three with nasal mucosa graft. The group treated with nasal mucosa showed no complications, while the patients treated with oral mucosa showed a delay in epithelialization and ischemic thinning.
Lamellar keratoplasty is another option available for the treatment of recurrent pterygium. Golchin et al. [3] reported a recurrence rate of 5'9%, in a series of 68 eyes with recurrent pterygium treated with lamellar keratoplasty, with a mean follow-up of 6 months. In the present case, we considered eccentric lamellar keratoplasty for several reasons. First, as proposed by Golchin in his work, the Bowman's membrane acts as a barrier to impede the growth of conjunctival tissue. The corneal lamellar grafts provide this membrane, restoring this barrier, thereby decreasing the risk of recurrence. Moreover, because it is a scarred and thinned cornea, the lamellar graft provides tectonic support and restores corneal thickness and transparency.
For all the exposed above, we consider the use of lamellar keratoplasty associated with nasal mucosal graft a good option for multiple recurrent pterygium treatment.
References
-
Ranty ML, Quintyn JC, Uro-Coste E, Delisle MB (2012) Ocular conjunctival pathology. A ten-year retrospective study in Toulouse-Rangueil University Hospital and literature review Ann Pathol 32: 170-176.
-
Luanratanakorn P, Ratanapakorn T, Suwan-Apichon O, Chuck RS (2006) Randomised controlled study of conjunctival autograft versus amniotic membrane graft in pterygium excision. Br J Ophthalmol 90: 1476-1480.
-
Golchin B, Butler TK, Robinson LP, Wechsler AW, Sutton G, et al. (2003) Long-term follow-up results of lamellar keratoplasty as a treatment for recurrent pterygium and for scleral necrosis induced by beta-irradiation. Cornea 22: 612-618.
-
Kheirkhah A, Blanco G, Casas V, Hayashida Y, Raju VK, et al. (2008) Surgical strategies for fornix reconstruction based on symblepharon severity. Am J Ophthalmol 146: 266-275.
-
Weinberg DA, Tham V, Hardin N, Antley C, Cohen AJ, et al. (2007) Eyelid mucous membrane grafts: a histologic study of hard palate, nasal turbinate, and buccal mucosal grafts. Ophthal Plast Reconstr Surg 23: 211-216.
-
Kim JH, Chun YS, Lee SH, Mun SK, Jung HS, et al. (2010) Ocular surface reconstruction with autologous nasal mucosa in cicatricial ocular surface disease. Am J Ophthalmol 149: 45-53.





