International Journal of Ophthalmology and Clinical Research
Glaucoma Functional Damage and Comparative Psychophysical Studies
Amal A. Elgohary1, Laila Hassan M. Elshazly2* and Iman A. Fahmy3
1Department of Ocular Physiology, Research Institute of Ophthalmology, Giza, Cairo 12511, Egypt
2Department of Ophthalmology, Memorial Institute of Ophthalmology, Giza, Cairo 12511, Egypt
3Department of Ophthalmology, Research Institute of Ophthalmology, Giza, Cairo 12511, Egypt
*Corresponding author: Laila Hassan M. El-shazly, Memorial institute of Ophthalmology, 3-Al-Ahram street, Giza, Cairo 12511, Egypt, E-mail: laihasan@gmail.com
Int J Ophthalmol Clin Res, IJOCR-2-039, (Volume 2, Issue 5), Research Article; ISSN: 2378-346X
Received: June 28, 2015 | Accepted: September 21, 2015 | Published: September 23, 2015
Citation: Elgohary AA, Elshazly LHM, Fahmy IA (2015) Glaucoma Functional Damage and Comparative Psychophysical Studies. Int J Ophthalmol Clin Res 2:039. 10.23937/2378-346X/1410039
Copyright: © 2015 Elgohary AA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: To determine the value of color vision, contrast sensitivity and stereopsis testing in measuring the extent of glaucomatous damage and how it helps in early diagnosis.
Methods: In a cross-sectional clinical study, 112 eyes of 56 glaucoma patients and 100 eyes of 50 normal control subjects underwent, automated perimetry, measurement of color vision [D15 test and city university color vision test (CUCV)], Lang stereoacuity test and binocular contrast sensitivity. Diagnosis of glaucoma was based on intraocular pressure, visual field (VF) and optic disc changes. Glaucoma patients were divided into two groups; group 1 with mild glaucoma VF changes and group 2 with advanced glaucoma VF changes.
Results: In early glaucoma group stereoacuity and binocular contrast sensitivity (at all spatial frequencies) were significantly decreased compared to control cases. There was significant difference between D15 and CUCV color tests in diagnosis of tritan defect in cases of glaucoma (P < 0.001). D15 was found more sensitive (P = 0.001) and more specific (P = 0.03), as compared to CUCV. The advanced glaucoma patients showed more significant defects in all these testing measures.
Conclusions: The binocular contrast sensitivity, stereoacuity and D15 color vision tests all together could help in detection of early glaucomatous nerve damage. These tests also could help in assessment of the glaucoma progress. CUCV is not the ideal test for discriminating patients with glaucoma.
Keywords
Glaucoma, Contrast sensitivity, Visual field, Color vision, Stereopsis
Introduction
Many clinical tests have been developed to determine the damage of visual function caused by glaucoma. These tests help in early detection of glaucoma, quantification of glaucomatous damage and progression. Currently, perimetry remains the measure of choice for studies of glaucoma progression. Visual field loss is the most prevalent and characteristic form of visual function loss associated with glaucoma [1]. High visual centers can combine the two disparate, inhomogeneous visual fields. Therefore it would be best to add binocular visual measures in patients with glaucoma to provide the most accurate representation of the patient's visual function that they normally use [2].
Visual function measurements in glaucoma cases include electrophysiology, visual acuity and psychophysical testing (i.e. color, contrast and depth perception) [3]. Visual acuity could not detect early changes in primary open angle glaucoma because the central macula is spared until late stages of glaucoma [3]. The higher order nature of color, contrast and stereopsis stimuli suggests that their responses are processed cortically and implies that their afferent signal is largely mediated by retinal ganglion cells (RGCs) supplying the main corticothalamic visual pathway [4]. Consequently, the percept of these types of stimuli is altered in glaucoma [5]. Damage to cortical regions involved in processing color, contrast and stereopsis visual percepts has been shown to result in reduced psychophysical functions [6]. Therefore, testing glaucoma subjects using different forms of psychophysiological tests has been shown to increase the diagnostic accuracy [6].
The purpose of the present study was to use color vision, contrast sensitivity and stereoacuity psychophysical tests in cases of glaucoma to determine how these tests are able to diagnose and evaluate the glaucomatous damage. These tests were performed binocularly in order to provide a better understanding of how a patient might be affected under natural viewing conditions. Also, we compared D15 and CUCV tests in early diagnosis of color vision loss in glaucoma cases.
Subjects and Methods
The study included 56 patients with glaucoma and 50 normal controls. Subjects were recruited from the outpatient clinic of the Research Institute of Ophthalmology. Informed written consent was given by all participants. All research adhered to the tenets of the Declaration of Helsinki. The diagnosis of glaucoma was established by slitlamp biomicroscopy, applanation tonometry, gonioscopy, intraocular pressure measurement by applanation tonometer and fundus examination using the 90D indirect lens to evaluate the optic disc. Evaluation of the optic disc was based on Disc Damage Likelihood Scale (DDLS) [7] assessing the neuroretinal rim area, cup-to-disc ratios and/or localized or diffuse retinal nerve fiber layer defects [8]. Automated VF analysis using the 24-2 SITA-standard program was chosen using Humphrey Field Analyzer [HFA II, Zeiss, Dublin, California]. Functional defects of glaucoma eyes were classified based on their HFA mean deviation (MD),as previously mentioned [9], as follows: mild , MD < -6 dB ; moderate , MD > -6 dB < -12 dB ; severe, MD > -12 dB . In the present study glaucoma cases were divided into two groups; group 1 represented mild cases with MD < -6 dB (early glaucoma), and Group 2 that included advanced glaucoma cases with MD > -12 dB. We excluded moderate cases because they were few cases.
Exclusion criteria for all subjects included high refractive errors, ocular disease such as cataract, corneal opacity and retinal disease. Also, cases with any systemic disease and medication that might impair vision or pupil responses were excluded.
Color vision
Color vision testing was carried out in good daylight conditions, supplemented when necessary with artificial lighting. Two color vision tests were used.
Farnsworth-Munsell D-15 test
This hue discrimination arrangement test uses 15 colored caps which are arranged in sequence from a fixed pilot cap. The results are plotted on a hue circle diagram, which allows tritan defects, red-green defects or generally poor hue discrimination to be identified.
The city university color vision (CUCV) test
The test consists of 10 plates in a book. Subjects were instructed to indicate which of the four test colors the closest match to the central color was. Each plate provides a single unique response corresponding to protan, duetran, tritan or normal responses. Number of subjects who were normal or tritans were detected and comparison of the two tests was done.
Stereoacuity (depth perception)
The stereoacuity was measured with the Lang-Stereo test; a random-dot stereo test uses a panographic technique to present disparity. Lang I: Disparity = Car 550", Star 600", Cat 1200" Lang II: Disparity = Moon 200", Car 400", Elephant 600". Subjects were tested under the same condition and they had no previous experience with this type of test, no glasses were required. The test cards were viewed binocularly at 40 cm distance. Patients were asked if they see a picture in the card and to point to its site. The lowest disparity which the patient can reliably discriminate was recorded and this stereothreshold was the measure of stereoacuity .Data were recorded as yes/no ,then scoring was done to facilitate statistical analysis.
Contrast Sensitivity
For each subject contrast sensitivity function (CSF) was determined using the Vistech Contrast Sensitivity Test chart (VCTS Vistech Consultants, Dayton, OH, USA) the chart is model 6000. It is similar to VCTS 6500 chart but is used to test near sensitivity. The chart consists of 5 rows of sine-wave gratings. The rows increase in spatial frequency from the top to bottom of the chart, and on each row the gratings decrease in contrast from left to right; it was viewed at 40 cm at a luminance level of 100 cd/m2. Patients were wearing their best distance correction and were tested binocularly. The lowest contrast grating determined the sensitivity score that used to plot a CSF for the patient. The test was repeated three times for every patient to ensure reproducibility.
Statistical analysis
The Students t-test and analysis of variance (ANOVA) test were used to analyze continuous variables such as age, vision, cup /disc ratio, IOP and MD. The Kruscalwalis test was used to determine differences among dichotomous variables, such as sex and answers to yes /no questions which were treated as dichotomous categorical variables in the analysis. When examining the scores of color and depth perception tests, we used the results for both eyes. The Spearman correlation coefficient was used to determine the relationships within the entire study population between binocular contrast sensitivity, mean deviation (both eyes), and stereopsis. Color vision was compared using wilcoxon signed ranks to compare between the two tests; CUCV and D15. The Kruscalwalis test was used to compare between normal, mild and advanced cases in each test type. Odd ratio and sensitivity and specificity were done using WinPepi statistic software version 11.44. A P-value less than 0.05 was considered statistically significant.
Results
A total of 106 participants, comprising 56 glaucoma patients and 50 non-glaucomatous normal controls, were included in this study. Glaucoma groups comprised 27 men and 29 women, mean age of 46.05 ± 10.89 years. Control cases comprised of 20 men and 30 women, mean age of 43.17 ± 9.46 years.
Mild glaucoma group showed significant difference in cup/disc ratio, as compared to control group (mean of right eyes was 0.45 ± 0.05 and mean of left eyes was 0.46 ± 0.03 (P = 0.001, t test). The mean deviation was 2.45 ± 1.5 and 2.56 ± 1.1 for right and left eyes respectively and it was significantly reduced as compared to the control group (P < 0.001, t test, (Table 1)).
![]()
Table 1: The Characteristics of the Examined Groups.
View Table 1
Advanced glaucoma group showed significant increase of C/D ratio (mean of right eyes was 0.78 ± 0.08 and mean of left eyes was 0.77 ± 0.07 (P = 0.001, t test). There was significant deterioration in visual fields (Right eye MD was 15.93 ± 0.80 and left eyes MD was 13.65 ± 0.72 (P = 0.001, t-test), as compared to the control group (Table 1).
Color vision
Responses to color testing are shown in table 2.
![]()
Table 2: Results of color vision and stereoacuity testing in control, mild glaucoma and advanced glaucoma cases.
View Table 2
CUCV in cases with mild glaucoma showed 30 (96.8%) normal color and one patient with tritan defect. Meanwhile, advanced glaucoma cases showed 17 (68%) normal color vision and 8 (32%) cases with tritan defect. Comparing these results with D15 test, tritan defect was found in 9 (29%) cases of mild glaucoma and 18 (72%) cases of advanced glaucoma. There was significant difference between these two tests in diagnosis of tritan defect (P < 0.001, kruscalwalis test). Mild glaucoma cases Odd ratio in CUCV was 4.97 (95% CI = 0.196:125.83, P = 0.33. Meanwhile, in D15 test odd ratio was 42.64 (95% CI = 2.37:764.97), P = 0.01. Advanced cases in CUCV odd ratio was 49.06 (95% CI = 2.69-894.76, P = 0.008). In D15, The odd ratio was 249.13 (95%CI = 13.55-4581.9, P = 0.0002).When comparing the sensitivity and specificity of both color tests, D15 was found more sensitive (P = 0.001) and more specific (P = 0.03), as compared to CUCV. CUCV sensitivity was 54%, meanwhile D15 sensitivity was 66%. Specificity of CUCV was 85% and specificity of D15 was 65%. Positive test in CUCV was 0.3 (10.6%), and positive test in D15 was 1.0 (28.7%), P = 0.0001. Predective value was higher in D15 test than CUCV, as shown in figure 1.
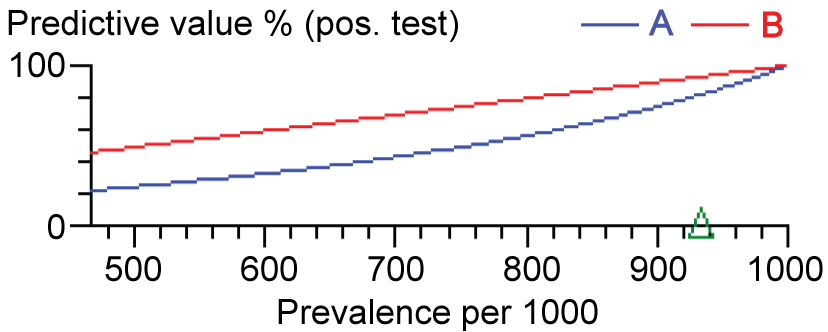
.
Figure 1: The predictive value of a positive test (or positive predictive value) is the post-test probability of the disease if the test is positive. A = CUCV test, B = D15 test.
View Figure 1
Stereoacuity
Patients in mild glaucoma group showed statically significant depth perception defects at all tested disparities [200, 550,600 and 1200 seconds of arc] (p < 0.05) as compared to the controls. The depth perception defects were increased in advanced glaucoma group. There was a statically significant difference between mild and advanced groups (Table 2).
Contrast sensitivity results
The mean values of binocular CSF of controls, mild and advanced glaucoma patients were summarized in table 3. The binocular CSF of mild and advanced glaucoma patients were significantly low at all spatial frequencies, as compared to control subjects. There was significant difference between mild and advanced cases at low and mid spatial frequencies (1.5, 3 and 6c/d), as shown in table 3 and figure 2.
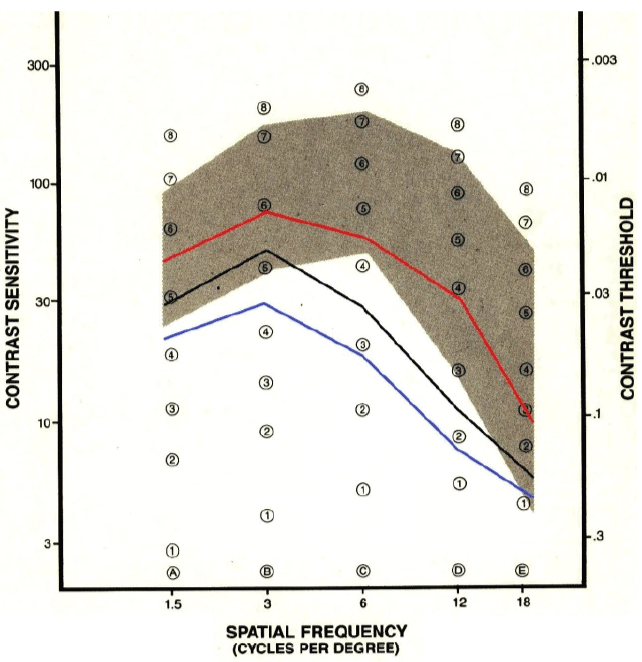
.
Figure 2: CSFcurves (mean values) of control group (the black line), group 1; patients with mild glaucoma (the red line) and group 3; patients with advanced glaucoma (the blue line).
View Figure 2
![]()
Table 3: Contrast sensitivity results in controls, mild glaucoma and advanced glaucoma cases.
View Table 3
There was significant correlation between the deterioration of visual field mean deviation and the reduced depth perception and contrast sensitivity .Also, as the contrast sensitivity was reduced, the depth perception was decreased (Table 4).
![]()
Table 4: Correlations of the studied variables.
View Table 4
Discussion
Glaucoma pathologically leads to the progressive damage of both large (magnocellular) and small ganglion cells (parvocellular and konicellular) retinal ganglion cells [10,11]. Also, glaucoma effects involve the lateral geniculate nucleus (LGN), geniculocortical pathway and visual cortex [4,12]. The LGN has three distinct visual channels, namely the magno-, parvo-, and koniocellular pathways for motion, red- green color and blue-yellow modalities, respectively [10]. The neural mechanisms that binocularly combine the compromised monocular inputs of contrast sensitivity, color and depth perception are integrated at the level of the striate and extrastriate visual cortex [13].
Sensitivity for detection of fine spatial detail, motion and colour signals can be selectively damaged in glaucoma and may precede visual field loss [14,15]. Various tests were used to detect the binocular contrast sensitivity function as well as stereoacuity and color vision deficits in early glaucoma [16]. In this study, we used Vistech contrast sensitivity test chart to explore for binocular contrast sensitivity reductions at different spatial frequencies. We observed deterioration of the contrast sensitivity at all spatial frequencies in early and advanced glaucoma cases. This was in agreement to work done by McKendrick et al. [11]. They observed that glaucoma patients demonstrated reduced sensitivity across the spatial frequency range, for which they suggested a combination of reduction of both magnocellular and parvocellular processing [11].
In the present study, we obtained reduced stereoacuity in both early and advanced glaucoma groups, and this was significantly related to reduce mean deviation in perimetry. Our study confirmed that stereopsis was reduced in glaucoma similar to previous studies that used the random dot [14], the line stereograms [2] and the Frisby depth perception tests [13]. Reche-Sainz JA et al. [17] found that in advanced glaucoma cases depth perception was reduced as examined by Titmus, and TNO depth perception tests, they did not find such changes in early cases with ocular hypertension [17]. The neuronal basis of stereovision depends on disparity cells sensitive to binocular disparity, located in the primary visual cortex and extrastriate areas. The explanation of loss of binocular vision was suggested to be due to relative delay of input from one eye compared to the input from the other eye, which may affect binocular interactions at the level of the visual cortex [12].
In the present study, there was a significant increase in number of tritan patients as detected by D15 test. This suggested that color vision testing is essential for every glaucoma patients. In glaucoma, blue color affection is related to involvement of small bistratified ganglion cells, which are fewer in number and comprise about 1% of ganglion cells and receive their input from the blue-cone bipolar cells [18]. The explanation for tritan defect in POAG was suggested to be due selective damage to blue-yellow sensitive ganglion cells or their axons either due to their larger receptive fields [19], or their relative scarcity [20]. In the present study we did not observe red green chromatic deficits. This is in agreement to the work done by Karwatsky et al. [21]. However, Rauscher et al. [16] described losses of the red-green chromatic mechanism in advanced POAG cases [22].
Many tests are used to detect tritan defect in POAG included H-R-R, Lanthony, F2 plates, D-15, and the City University tests. In general, the CUCV and D15 tests are useful for acquired color vision defects [23]. However, in the present study we observed low sensitivity of the CUCV test and moderate sensitivity of D15 test in glaucoma patients. This is relatively in agreement with previous studies suggested that these tests individually are not considered to be very sensitive for screening POAG. The results from a combination of these tests may be a useful addition to other data collected in glaucoma screening programs [18].
Conclusion
We found that glaucoma patients showed significant reduction in binocular contrast sensitivity scores and depth perception which may have utility in identifying early glaucomatous nerve damage. Also, color vision screening using D15 test may help in discriminating patients with glaucoma. Future use of a simple office test that may combine color, contrast and depth perception in one test may provide an easy way for screening of early glaucomatous damage.
References
-
Lieberman MF, Congdon NG, He M (2011) The value of tests in the diagnosis and management of glaucoma. Am J Ophthalmol 152: 889-899.
-
Bassi CJ, Galanis JC (1991) Binocular visual impairment in glaucoma. Ophthalmology 98: 1406-1411.
-
Sharma P, Sample PA, Zangwill LM, Schuman JS (2008) Diagnostic tools for glaucoma detection and management. Surv Ophthalmol 53: 17-32.
-
Nickells RW, Howell GR, Soto I, John SW (2012) Under pressure: cellular and molecular responses during glaucoma, a common neurodegeneration with axonopathy. Annu Rev Neurosci 35: 153-179.
-
Adams AJ, Rodic R, Husted R, Stamper R (1982) Spectral sensitivity and color discrimination changes in glaucoma and glaucoma-suspect patients. Invest Ophthalmol Vis Sci 23: 516-524.
-
Carle CF, James AC, Kolic M, Essex RW, Maddess T (2014) Luminance and colour variant pupil perimetry in glaucoma. Clin Experiment Ophthalmol 42: 815-824.
-
Bayer A, Harasymowycz P, Henderer JD, Steinmann WG, Spaeth GL (2002) Validity of a new disk grading scale for estimating glaucomatous damage: correlation with visual field damage. Am J Ophthalmol 133: 758-763.
-
Jonas JB, Fernandez MC, Naumann GO (1992) Glaucomatous parapapillary atrophy. Occurrence and correlations. Arch Ophthalmol 110: 214-222.
-
Hodapp E, Parrish R II, Anderson D (1993) Clinical Decisions in Glaucoma. St Louis, MO Mosby-Year Book, Inc, 52-59.
-
Calkins DJ (2012) Critical pathogenic events underlying progression of neurodegeneration in glaucoma. Prog Retin Eye Res 31: 702-719.
-
McKendrick AM, Sampson GP, Walland MJ, Badcock DR (2007) Contrast sensitivity changes due to glaucoma and normal aging: low-spatial-frequency losses in both magnocellular and parvocellular pathways. Invest Ophthalmol Vis Sci 48: 2115-2122.
-
Gupta N, Ang LC, Noel de Tilly L, Bidaisee L, Yucel YH (2006) Human glaucoma and neural degeneration in intracranial optic nerve, lateral geniculate nucleus, and visual cortex. Br J Ophthalmol 90: 674-678.
-
Gupta N, Krishnadev N, Hamstra SJ, Yucel YH (2006) Depth perception deficits in glaucoma suspects. Br J Ophthalmol 90: 979-981.
-
El-Gohary AA, Siam GA (2009) Stereopsis and contrast sensitivity binocular summation in early glaucoma. Research Journal of Medicine and Medical Sciences 4: 85.
-
Banitt MR, Ventura LM, Feuer WJ, Savatovsky E, Luna G, et al. (2013) Progressive loss of retinal ganglion cell function precedes structural loss by several years in glaucoma suspects. Invest Ophthalmol Vis Sci 54: 2346-2352.
-
Rauscher FG, Chisholm CM, Edgar DF, Barbur JL (2013) Assessment of novel binocular colour, motion and contrast tests in glaucoma. Cell Tissue Res 353: 297-310.
-
Reche-Sainz JA, Gomez de Liano R, Toledano-Fernandez N, Garcia-Sanchez J (2013) Binocular vision in glaucoma. Arch Soc Esp Oftalmol 88: 174-178.
-
Pacheco-Cutillas M, Edgar DF, Sahraie A (1999) Acquired colour vision defects in glaucoma-their detection and clinical significance. Br J Ophthalmol 83: 1396-1402.
-
Quigley HA, Sanchez RM, Dunkelberger GR, L'Hernault NL, Baginski TA (1987) Chronic glaucoma selectively damages large optic nerve fibers. Invest Ophthalmol Vis Sci 28: 913-920.
-
Calkins DJ, Tsukamoto Y, Sterling P (1998) Microcircuitry and mosaic of a blue-yellow ganglion cell in the primate retina. J Neurosci 18: 3373-3385.
-
Karwatsky P, Overbury O, Faubert J (2004) Red-green chromatic mechanisms in normal aging and glaucomatous observers. Invest Ophthalmol Vis Sci 45: 2861-2866.
-
O'Neill-Biba M, Sivaprasad S, Rodriguez-Carmona M, Wolf JE, Barbur JL (2010) Loss of chromatic sensitivity in AMD and diabetes: a comparative study. Ophthalmic Physiol Opt 30: 705-716.
-
Heron G, Erskine NA, Farquharson E, Moor AT,White H (1994) Color-vision screening in glaucoma: the tritan album and other simple tests. Ophthalmic Physiol Opt 14: 233-238.





