International Journal of Ophthalmology and Clinical Research
Treatment of Orbital IgG4-Related Disease
Rachel Lee1, Craig A MacKinnon1, Neil Aburn2 and Swee T Tan1,3*
1Head & Neck and Skull Base Surgery/Oncology Programme, Wellington Regional Plastic, Maxillofacial & Burns Unit, Hutt Hospital, New Zealand
2Department of Ophthalmology, Wellington Regional Hospital, New Zealand
3Gillies McIndoe Research Institute, Wellington, New Zealand
*Corresponding author: Swee T Tan, ONZM, MBBS, PhD, FRACS, Gillies McIndoe Research Institute, PO Box 7184, Newtown, Wellington 6242, New Zealand, Tel: +64 4 2820366, E-mail: swee.tan@gmri.org.nz
Int J Ophthalmol Clin Res, IJOCR-2-019, (Volume 2, Issue 2), Review Article; ISSN: 2378-346X
Received: February 26, 2015 | Accepted: March 28, 2015 | Published: March 31, 2015
Citation: Lee R, MacKinnon CA, Aburn N, Tan ST (2015) Treatment of Orbital IgG4-Related Disease. Int J Ophthalmol Clin Res 2:019. 10.23937/2378-346X/1410019
Copyright: © 2015 Lee R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
IgG4-related disease (IRD) is a distinct group of disorders with specific pathological and clinical features that can manifest in various organs. It is characterised by intense lymphoplasmocytic inflammation with IgG4+ cells of the involved organ, subsequently leading to fibrosis and formation of tumefactive masses. Its pathophysiology is unclear but the involvement of a T Helper2-dominant immune response and significant numbers of CD4+, CD25+ and Foxp3+ regulatory T cells have been implicated. Corticosteroids are currently the mainstay treatment for IRD but are associated with adverse side effects, recurrence on discontinuation of medication, and limited response if significant fibrosis is already present. Pentoxyphylline (POF) and α-tocopherol (AT) are anti-inflammatory and anti-fibrosing agents that can be effective in treating both the acute inflammatory and chronic fibrosclerosing components of IRD. This article reviews IRD and its treatment including the emerging role of POF and AT for this inflammatory fibrosclerosing condition.
Keywords
IgG4-related disease, Orbital, Pentoxifylline, α-Tocopherol, Inflammatory, Fibro-sclerotic, Steroids, Treatment, Autoimmune
Introduction
IgG4-related disease (IRD) is a newly recognised, distinct group of disorders with a common pathophysiology but variable manifestations [1]. It was first described in the Japanese literature in 2003, in a case series that identified extra-pancreatic manifestations of patients with auto-immune pancreatitis [2]. It has since been widely reported by other Japanese, other Asian, and North American groups who propose useful diagnostic guidelines and criteria for different subsets of the disease [3,4]. However, there remains a paucity of literature about this condition and its treatment outside Japan, mainly due to the lack of awareness of this new entity and its variable manifestations.
Disease Characteristics
IRD is characterised by intense IgG4+ plasma cell infiltration of the involved organ(s), storiform fibrosis, formation of tumour-like masses, 5 - 50 folds elevated serum levels of IgG4 and high sensitivity to corticosteroids [1,5]. Symptoms depend on the organ(s) affected, most commonly the pancreas (autoimmune pancreatitis) [6], biliary system (sclerosing cholangitis) [7], lacrimal and salivary glands (Mikulicz�s disease) [8]. Involvement of the orbit [9], thyroid (Riedel�s thyroiditis) [10], kidneys (tubulointerstitial nephritis) [11], retroperitoneum (retroperitoneal fibrosis) [12], aorta (aortitis) [13], bowel (mesenteritis) [14], prostate [15], breast [16], lungs [17], pericardium [18], meninges [19], hypophysis [20], lymph nodes [9] and skin [21] have also been described. Any number of organs can be affected simultaneously or metachronously [5].
In IRD, the extensive infiltration of IgG4+ plasma cells and T lymphocytes in the affected organ(s) disrupts tissue form and function, and also initiates an intense inflammatory response [5,22]. These processes lead to fibrosis, obliterative phlebitis and formation of masses within the affected organ [5,22], which are often mistaken for tumors. As the disease progresses slowly and insidiously, the patient may present with mild and non-specific symptoms relating to organ swelling and disruption of function [5].
Orbital IRD
IRD can affect the orbit and ocular structures, causing swelling of the lacrimal glands (dacryoadenitis) [9,23], orbital septum [24], extra-ocular muscles (myositis) [25], optic nerve sheath [9], and xanthogranulomatous changes of the orbit [26], all of which can be unilateral or bilateral. In the retrobulbar region, the disease is associated with the formation of pseudotumors [25], which can cause proptosis, pain, restriction in ocular movement with subsequent diplopia, and reduced visual acuity. It has also been reported to extend beyond the orbit to infiltrate the infra-orbital nerve [25], and intracranially to involve the cavernous sinus [27]. Interestingly, the conjunctiva is not affected [9,23]. It is important to consider this diagnosis in the work-up of orbital inflammatory disease, and to distinguish it from other conditions with similar presentations, such as idiopathic orbital disease, ocular adnexal marginal zone B-cell lymphoma, systemic vasculitic conditions such as Wegener�s granulomatosis, and ocular myositis. In IRD, in addition to the heavily increased presence of IgG4+ cells in biopsy specimens or serum, there may also be accompanying systemic symptoms if there is concurrent involvement of other organs such as the pancreas or peripheral lymph nodes [9], which may help in the diagnosis of the disease.
Pathogenesis and Pathophysiology
The pathogenesis and pathophysiology of IgG4 activation and infiltration in IRD is not well understood. There are two main immunological characteristics that have been observed both locally in the affected tissues and peripherally. The first favors that of a T helper2 (Th2)-dominant immune response, which favours the production of Th2 cytokines including IL-4, IL-5, IL-10 and IL-13 [28,29]. These cytokines also trigger antibody class switch to IgE and eosinophilic migration and infiltration [29], which together with the high incidence of allergic manifestations observed in patients with IRD [30], suggest the involvement of an allergic mechanism in the pathogenesis of the disease [30].
The second is that of significantly higher numbers of regulatory T cells expressing CD4, CD25, and transcription factor Foxhead Box P3 (Foxp3) [28], along with over-expression of regulatory cytokines IL-10 and transforming growth factor-β1 (TGF-β1) [28]. IL-10 directs B cells to produce IgG4 [31], while TGF-β1, a key pro-fibrotic cytokine, induces the deposition of extracellular matrix proteins and Type-1 collagen, leading to fibroplasia [32]. The intense inflammatory process generated by the intense infiltration of IgG4 antibodies leads to the pathological production of oxidative stress and free oxygen radicals, which may overwhelm the cellular response mechanism, resulting in tissue injury [27].
Serological Findings
The hallmark of IRD is an elevation in serum levels of IgG4 [5]. While useful in confirming the diagnosis of IRD, it is not a specific diagnostic marker [33], and has to be correlated with histopathological analysis [3]. In addition, it is difficult to co-relate serum levels to the disease severity of a specific organ [34], as it depends on an estimate of the amount of plasma cells within a particular organ [34]. Similarly, serum levels of IgG4 may remain elevated following treatment despite regression of the lesion [33], due to residual IgG4 secreting cells located sub-clinically elsewhere. Clinical improvement remains the best indicator for monitoring therapeutic efficacy.
Treatment
Corticosteroids are the mainstay treatment for IRD [35] based on their immunomodulating and anti-inflammatory properties [36], specifically, inhibition of B and T cell activity, cytokine production, and inflammatory cascades [36]. However, their effects are often short-lived with recurrence upon discontinuation or early tapering of the medication [35]. In addition, they are associated with significant adverse effects including immunosuppression, osteoporosis, Cushingoid facies, glucose intolerance and hypertension [36]. Immuno-modulators, such as methotrexate, azathioprine, mycophenolate, rituximab and bortezomib have been used in recurrent or refractory cases [35]. While prolonged remission and successful treatment of the disease has been reported, it is confined to a few case studies and small retrospective series with short follow-up [35]. Hence their long term efficacy remains unknown.
Pentoxyphylline and α-Tocopherol as A Novel Treatment for IRD
Pentoxyphylline (POF) is a methylxanthine derivative which acts as a non-specific phosphodiesterase inhibitor with anti-inflammatory [37], anti-fibrotic [38] and anti-oxidant properties [39]. Specifically, it inhibits TNF-α production [37], fibroblast proliferation and collagen deposition, while stimulating collagenase activity [38]. It has been used for the treatment of a variety of inflammatory and fibrotic conditions, including peripheral vascular disease, steatohepatitis, sarcoidosis, radiation-induced fibrosis and osteoradionecrosis (ORN) [27,40,41].
α-Tocopherol (AT), more commonly known as vitamin E, is a lipid-soluble vitamin with anti-oxidant properties [42]. It scavenges free oxygen radicals produced during oxidative stress. [42], and protects Lee R, et al. membranes against lipid peroxidation [42], AT may also act as an anti-fibrotic agent [43], as it has been shown to inhibit the transcription of procollagen type 1 and over-expression of TGF-β1, both of which induce fibrosis [43]. It has been used in a variety of conditions where there is sensitivity to oxidative reactions, including delaying age-related macular degeneration, atherosclerosis, functional impairment or decline in patients with Alzheimer�s� disease, and non-alcoholic fatty liver disease [42].
A recent case report demonstrates the novel use of these two agents in the treatment of orbital IRD in a 32 year-old woman who presented with an 18-month history of diplopia, proptosis and headaches (Figure 1A, Supplemental Figure 1A) [44]. An MRI scan showed an ill-defined, intra-conal lesion causing 5mm of proptosis and displacement of the optic nerve supero-medially (Figure 2).
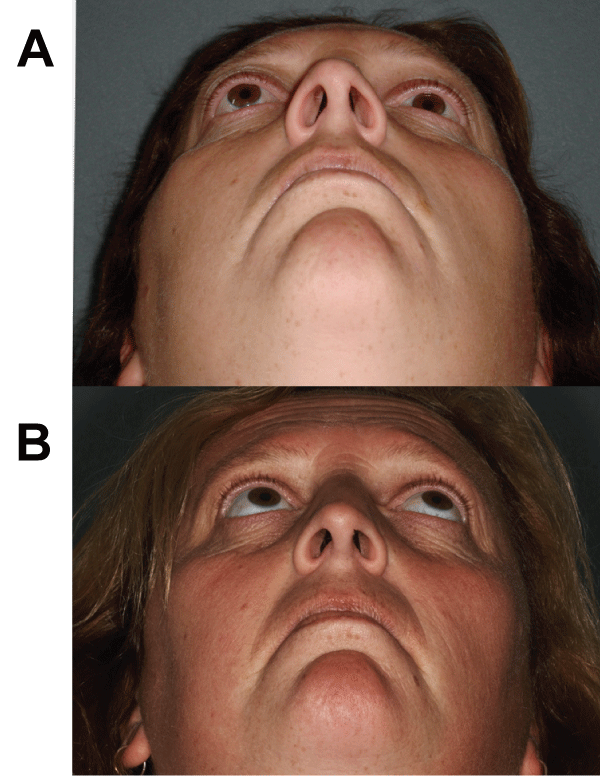
Figure 1: (A) Worm�s eye view of a 32-year old female referred with on-going
symptoms of right-sided diplopia, proptosis, retrobulbar pain and headaches.
Full ophthalmological assessment showed 6/18 vision on right compared to
6/9 on left (aided), 5mm of proptosis and markedly reduced elevation of the
right eye. (B) Complete resolution of symptoms and proptosis with normal
extraocular movements without diplopia, and return of visual acuity to the
pretreatment state, one month after a 22-month course of pentoxifylline and
α-tocopherol. Reproduced with permission from BJOMFS [44].
View Figure 1
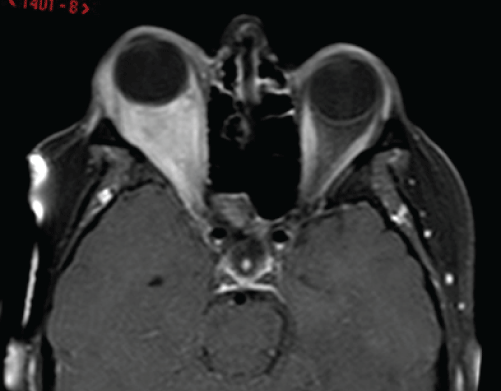
Figure 2: Axial T1 post-contrast MRI sequence showing a large ill-defined
intra-orbital mass that occupied most of the intra-conal space extending from
the globe to the orbital apex displacing the optic nerve supero-medially and
5mm of proptosis. Reproduced with permission from BJOMFS [44].
View Figure 2
The lesion initially responded to steroid therapy which was discontinued because it interfered with her diabetic control. An open biopsy showed a fibro-sclerotic lesion with B and T cell infiltration, increased IgG4+ plasma cells, up to 135/high power field, and IgG4+/IgG+ cell ratio of 41.8%, which confirmed the diagnosis of orbital IRD. The levels of serum IgG4 were not measured. Following a 22-month course of POF 800mg and AT 1,000IU orally twice daily, there was complete resolution of the symptoms and proptosis clinically (Figure 1B, Supplemental Figure 1B), as well as marked reduction of the lesion with minimal enhancement on repeat MRI scanning (Supplemental Figure 2).
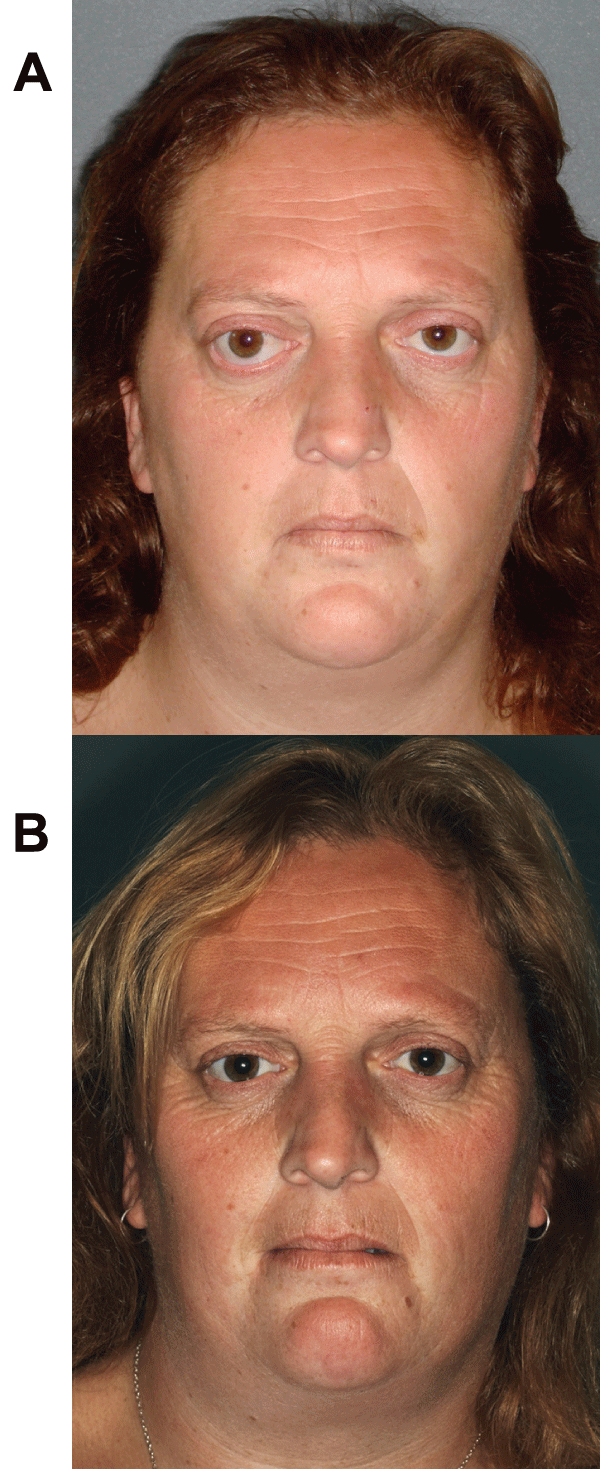
Supplemental Figure 1: (A) Frontal view of a 32-year old female referred
with on-going symptoms of diplopia, right-sided proptosis, retrobulbar pain
and headaches. Full ophthalmological assessment showed 6/18 vision
on right compared to 6/9 on left (aided), 5mm of proptosis and markedly
reduced elevation of the right eye. (B) Complete resolution of symptoms
and proptosis with normal extraocular movements without diplopia, and
return of visual acuity to the pre-treatment state, one month after a 22-month
course of pentoxifylline and α-tocopherol. Reproduced with permission from
BJOMFS [44].
View Supplemental Figure 1
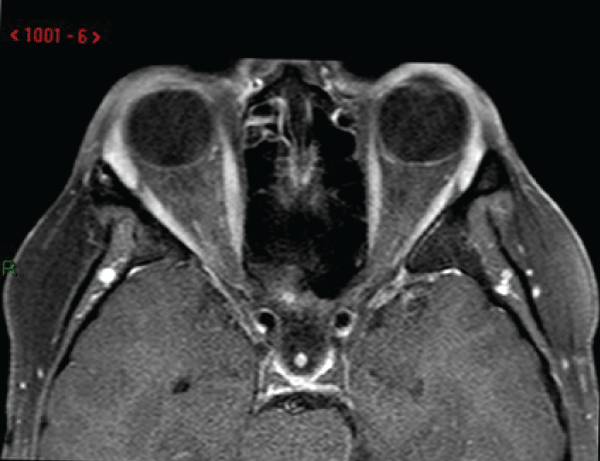
Supplemental Figure 2: Axial T1 post-contrast MRI sequence 22 months
following treatment with pentoxifylline and α-tocopherol, showing reduction in
the volume of the lesion that enhanced minimally and complete resolution of
proptosis. Reproduced with permission from BJOMFS [44].
View Supplemental Figure 2
The proposed mechanism of action of these two agents is that being anti-oxidant and anti-inflammatory, POF and AT trap free oxygen radicals to mitigate the inflammatory cascade in IRD [27]. They also block the expression of TGF-β1, as well as other cytokines and growth factors secreted by fibroblasts involved in inflammation, thus preventing the deposition of Type-1 collagen responsible for tissue fibrosis [45]. Therefore, by inhibiting both the inflammatory and fibrotic processes in IRD, they can effectively abrogate the acute as well as ongoing inflammatory response [27]. In contrast, corticosteroids and immunomodulators only target the lymphoid component but not the fibrous component of IRD. This may account for the limited response of IRD lesions to these agents, especially if there is already significant sclerosis within the lesion, as in the case reported [44].
An excellent example in the literature where this combination of POF and AT has also been shown to be effective is in the treatment of osteoradianecrosis (ORN), a condition in which there is a delayed irradiation-induced failure of bone and soft tissue healing [41]. The affected tissues in ORN show hypovascular-hypoxic effects and bony fibroatrophy [41]. Delmanian et al. [41] demonstrate that when used in combination, POF and AT result in significant reduction of exposed bone, improvement in mucosal healing, and improvement of local symptoms. They have also shown that POF or AT is ineffective when used alone [46], suggesting a synergistic effect of the Lee R, et al. agents. Interestingly, the authors find that two years is the duration needed to achieve two-thirds of the maximum response to the treatment [47]. A similar period of treatment with POF and AT was required for the patient with orbital IRD, resulting in complete resolution of proptosis was observed clinically and marked reduction in the volume of the lesion that enhanced minimally on repeat MRI scanning [44].
POF and AT have both been demonstrated to be safe and well tolerated by patients in the short and long term [46,47]. There is conflicting data on the safety of high dose AT with regards to mortality [48,49], although the general consensus is that it has neither significant beneficial nor adverse effects on survival regardless of dose [48]. The main side effects of POF experienced by a small number of patients in the series reported by Delanian et al. [46,47] include epigastralgia, headaches, asthenia, vertigo, insomnia, and hypotension. However, these were easily controlled with reduction in POF dosage and symptomatic treatment, and no participant discontinued the treatment because of these side effects [47]. The dosage of POF and AT used in these trials was similar to the dosage we used for the patient with orbital IRD [44].
It will be useful to conduct clinical trials using POF and AT on IRD affecting the orbit and/or other organs.
References
-
Stone JH, Zen Y, Deshpande V (2012) IgG4-related disease. N Engl J Med 366: 539-551.
-
Kamisawa T, Funata N, Hayashi Y, Eishi Y, Koike M, et al. (2003) A new clinicopathological entity of IgG4-related autoimmune disease. J Gastroenterol 38: 982-984.
-
Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, et al. (2012) Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD). Mod Rheumatol 22: 21-30.
-
Chari ST, Smyrk TC, Levy MJ, Topazian MD, Takahashi N, et al. (2006) Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin Gastroenterol Hepatol 4: 1010-1016.
-
Cheuk W, Chan JK (2010) IgG4-related sclerosing disease: a critical appraisal of an evolving clinicopathologic entity. Adv Anat Pathol 17: 303-332.
-
Deshpande V, Gupta R, Sainani N, Sahani DV, Virk R, et al. (2011) Subclassification of autoimmune pancreatitis: a histologic classification with clinical significance. Am J Surg Pathol 35: 26-35.
-
Kamisawa T, Takuma K, Anjiki H, Egawa N, Kurata M, et al. (2009) Sclerosing cholangitis associated with autoimmune pancreatitis differs from primary sclerosing cholangitis. World J Gastroenterol 15: 2357-2360.
-
Stone JH (2012) IgG4-related disease: nomenclature, clinical features, and treatment. Semin Diagn Pathol 29: 177-190.
-
Plaza JA, Garrity JA, Dogan A, Ananthamurthy A, Witzig TE, et al. (2011) Orbital inflammation with IgG4-positive plasma cells: manifestation of IgG4 systemic disease. Arch Ophthalmol 129: 421-428.
-
Dahlgren M, Khosroshahi A, Nielsen GP, Deshpande V, Stone JH (2010) Riedel's thyroiditis and multifocal fibrosclerosis are part of the IgG4-related systemic disease spectrum. Arthritis Care Res (Hoboken) 62: 1312-1318.
-
Saeki T, Nishi S, Ito T, Yamazaki H, Miyamura S, et al. (2007) Renal lesions in IgG4-related systemic disease. Intern Med 46: 1365-1371.
-
Fujimori N, Ito T, Igarashi H, Oono T, Nakamura T, et al. (2013) Retroperitoneal fibrosis associated with immunoglobulin G4-related disease. World J Gastroenterol 19: 35-41.
-
Stone JH, Khosroshahi A, Hilgenberg A, Spooner A, Isselbacher EM, et al. (2009) IgG4-related systemic disease and lymphoplasmacytic aortitis. Arthritis Rheum 60: 3139-3145.
-
Wong DD, Pillai SR, Kumarasinghe MP, McGettigan B, Thin LW, et al. (2012) IgG4-related sclerosing disease of the small bowel presenting as necrotizing mesenteric arteritis and a solitary jejunal ulcer. Am J Surg Pathol 36: 929-934.
-
Nishimori I, Kohsaki T, Onishi S, Shuin T, Kohsaki S, et al. (2007) IgG4-related autoimmune prostatitis: two cases with or without autoimmune pancreatitis. Intern Med 46: 1983-1989.
-
Cheuk W, Chan AC, Lam WL, Chow SM, Crowley P, et al. (2009) IgG4-related sclerosing mastitis: description of a new member of the IgG4-related sclerosing diseases. Am J Surg Pathol 33: 1058-1064.
-
Zen Y, Inoue D, Kitao A, Onodera M, Abo H, et al. (2009) IgG4-related lung and pleural disease: a clinicopathologic study of 21 cases. Am J Surg Pathol 33: 1886-1893.
-
Atallah PC, Kassier A, Powers S (2014) IgG4-related disease with effusive-constrictive pericarditis, tamponade, and hepatopathy: a novel triad. Int J Cardiol 176: 516-518.
-
Lindstrom KM, Cousar JB, Lopes MB (2010) IgG4-related meningeal disease: clinico-pathological features and proposal for diagnostic criteria. Acta Neuropathol 120: 765-776.
-
Leporati P, Landek-Salgado MA, Lupi I, Chiovato L, Caturegli P (2011) IgG4-related hypophysitis: a new addition to the hypophysitis spectrum. J Clin Endocrinol Metab 96: 1971-1980.
-
Cheuk W, Lee KC, Chong LY, Yuen ST, Chan JK (2009) IgG4-related Sclerosing disease: a potential new etiology of cutaneous pseudolymphoma. Am J Surg Pathol 33: 1713-1719.
-
Zen Y, Nakanuma Y (2010) IgG4-related disease: a cross-sectional study of 114 cases. Am J Surg Pathol 34: 1812-1819.
-
Sato Y, Ohshima K, Ichimura K, Sato M, Yamadori I, et al. (2008) Ocular adnexal IgG4-related disease has uniform clinicopathology. Pathol Int 58: 465-470.
-
Mehta M, Jakobiec F, Fay A (2009) Idiopathic fibroinflammatory disease of the face, eyelids, and periorbital membrane with immunoglobulin G4-positive plasma cells. Arch Pathol Lab Med 133: 1251-1255.
-
Wallace ZS, Khosroshahi A, Jakobiec FA, Deshpande V, Hatton MP, et al. (2012) IgG4-related systemic disease as a cause of "idiopathic" orbital inflammation, including orbital myositis, and trigeminal nerve involvement. Surv Ophthalmol. 57: 26-33
-
Singh K, Rajan KD, Eberhart C (2010) Orbital necrobiotic xanthogranuloma associated with systemic IgG4 disease. Ocul Immunol Inflamm 18: 373-378.
-
Delanian S, Lefaix JL (2004) The radiation-induced fibroatrophic process: therapeutic perspective via the antioxidant pathway. Radiother Oncol 73: 119-131.
-
Zen Y, Fujii T, Harada K, Kawano M, Yamada K, et al. (2007) Th2 and regulatory immune reactions are increased in immunoglobin G4-related sclerosing pancreatitis and cholangitis. Hepatology 45: 1538-1546.
-
Kudo-Tanaka E, Nakatsuka S, Hirano T, Kawai M, Katada Y, et al. (2009) A case of Mikulicz's disease with Th2-biased cytokine profile: possible feature discriminable from Sj�gren's syndrome. Mod Rheumatol 19: 691-695.
-
Kamisawa T, Anjiki H, Egawa N, Kubota N (2009) Allergic manifestations in autoimmune pancreatitis. Eur J Gastroenterol Hepatol 21: 1136-1139.
-
Jeannin P, Lecoanet S, Delneste Y, Gauchat JF, Bonnefoy JY (1998) IgE versus IgG4 production can be differentially regulated by IL-10. J Immunol 160: 3555-3561.
-
Detlefsen S, Sipos B, Zhao J, Drewes AM, Kl�ppel G (2008) Autoimmune pancreatitis: expression and cellular source of profibrotic cytokines and their receptors. Am J Surg Pathol 32: 986-995.
-
Sah RP, Chari ST (2011) Serologic issues in IgG4-related systemic disease and autoimmune pancreatitis. Curr Opin Rheumatol 23: 108-113.
-
Strehl JD, Hartmann A, Agaimy A (2011) Numerous IgG4-positive plasma cells are ubiquitous in diverse localised non-specific chronic inflammatory conditions and need to be distinguished from IgG4-related systemic disorders. J Clin Pathol 64: 237-243.
-
Khosroshahi A, Stone JH (2011) Treatment approaches to IgG4-related systemic disease. Curr Opin Rheumatol 23: 67-71.
-
Rhen T, Cidlowski JA (2005) Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med 353: 1711-1723.
-
Schandene L, Vandenbussche P, Crusiaux A, Al�gre ML, Abramowicz D, et al. (1992) Differential effects of pentoxifylline on the production of tumour necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6) by monocytes and T cells. Immunology 76: 30-34.
-
Berman B, Duncan MR (1989) Pentoxifylline inhibits normal human dermal fibroblast in vitro proliferation, collagen, glycosaminoglycan, and fibronectin production, and increases collagenase activity. J Invest Dermatol. 92: 605-610.
-
Horvath B, Marton Z, Halmosi R, Alexy T, Szapary L, et al. (2002) In vitro antioxidant properties of pentoxifylline, piracetam, and vinpocetine. Clin Neuropharmacol 25: 37-42.
-
Ward A, Clissold SP (1987) Pentoxifylline. A review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy. Drugs 34: 50-97.
-
Delanian S, Chatel C, Porcher R, Depondt J, Lefaix JL (2011) Complete restoration of refractory mandibular osteoradionecrosis by prolonged treatment with a pentoxifylline-tocopherol-clodronate combination (PENTOCLO): a phase II trial. Int J Radiat Oncol Biol Phys. 80: 832-839.
-
Tucker JM, Townsend DM (2005) Alpha-tocopherol: roles in prevention and therapy of human disease. Biomed Pharmacother 59: 380-387.
-
Lefaix JL, Delanian S, Vozenin MC, Leplat JJ, Tricaud Y, et al. (1999) Striking regression of subcutaneous fibrosis induced by high doses of gamma rays using a combination of pentoxifylline and alpha-tocopherol: an experimental study. Int J Radiat Oncol Biol Phys 43: 839-847.
-
Lee R, MacKinnon CA, Aburn N, Tan ST (2015) Orbital immunoglobulin IgG4-related inflammatory fibrosclerosing lesion treated with pentoxifylline and a-tocopherol: case report. Br J Oral Maxillofac Surg 53: 197-199.
-
Delanian S, Lefaix JL (2004) The radiation-induced fibroatrophic process: therapeutic perspective via the antioxidant pathway. Radiother Oncol 73: 119-131.
-
Delanian S, Porcher R, Balla-Mekias S, Lefaix JL (2003) Randomized, placebo-controlled trial of combined pentoxifylline and tocopherol for regression of superficial radiation-induced fibrosis. J Clin Oncol 21: 2545-2550.
-
Delanian S, Porcher R, Rudant J, Lefaix JL (2005) Kinetics of response to long-term treatment combining pentoxifylline and tocopherol in patients with superficial radiation-induced fibrosis. J Clin Oncol 23: 8570-8579.
-
Berry D, Wathen JK, Newell M (2009) Bayesian model averaging in meta-analysis: vitamin E supplementation and mortality. Clin Trials 6: 28-41.
-
Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, et al. (2005) Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med 142: 37-46.





