International Journal of Ophthalmology and Clinical Research
Study of Critical Flicker Fusion (CFF) Function and P100 latency of Visual Evoked Potential (VEP) in Normal subjects and Patients who Recovered from Acute Optic Neuritis
Yanjun Chen1*, Kevin A Buhr2 and James Ver Hoeve1
1Department of Ophthalmology & Visual Sciences, University of Wisconsin, USA
2Department of Biostatistics and Medical Informatics, University of Wisconsin School of Medicine and Public Health, USA
*Corresponding author: Yanjun Chen, M.D, Ph.D, Department of Ophthalmology and Visual Sciences University of Wisconsin School of Medicine and Public Health, 2828 Marshall Ct. Suite 200, Madison, WI 53705, USA, Tel: 608263148, E-mail: ychen344@wisc.edu
Int J Ophthalmol Clin Res, IJOCR-4-067, (Volume 4, Issue 1), Research Article; ISSN: 2378-346X
Received: October 14, 2016 | Accepted: February 25, 2017 | Published: February 28, 2017
Citation: Chen Y, Buhr KA, Hoeve JV (2017) Study of Critical Flicker Fusion (CFF) Function and P100 latency of Visual Evoked Potential (VEP) in Normal subjects and Patients who Recovered from Acute Optic Neuritis. Int J Ophthalmol Clin Res 4:067. 10.23937/2378-346X/1410067
Copyright: © 2017 Chen Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Critical Flicker Fusion (CFF) function has been used to assess visual function in patients with impaired vision. The purpose of this study is to seek further evidence to support the role of the CFF in the assessment of optic nerve function by comparing CFF to implicit time of the P100 of Pattern Reversal Visual Evoked Potential (PRVEP) in normal subjects and patients who recovered from acute optic neuritis.
Methods: 4 healthy adult subjects and 11 patients (14 affected eyes) who recovered from acute optic neuritis underwent PRVEP and CFF, as well as Snellen visual acuity and Ishihara color testing. For normal subjects, neutral density filters (NDF) of 0.6 to 3.0 log unit were used to control stimulus luminance to obtain response vs. stimulus intensity curve for each measurement. The CFF and PRVEP P100 latency were compared between the normal subjects and patients.
Results: In normal subjects, CFF decreased 7.8 Hz (95% CI [7.4, 8.3], p < 0.001) and P100 latency increased 17.9 ms (95% CI [9.7, 26.0], p < 0.001) per log unit increase in NDF. Increasing NDF was also associated with decreasing logMAR acuity and decreasing P100 amplitude (both p < 0.001). In patients recovering from unilateral acute optic neuritis, CFF was significantly lower in affected than unaffected eyes. For all patients, affected eyes had significantly lower CFF than healthy eyes in normal subjects. PRVEP P100 latency did not differ significantly in the affected eyes of patients from the unaffected eyes of patients and normal subjects.
Conclusion: Both CFF and PRVEP P100 latency are linearly correlated with log luminance. CFF is significantly decreased in the affected eyes of patients who recovered from acute optic neuritis. CFF may complement the currently used office tests to facilitate the assessment of optic nerve dysfunction.
Keywords
Critical flicker fusion, Visual evoked potential, P100 latency, Neutral density filter, Optic neuritis
Introduction
When the eye is exposed to rapidly alternating light a sense of flickering is produced. As the frequency of the intermittent light increases to a threshold value, the flickering light becomes indistinguishable from a steady light. The threshold frequency beyond which flickering light transforms to steady light is called critical flicker fusion (CFF) function [1]. Multiple levels of the visual pathway and cortical involvement have been proposed to contribute the physiologic mechanism of CFF, such the retinal photoreceptors and ganglion cells [2-5], primary visual cortex [6], and temporal, parietal and other extra striate cortices [6-9].
Using CFF to assess optic nerve function has been studied in cataract [7-9], age-related macular degeneration [10,11], glaucoma [12-15], and optic neuropathies [12,16-18] with varying results. Study of CFF in cataract found little impact of dense cataract on CFF [8-10]. Studies on multiple sclerosis (MS) associated demyelinating optic neuritis found decrements of CFF in not only the acute stage of visual loss, but also after visual acuity recovered to normal, as well as in those who do not report a history of optic neuritis [19,21-23]. The above results suggest that CFF could be a sensitive measure of optic nerve function.
The purpose of the present study was to compare CFF with PRVEP, a widely accepted electrophysiologic measurement of optic nerve function [19,20], in normal subjects and patients who recovered from acute optic neuritis. In addition, CFF was compared to logMAR acuity and color vision, the two most commonly used visual function testing in clinical practice.
Materials and Methods
Subjects
The study recruited two groups of subjects. Group 1 included 4 healthy adult subjects (2 males and 2 females) ranging in age from 31 to 60 years (mean ± SD, 43.8 ± 12.0). Group 2 included eleven patients (3 males and 8 females, fourteen affected eyes) who recovered from acute optic neuritis, with age ranging from 33 to 66 years (mean ± SD, 48.1 ± 10.3) (Table 1). The exclusion criteria included history of ocular disease other than refractive error, eye trauma or ocular surgery, or concurrent use of ophthalmic solutions other than artificial tears. Those who had a history of ocular disease other than optic neuritis were excluded from the patient group. All healthy subjects had normal eye examination including visual acuity, slit lamp and undilated fundus exam. The procedures conformed to the tenets of the Declaration of Helsinki. The study was approved by the University of Wisconsin-Madison Institutional Review Board. Written consent form was obtained after the details of the tests were explained in detail.
CFF and PRVEP testing
CFF function was measured at a fixed luminance level using a white light flicker fusion device (Flicker Fusion Analyzer, Lafayette Inc., Loughborough, UK). The white electroluminescent lamps produce even illumination over a ½ inch diameter viewing area on a dark background with luminance of 58 cd/m2. The stimulus subtends a visual angle of 1.9 deg in the area of fixation and was viewed separately by each eye through viewing chamber from 15 inch away. CFF threshold was assessed using a standard ascending and descending method. For ascending method, the stimulus was set at 0 Hz, increased by 1 Hz/sec, and the subjects indicated when the stimulus stopped flickering and fused to a uniform, steady light stimulus. For descending method, the stimulus was set at 60 Hz, which is well above the fusion threshold and appears as a uniform, steady light stimulus. The stimulus decreased by 1 Hz/sec, and the subjects indicated when stimulus began flickering. In each trial, the CFF threshold was tested twice by both ascending and descending method; the average between ascending and descending threshold of the two repeats was taken as the CFF threshold for that trial [19].
PRVEP recordings were made in accordance with the respective ISCEV standards [20,21]. The PRVEP was recorded from gold cup electrodes place at O1, Oz, and O2 sites, with the reference electrode placed at Fz and ground at Cz. The PERG was recorded from DTL fiber electrodes [22] draped over the inferior fornix and referenced to a gold cup electrode near the ipsilateral outer canthus. Amplification gain was 20 K with a bandpass filter of 1 to 100 Hz. The stimuli were high contrast (> 90%) checkerboard patterns. Stimuli were displayed on a customized LED display of the E3 electrodiagnostic system (Diagnosys, LLC, Lowell, MA). The luminance of the white squares was 100 cd/m2; the dark squares were < 0.1 cd/m2. Check sizes subtended 30 min arc and the reversal rate was at 2 Hz. The display monitor is 81 cm wide × 65 cm high and placed 1.5 meters away from subjects, subtending a visual angle of 30.2 by 24.8 deg. The average of 100 sweeps was obtained for each trial. During the visit, two immediate PRVEPs were recorded in normal subjects and one in patients.
For CFF and PRVEP testing, the subjects were dark adapted for 15 min before recording began.
Testing protocol
For group 1 normal subjects, Snellen visual acuity, color vision (Ishihara plate) and CFF were measured in the left and right eye in routine eye exam room illuminated by ceiling light. Snellen visual acuity was transformed to logarithm of the minimum angle of resolution (logMAR) for further analysis. PRVEP were conducted in both the left and right eye in two subjects, and in the right eye for the other two. The PRVEP recordings were similar between the left and right eye for a given subject; the PRVEP recordings from the right eye from each of the four normal subjects were used for the analysis. During each test, a series of neutral density filter (NDF) of 0.6, 1.2, 1.8, 2.4 and 3.0 log unit were placed over the tested eye to modulate the luminance level of the stimulus, with higher NDF producing greater reduction in stimulus luminance. For each test, the sequence of the neutral density filter was first randomized using a random number generator, followed by randomization of the eye tested and the sequence of ascending/descending method (for CFF). The sequence of neutral density filter and the order of eye tested were counterbalanced to minimize any effect of sequence (e.g., fatigue or practice effect).
For group 2 patients, Snellen visual acuity, color vision (Ishihara plate) and CFF were measured in the left and right eye in routine eye exam room illuminated by ceiling light. PRVEP were recorded in the left and right eye without neutral density filter. The sequence of the eye tested as well as ascending/descending method for CFF was randomized using a random number generator for each measurement.
Statistics
For group 1 normal subjects, intraclass correlation coefficients (ICC) were calculated for CFF threshold, logMAR acuity, and Ishihara color vision. ICCs were calculated using an ICC (1,1) model in the sense of Shrout, et al. for classes defined by subject and NDF level within subject (evaluating the correlation across eyes and repeats within eyes) [23] Based on these results, all further analyses used data from normal subjects' right eyes averaging over repeats.
CFF, logMAR acuity, Ishihara color plate testing, and PRVEP P100 latency and amplitude measurements were plotted against NDF by subject. Means and 95% confidence intervals (CIs) for CFF and PRVEP P100 latency at baseline (NDF = 0) were calculated, and ANCOVA models (with a single fixed subject effect and a continuous NDF covariate) were used to evaluate the relationships between NDF and each of CFF, logMAR acuity, and P100 latency and amplitude with point estimates and 95% CIs given for selected coefficients. The relationship between NDF and Ishihara scores was summarized descriptively.
Statistical analysis was performed with R version 3.3.0. Testing was conducted at an α = 0.05 significance level. For group 2 patients, critical flicker fusion and PRVEP P100 latency were plotted for unaffected and affected eyes, with results for normal (group 1) subjects at NDF = 0 provided for comparison. Two-group t-tests were used to compare CFF and P100 latency values between right eyes in normal subjects (at NDF = 0) with unaffected eyes in unilaterally affected patients and with all affected eyes (averaging for bilaterally affected patients). For unilateral patients, paired t-tests were used to compare CFF and P100 latency values between affected and unaffected eyes. Mean differences and 95% CIs were summarized.
CFF and P100 latency were plotted for normal eyes across NDF values ranging from 0 to 2.4, as well as for affected and unaffected eyes in patients. For normal eyes, a multivariable regression of the CFF and P100 latency dependent variables against the independent NDF variable was performed to construct a regression line relating CFF and P100 latency for normal eyes under varying NDF. This regression was used to construct 95% and 99% profile prediction bands (i.e., prediction bands for the CFF/latency relationship profiled across possible NDF values) to serve as a reference range for the CFF/latency relationship in normal eyes subject to varying NDF conditions. Affected and unaffected eyes (n = 22) in patients were compared against this model using a profile Z-test against the prediction distribution, using a Bonferroni corrected α = 0.05/22 = 0.002 adjusted for n = 22 patient eyes.
Results
Part 1. Normal subjects
CFF, Ishihara color plate and logMAR acuity vs. log stimulus luminance: Across classes defined by subject and NDF filter, the intraclass correlation for CFF, logMAR, and Ishihara measurements on left and right eyes and repeat measurements on the same eye was high (ICC = 0.994, 0.982, and 0.986 for CFF, logMAR, and Ishihara respective). For this reason and for simplicity of presentation, measurements for normal subjects' right eyes averaged across repeats were used for all subsequent analysis.
Figure 1 shows the CFF, logMAR acuity and Ishihara color plate testing measurements by NDF for four normal subjects. At baseline (NDF = 0), the mean CFF was 34.5 Hz (95% CI [29.5, 39.5]). An ANCOVA analysis of CFF versus NDF indicated no evidence of different slope by subject (p = 0.19) with evidence of different per-subject intercepts (p < 0.001); the shared slope of the fitted model represented a 7.8 Hz decrease (95% CI [7.4, 8.3], p < 0.001) per log unit increase in NDF. The logMAR acuity increased with increasing NDF (p < 0.001), with minimal increase between NDF = 0.0 and 1.2 and rapid increase for NDF > 1.2. For the Ishihara color plate test, three of four subjects experienced no decrease in score except at NDF = 3.0. The fourth subject experiences a decrease for NDF > 1.2.
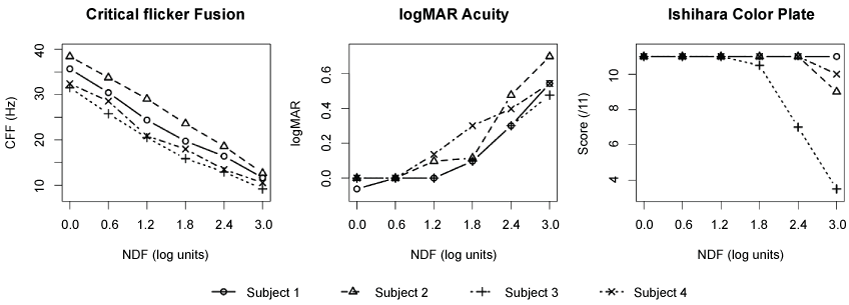
.
Figure 1: Measurements of CFF, logMAR acuity and Ishihara color plate by NDF observed for each of four subjects (average of both repeats for right eye). The CFF vs. log stimulus luminance function (left) demonstrated a linear correlation, showing a 7.8 Hz decrease (95% CI [7.4, 8.3], p < 0.001) per log unit increase in NDF. Color vision was least affected by changes in log stimulus luminance (right), and logMAR acuity increased minimally as NDF increased up to 1.2 log, above which acuity increased precipitously (middle).
View Figure 1
PRVEP P100 latency and amplitude vs. log stimulus luminance: Figure 2 shows PRVEP recordings for a particular subject, indicating the effect of decreasing stimulus luminance/increasing NDF on P100 latency and amplitude. Increasing NDF caused progressive delay of arrival of P100, i.e., prolongation of the P100 latency, as marked by the dashed line drawing through the peak of the P100 waves, as well as decrease in resulting P100 response amplitude. At the highest NDF of 3.0 log (dimmest stimulus), the P100 response is often difficult to discern. For this reason, PRVEP response at NDF = 3.0 was excluded from subsequent analysis.
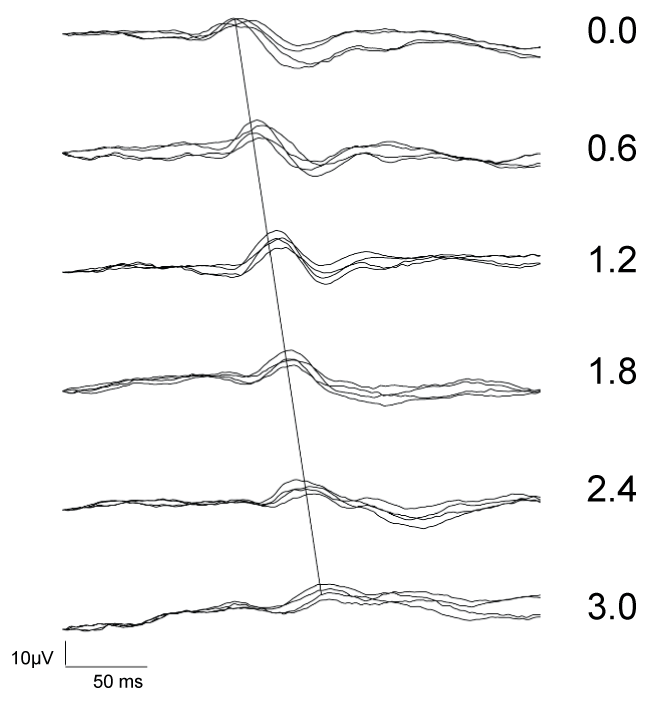
.
Figure 2: Examples of PRVEP recording to 30 min arc checkerboard stimulus in S1 (right eye). Four repeats of PRVEP were overlapped for log stimulus intensity produced by NDF (marked in numbers to the right of each recording). P100 response is demonstrated as distinct positive deflection. Gradual prolongation of the P100 latency with increasing NDF is marked by the line traversing the P100 responses.
View Figure 2
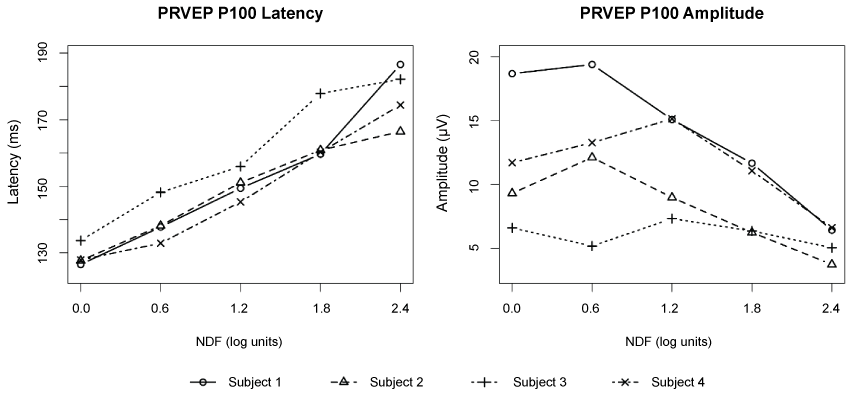
.
Figure 3: Plots of PRVEP P100 latency and amplitude as function of log stimulus luminance (NDF) for four subjects. The PRVEP were recorded in the right eye for each subject with two repeats. The averages of the repeats are shown here. Data for 3.0 log NDF are excluded. The PRVEP P100 latency vs. NDF function demonstrated a linear correlation, with increasing NDF associated with increasing P100 latency (p < 0.001) (left). Similarly, a decrease in P100 amplitude was associated with increasing NDF (p < 0.001), although individual subjects had markedly different trajectories (right).
View Figure 3
For PRVEP P100 latency, the average baseline (NDF = 0) value was 129 ms (95% CI [124, 134]). Increasing NDF was associated with increasing P100 latency (p < 0.001) (Figure 3). An ANCOVA analysis indicates a mild trend of different slope by subject (p = 0.04) with an average fitted slope representing a 17.9 ms increase in latency (95% CI [9.7, 26.0]) for each log unit increase in NDF. Similarly, an ANCOVA analysis indicated a decrease in P100 amplitude with increasing NDF (p < 0.001), although individual subjects had markedly different trajectories.
Patients
The patient group included 14 affected and 8 unaffected eyes from 11 patients (3 affected bilaterally, and 8 affected unilaterally) who recovered from acute optic neuritis (Table 1). Patients were diagnosed with acute optic neuritis based on the history of acute visual loss, normal fundus examination at the presentation, and improvement of vision after steroid therapy. Nine of eleven patients had contrast enhancement of the affected optic nerve on orbital magnetic resonance imaging (MRI). The MRI is not available in one patient and did not reveal contrast enhancement of the affected optic nerve in another. Multiple sclerosis (MS) was diagnosed in four and idiopathic optic neuritis in six patients. The diagnosis of MS was made by neurologists shortly after the onset of acute optic neuritis and none of the patients was diagnosed in later follow-up. Testing to rule out other systemic inflammatory and infectious etiology that may cause optic neuritis, such as neuromyelitis optica (NMO) antibody, antinuclear antibody (ANA), rheumatoid factor (RF), angiotensin converting enzyme (ACE), rapid plasma reagin (RPR), Lyme disease antibodies, as well as cerebrospinal fluid analysis, were pursued at the discretion of neurologists based on clinical suspicions. NMO antibody was tested in 6 of 11 patients, all negative. One patient had a known diagnosis of follicular lymphoma but with no cancer cells were found in the spinal fluid analysis. All patients were treated with systemic steroids due to moderate to severe visual decline based on visual acuity, color vision, or visual field assessment. At the presentation of acute optic neuritis, the Snellen visual acuity ranged from 20/20 to light perception with median of 20/60. At the time of the study, the Snellen visual acuity ranged from 20/20 to 20/800 with median of 20/20. The mean interval between the initial presentation and the time of the study is 34.2 months (3 to 204 months). The patients also underwent automated perimetry testings (Humphrey Field Analyzer using the SITA-standard 24-2 or 30-2 protocol [Humphrey visual field (HVF)], Carl Zeiss Meditec, Dublin, CA) at the time of presentation and within one year of the study as part of their clinical assessment. The mean deviation (MD) ranged from -33.86 to -9.71 dB (mean ± SD: -24.47 ± 8.21) at presentation and from -20.68 to -0.79 dB (mean ± SD: -7.60 ± 6.50) at the time of the study.
![]()
Table 1: The demographic and clinical features of the patients group.
View Table 1
Figure 4 shows CFF (left) and PRVEP P100 latency (right) for normal right eyes for the four healthy subjects, unaffected eyes from the unilaterally affected patients, and affected eyes from the unilaterally and bilaterally affected patients, all assessed at NDF = 0. There was no significant difference in CFF (p = 0.33) or PRVEP P100 latency (p = 0.38) between eyes of healthy subjects and unaffected eyes of (unilaterally affected) patients. Patients' affected eyes had significantly lower CFF than healthy eyes (p = 0.002), but there was no observed P100 latency difference (p = 0.17). Within unilaterally affected patients, the affected eye had CFF an average 7.2 Hz lower (95% CI [0.1, 14.3]) than the unaffected eye (p = 0.05) but with no evidence of difference in P100 latency (p = 0.14).
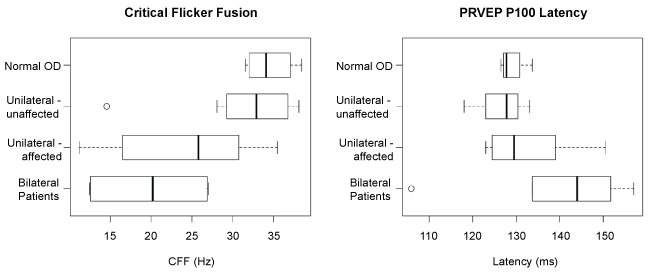
.
Figure 4: Box-and-Whisker plot of CFF (left) and P100 latency (right) in normal subjects, unaffected eyes of unilaterally affected patients, and affected eyes in unilaterally and bilaterally affected patients, all assessed at NDF = 0. The median value, 1st and 3rd quartiles of the CFF and P100 latency are shown in box and the ranges are shown in Whisker plot. The extreme values are shown in open circles. There was no significant difference in CFF (p = 0.33) or PRVEP P100 latency (p = 0.38) between eyes of healthy subjects and unaffected eyes of (unilaterally affected) patients. Patients' affected eyes had significantly lower CFF than healthy eyes (p = 0.002), but there was no observed P100 latency difference (p = 0.17).
View Figure 4
Figure 5 shows a plot of CFF versus P100 latency for normal subjects (black squares) at varying NDF levels from 0 to 2.4, together with affected patient eyes (crosses for non-MS patients, triangles for MS patients) assessed at NDF = 0. A multivariable regression line (of both dependent variables against NDF) and associated 95% and 99% profile prediction bands calculated using data from normal subjects only (n = 20 eyes) show the expected relationship between CFF and P100 latency for normal eyes. Note that, in normal eyes, CFF is inversely correlated with P100 latency due to modulation by NDF, with the area of high CFF and low P100 latency corresponding to 0 NDF. In contrast, five of fourteen affected patient eyes exhibit a combination of low CFF with relatively low latency that falls outside the 99% profile prediction bands of normal eyes at varying NDF levels. These five affected patient eyes exhibit a pattern that is significantly different from that exhibited by normal eyes (p < 0.001 for each of the five eyes, tested against a Bonferroni α = 0.05/22 = 0.002 adjusted for n = 22 patient eyes including both affected and unaffected eyes to be maximally conservative).
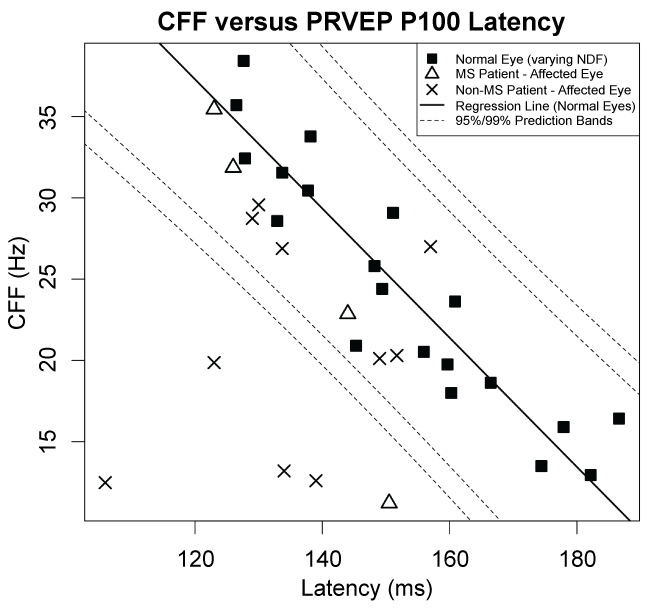
.
Figure 5: Distribution of CFF vs. PRVEP P100 latency in normal subjects and affected patient eyes. For normal subjects, the CFF and PRVEP P100 latency for the right eye (average of two repeats) are plotted for all NDF. A regression line and 95% and 99% prediction bands are shown, based on a multivariable regression of both dependent variables against NDF using data from normal subjects only. For patients, CFF and latency for affected eyes (average of two repeats) are shown using different symbols for MS and non-MS patients. Five of the fourteen affected patient eyes exhibit a pattern that is significantly different (p < 0.001) than the prediction band fit to normal eyes.
View Figure 5
Discussion
In this study, we measured CFF in normal subjects and patients who recovered from acute optic neuritis, and compared CFF with PRVEP to explore the relation between the two measurements. To our knowledge, this is the first study to directly compare CFF and PRVEP in such a clinical setting. The study revealed several findings:
First, both CFF and PRVEP P100 latency are linearly correlated with log stimulus luminance in normal subjects (Figure 1, left and Figure 3, left). In contrast, logMAR acuity and Ishihara color test are less affected by reduction of log stimulus luminance, evidenced by the findings that the NDF of less than 1.2 log had minimal influence on logMAR acuity, and Ishihara color function only declined at high NDF level (Figure 1, middle and right), suggesting its greatest resistance to reduction in stimulus luminance. CFF uses high intensity white light stimuli oscillating at a high temporal frequency which recruits exclusively cone response and foveal projection to the occipital cortex [24]. Similarly, the stimulus used for PRVEP in this study likely also activates cone response given high stimulus intensity and a high luminance background which suppresses rod contribution to the VEP response. Studies have shown that reduced retinal illumination causes delay in the cone ERG response. It is therefore possible that the linear correlation of CFF and PRVEP P100 latency with stimulus luminance is a result of a common dependence on the cone response in the retina [25,26]. In this study, we found a significant correlation between both the latency and amplitude of the PRVEP P100 response with log stimulus luminance, although the response vs. stimulus intensity function appears more homogeneous for P100 latency than amplitude. The finding supports the results from other studies that PRVEP P100 latency is a more sensitive measure than amplitude for optic nerve dysfunction in optic neuritis [27,28].
Second, CFF appears to linearly correlate with PRVEP P100 latency in normal subjects exposed to varied stimulus luminance (Figure 5). The CFF is a psychophysiological measurement of temporal property of perception at which a flickering spot of light appears steady, whereas PRVEP is an electrophysiological measurement of the latency of a cortical electrical field. As mentioned before, the possible mechanism for the correlation between these two fundamentally different measurements likely relies on the shared anatomical substrate for CFF and PRVEP pathway, the cones. However, the response-stimulus intensity curve for CFF and PRVEP P100 latency should also receive contribution from the visual pathway beyond the level of cones, otherwise one would expect similar slope between the CFF, P100 latency and the cone response-stimulus intensity curve. The exact mechanism underlying the stimulus-intensity function for CFF requires further investigation. Nevertheless, the strong linear correlation between CFF and log stimulus luminance as well as CFF and PRVEP P100 latency supports the role of CFF, as a supplement for VEP, in the assessment of optic nerve function.
Furthermore, when CFF and VEP are recorded with zero NDF, i.e., no diminish in stimulus luminance, the CFF vs. PRVEP P100 latency distribution in at least some affected eyes of patients appears to follow the linear CFF-PRVEP P100 latency curve observed in normal subjects, whereas others deviate from that of normal subjects (Figure 5). The inhomogeneity of the CFF vs. PRVEP P100 latency distribution among the study patients could potentially be due to varying pathophysiologic process underlying their visual loss (e.g., demyelinating vs. axonal injury), even though they seemed to follow a similar clinical course. Further research is needed to show how CFF vs. P100 latency changes in different types of optic neuropathy to elucidate such a speculation. One would further speculate that an optic nerve that recovered from previous optic neuritis may have more impaired ability to differentiate temporal than spatial property of the light stimulation embedded in the CFF and VEP stimulus, given that the majority of the affected eyes of patients demonstrated relatively good Snellen acuity and visual field function, and yet significantly decreased CFF and prolonged PRVEP P100 latency at the time of the study (Table 1).
Lastly, CFF appears to be more sensitive in identifying previous optic nerve injury in this group of patients who recovered from acute optic neuritis. The CFF is significant lower in the affected eyes than the unaffected eyes of patients and normal subjects (Figure 4, left), even though the visual acuity recovered to near normal in most patients. It is somewhat unexpected that the PRVEP P100 latency was not significantly different in the affected eyes of patients from that in the unaffected eyes of patients or normal subjects (Figure 4, right), since VEP is generally regarded as a sensitive neurophysiologic testing for optic nerve function [29]. These findings suggest that CFF may add to low-contrast visual acuity testing as a valuable tool to measure persistent optic nerve dysfunction after recovery of acute optic neuritis [30]. Compared to PRVEP, CFF is faster to administer, less demanding on testing environment and technical support, and more cost-effective.
One must be aware that, unlike the PRVEP, CFF is a subjective test and could thus be influenced by subject's attention and cognition. However, CFF change contributed by age, fatigue, sleep deprivation, drug use (antidepressants or sedatives), and cognition (Alzheimer's, Parkinson's or hepatic encephalopathy) [31-37] appears small and is usually on a scale of several Hz [31,32,35,38-48], compared to a much greater change of CFF observed in diseases of the optic nerve [49,50] and to the large drop of CFF (7.8 Hz) per log unit decrement in stimulus intensity demonstrated in this study. Furthermore, one would expect that CFF change from attention and cognition should be more symmetrical between the two eyes; the attention and cognitive factor should thus have little impact on CFF especially when unilateral optic nerve dysfunction is concerned.
We acknowledge several limitations of the study. (1) In this retrospective study, the patients underwent a varying collection of clinical testing for systemic inflammatory and infectious etiologies contributing to optic neuritis, resulting in a heterogeneous group contained in "idiopathic optic neuropathy". However, the differentiation between MS and "idiopathic" optic neuritis is felt to be fairly distinct as the diagnosis of MS was established by MS neurologists using a standard criterion, (2) The normal control group only included four subjects. However, the visual function testing (visual acuity, color vision, CFF, and VEP) among the four normal subjects demonstrated a strong consistency (Figure 1, Figure 3 and Figure 4), and (3) the longitudinal measurements of CFF among patients during their recovery of visual function are not available. Such information will help assess the value of CFF in monitoring optic nerve function and should be evaluated in future study.
Conclusion
Both CFF and PRVEP P100 latency are linearly correlated with log luminance. CFF is significantly decreased in the affected eyes of the patients who recovered from acute optic neuritis. CFF may complement the existing technology for the evaluation of optic nerve dysfunction. The clinical utility of CFF in assessing optic nerve function will be explored in future studies comparing the sensitivity of CFF and PRVEP P100 in patients with different types of optic nerve disease.
Acknowledgement
This project was supported in part by an unrestricted research grant to the Department of Ophthalmology and Visual Sciences at the University of Wisconsin from Research to Prevent Blindness, Inc.
Conflict of Interest Statement
No interest to disclose.
Ethical Statement
The procedures conformed to the tenets of the Declaration of Helsinki.
References
-
Hecht S, Shlaer S, Verrijp CD (1933) Intermittent Stimulation By Light : Ii. The Measurement Of Critical Fusion Frequency For The Human Eye. J Gen Physiol 17: 237-249.
-
Foley PJ (1961) Interrelationships of background area, target area, and target luminance in their effect on the critical flicker frequency of the human fovea. J Opt Soc Am 51: 737-740
-
Giorgi A (1963) Effect of wavelength on the relationship between critical flicker frequency and intensity in foveal vision. J Opt Soc Am 53: 480-486.
-
Harvey LO Jr (1970) Critical flicker frequency as a function of viewing distance, stimulus size and luminance. Vision Res 10: 55-63.
-
Raninen A, Franssila R, Rovamo J (1991) Critical flicker frequency to red targets as a function of luminance and flux across the human visual field. Vision Res 31: 1875-1881.
-
Wells EF, Bernstein GM, Scott BW, Bennett PJ, Mendelson JR (2001) Critical flicker frequency responses in visual cortex. Exp Brain Res 139: 106-110.
-
Vianya-Estopà M, Douthwaite WA, Pesudovs K, Noble BA, Elliott DB (2004) Development of a critical flicker/fusion frequency test for potential vision testing in media opacities. Optom Vis Sci 81: 905-910.
-
del Romo GB, Douthwaite WA, Elliott DB (2005) Critical flicker frequency as a potential vision technique in the presence of cataracts. Invest Ophthalmol Vis Sci 46: 1107-1112.
-
Douthwaite WA, Vianya-Estopà M, Elliott DB (2007) Predictions of postoperative visual outcome in subjects with cataract: a preoperative and postoperative study. Br J Ophthalmol 91: 638-643.
-
Vianya-Estopà M, Douthwaite WA, Noble BA, Elliott DB (2006) Capabilities of potential vision test measurements: clinical evaluation in the presence of cataract or macular disease. J Cataract Refract Surg 32: 1151-1160.
-
Maier M, Groneberg T, Specht H, Lohmann CP (2010) Critical flicker-fusion frequency in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 248: 409-413.
-
Tyler CW (1981) Specific deficits of flicker sensitivity in glaucoma and ocular hypertension. Invest Ophthalmol Vis Sci 20: 204-212.
-
Van Toi V, Grounauer PA, Burckhardt CW (1990) Artificially increasing intraocular pressure causes flicker sensitivity losses. Invest Ophthalmol Vis Sci 31: 1567-1574.
-
Yoshiyama KK, Johnson CA (1997) Which method of flicker perimetry is most effective for detection of glaucomatous visual field loss? Invest Ophthalmol Vis Sci 38: 2270-2277.
-
Rota-Bartelink A (1999) The diagnostic value of automated flicker threshold perimetry. Curr Opin Ophthalmol 10: 135-139.
-
Al Khamis AR, Easterbrook M (1983) Critical flicker fusion frequency in early chloroquine retinopathy. Can J Ophthalmol 18: 217-219.
-
Galvin RJ, Regan D, Heron JR (1976) Impaired temporal resolution of vision after acute retrobulbar neuritis. Brain 99: 255-268.
-
Okamoto Y, Okamoto F, Yamada S, Honda M, Hiraoka T, et al. (2010) Vision-related quality of life after transsphenoidal surgery for pituitary adenoma. Invest Ophthalmol Vis Sci 51: 3405-3410.
-
Landis C (1954) Determinants of the critical flicker-fusion threshold. Physiol Rev 34: 259-286.
-
Bach M, Brigell MG, Hawlina M, Holder GE, Johnson MA, et al. (2013) ISCEV standard for clinical pattern electroretinography (PERG): 2012 update. Doc Ophthalmol 126: 1-7.
-
Odom JV, Bach M, Brigell M, Holder GE, McCulloch DL, et al. (2010) ISCEV standard for clinical visual evoked potentials (2009 update). Doc Ophthalmol 120: 111-119.
-
Dawson WW, Trick GL, Litzkow CA (1979) Improved electrode for electroretinography. Invest Ophthalmol Vis Sci 18: 988-991.
-
Shrout PE, Fleiss JL (1979) Intraclass correlations: uses in assessing rater reliability. Psychol Bull 86: 420-428.
-
Shpak AA (1992) Critical frequency of photic driving: which part of the visual pathway do we examine? Acta Ophthalmol (Copenh) 70: 587-592.
-
Hecht S, Smith EL (1936) intermittent stimulation by light : vi. area and the relation between critical frequency and intensity. J Gen Physiol 19: 979-989.
-
Ooba Y, Suzuki E (1956) Critical fusion frequency of flicker and the electrical excitability of the retina. Tohoku J Exp Med 63: 389-397.
-
Brusa A, Jones SJ, Plant GT (2001) Long-term remyelination after optic neuritis: A 2-year visual evoked potential and psychophysical serial study. Brain 124: 468-479.
-
Esen E, Sizmaz S, Balal M, Yar K, Demirkiran M, et al. (2016) Evaluation of the Innermost Retinal Layers and Visual Evoked Potentials in Patients with Multiple Sclerosis. Curr Eye Res 41: 1353-1358.
-
Bodis-Wollner Ivam, Andrea Antal (2006) Optic nerve and central nervous dysfunctions: Parkinson's disease and multiple sclerosis. In: Heckenlively John R, Arden Geoffrey B, Principles and Practice of Clinical Electrophysiology of Vision. Cambridge, The MIT Press, Massachusetts, 977.
-
Galetta SL, Villoslada P, Levin N, Shindler K, Ishikawa H, et al. (2015) Acute optic neuritis: Unmet clinical needs and model for new therapies. Neurol Neuroimmunol Neuroinflamm 2: e135.
-
Akos K, Akos M (1970) Age changes in men of critical flicker frequency series effect (CFFSE). Gerontologia 16: 41-47.
-
Ben'kovich B, Marshak OV (1984) Effect of tranquilizers on critical flicker fusion frequency in the visual system. Hum Physiol 10: 435-439.
-
Cronin-Golomb A, Corkin S, Rizzo JF, Cohen J, Growdon JH, et al. (1991) Visual dysfunction in Alzheimer's disease: relation to normal aging. Ann Neurol 29: 41-52.
-
Luczak A, Sobolewski A (2005) Longitudinal changes in critical flicker fusion frequency: an indicator of human workload. Ergonomics 48: 1770-1792.
-
Riklan M, Levita E, Misiak H (1970) Critical flicker frequency and integrative functions in parkinsonism. J Psychol 75: 45-51.
-
Smith JM, Misiak H (1976) Critical flicker frequency (CFF) and psychotropic drugs in normal human subjects-a review. Psychopharmacologia 47: 175-182.
-
Torlot FJ, McPhail MJ, Taylor-Robinson SD (2013) Meta-analysis: The diagnostic accuracy of critical flicker frequency in minimal hepatic encephalopathy. Aliment Pharmacol Ther 37: 527-536.
-
Azcona O, Barbanoj MJ, Torrent J, Janè F (1995) Evaluation of the central effects of alcohol and caffeine interaction. Br J Clin Pharmacol 40: 393-400.
-
Chang TT, Ciuffreda KJ, Kapoor N (2007) Critical flicker frequency and related symptoms in mild traumatic brain injury. Brain Inj 21: 1055-1062.
-
Hemelryck W, Rozloznik M, Germonpré P, Balestra C, Lafère P (2013) Functional comparison between critical flicker fusion frequency and simple cognitive tests in subjects breathing air or oxygen in normobaria. Diving Hyperb Med 43: 138-142.
-
Jansen AA, de Gier JJ, Slangen JL (1986) Diazepam-induced changes in signal detection performance. A comparison with the effects on the Critical Flicker-Fusion Frequency and the Digit Symbol Substitution Test. Neuropsychobiology 16: 193-197.
-
Kircheis G, Bode JG, Hilger N, Kramer T, Schnitzler A, et al. (2009) Diagnostic and prognostic values of critical flicker frequency determination as new diagnostic tool for objective HE evaluation in patients undergoing TIPS implantation. Eur J Gastroenterol Hepatol 21: 1383-1394.
-
Landis C, Hamwi V (1954) The effect of certain physiological determinants on the flicker-fusion threshold. J Appl Physiol 6: 566-572.
-
Lind NA, Turner P (1968) The effect of chlordiazepoxide and fluphenazine on critical flicker frequency. J Pharm Pharmacol 20: 804.
-
MacNab MW, Foltz EL, Sweitzer J (1985) Evaluation of signal detection theory on the effects of psychotropic drugs on critical flicker-fusion frequency in normal subjects. Psychopharmacology (Berl) 85: 431-435.
-
Maeda E, Yoshikawa T, Hayashi N, Akai H, Hanaoka S, et al. (2011) Radiology reading-caused fatigue and measurement of eye strain with critical flicker fusion frequency. Jpn J Radiol 29: 483-487.
-
Schnider P, Maly J, Grunberger J, Aull S, Zeiler K, et al. (1995) Improvement of decreased critical flicker frequency (CFF) in headache patients with drug abuse after successful withdrawal. Headache 35: 269-272.
-
Sundqvist A, Nilsson BY, Tomson T (1999) Valproate monotherapy in juvenile myoclonic epilepsy: dose-related effects on electroencephalographic and other neurophysiologic tests. Ther Drug Monit 21: 91-96.
-
Salmi T (1985) Critical flicker frequencies in MS patients with normal or abnormal pattern VEP. Acta Neurol Scand 71: 354-358.
-
Sandry M (1963) Critical flicker frequency in multiple sclerosis. Percept Mot Skills 16: 103-108.





