International Journal of Pathology and Clinical Research
Ovarian Neoplasm in a Patient with Prior Breast Carcinoma - Challenges and Pitfalls in Diagnostic Process
Elena Arabadzhieva1*, Sasho Bonev1, Lyubomira Dimitrova2, Atanas Yonkov1, Dimitar Bulanov1, Vesela Ivanova2 and Violeta Dimitrova1
1Department of General and Hepato-Pancreatic Surgery, University Hospital "Alexandrovska" - Sofia, Medical University, Bulgaria
2Department of General and Clinical Pathology, Medical University - Sofia, Bulgaria
*Corresponding author:
Elena Arabadzhieva, Department of General and Hepato-Pancreatic Surgery, University Hospital "Alexandrovska" - Sofia, Medical University, Sofia, Bulgaria, Tel: +359886376071, Fax: +35929230307, E-mail: elena_arabadjieva@abv.bg
Int J Pathol Clin Res, IJPCR-1-008, (Volume 1, Issue 1), Case Report; ISSN: 2469-5807
Received: July 21, 2015 | Accepted: August 30, 2015 | Published: September 02, 2015
Citation: Arabadzhieva E, Bonev S, Dimitrova L, Yonkov A, Bulanov D, et al. (2015) Ovarian Neoplasm in a Patient with Prior Breast Carcinoma - Challenges and Pitfalls in Diagnostic Process. Int J Pathol Clin Res 1:008. 10.23937/2469-5807/1510008
Copyright: © 2015 Arabadzhieva E, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Metastatic lesions account for approximately 8% of all ovarian neoplasms. About 12% of clinically apparent metastases from non-gynecological primary site originate from breast carcinomas with a variable period of manifestation and detection.
Report of Case: We present a case of 58-year-old woman who underwent bilateral adnexectomy with small bowel resection during urgent operation for an incarcerated umbilical hernia. There was a slight enlargement in both ovaries and presence of cystic formation with some papillary structures in the right one. Histologically, bilateral infiltration of tumor cells with moderate cytological and nuclear atypia and tubular, nested and cystic growth pattern was observed. The patient had a long list of co-morbidities: liver cirrhosis, ascites, isolated cytology with presence of tumor cells, elevated level of CA-125 and quadrantectomy of the right breast, performed 15 years ago. Diagnostic examination continued in differing primary ovarian carcinoma from metastatic process using immunohistochemical markers - Gross cystic disease fluid protein-15 (GCDFP-15), Mammaglobin, Wilms tumor protein (WT1), p53, Ki67 and hormone receptors for estrogen (ER), progesterone (PR) and human epidermal growth factor (HER2). Considering the clinical data and the results from the pathological examination we concluded that the patient had bilateral ovarian metastases from breast carcinoma. We share our stepwise approach to the diagnosis discussing some of the difficulties emerged in the process and some of the pitfalls reaching the final conclusion.
Keywords
Ovarian neoplasm, Breast carcinoma, Metastases, Immunohistochemistry
Introduction
Tumors located in the ovaries include very diverse groups of neoplasms. In addition to the immense spectrum of primary lesions, almost 8% of all tumor masses in the ovaries are metastatic. The most common sites of origin are gastrointestinal tract (stomach and colon), pancreas and biliary tract, as well as breast [1]. Metastatic lesions in the ovaries are detected in 10-20% of the autopsies and almost 30% of the ovariectomy specimens of patients with breast carcinoma [2,3]. The interval between the detection of the adnexal masses and the initial breast cancer is wide-ranging- from 9 months to 20 years with median of 5 years [4]. These metastases tend to mimic primary ovarian neoplasms in many aspects - clinical, histopathological and immunohistochemical - which sometimes makes their recognition challenging.
Case report
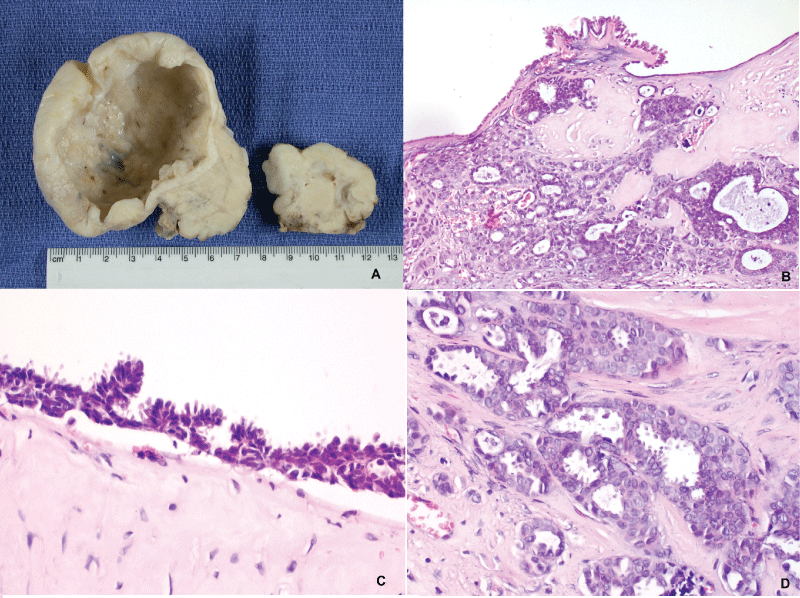
We present a case of a 58-year-old woman who underwent an urgent laparotomy because of an incarcerated umbilical hernia. During the operation, a resection of a necrotic segment of the small bowel was performed as well as a bilateral adnexectomy due to observed changes in both ovaries. Abdominal exploration revealed liver cirrhosis, ascites and disseminated nodular lesions on the peritoneal surface compatible with peritoneal carcinomatosis. Both ovaries were slightly enlarged with firm consistency and white-tan nodular surface. In the right one, there was a thin-walled cystic formation measuring 8 cm in diameter with focal papillary proliferations on the inside (Figure 1A).
.
Figure 1: Ovaries - specimen; B, C, D: Ovaries - histology, H&E, (B - original objective x20, C and D - original objective x40)
View Figure 1
The specimens were formalin-fixed, paraffin-embedded, sliced and stained with hematoxylin-eosin (H&E). The histologic findings consisted of diffuse infiltration in both ovaries of tumor cells with moderate cytological and nuclear atypia and tubular, nested and cystic growth pattern. They were inlayed in abundant hyaline stroma with multiple microcalcifications (Figure 1B and Figure 1D). The cyst in the right ovary showed serous epithelial covering with minute papillary projections and cell stratifications without atypical cytological characteristics (Figure 1B and Figure 1C).
The constellation of findings led to two divergent differential diagnoses - low grade serous ovarian carcinoma and metastases from other primary site - considering the histology - ductal breast carcinoma and intestinal stomach cancer were the first to be excluded.
The patient had been hospitalized numerous times during the past few years because of alcohol-related liver cirrhosis, diagnosed in 2011 with liver biopsy, and she had frequent laparocenteses due to ascites. Each of these punctures had been accompanied with cytological examination and in one of the last there had been atypical cells, suspicious of carcinoma. Searching for the origin of the latter, an elevated level of CA 125 of 290 U/ml had been measured and an ultrasound examination of the abdomen had detected a cyst in her right ovary. By this time the patient had been assumed to have primary ovarian carcinoma with peritoneal dissemination. She had not been recommended for a surgery due to her end-stage liver disease and other co-morbidities such as epilepsy. Going back to the very beginning of her medical history a protocol of a quadrantectomy performed in the year 2000 was found. She had been diagnosed with invasive ductal carcinoma 15 years ago. The tumor was staged pT2 NO and radionuclide ligand-binding assays confirmed that it had been hormone receptor-positive. For unknown reasons only adjuvant hormonal therapy was performed during the first 5 years. As a respond to the treatment, a locoregional recurrence and metastases have not been observed before diagnosis of the current process.
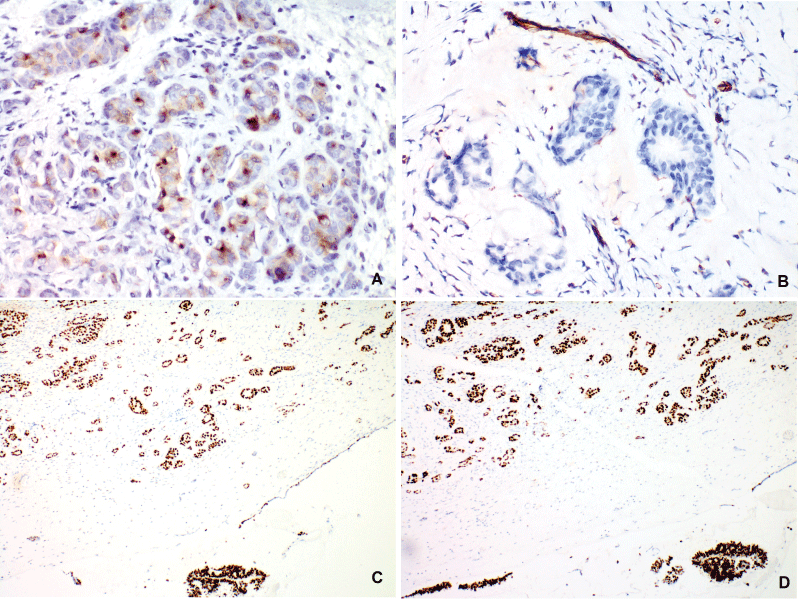
The next step in the diagnostic process was the ancillary immunohistochemical study performed with standart avidin-biotin immunoperoxidase method using commercial RTU DAKO antibodies, DAKO Autostainer Link 48 and 3,3'-diaminobenzidine (DAB) based detection system (Envision ™FLEX) according to the established protocols. The observed results are summarized in Table 1 and Figure 2.
![]()
Table 1: Immunohistochemical markers
View Table 1
.
Figure 2: Immunohistochemistry. Avidin-biotin peroxidase method was applied for the stains. A. GCDFP-15 (original objective x40): Focal cytoplasmic staining. B. WT1 (original objective x40): Negative expression in the tumor; positive control in blood vessels (endothelium). C, D. Estrogen and progesterone receptors (original objective x10): Diffuse, intense nuclear staining in the metastases and in the cystadenoma.
View Figure 2
Analyzing the accumulated clinical and pathological data we reached to the diagnosis of bilateral ovarian metastases from hormone receptor-positive ductal breast carcinoma. This case attracted our attention because of the late occurrence of the metastatic adnexal masses after the primary cancer with no other clinical and laboratory signs of local recurrence of the breast tumor. The cystic formation in the right ovary was designated as a serous cystadenoma. After presenting the case to the cancer committee of the hospital, chemotherapy was initiated.
Discussion
Adnexal masses in patients with medical history of breast carcinoma tend to be benign in the majority of cases. If the lesion is malignant, the chances of being primary ovarian carcinoma are 3 to 7 times greater than being a metastasis [5,6,8-10]. The relative risk can be even higher in BRCA 1 and 2 mutation carriers [7,8]. The other problematic issue is the wide-ranging interval of detection of breast cancer metastases to the ovaries - it varies in the different studies depending on the features of : 1/ the patient - age, menopausal status, BRCA mutation status; 2/ the primary tumor - size, lymph node metastases, steroid receptors and HER2 expression, and the 3/surgery - prophylactic (risk-reducing) adnexectomy, hormonal castration or detectable ovarian mass [3,8,23] There is not a strictly defined correlation between the listed characteristics and the clinico-morphological manifestation of the metastatic lesions which makes their recognition challenging.
Metastases of breast carcinoma, located in the ovaries, have no specific clinical presentation. Most of the patients have no symptoms, some present with complaints of abdominal distension, pain or rarely - a palpable mass [10]. In our case these symptoms could be result from the decompensated liver cirrhosis. CA 125 and CA 15-3 are not recommended as sole screening markers because of their low sensibility - if used for assessment of adnexal masses they should be interpreted in correlation with some of the subsequently discussed features [11,12].
Metastatic lesions tend to have macroscopic and ultrasound characteristics which could facilitate their recognition - such as bilateral engagement of the ovaries, solid nodular growth and small dimensions < 10 cm [10]. However, these features are not absolute - for example 80% of breast carcinoma metastases are bilateral but the same refers to 66% of primary serous ovarian carcinomas. Primary ovarian carcinomas tend to be more than 5 cm in diameter, cystic with complex ultrasound architecture [6,7]. Peritoneal dissemination and ascites can be observed in both primary and metastatic ovarian lesions with a similar frequency [13,14].
The differential diagnosis between primary ovarian carcinoma and breast metastases remains difficult even when immunohistochemistry is applied. Both tumors stain positive for Cytokeratin 7 (CK7), ER and PR [15-17]. CA125 and WT1 are identified as markers for ovarian carcinomas (serous in particular) with positive expression reaching 90% for the former and about 78-86% for the latter [18,19]. GCDFP -15 and Mammaglobin are commonly used and accepted markers for identification of breast epithelia which defines their presence in the panel for differentiation of metastatic ovarian neoplasms. Their sensitivity and specificity are quite variable ranging from 20 to 80% and for this reason they are commonly used in combination [17,20,21].
75% of the secondary ovarian tumors with mammary origin have the histological features of ductal invasive carcinoma (NOS). Regarding the fact that hormone receptor status of metastases may differ from the primary tumor (receptor conversion), the metastatic lesions should be biopsied and tested [22]. In small series, the immunohistochemical staining for ER, PR and HER2 in ovarian metastases is similar to this encountered in primary breast cancers - up to 75% of metastatic lesions are ER+ and up to 20% are HER2-positive [10]. In our case the metastases were ER+/PR+ which correlated to the results from ligand-binding essay results 15 years ago. HER2 expression was examined for the first time in the metastases and it was negative. There is not a specific group of breast carcinomas prone to distant recurrence in the ovary, but some studies demonstrate that ER+/PR+/HER2 (-) immunohistochemical profile is related to better prognosis and therefore increased overall survival [23]. The latter findings combined with the observed constellation of low proliferative index Ki67 < 10% and wild type p53 expression corresponded to the less aggressive and prolonged course of the disease in our case.
Apart from aforementioned diagnostic issues some aspects concerning the role of surgery in metastatic ovarian lesions are also matter of discussion. There is not a strict guideline concerning the volume of the operative procedure in these cases [4,10,24]. Considering the multiple co-morbidities of our patient, the emergency character of the surgery and the clinical suspicion of primary ovarian carcinoma, the bilateral adnexectomy was established as the ultimate choice. It led to the verification of the metastatic nature of the ovarian masses and provided a significant cytoreduction of the tumor load.
Conclusion
Breast carcinomas and ovarian neoplasms are related. This relation could be demonstrated either as synchronous or metachronous metastases of the breast cancer in the ovaries, or as co-existing primary carcinomas. The discrimination between these cases and the establishment of the accurate diagnosis could be challenging but it is essential since there is a difference in the consequent therapeutic behavior. Each step of this complex process has issues which still demand to be validated because of their further influence on the multidisciplinary approach to these lesions.
Conflicts of interest
The authors declare that they have no conflicts of interest to disclose.
The patient's approval has been given for this case report.
References
-
Petru E, Pickel H, Heydarfadai M, Lahousen M, Haas J, et al. (1992) Nongenital cancers metastatic to the ovary. Gynecol Oncol 44: 83-86.
-
Gavriilidis P, Ananiadis A, Theodoulidis V, Toskas I, Barbanis S (2012) Ovarian metastasis from breast invasive ductal carcinoma. Hippokratia 16: 391.
-
Gagnon Y, Têtu B (1989) Ovarian metastases of breast carcinoma. A clinicopathologic study of 59 cases. Cancer 64: 892-898.
-
Abu-Rustum NR, Aghajanian CA, Venkatraman ES, Feroz F, Barakat RR (1997) Metastatic breast carcinoma to the abdomen and pelvis. Gynecol Oncol 66: 41-44.
-
Simpkins F, Zahurak M, Armstrong D, Grumbine F, Bristow R (2005) Ovarian malignancy in breast cancer patients with an adnexal mass. Obstet Gynecol 105: 507-513.
-
Tuncer ZS, Boyraz G, Selcuk I, Sahin N, Kaynaroglu V, et al. (2012) Adnexal masses in women with breast cancer. Aust N Z J Obstet Gynaecol 52: 266-269.
-
Bergfeldt K, Rydh B, Granath F, Grönberg H, Thalib L, et al. (2002) Risk of ovarian cancer in breast-cancer patients with a family history of breast or ovarian cancer: a population-based cohort study. Lancet 360: 891-894.
-
Tserkezoglou A, Kontou S, Hadjieleftheriou G, Apostolikas N, Vassilomanolakis M, et al. (2006) Primary and metastatic ovarian cancer in patients with prior breast carcinoma. Pre-operative markers and treatment results. Anticancer Res 26: 2339-2344.
-
Curtin JP, Barakat RR, Hoskins WJ (1994) Ovarian disease in women with breast cancer. Obstet Gynecol 84: 449-452.
-
Bigorie V, Morice P, Duvillard P, Antoine M, Cortez A, et al. (2010) Ovarian metastases from breast cancer: report of 29 cases. Cancer 116: 799-804.
-
Perrotin F, Marret H, Bouquin R, Fischer-Perrotin N, Lansac J, et al. (2001) [Incidence, diagnosis and prognosis of ovarian metastasis in breast cancer]. Gynecol Obstet Fertil 29: 308-315.
-
Le Thai N, Lasry S, Brunet M, Harvey P, Rouesse J (1991) [Cancer of the ovary after cancer of the breast. 45 cases]. J Gynecol Obstet Biol Reprod (Paris) 20: 1043-1048.
-
Ulbright TM, Roth LM, Stehman FB (1984) Secondary ovarian neoplasia. A clinicopathologic study of 35 cases. Cancer 53: 1164-1174.
-
Hann LE, Lui DM, Shi W, Bach AM, Selland DL, et al. (2000) Adnexal masses in women with breast cancer: US findings with clinical and histopathologic correlation. Radiology 216: 242-247.
-
Lagendijk JH, Mullink H, van Diest PJ, Meijer GA, Meijer CJ (1999) Immunohistochemical differentiation between primary adenocarcinomas of the ovary and ovarian metastases of colonic and breast origin. Comparison between a statistical and an intuitive approach. J Clin Pathol. 52: 283-290.
-
Kaufmann O, Deidesheimer T, Muehlenberg M, Deicke P, Dietel M (1996) Immunohistochemical differentiation of metastatic breast carcinomas from metastatic adenocarcinomas of other common primary sites. Histopathology. 29: 233-240.
-
Noriega M, Paesani F, Perazzo F, Lago N, Krupitzki H, et al. (2012) Immunohistochemical characterization of neoplastic cells of breast origin. Diagn Pathol 7: 73.
-
Tornos C, Soslow R, Chen S, Akram M, Hummer AJ, et al. (2005) Expression of WT1, CA 125, and GCDFP-15 as useful markers in the differential diagnosis of primary ovarian carcinomas versus metastatic breast cancer to the ovary. Am J Surg Pathol. 29: 1482-1489.
-
Lee BH, Hecht JL, Pinkus JL, Pinkus GS (2002) WT, estrogen receptor, and progesterone receptor as markers for breast or ovarian primary sites in metastatic adenocarcinoma to body fluids. Am J Clin Pathol 117: 745-750.
-
Bhargava R, Beriwal S, Dabbs DJ (2007) Mammaglobin vs GCDFP-15: an immunohistologic validation survey for sensitivity and specificity. Am J Clin Pathol 127: 103-113.
-
Fritzsche FR, Thomas A, Winzer KJ, Beyer B, Dankof A, et al. (2007) Co-expression and prognostic value of gross cystic disease fluid protein 15 and mammaglobin in primary breast cancer. Histol Histopathol 22: 1221-1230.
-
Hoefnagel LD, van de Vijver MJ, van Slooten HJ, Wesseling P, Wesseling J, et al. (2010) Receptor conversion in distant breast cancer metastases. Breast Cancer Res 12: R75.
-
Tazhibi M, Fayaz M, Mokarian F (2013) Detection of prognostic factors in metastatic breast cancer. J Res Med Sci 18: 283-290.
-
Eitan R, Gemignani ML, Venkatraman ES, Barakat RR, Abu-Rustum NR (2003) Breast cancer metastatic to abdomen and pelvis: role of surgical resection. Gynecol Oncol 90: 397-401.





