International Journal of Pathology and Clinical Research
Pathologic Reporting of the Surgical Margin after Radical Prostatectomy: Considerations for Improving Oncologic Prognostication
Michael J. Whalen1* and Michael B. Rothberg2
1Department of Urology, Yale School of Medicine, New Haven, CT, USA
2Department of Urology, Wake Forest School of Medicine, Winston-Salem, NC, USA
*Corresponding author:
Michael J. Whalen, MD, Dept of Urology, Yale School of Medicine, Yale-New Haven Hospital, PO Box 208058, New Haven, CT 06520-8058, USA, E-mail: Mjw2117@gmail.com
Int J Pathol Clin Res, IJPCR-2-018, (Volume 2, Issue 1), Review Article; ISSN: 2469-5807
Received: November 26, 2015 | Accepted: January 04, 2015 | Published: January 06, 2016
Citation: Whalen MJ, Rothberg MB (2016) Pathologic Reporting of the Surgical Margin after Radical Prostatectomy: Considerations for Improving Oncologic Prognostication. Int J Pathol Clin Res 2:018. 10.23937/2469-5807/1510018
Copyright: © 2016 Whalen MJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Positive surgical margins (PSM) after radical prostatectomy have been identified as an adverse pathological feature predictive of higher rates of biochemical recurrence, distant metastasis, and cancer-specific mortality compared to negative margins. Such observations underlie the rationale for adjuvant radiotherapy for locally advanced disease or positive margins. Even without adjuvant radiotherapy, however, not all patients who have a positive margin will recur. While other adverse pathological features such as pathologic stage, pathologic Gleason score, and PSA likely play a role, there has been recent evidence demonstrating the importance of nuanced examination of the positive margin to enhance risk stratification and prognosis. Such features include extent involvement or multifocality, predominant Gleason grade at the PSM, and even close surgical margins. The impact of these features on oncologic outcomes is explored in this review, along with the potential role of preoperative multiparametric magnetic resonance imaging and intraoperative frozen sections to predict and reduce the rates of PSM.
Keyword
Radical prostatectomy, Surgical margins, Biochemical failure, Pathologic reporting
Introduction
Positive surgical margins
The objective of radical prostatectomy (RP) for the treatment of clinically localized prostate cancer is complete surgical extirpation, performance of pelvic lymphadenectomy when indicated, and the achievement of a negative surgical margin. Positive surgical margin (PSM) status has been identified as an adverse pathologic feature, associated with increased rates of biochemical recurrence (BCR)/local progression [1], metastasis [1], and cancer-specific mortality [2,3]. Consequently, PSM has been incorporated into multiple nomograms for post-operative prostate cancer prognosis, serving as an independent risk factor for biochemical recurrence after radical prostatectomy [4] and for disease-free survival after post-prostatectomy salvage radiation therapy [5].
Role of adjuvant radiotherapy for positive surgical margins
The adverse impact of the PSM on oncologic outcomes underlies the rationale for adjuvant radiation therapy after RP for men with adverse pathology (i.e. ≥ pT3 or PSM). Three major phase 3 randomized controlled trials (EORTC 22911 [6], SWOG 8794 [7] & ARO 96-02 [8]) have investigated the impact of adjuvant radiotherapy on various oncologic outcomes such as BCR-free survival, metastasis-free survival, clinical progression-free survival (i.e. BCR + metastasis), and overall survival. Although the impact on BCR has been confirmed by each of these studies, the influence on metastasis-free survival and overall mortality was found to be significant only for SWOG 8794. [7] While EORTC 22911 reported that improvements in clinical progression-free survival did not persist at the longer interval follow-up of median 10.6 years, post-hoc analysis of the data did reveal improved clinical progression-free survival for patients with positive margins, regardless of final pathologic stage (Figure 1A) [6]. Similarly, ARO 96-02 illustrated the differential improvement in recurrence-free survival for patients with positive surgical margins (Figure 1B) [8]. Furthermore, SWOG 8794 allowed positive surgical margins as an independent inclusion criterion for adjuvant therapy. More nuanced examination of the surgical margin, such as predominant Gleason grade or extent/multifocality of the PSM, was not explored in these studies, however.
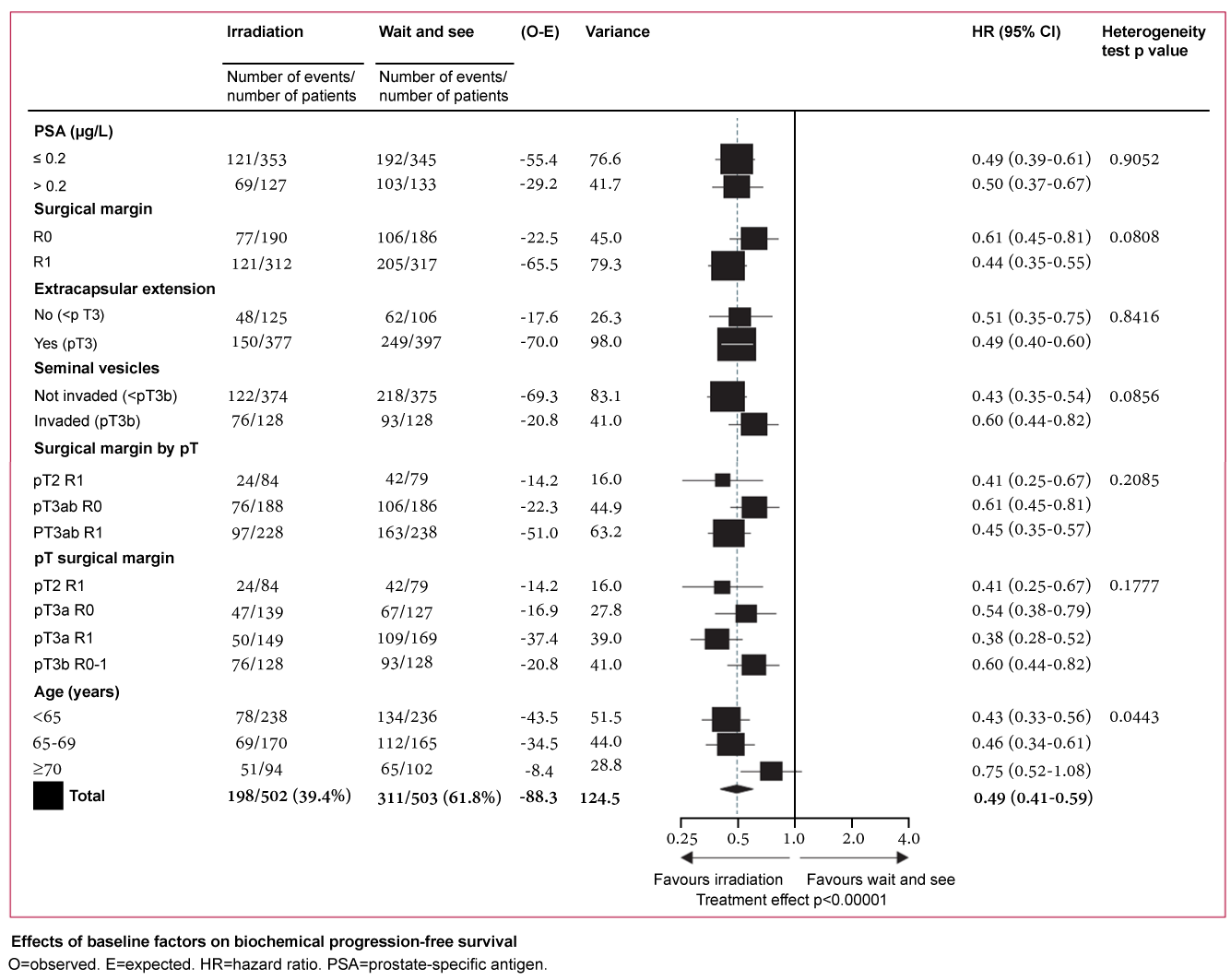
.
Figure 1A: Forrest plot showing impact of patient clinicopathologic features on biochemical progression-free survival. Derived from Bolla et al., Lancet, 2012 [6].
View Figure 1A
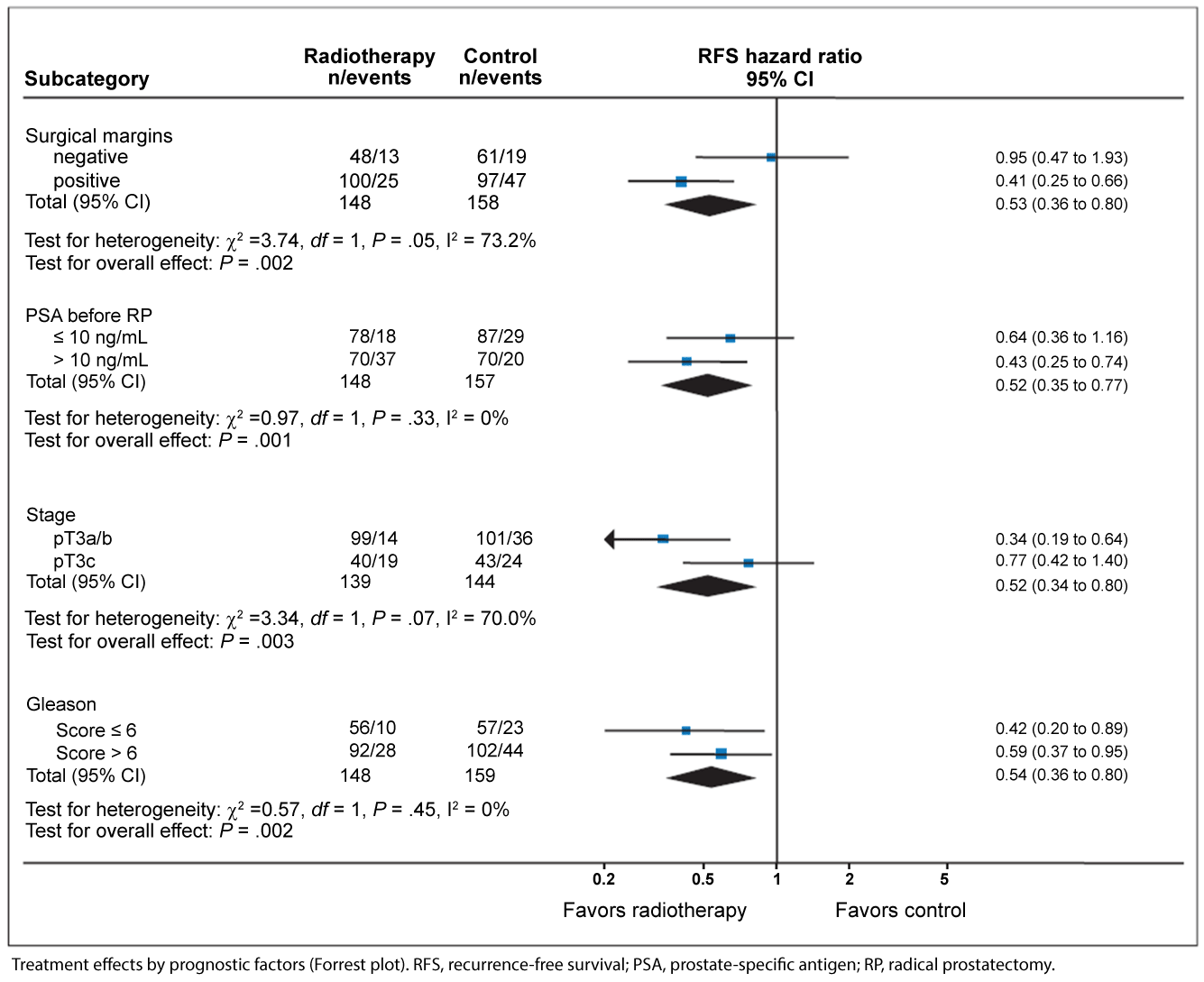
.
Figure 1B: Forrest plot showing impact of patient clinicopathologic features on recurrence-free survival. Derived from Wiegel et al., J Clin Oncol, 2009 [8].
View Figure 1B
Risk factors for positive margins
Rates of positive margins in contemporary series of radical prostatectomy range from 11-38% [9], and are dependent on pathologic stage and Gleason score [10], surgeon experience as well as institutional operative-volume [11], and may be influenced by the surgical modality itself (i.e. open vs. laparoscopic vs. robotic-assisted) [11]. The influence of the learning curve has been demonstrated in a series of n = 450 consecutive robotic-assisted radical prostatectomy (RARP) cases demonstrating PSM rates of 36% in the first 50 patients, 17.6% in patients 51-250, and 7.5% in patients 251-450 [12]. A recent large, multi-institutional, retrospective study of 22,393 open, laparoscopic, and robotic-assisted RP procedures from 2000-2011 reported PSM range from 13.8%-22.8%, with superior rates for RARP and laparoscopic procedures compared to open surgery when controlled via propensity scoring for prostate specific antigen (PSA), Gleason score, pathologic stage, and year of surgery covariates [11]. This study corroborates an earlier single-institution series of 200 sequential RARP vs. 200 sequential retropubic RP, showing superior PSM rates for RARP for stage pT2 disease (9.4% vs. 24.1%, p < 0.001) [13].
Despite refinements in surgical technique, including the advent of robotic surgery, surgical margin status remains under active investigation throughout the literature. Indeed, PSM does not always lead to BCR, with rates reported to be 19-48% at a median five years post-operatively [2,14,15]. Furthermore, the use of BCR as a surrogate measure of survival is controversial, as many men experiencing BCR will not die of prostate cancer [2,16]. It has been demonstrated that prostate-cancer specific mortality is worse for men with BCR only in the setting of short latency to BCR or short PSA doubling time [17,18]. These observations invite the possibility that other pathologic characteristics of the PSM may underlie the risk for disease recurrence and progression.
Standardized reporting of the positive surgical margin
Disparate results regarding the prognostic impact of the PSM may also be due to variability and inconsistency in the detail of reporting across surgeons and institutions [19,20]. Indeed, rather than a binary result of positive versus negative, more nuanced methods of reporting surgical margins have been proposed. While the standard definition of PSM consists of tumor that extends to the surface of the prostate wherein the surgeon has cut across the tissue plane [21], refinements in the understanding of the location of the positive margin, length of the positive margin, presence of PSM multifocality, predominant Gleason score at PSM, and even close but negative PSM have been investigated. To analyze the literature in a systematic manner in an effort to propose standardized reporting guidelines for the clinical pathologic community, the International Society of Urological Pathology (ISUP) Consensus Conference on Handling and Staging of Radical Prostatectomy Specimens was convened and met most recently in 2009 in Boston, MA. Five working groups were tasked with various aspects of RP specimen processing, including cancer volume, extraprostatic extension and lymphovascular invasion, and surgical margins. The most recent publication from this conference appeared in 2011 in Modern Pathology [21]. The recommendations of this group regarding the previously mentioned aspects of PSM will be discussed in this review. The impact of the various PSM features on biochemical recurrence, metastatic progression, prostatic cancer-specific, and overall survival have only been recently examined, remain a source of controversy, and will be explored as well.
Margin Location
The most common site of PSM is the apex in both cT1c and cT2 disease [21]. The posterior and posterolateral aspects of the prostate are other common sites [20]. The location of the PSM may be influenced by pathologic stage as well. A recent study by Pagano et al. examined PSM rate and location in patients with pathologic seminal vesicle invasion at RP [22]. Overall PSM rate was 41% and were more common at the peripheral zone (57%) than the apex (32%). While bladder neck and anterior positive margins were more common with RP via a perineal approach, the apex is the most common site of PSM in the contemporary era [13]. The oncologic impact of PSM location is controversial, with multiple studies demonstrating an inability to independently predict BCR on multivariable regression analysis [11,23-25]. Obek et al. concluded that PSM at multiple sites, posterolateral surface, or bladder neck increased risk of disease progression [26]. The bladder neck is an uncommon site for PSM (4-6%), and usually occurs in addition to other PSM. [22,27] The presence of bladder neck involvement has been identified as an independent predictor of early BCR, tantamount to the risk of locally advanced disease, such as pT3a [28] or seminal vesical invasion [29]. The study by Pagano et al. reported a seven-fold greater risk of BCR with bladder neck PSM on multivariable regression analysis [22]. After evaluation of contemporary evidence, the ISUP 2009 Conference promoted routine reporting of the location of the PSM, including cases of intraprostatic incision into tumor and extraprostatic extension. Consensus was reached regarding reporting as "posterior, posterolateral, lateral, anterior at the apex and mid-portion of the prostate, and the bladder neck" [21]. Although not based on level I evidence, this consensus statement may serve to provide surgeon feedback and aid in modification of surgical technique.
Close Margins
Data from other domains of oncology, such as breast cancer, have demonstrated that decreasing distance from tumor to surgical margin is associated with both local recurrence and distant metastasis [30]. The correlation between a "close" surgical margin (CSM) and BCR after RP has been previously examined in the pre-PSA era. These studies reported similar rates of BCR for negative surgical margin (NSM) vs. CSM and were cited by the ISUP 2010 Consensus statement. In 1997, Epstein et al. investigated n = 101 margin-negative patients after RP with at least 5 years follow-up. The authors found that mean distance from tumor to inked margin was similar between patients who experienced BCR (0.46 mm) and those who did not (0.33 mm) [31]. Measured distance was also not found to be an independent predictor of BCR. Later in 2005, Emerson et al. reported on n = 278 margin-negative cases and found that the closest measured distance between tumor and inked margin (median = 0.5 mm) did not predict BCR on multivariable analysis [32]. Bong et al. in 2009 similarly found no difference in BCR between negative margins and CSM, defined as tumor < 1 mm from inked margin [33].
The above studies had several limitations, however. The Epstein and Emerson studies were conducted in the pre-PSA era, with > 80% of patients having cT2 disease. Also, they relied on analysis of tumor to margin distance as a continuous variable in the multivariable regression models. The assumption of a linear relationship between the closeness of the margin and BCR may not be accurate, and may underestimate the true risk of CSM. In addition, the study by Bong et al. reported follow-up data of only 12 months, which is premature to accurately capture BCR. Nevertheless, the ISUP 2010 stated, "Pathologists agree that tumor extending close to the capsular margin, yet not to it, should be reported as a negative margin" [21]. Standard pathologic reporting at most institutions reflects this consensus statement.
Subsequent evidence from more contemporary series suggests close margins more closely resemble PSM and are an independent predictor of BCR. The definition of a "close" surgical margin (CSM) has varied from < 0.1 mm to < 1 mm. Most recently, Whalen et al. reported on n = 609 patients who underwent RP and concluded the presence of both PSM and CSM (defined as < 1 mm) were associated with higher rates of BCR relative to negative margins on multivariable logistic regression (HR = 2.7 and 2.72, respectively) [34]. Moreover, estimated 3-year freedom from BCR following a CSM was similar to that of PSM (70.4% and 74.5%, respectively; log-rank p = 0.66) when compared to a 90% 3-year freedom from BCR for patients with negative margins (log-rank p < 0.001 for NSM vs. PSM and p = 0.01 for NSM vs. CSM) (Figure 2) [34]. Additional evidence comes from Lu et al. (n = 894, 11% CSM) and Izard et al. (n = 1588, 15% CSM), who both defined CSM as < 0.1 mm and concluded CSM not only significantly predicted BCR, but effectively doubled BCR risk when compared to patients with negative margin status (HR = 1.5-2.1) [35,36]. These reports indicate the strong predictive value of CSM for disease recurrence and demonstrate similar risk profiles for BCR for CSM and PSM. Although further studies are needed to assess the impact of close margins on cancer-specific and overall survival, the impact on BCR indeed warrants reexamination of the ISUP recommendations.
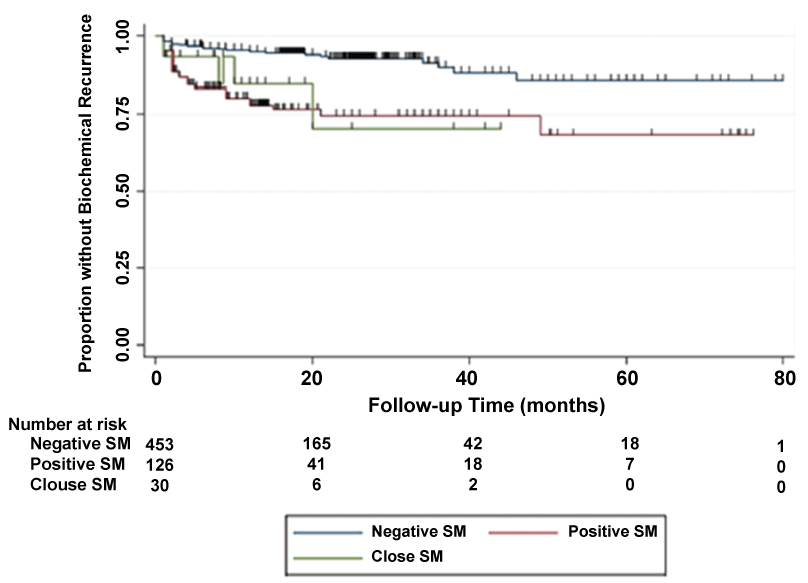
.
Figure 2: Kaplan Meier Curve of Biochemical Recurrence by Surgical Margin Status. Derived from Whalen et al., Urol Oncol, 2015 [34].
View Figure 2
Gleason Grading at Positive Margin
Higher incidences of PSM have been reported for patients with higher pathologic staging and Gleason score. A large series of 65,633 patients who underwent RP were found to have PSM in 21.2%. PSM was greater in pT3 patients vs. pT2 (44% vs. 18%, p < 0.001), as well as Gleason 7-10 vs. Gleason 6 tumors (28% vs. 18%, p < 0.001) [2]. The multivariable analysis from this study concluded PSM is an independent significant predictor of prostate-cancer specific mortality; however, this finding was observed only for patients with higher grade and pT3 tumors. In a survey from the ISUP 2009 Conference, the majority of pathologists do not report Gleason score of the tumor at the site of margin positivity, although a small proportion indicated that they report Gleason score in the setting of high grade disease. In addition, 85% reported that they did not grade a margin-positive tumor in the setting of extraprostatic extension. The consensus was for inclusion of Gleason grade at the positive surgical margin at the pathologist's discretion, based on a paucity of evidence [21].
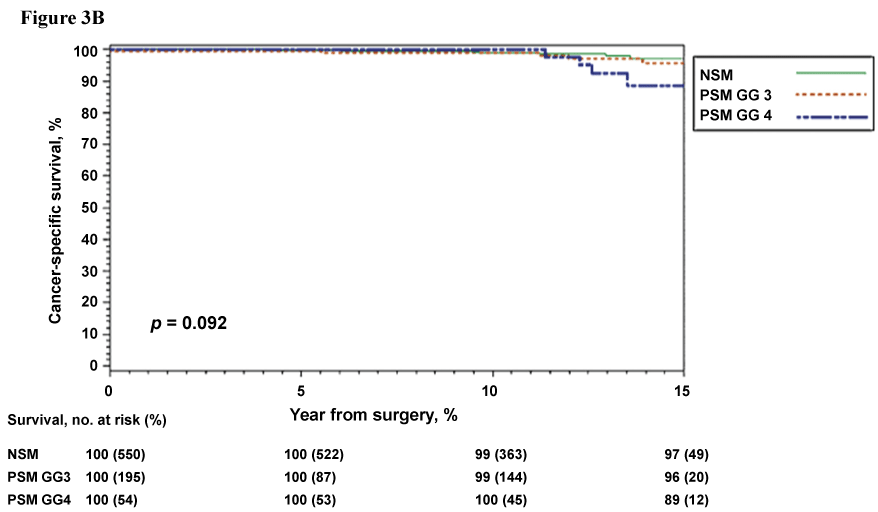
Prior inquiries into the significance of the Gleason score, or predominant Gleason grade, at the site of a positive surgical margin have yielded inconsistent results with respect to prognostication and need for adjuvant therapies. Recent evidence does support pathologic disclosure of the predominant Gleason grade at the PSM. Viers et al. reported on n = 1036 patients with stage pT2-3a, Gleason 7 prostate cancer who underwent RP, of which, n = 338 had PSM [37]. From these patients with PSM, 242 (72%) and 96 (28%) had primary Gleason grades 3 and 4 at the site of PSM, respectively [37]. After long median follow-up of 13 years, the presence of Gleason 4 disease at a PSM resulted in significantly worse 15-year systemic progression-free (74% vs. 90%) and cancer-specific survival (86% vs. 96%) outcomes when compared to patients with Gleason 3 disease at the PSM (Figure 3A & Figure 3B) [37]. Moreover, on multivariate analysis Gleason 4 disease at the PSM was independently associated with a significantly higher risk of systemic progression (HR = 2.74, p = 0.004) and prostate cancer-related death (HR = 3.91, p = 0.02) [37]. Of note, a similar number of patients with PSM primary Gleason 3 and 4 received adjuvant radiation therapy (14% vs. 20%). Therefore, for patients with clinically significant prostate cancer (Gleason > 7) and PSM following RP, inclusion of Gleason grading at the site of PSM in pathologic reporting may provide urologists with important information regarding the need for additional therapies and postoperative monitoring.
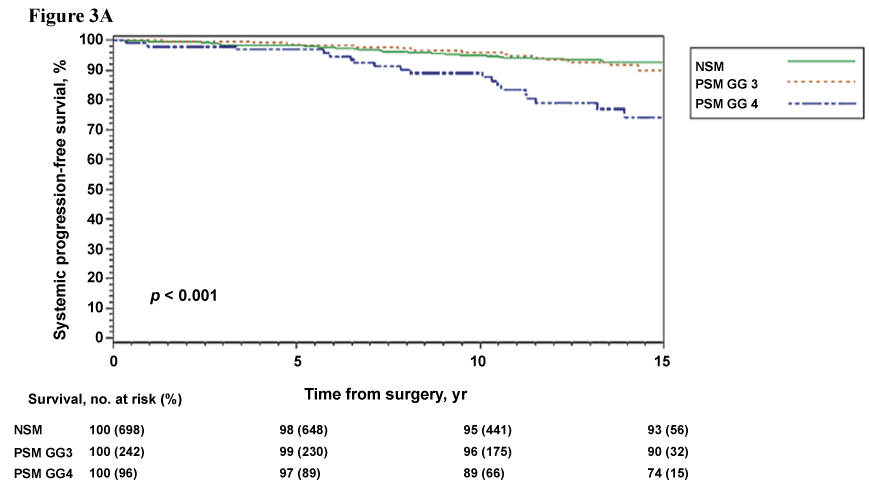
.
Figure 3A: Systemic progression-free survival stratified by primary Gleason score at the positive surgical margin and margin status. Derived from Viers et al., Eur Urol, 2014 [37].
View Figure 3A
.
Figure 3B: Cancer-specific survival stratified by primary Gleason score at the positive surgical margin and margin status. Derived from Viers et al., Eur Urol, 2014 [37].
View Figure 3B
PSM Extent of Involvement
The extent of involvement of tumor at PSMs influences the likelihood of BCR. Indeed, the ISUP 2009 consensus argued for reporting of extent of positive margin as mm of involvement, rather than subjective quantification (i.e. "focal" vs. "extensive") [21]. There are several published series that corroborate this consensus. Sooriakumaran et al. reported on n = 893 patients, of which, n = 189 (21.2%) had PSM following RP [11]. Investigators stratified patients by PSM length and anatomical location. Overall, PSMs that were either > 3 mm in length or multifocal along the inked margin were independent predictors of BCR (HR = 2.84, p < 0.001) compared to PSMs < 3 mm in length, unifocal PSMs, or NSMs; however, anatomical location of the PSM was not significantly associated with an increased likelihood of BCR [11]. Interestingly, PSMs > 3 mm or those with multifocality were associated with a higher likelihood of BCR for pT2 disease (HR = 5.26, p < 0.001) than for pT3 disease (HR = 1.83, p = 0.076) [11]. Similar findings were reported by Emerson et al., who described on n = 86 (23%) patients with PSMs from their series of n = 369 RP specimens, and report that the number of PSM sites was independently associated with risk of BCR on multivariate analysis when adjusting for Gleason score (OR = 1.63, p = 0.002) [38]. Most recently, Servoll et al. reported on n = 300 patients undergoing RP, of whom n = 163 (54%) had at least one PSM [39]. On multivariate analysis, PSM length > 3 mm was independently associated with an increased risk of BCR when compared to patients with PSM length < 3 mm (HR = 1.95, p = 0.017) and NSM (HR = 2.49, p = 0.001) [39]. Moreover, linear extent of PSM > 3 mm demonstrated significantly worse BCR-free survival when compared to PSM < 3 mm at a median follow-up time of 68 months (p = 0.005) [39].
MRI as an adjunct to decrease positive margin rates
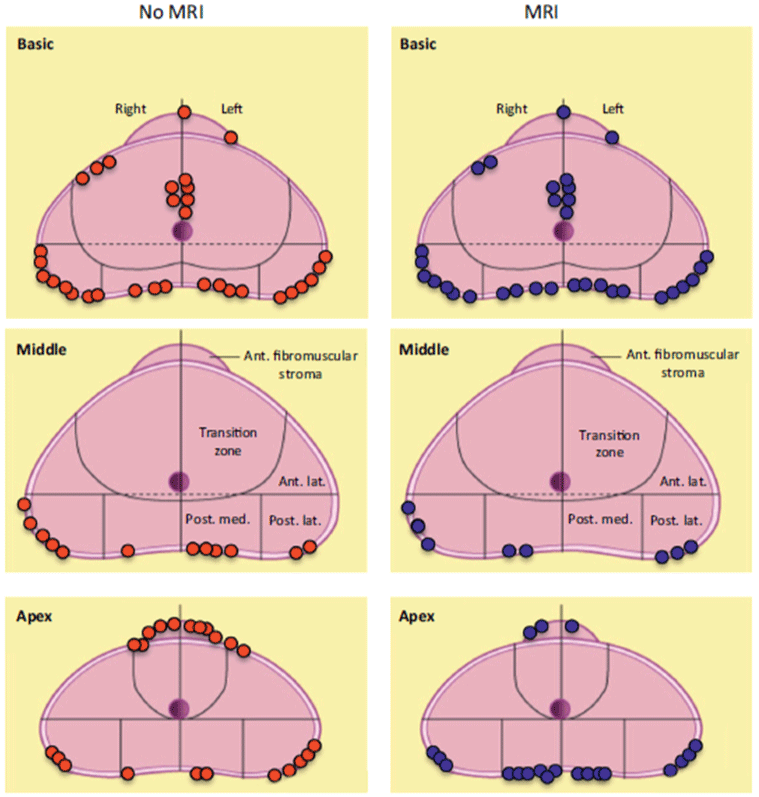
The utility of preoperative MRI on surgical outcomes, especially rates of PSM, is controversial. A Norwegian group recently reported a prospective randomized controlled trial in which patients were assigned to either MRI (n = 222) or non-MRI (n = 216) prior to undergoing RP [40]. Although there was no reduction in overall risk for PSM afforded by preoperative MRI, investigators do report a subgroup analysis where patients with cT1 disease experienced reduced rates of PSM (16% vs. 27%), with relative and absolute risk reductions of 41% and 11%, respectively. Figure 4 illustrates the similar anatomical distribution of PSM for patients who underwent preoperative MRI vs. those that did not. Of the 126 reported sites of PSM, 89% extended from the index tumor, which preoperative MRI was able to identify in 92% of patients. Most interestingly, the availability of preoperative MRI resulted in urologists making wider excisions in 29% of patients, although no improvement in PSM rate was noted for the entire cohort [40]. The role of even wider surgical resection based on preoperative MRI findings has yet to be explored.
.
Figure 4: Distribution of positive surgical margins sites on radical prostatectomy specimens. Adapted from Rud et al., Eur Urol, 2015 [40].
View Figure 4
Intraoperative Frozen Section Impact on PSM Rates
Intraoperative frozen section analysis aims to confirm margin negativity while achieving a balance between an adequate surgical excision for oncologic control and nerve sparing to optimize functional outcomes. Recently, von Bodman et al. reported on n = 236 patients who underwent whole surface frozen section during RP, yielding a 22% (52/236) positivity rate [41]. A vast majority of these cases (92.3%) were subsequently converted to NSM, producing an overall PSM rate of 3% (7/236) with false negative rate of 1.6% (3/184) [41]. About 15% of these patients had the preoperative nerve sparing plan altered by frozen section results. Fromont et al. reported on n=100 patients who underwent nerve sparing laparoscopic RP and a standardized intraoperative wedge tissue biopsy near the NVBs for frozen section analysis [42]. In the event of a positive frozen section the ipsilateral NVB would be excised. Positive frozen sections were identified in n = 24 (24%) patients, leading to an overall reduction in PSM rate from 33% to 12% (p < 0.001), from 26.1% to 7.9% (p < 0.005) for pT2 lesions, and from 83.3% to 41.7% (p < 0.05) for pT3 lesions [42]. Moreover, when compared to final RP pathology, intraoperative frozen section demonstrated 100% sensitivity and 96% specificity [42]. Other studies, however, have opted to only sample suspicious peripheral regions based on visual inspection. Tsubio et al. report their experience with n = 760 patients who underwent RP in which areas on the surface of the prostate that appeared to be suspicious for PSM were intraoperatively biopsied for frozen section analysis [43]. For the n = 259 (34.1%) patients who had intraoperative biopsy on suspicious lesions, frozen section analysis resulted in a sensitivity of 41.8% (23/55), specificity of 100% (204/204), positive predictive value of 100% (23/23), and negative predictive value of 86.4% (204/259) when compared to permanent section analysis [43]. These results demonstrate the technical feasibility and accuracy of this technique; the implications for increased rates of nerve sparing for intermediate- or high-risk disease-who might otherwise be treated with wide resection to ensure oncologic control-remains to be explored.
Conclusion
Recent evidence has allowed for refined understanding of the role of various pathological aspects of the positive surgical margin, as well as a close, but negative surgical margin. Contemporary pathologic reporting of surgical specimens after radical prostatectomy should routinely include disclosure of length of positive margin, margin multifocality, predominant Gleason grade, and margin to tumor distance for negative margins less than 1mm. The added prognostic value of incorporating these pathologic features into contemporary cancer nomograms has yet to be defined. The roles of multiparametric MRI and intraoperative frozen section on reducing positive surgical margin rates and broadening the potential for safe nerve sparing remain under investigation. More inclusive pathologic reporting will enable enhanced risk stratification and improved identification of those patients that will benefit from adjuvant radiotherapy.
References
-
Pfitzenmaier J, Pahernik S, Tremmel T, Haferkamp A, Buse S, et al. (2008) Positive surgical margins after radical prostatectomy: do they have an impact on biochemical or clinical progression? BJU Int 102: 1413-1418.
-
Wright JL, Dalkin BL, True LD, Ellis WJ, Stanford JL, et al. (2010) Positive surgical margins at radical prostatectomy predict prostate cancer specific mortality. J Urol 183: 2213-2218.
-
Chalfin HJ, Dinizo M, Trock BJ, Feng Z, Partin AW, et al. (2012) Impact of surgical margin status on prostate-cancer-specific mortality. BJU Int 110: 1684-1689.
-
Kattan MW, Wheeler TM, Scardino PT (1999) Postoperative nomogram for disease recurrence after radical prostatectomy for prostate cancer. J Clin Oncol 17: 1499-1507.
-
Stephenson AJ, Scardino PT, Kattan MW, Pisansky TM, Slawin KM, et al. (2007) Predicting the outcome of salvage radiation therapy for recurrent prostate cancer after radical prostatectomy. J Clin Oncol 25: 2035-2041.
-
Bolla M, van Poppel H, Tombal B, Vekemans K, Da Pozzo L, et al. (2012) Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet 380: 2018-2027.
-
Thompson IM, Tangen CM, Paradelo J, et al. (2009) Adjuvant radiotherapy for pathologic T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J Urol 181: 956-962.
-
Weigel T, Bottke D, Steiner U, et al. (2009) Phase III postoperative adjuvant radiotherapy after radical prostatectomy compared with radical prostatectomy alone in pT3 prostate cancer with postoperative undetectable prostate-specific antigen: ARO 96-02/AUO AP 09/95. J Clin Oncol 27: 2924-2930.
-
Yossepowitch O, Bjartell A, Eastham JA, et al. (2009) Positive surgical margins in radical prostatectomy: outlining the problem and its long-term consequences. Eur Urol 55: 87-99.
-
Albadine R, Hyndman ME, Chaux A, Jeong JY, Saab S, et al. (2012) Characteristics of positive surgical margins in robotic-assisted radical prostatectomy, open retropubic radical prostatectomy, and laparoscopic radical prostatectomy: a comparative histopathologic study from a single academic center. Hum Pathol; 43.
-
Sooriakumaran P, Ploumidis A, Nyberg T, Olsson M, Akre O, et al. (2015) The impact of length and location of positive margins in predicting biochemical recurrence after robot-assisted radical prostatectomy with a minimum follow-up of 5 years. BJU Int, 115: 106-113.
-
Yee DS, Narula N, Amin MB, Skarecky DW, Ahlering TE (2009) Robot-assisted radical prostatectomy: current evaluation of surgical margins in clinically low-, intermediate-, and high-risk prostate cancer. J Endourol 23: 1461-1465.
-
Smith Jr JA, Chan RC, Chang SS, Herrell SD, Clark PE, et al. (2007) A comparison of the incidence and location of positive surgical margins in robotic assisted laparoscopic radical prostatectomy and open retropubic radical prostatectomy. J Urol, 178: 2385-2389.
-
Swindle P, Eastham JA, Ohori M, Kattan MW, Wheeler T, et al. (2005) Do margins matter? The prognostic significance of positive surgical margins in radical prostatectomy specimens. J Urol 174: 903-907.
-
Kausik SJ, Blute ML, Sebo TJ, Leibovich BC, Bergstralh EJ, et al. (2002) Prognostic significance of positive surgical margins in patients with extraprostatic carcinoma after radical prostatectomy. Cancer 95: 1215-1219.
-
Boorjian SA, Tollefson MK, Rangel LJ, Bergstralh EJ, Karnes RJ (2012) Clinicopathological predictors of systemic progression and prostate cancer mortality in patients with a positive surgical margin at radical prostatectomy. Prostate Cancer Prostatic Dis 15: 56-62.
-
D'Amico AV, Moul J, Carroll PR, Sun L, Lubeck D, et al. (2005) Surrogate end point for prostate cancer specific mortality in patients with nonmetastatic hormone refractory prostate cancer. J Urol 173: 1572-1576.
-
Freedland SJ, Humphreys SB, Mangold LA, Eisenberger M, Dorey FJ, et al. (2005) Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA 294: 433-439.
-
Eastham JA, Kattan MW, Riedel E, Begg CB, Wheeler TM, et al. (2003) Variations among individual surgeons in the rate of positive surgical margins in radical prostatectomy specimens. J Urol 170: 2292-2295.
-
Epstein JI, Amin M, Boccon-Gibod L, Egevad L, Humphrey PA, et al. (2005) Prognostic factors and reporting of prostate carcinoma in radical prostatectomy and pelvic lymphadenectomy specimens. Scand J Urol Nephrol Suppl : 34-63.
-
Tan PH, Cheng L, Srigley JR, et al. (2011) International Society of Urological Pathology (ISUP) Consensus Conference on Handling and Staging of Radical Prostatectomy Specimens. Working group 5: surgical margins. Mod Path 24: 48-57.
-
Pagano MJ, Whalen MJ, Paulucci DJ, et al. (2015) Predictors of biochemical recurrence in pT3b prostate cancer after radical prostatectomy without adjuvant radiotherapy. Prostate.
-
Pettus JA, Weight CJ, Thompson CJ, Middleton RG, Stephenson RA (2004) Biochemical failure in men following radical retropubic prostatectomy: impact of surgical margin status and location. J Urol 172: 129-132.
-
Saether T, Sørlien LT, Viset T, Lydersen S, Angelsen A (2008) Are positive surgical margins in radical prostatectomy specimens an independent prognostic marker? Scand J Urol Nephrol 42: 514-521.
-
Sofer M, Hamilton-Nelson KL, Civantos F, Soloway MS (2002) Positive surgical margins after radical retropubic prostatectomy: the influence of site and number on progression. J Urol 167: 2453-2456.
-
Obek C, Sadek S, Lai S, et al. (1999) Positive surgical margins with radical retropubic prostatectomy: anatomic site-specific pathologic analysis and impact on prognosis. Urol 54: 682-688.
-
Poulos CK, Koch MO, Eble JN, Daggy JK, Cheng L (2004) Bladder neck invasion is an independent predictor of prostate-specific antigen recurrence. Cancer 101: 1563-1568.
-
Zhou M, Reuther AM, Levin HS, et al. (2009) Microscopic bladder neck involvement by prostate carcinoma in radical prostatectomy specimens is not a significant independent prognostic factor. Mod Pathol 22: 385-392.
-
Buschemeyer WC 3rd, Hamilton RJ, Aronson WJ, Presti JC Jr, Terris MK, et al. (2008) Is a positive bladder neck margin truly a T4 lesion in the prostate specific antigen era? Results from the SEARCH Database. J Urol 179: 124-129.
-
Goldstein NS, Kestin L, Vicini F (2003) Factors associated with ipsilateral breast failure and distant metastases in patients with invasive breast carcinoma treated with breast-conserving therapy. A clinicopathologic study of 607 neoplasms from 583 patients. Am J Clin Pathol 120: 500-527.
-
Epstein JI, Sauvageot J (1997) Do close but negative margins in radical prostatectomy specimens increase the risk of postoperative progression? J Urol 157: 241-243.
-
Emerson RE, Koch MO, Daggy JK, Cheng L (2005) Closest distance between tumor and resection margin in radical prostatectomy specimens: lack of prognostic significance. Am J Surg Pathol 29: 225-229.
-
Bong GW, Ritenour CW, Osunkoya AO, Smith MT, Keane TE (2009) Evaluation of modern pathological criteria for positive margins in radical prostatectomy specimens and their use for predicting biochemical recurrence. BJU Int 103: 327-331.
-
Whalen MJ, Shapiro EY, Rothberg MB, et al. (2015) Close surgical margins after radical prostatectomy mimic biochemical recurrence rates of positive margins. Urol Oncol 33:494.e9-e14.
-
Lu J, Wirth GJ, Wu S, Chen J, Dahl DM, et al. (2012) A close surgical margin after radical prostatectomy is an independent predictor of recurrence. J Urol 188: 91-97.
-
Izard JP, True LD, May P, et al. (2014) Prostate cancer that is within 0.1mm of the surgical margin of a radical prostatectomy predicts greater likelihood of recurrence. Am J Surg Pathol 38: 333-338.
-
Viers BR, Sukov WR, Gettman MT, et al. (2014) Primary Gleason grade 4 at the positive margin is associated with metastasis and death among patients with Gleason 7 prostate cancer undergoing radical prostatectomy. Eur Urol 66: 1116-1124.
-
Emerson RE, Koch MO, Jones TD, Daggy JK, Juliar BE, et al. (2005) The influence of extent of surgical margin positivity on prostate specific antigen recurrence. J Clin Pathol 58: 1028-1032.
-
Servoll E, Vlatkovic L, Saeter T, et al. (2014) The length of a positive surgical margin is of prognostic significance in patients with clinically localized prostate cancer treated with radical prostatectomy. Urol Int 93: 289-295.
-
Rud E, Baco E, Klotz D, et al. (2015) Does preoperative magnetic resonance imaging reduce the rate of positive surgical margins at radical prostatectomy in a randomized clinical trial? Eur Urol 68: 487-496.
-
von Bodman C, Brock M, Roghmann F, et al. (2013) Intraoperative Frozen Section of the Prostate Decreases Positive Margin Rate While Ensuring Nerve Sparing Procedure During Radical Prostatectomy. J Urol 190: 515-520.
-
Fromont G, Baumert H, Cathelineau X, Rozet F, Validire P, et al. (2003) Intraoperative frozen section analysis during nerve sparing laparoscopic radical prostatectomy: feasibility study. J Urol 170: 1843-1846.
-
Tsuboi T, Ohori M, Kuroiwa K, Reuter VE, Kattan MW, et al. (2005) Is intraoperative frozen section analysis an efficient way to reduce positive surgical margins? Urology 66: 1287-1291.





