International Journal of Pathology and Clinical Research
A De Novo Malignant Mixed Tumor (Carcinosarcoma) of the Parotid Gland: Case Report
Hussam F Tallab1*, David E Ricklan2 and Peter J Catalano1
1Department of Otolaryngology, Tufts Medical School, SEMC, Boston, USA
2Department of Pathology, Steward St. Elizabeth's Medical Center, Steward Health Care, USA
*Corresponding author:
Hussam F Tallab, Clinical Associate, Department of Otolaryngology, St. Elizabeth's Medical Center/Tufts Medical School, 736 Cambridge Street, Boston, MA 02135, USA, Tel: (617) 779-6441; E-mail: hussam.tallab@steward.org
Int J Pathol Clin Res, IJPCR-2-020, (Volume 2, Issue 1), Case Report; ISSN: 2469-5807
Received: October 11, 2015 | Accepted: January 26, 2016 | Published: January 29, 2016
Citation: Tallab HF, Ricklan DE, Catalano PJ (2016) A De Novo Malignant Mixed Tumor (Carcinosarcoma) of the Parotid Gland: Case Report. Int J Pathol Clin Res 2:020. 10.23937/2469-5807/1510020
Copyright: © 2016 Tallab HF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution,
and reproduction in any medium, provided the original author and source are credited.
Abstract
True malignant mixed tumors (carcinosarcomas) of salivary gland origin are exceedingly rare and demonstrate malignant epithelial and stromal components. The subject of this case study is a 61 year-old male who presented with mild left facial swelling with minimal discomfort and without a mass of the parotid gland. He underwent CT imaging which demonstrated a cystic lesion of the parapharyngeal space. CT guided fine needle aspiration of the lesion failed to demonstrate malignancy. The patient ultimately underwent two surgical procedures to fully excise the cancer. Histological and immunological evaluation revealed a carcinosarcoma. Carcinosarcoma (true malignant mixed tumor) of the parotid gland is an extremely uncommon tumor composed of both carcinomatous and sarcomatous elements coexisting together. An understanding of this manifestation is crucial to the management and treatment of this disease. Review of the literature supports combined modality of surgery with postoperative radiation as standard of care for this malignancy although the long-term prognosis for these patients is unclear.
Keyword
Carcinosarcoma, Parotid gland, Malignant mixed tumor, Parotid neoplasm, Salivary gland neoplasm
Introduction
Carcinosarcoma of the parotid gland is a rare tumor composed of both malignant epithelial (carcinomatous) and malignant mesenchymal (sarcomatous) elements. These tumors represent approximately 0.04% to 0.16% of salivary gland neoplasm and 0.4% of all salivary gland malignancies [1]. They were first described by Kirklin et al. in 1951 [2]; however, in 1967, King first used the term true malignant mixed tumor in the salivary gland in a study that helped to raise the attention to this tumor type [3]. In the previous study, there was no age specific-reference, and patients usually ranged from 15-85 years with no predilection in regard to gender [4]. Many of the patients had a history of pleomorphic adenoma, recurrent pleomorphic adenoma, or recurrent carcinoma ex pleomorphic adenoma. Some cases, however, seem to arise de novo in the absence of a previous pleomorphic adenoma [5].
The present study reports a case of the parotid gland de novo carcinosarcoma which contained carcinomatous components and sarcoma that was ultimately treated with primary surgical resection and adjunctive radiation. To the authors' knowledge, de Nova parotid gland carcinosarcoma has been described only a few times in the literature.
Case Presentation
A 61 year-old male presented with a left side facial swelling that had slowly increased in size and had become locally painful. He first noticed the swelling in January 2015. He was evaluated by an otolaryngologist at a local hospital where he was found to have a slightly swollen left parotid gland without a palpable mass. There was no facial weakness or palpable lymph-adenopathy. The patient denied trismus, dysphagia, odynophagia, otalgia, hoarseness of voice, dyspnea, weight loss and facial weakness. He has a ten-year history of smoking and is an active smoker. The patient had no other symptoms, and the medical history was not contributory. A contrast enhanced CT scan was obtained revealing a non-enhancing smooth walled cystic mass located in the left parapharyngeal space measuring 3 × 5 cm without definitive connection to the deep lobe of the left parotid gland. No lymphadenopathy was noted. Fine needle aspiration was performed with CT guidance and four cc of orange colored serosangious fluid was obtained. No malignant cells were seen on cytology.
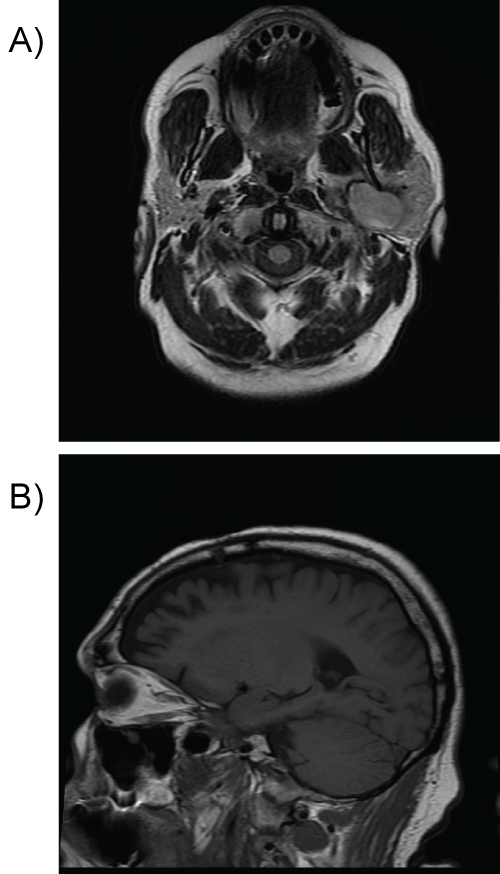
The patient was referred to our institution where his exam was unchanged; however, the pressure from the lesion has increased after the fine needle aspiration, causing some pain. To further evaluate the lesion, a gadolinium enhanced magnetic resonance scan was obtained demonstrating a smooth cystic lesion with a uniformly enhancing wall, thin internal septation, and an eccentric 5-mm nodular component (Figure 1). Cervical lymph-adenopathy was absent. The lesion still had no definitive connection to the deep lobe of the parotid gland. The patient underwent a left neck exploration in an attempt to excise the cystic parapharyngeal space lesion. During surgery, the cyst was extremely adherent to the surrounding structures, most notably the skull base. No enlarged or suspicious cervical adenopathy was encountered. Because the lesion was presumed benign and could not be excised via this approach, the cyst fluid was drained, the cyst capsule biopsied, and the procedure terminated.
.
Figure 1: A) Axial T2-MR and sagital T1-MRI images deep lobe of the left parotid gland, there is a mass which measures 5 × 2.8 × 3.3 cm. There is a curvilinear margin of dark signal around the lesion on T2-weighted images; B) The internal contents of the lesion are largely isointense to the surrounding parotid gland. The lesion is extensively but heterogeneously enhancing.
View Figure 1
The pathology report of the cyst wall read "highly suspicious for malignancy", but could not be more specific due to the limited tissue sample provided the pathologist. Cytology was again negative. The patient was counseled regarding these findings and a more definitive surgical procedure was planned via a parotid approach. The patient was counseled about the likelihood of facial nerve sacrifice during the second procedure. In the two weeks between the initial and definitive procedure, the patient developed severe facial and neck pain, trismus, increased facial swelling, loss of appetite, and some weight loss. He required nutritional supplements and narcotics for pain control.
During the second surgical procedure, a lateral parotid approach was used and the facial nerve was identified. However, the lesion was now easily palpable as a firm mass that grossly involved the parapharyngeal space and deep lobe of the parotid. It was adherent to the skull base and internal carotid artery. A radical parotidectomy with resection of the left facial nerve was performed along with a jugular lymph node dissection, without injury to the neurovascular structures of the carotid sheath. The patient convalesced well from surgery with reduction in pain, trismus, and facial swelling. A gold weight was inserted into his left upper eyelid to prevent corneal abrasion.
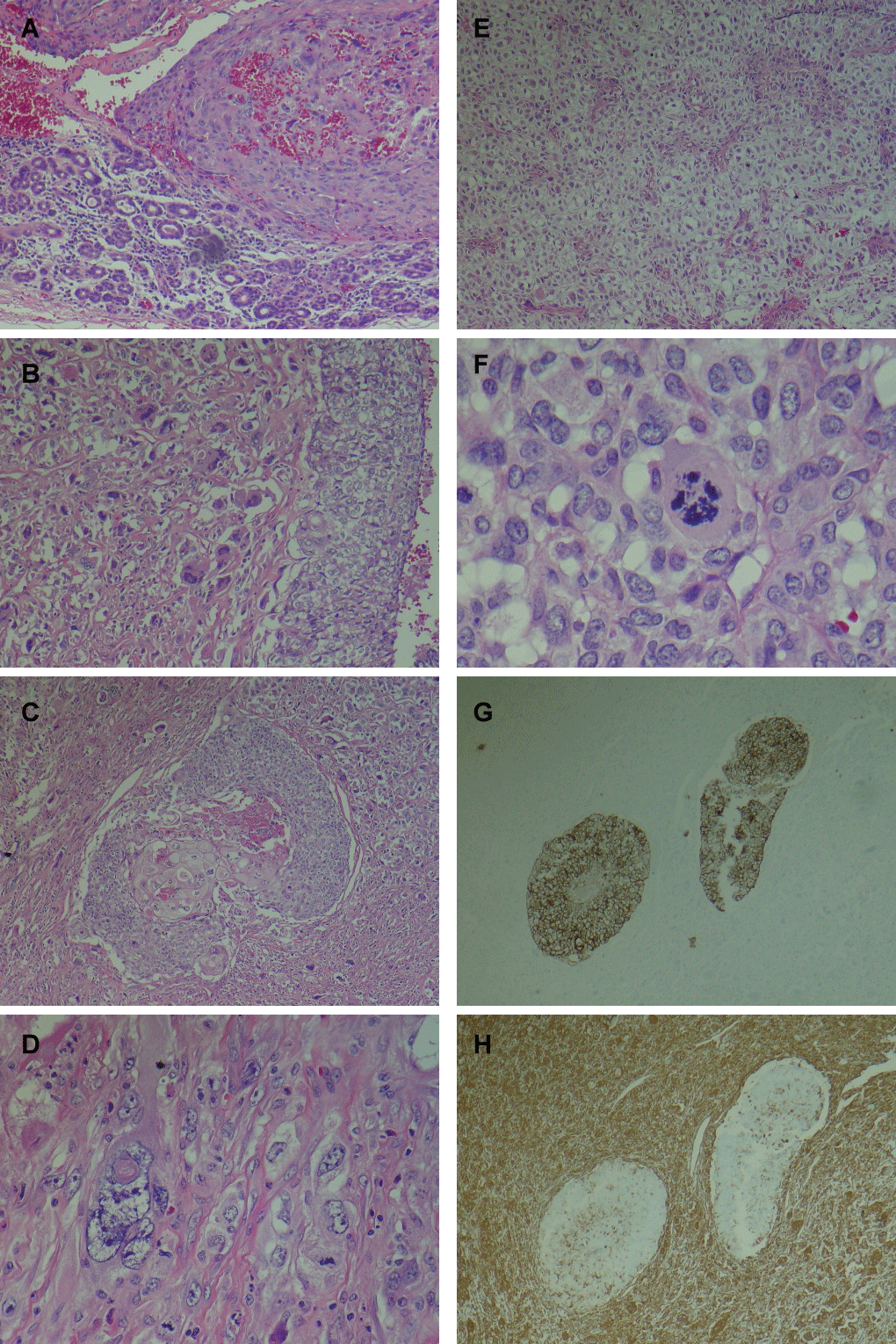
Histological examination shows a malignant biphasic tumor arising in a background of partially atrophic parotid gland. The tumor shows a carcinomatous component with focal squamous differentiation, and a sarcomatous component, with spindle cells, pleomorphic cells, and focal chondroid differentiation. Immunohistochemical staining with pankeratin shows positivity within the epithelial component, while staining with vimentin shows positivity predominantly in the sarcomatous component (Figure 2).
.
Figure 2: (A) Tumor (upper) showing sarcomatoid component, in association with atrophic parotid gland (lower); (B) Biphasic tumor with malignant epithelial component (right) and sarcomatous component (left); (C) Biphasic tumor showing central carcinomatous component with focal squamous differentiation, surrounded by sarcomatous component; (D) Sarcomatoid component with bizarre tumor giant cell (left) and mitosis (right); (E) Chondroid differentiation within sarcomatoid component; (F) Atypical mitosis in sarcomatoid component; (G) Immunohistochemical stain for pankeratin highlights the epithelial component; (H) Immunohistochemical stain for vimentin highlights the sarcomatoid component.
View Figure 2
Based on these results, the patient was referred for post-operative adjuvant chemo-radiotherapy to the left neck and skull base.
Discussion
Carcinosarcoma is an exceptionally rare and poorly differentiated neoplasm characterized by biphasic cell differentiation and possessing both carcinomatous components and malignant mesenchymal elements without definite evidence of transition between the two. It has been described in various organs throughout the body including the upper and lower respiratory tracts [6], gastrointestinal tract [7], urogenital tract [8], breast [9] and skin [10]. Most parotid tumors are benign (80%); however, 20% are malignant [11]. Carcinosarcoma usually represent approximately 0.04% to 0.16% of all salivary gland neoplasms and 0.4% of all salivary gland malignancies.
The clinical presentation of the malignant and the benign parotid tumors are often dissimilar. For benign lesions, the patient will usually present with a painless, palpable mass in the parotid area. Signs of facial nerve involvement, pain, and enlarged lymph nodes may reserve for malignancies. Benign lesions of the parapharyngeal space can often be occult until they reach 5-7 cm in size and present as a neck or tonsil mass. Malignant lesions of the parapharyngeal space are exceedingly rare and include lymphoma, melanoma, metastases, and sarcomas. Nonetheless, the large variation in histological types makes these tumors challenging to diagnose radiographically and clinically. In this case, the patient's early clinical presentation was not suggestive of malignancy, and both CT imaging and fine needle aspiration results were consistent with a benign lesion. At his initial surgery, the cyst fluid was drained and a biopsy of the cyst was obtained because of its adherence to the skull base. It was this small specimen from the cyst wall that demonstrated cells with malignant features. Fine needle aspiration (FNA) is a valuable diagnostic tool; however, if the tumor is located in the deep loop of the parotid, FNA can be challenging, even with CT guidance.
Cross sectional imaging, such as computed tomography (CT) or magnetic resonance imaging (MRI), is standard in the evaluation of parotid lesions. CT is the method of choice in most patients, especially those suspected of having a benign superficial lesion. A CT scan is also helpful to detect calcifications (pre-contrast) and a pattern of enhancement (post contrast). However, MRI is the method of choice for patients with a strong suspicion for malignancy [12]. MRI can identify the exact location of the lesions (superficial or deep lobes), differentiate cystic from solid components, help identify perineural spread, as well as lymphadenopathy. More recently, new MRI technologies, such as dynamic contrast-enhanced (DCE-MRI), diffusion-weighted MRI (DW-MRI) and proton MR spectroscopy (MRS), have shown promising results in the differentiation between benign and malignant salivary gland neoplasm. While pre-operative imaging serves a major role in surgical planning of all salivary neoplasms, in this case there was no evidence of a solid mass on CT or MR, and no features of the cyst, or its fluid, that would suggest malignancy. Thus pointing out the limits of even the most advanced imaging technlogies.
Since the earliest reports of the mixed malignant tumor, research studies have presented several theories as to the different origins of these tumors including the following hypotheses: (1) the "collision theory", i.e., the origin from two distinct malignant cell populations which then unite; (2) the "combination theory", i.e., the origin from a common cell with the ability to differentiate along both lines; (3) the "metaplastic monoclonal or conversion theory", i.e., the origin from metaplastic transformation of a single neoplastic cell type [13]. To analyze the clonality of the different morphologic tumor components, Vekony et al. performed oligonucleotide microarray-based comparative genomic hybridization (oaCGH) on the carcinoma and the sarcoma components separately and found 75% overall homology between the two genomic profiles. These results support the hypothesis that the carcinomatous and sarcomatous components of carcinosarcomas are clonally related.
Clinically, carcinosarcomas are very aggressive, locally recurrent, and, in up to 50% of patients, metastasis via lymphatic and hematological routes to local and regional sites is frequently seen. The mainstay of treatment consists of surgery alone, surgery and postoperative radiotherapy, or surgery with postoperative chemo-radiation depending upon the location and size of the lesion, and nodal status. As in this case, radical parotidectomy results in facial nerve palsy, therefore, subsequent measures have to be taken to protect the affected eye from corneal ulceration. Neck dissection is appropriate in those with lymphadenopathy, and limited neck dissection can be useful in determining occult lymphatic disease. The prognosis often depends on the clinical and histological stage of the disease; however, it is difficult to predict long-term survival due to the low prevalence of the tumor.
Several studies have evaluated the relevance of the presence of p53 in carcinosarcoma. In 1995, Gallo et al. found p53 to correlate with clinical aggressiveness in patients with carcinoma of the parotid gland [14]. Our patient did not have p53 present in his tumor, which is a favorable finding. In 2006, Luukka et al. reviewed a series of 212 patients with carcinosarcoma and discovered a correlation between a five year survival rate and a high volume corrected index (VCI Ki-69) [15]. This evaluation was not performed on our patient. Future research studies are focusing on understanding the pathogenesis of carcinosarcoma, the use of improved immunohistochemistry to stage the disease, and advancements made in molecular genetics to improve our understanding of the clonal origin of this tumor.
Declaration of Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
-
Gnepp DR (1993) Malignant mixed tumors of the salivary glands: a review. Pathol Annu 28 Pt 1: 279-328.
-
Kirklin JW, McDonald JR, Harrington SW, New GB (1951) Parotid tumors; histopathology, clinical behavior, and end results. Surg Gynecol Obstet 92: 721-733.
-
King OH Jr (1967) Carcinosarcoma of accessory salivary gland. First report of a case. Oral Surg Oral Med Oral Pathol 23: 651-659.
-
Liess BD, Hirschi S, Zitsch RP 3rd, Frazier S, Konrad A (2007) Carcinosarcoma of the parotid gland: report of a case with immunohistochemical findings. Ann Otol Rhinol Laryngol 116: 702-704.
-
Velez-Perez A, Huang RS, Everett JM, Guo S, Buryanek J, et al. (2015) Carcinosarcoma of the parotid: investigating its biology with morphoproteomics. Int J Surg Pathol 23: 116-121.
-
Zhou XM, Li P, Zhao L, Shi WJ (2015) Lung Carcinosarcoma Masked by Tracheobronchial Aspergillosis. Intern Med 54: 1905-1907.
-
Cavallin F, Scarpa M, Alfieri R, Cagol M, Ruol A, et al. (2014) Esophageal carcinosarcoma: management and prognosis at a single Italian series. Anticancer Res 34: 7455-7459.
-
Newsom K, Tojuola B, Al-Quran S, Parekattil S, Hamilton W, et al. (2014) Carcinosarcoma of the ureter with a small cell component: report of a rare pathologic entity and potential for diagnostic error on biopsy. Case Rep Pathol 2014: 391615.
-
Mele M, Jensen LL, Vahl P, Funder JA (2015) Breast carcinosarcoma: clinical and pathological features. Breast Dis 35: 211-215.
-
Loh TL, Tomlinson J, Chin R, Eslick GD (2014) Cutaneous carcinosarcoma with metastasis to the parotid gland. Case Rep Otolaryngol 2014: 173235.
-
Bussu F, Parrilla C, Rizzo D, Almadori G, Paludetti G, et al. (2011) Clinical approach and treatment of benign and malignant parotid masses, personal experience. Acta Otorhinolaryngol Ital 31: 135-143.
-
Thoeny HC (2007) Imaging of salivary gland tumours. Cancer Imaging 7: 52-62.
-
McCluggage WG (2002) Malignant biphasic uterine tumours: carcinosarcomas or metaplastic carcinomas? J Clin Pathol 55: 321-325.
-
Gallo O, Franchi A, Bianchi S, Boddi V, Giannelli E, et al. (1995) p53 oncoprotein expression in parotid gland carcinoma is associated with clinical outcome. Cancer 75: 2037-2044.
-
Luukkaa H, Klemi P, Leivo I, Vahlberg T, Grenman R (2006) Prognostic significance of Ki-67 and p53 as tumor markers in salivary gland malignancies in Finland: an evaluation of 212 cases. Acta Oncol 45: 669-675.





