International Journal of Pathology and Clinical Research
Basaloid Salivary Gland-Analogue Tumors of the Mammary Gland: Clinicopathologic Assessment of a Rare Subtype
Andrew L Walls, Lavinia P Middleton, Adel K El-Naggar and Aysegul A Sahin*
Division of Pathology, The University of Texas MD Anderson Cancer Center, Houston, USA
*Corresponding author:
Aysegul A Sahin, MD, Professor/Faculty, Division of Pathology, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd., Houston, TX, 77030, USA, E-mail: asahin@mdanderson.org
Int J Pathol Clin Res, IJPCR-2-042, (Volume 2, Issue 3), Review Article; ISSN: 2469-5807
Received: March 08, 2016 | Accepted: July 28, 2016 | Published: July 31, 2016
Citation: Walls AL, Middleton LP, El-NaggarAK, Sahin AA (2016) Basaloid Salivary Gland-Analogue Tumors of the Mammary Gland: Clinicopathologic Assessment of a Rare Subtype. Int J Pathol Clin Res 2:042. 10.23937/2469-5807/1510042
Copyright: © 2016 Walls AL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Salivary and mammary glands share common ontology, but variable prevalence of tumor subtype frequency. Common primary salivary gland-type tumors are rare and less recognized in breast. Analogous to primary basaloid salivary tumors, basaloid mammary carcinomas, as in primary salivary, exhibit a spectrum of features from benign to malignant. The rarity and unfamiliarity of these tumors may lead to diagnostic and management challenges.
We present the clinicopathologic characteristics of 11 tumors of basaloid salivary-analogue breast carcinomas identified over a 10-year period. All 11 were female patients ranged in age from 34-78 (mean 58 years). Tumor size (gross measurement) ranged from 0.9-6.0 cm (mean 2.6 cm). Two patients had axillary lymph node metastases and one developed a distant metastasis. Three tumors each were estrogen receptor and progesterone receptor positive, and none had amplification of HER2. Follow-up information was available for 9 patients; seven had no evidence of disease at last available follow-up (8-124 months, mean 59 months). One patient had no evidence of disease at 14 months but was subsequently lost to follow-up, one died of unknown causes 17 months after diagnosis and one patient developed lung metastases after 11 months but had no evidence of disease after 124 months of follow-up. Histologically, all tumors showed basaloid tumor nodules with infiltrative borders and extension into surrounding adipose tissue. Two tumors were well differentiated with greater than 75% gland formation, 4 were moderately differentiated with up to 75% gland formation, and 5 were poorly differentiated with less than 10% gland formation.
Conclusions: Malignant basaloid salivary gland-like tumors of the breast are rare and show a spectrum of phenotypic features. The results show that basal-cell adenocarcinomas behave in a manner similar to other salivary gland-type tumors of the breast and have a better outcome than their counterparts in the salivary gland. These tumors resemble other salivary gland-type tumors, including neuroendocrine small cell neoplasms, and may lead to differential diagnostic challenges. Immunohistochemical analysis can be used in the differential diagnosis.
Introduction
Mammary and salivary glands have remarkably similar ontogeny and overlapping features of tumorigenesis [1]. However, different incidence of tumor types exists between the organs. Although ductal-derived carcinomas are the most frequent tumors in mammary glands, glandular-derived tumors are most common in salivary glands. The underlying factors associated with these different tumorigenic patterns are uncertain. Although uncommon, salivary gland-type tumors occur in the mammary gland, and mammary gland-type tumors not uncommonly occur in the salivary gland (i.e., salivary duct and secretory carcinomas). The majority of salivary gland-type tumors of the breast are adenoid cystic carcinomas (ACC) [2], but rare instances of basaloid tumors, have been described [1-3].
Reported basaloid mammary carcinomas manifest a spectrum of histologic, differentiation, and biological behavior. In general, malignant salivary gland-type tumors in the breast have a better prognosis than their counterparts in the salivary gland and other anatomic sites [2,4-6]. The infrequency of basaloid salivary gland-type carcinomas in the mammary gland may lead to differential diagnostic and management difficulties. Central to this issue is whether salivary gland-type tumors in the breast mimic the biologic behavior of their counterparts in the salivary glands. We report a series of rare salivary gland-type tumors with basaloid morphology in mammary glands and discuss their differential diagnosis and clinical behavior.
Materials and Methods
We searched the pathology database at our institution to identify all basaloid salivary gland-type tumors diagnosed from 2003 through 2013, identified 11 patients with this diagnosis. Eight tumors were received from outside institutions, and 3 were initially diagnosed locally. Histologic features of each tumor were evaluated by breast and head and neck pathologists independently for purposes of this study. Clinical data and pathologic slides, and in some cases paraffin blocks, were provided by referring pathologists and other physicians. All tissue was formalin-fixed and paraffin-embedded (FFPE) with hematoxylin and eosin (H&E) staining. Some cases included immunohistochemical stains from the referring institution, and in other cases initial or additional immunohistochemical analysis was performed at our institution. Institutional Review (IRB) approval was obtained for the study.
For tumors in which materials were available, immunohistochemical analysis was performed using a polymeric biotin-free horseradish peroxide method with a Bond Max stainer (Leica Microsystems, Bannockburn, IL) for all markers except epidermal growth factor receptor (EGFR). Unstained tissue sections 4 μm thick were prepared from FFPE sections and placed in the automated Bond Max stainer. Antibody source, clone, dilution, and antigen retrieval methods are given for each antibody in table 1. The Refine Polymer Detection kit was used for immunostaining, and 3,3-diaminobenzidine was used as a chromogen. Slides were counterstained with Mayer's hematoxylin. Antibody stains were evaluated using known positive and negative tissue controls. For EGFR immunohistochemical staining, the polymeric biotin-free horseradish peroxide method was used with a Dako Autostainer. Unstained sections 4 μm thick were prepared from FFPE sections. Enzyme retrieval was performed with Dako Proteinase K for 10 minutes, and slides were incubated for 1 hour with EGFR antibody (Table 1). Thirty-minute incubation with the mouse EnVision kit (Dako) was used for secondary antibody staining. An EnVision Flex Detection kit (Dako) was used for detection of bound antibody, with 3,3-diaminobenzidine serving as the chromogen (Dako). Slides were counterstained with Mayer's hematoxylin. Known positive and negative tissue controls were included in the analysis.
![]()
Table 1: Antibody source, clone, and dilution and antigen retrieval method for immunohistochemical analysis.
View Table 1
Clinical and pathologic data and follow-up information were extracted from pathology reports and patients' medical records and for those outside sources by direct contact with the corresponding institutions.
Results
Clinical presentation
Table 2 presents a summary of clinical and pathological characteristics. The age of the 11 female patients ranged from 34-78 (mean 58 years). One 51-year-old woman had a history of invasive ductal carcinoma of the breast 5 years prior to her diagnosis of basaloid breast cancer, and one had asynchronous contralateral invasive ductal breast carcinoma, as well as a history of high-grade serous carcinoma of the ovary 3 years prior to her diagnosis of basaloid breast cancer. A third patient, a 61-year-old woman, had a history of gastric non-Hodgkin lymphoma prior to her diagnosis of basaloid breast cancer. The remaining patients had no known history of prior malignancy, and none of the patients were known to have a history of a salivary gland tumor or a tumor of basaloid morphology at any other site. A 34-year-old woman had tested positive for a BRCA2 mutation.
![]()
Table 2: Clinical and pathological characteristics in 11 cases.
View Table 2
Gross appearance
Six tumors were in the right breast, and 5 in the left. All tumors presented as a solitary mass. Three tumors with mammographic studies were reported as a high-density mass, with "microlobulated" margins in one caseand "lobular" margins in the other 2. The tumors arose in different quadrants of the breast and ranged in size from 0.9 cm to 6.0 cm (mean 2.6 cm).
Microscopic pathologic findings
All tumors were unicentric, with microscopically infiltrative borders and extension into the surrounding adipose tissue. All tumors were composed of basaloid tumor cells forming nodules and nests, with peripheral palisading. The basaloid cell population consisted of small- to medium-sized uniform cells with scant to moderate, pale, eosinophilic or amphiphilic cytoplasm; ill-defined cell borders; and round to oval nuclei with indistinct nucleoli.
All tumors had variable degrees of glandular structures (Figure 1, Figure 2 and Figure 3). Two cases were well differentiated with the majority of each tumor formed of small uniform basaloid cells and > 75% gland formation. Four tumors showed moderately enlarged cells with more prominent nuclei and less than 50% gland formation. Five cases had poorly differentiated areas of solid growth with, at most, rare residual gland formation and large solid sheets of cells with necrosis. One of these poorly differentiated cases contained a prominent area of squamous differentiation with basaloid squamous features, including high-grade nuclei and prominent necrosis. The basaloid squamous nests could be seen emanating from the rare residual glands.
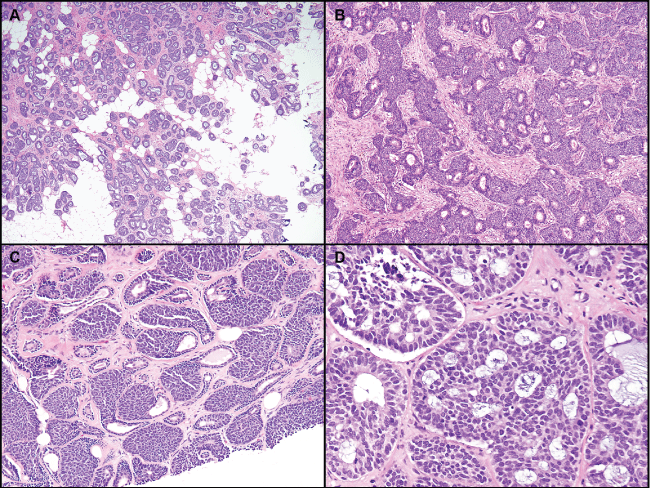
.
Figure 1: Well or moderately differentiatedbasaloid tumors, with various degrees of gland formation. A) Prominent gland formation with infiltration into adipose tissue; B,C) Small nests of basaloid cells with peripheral palisading surrounding and arising from glands; D) Case with features focally overlapping with adenoid cystic carcinoma. Some peripheral palisading is still apparent in this area.
View Figure 1
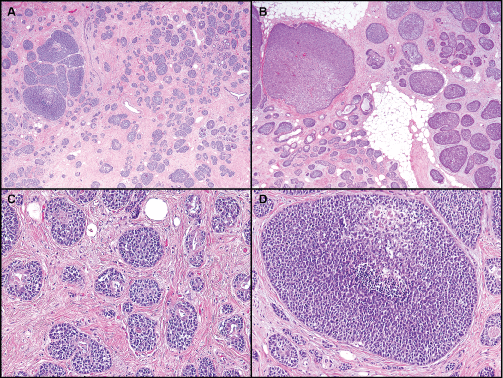
.
Figure 2: Basaloid tumors with intratumoral heterogeneity. A) Mixture of small nests with some gland formation and clusters of larger solid nests; B) Mixture of small and large solid nests. Peripheral palisading is prominent; C) Residual gland formation with surrounding basaloid proliferation; D) Larger nest of basaloid cells with focal necrosis.
View Figure 2
Mitoses ranged from 1 to 29 per 10 high-powered fields (HPF) (mean 12.2), with 5 cases having less than 10/10 HPF and 6 cases having more than 10/10 HPF. The cases with the highest mitotic rates corresponded with the poorly differentiated cases with predominantly solid growth.
Immunohistochemistry
Table 3 presents the immunohistochemical findings. Two of the 10 cases tested were estrogen receptor (ER)- and progesterone receptor (PR)- positive, one was ER-positive only, and one was PR-positive only. None of the 10 cases tested for HER2 protein over expression were positive (less than or equal to 1+ by immunohistochemistry or non amplified for Her 2 by fluorescence in situ hybridization). Overall, 6 cases were triple-negative. The case that was not tested for HER2/neu amplification was ER/PR-negative.
![]()
Table 3: Immunohistochemical data, including hormone receptor and HER2/neu status.
View Table 3
Immunohistochemical staining for p63 and cytokeratin 5/6 was variable. Five of 8 cases were negative for p63, and one showed staining only in the areas of squamous differentiation. In one case, heterogeneous staining in a minority of the cells was noted, in another case, a positive peripheral staining in small nests was noted (Figure 4A). Four of 6 cases were positive for cytokeratin 5/6, but all positive cases showed only patchy staining and no diffuse positivity (except in areas of squamous differentiation, as with p63) (Figure 4B).
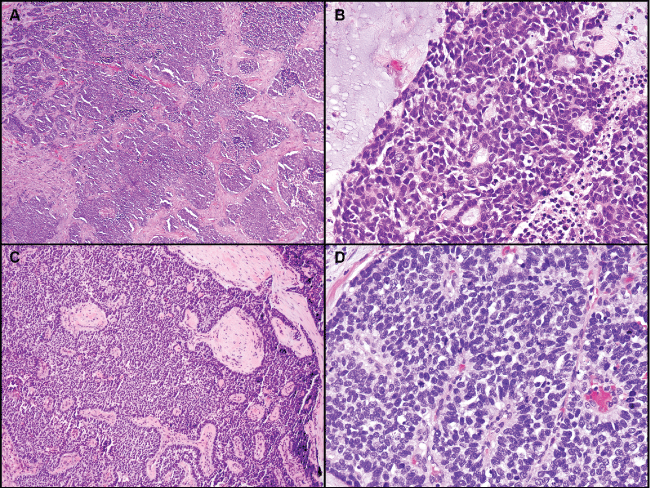
.
Figure 3: Poorly differentiated basaloid tumors. A) Low power view showing irregular solid nests of basaloid tumor cells; B) Focal residual gland formation within solid nests; C) Prominent peripheral palisading is still present within solid growth; D) Rosette-like structures and mitotic figures are present. As in all of the cases tested, neuroendocrine markers were negative.
View Figure 3
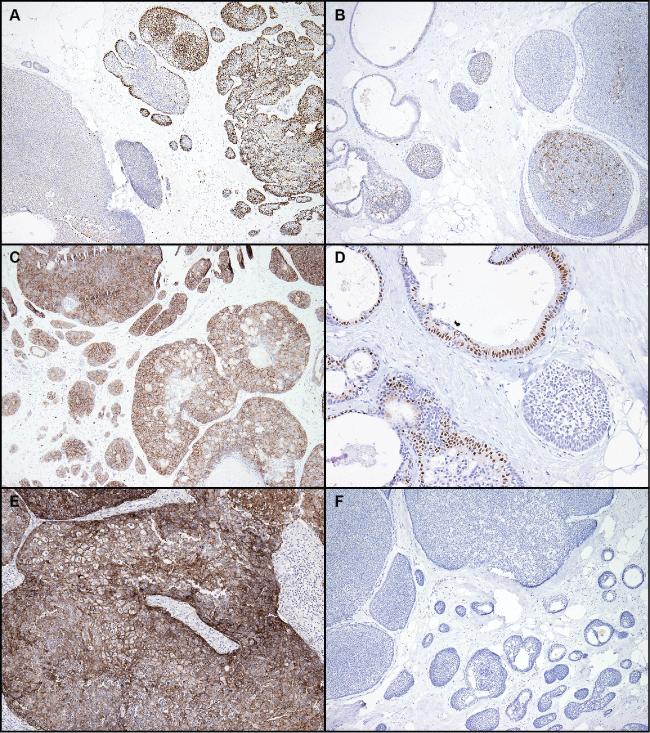
.
Figure 4: Immunohistochemical staining. A) Variable staining for p63; this case showed the strongest p63 staining; B) Patchy staining for cytokeratin 5/6; C) Strong positive staining for c-kit. Other cases showed weak or no staining; D) Variable staining for GATA-3; E) No EGFR staining in tumor cells; F) No synaptophysin staining. All cases tested for synaptophysin and chromogranin were negative.
View Figure 4
Staining for C-kit was generally negative, except for 1 case with strong membranous staining (Figure 4C). GATA-3 staining was positive in 3 of 6 cases tested (Figure 4D). Three cases showed only focal weak staining. Staining for EGFR was strongly positive in one tumor with squamous features (Figure 4E). Staining for the neuroendocrine markers synaptophysin and chromogranin was negative in all of the 10 cases tested (Figure 4F).
Treatment and follow-up
All patients were surgically treated, six with local excision, 5 patients underwent mastectomy, two patients had modified radical mastectomies, and 3 had simple mastectomies.
Five patients had adjuvant chemotherapy, and 3 patients received neoadjuvant chemotherapy. One patient experienced progression of tumor growth while receiving neoadjuvant chemotherapy, and one is currently receiving adjuvant chemotherapy at the time of this study. Five patients received radiation therapy. No treatment information was available for 3 patients.
Of the 10 patients with axillary lymph node dissection, two had axillary metastases. Three patients had only sentinel lymph nodes examined (1 node in 1 patient and 4 nodes in 2 patients), and all were negative. Seven patients had an axillary dissection, and the number of lymph nodes examined ranged from 8 to 36 (in 1 patient, the number of nodes removed is not known). One of the patients had 3 sentinel nodes biopsied, 2 of which were positive for micrometastases. One of the patients with axillary dissection showed lymph node metastases, with 7 of 33 nodes positive.
Follow-up information was available for 9 patients. Seven patients had no evidence of disease during their follow-up periods, which ranged from 8 to 124 months (mean 59 months). One patient had no evidence of disease at 14 months but was subsequently lost to follow-up. One patient developed non-small cell lung cancer with brain and spine metastases and died 17 months after her breast cancer diagnosis. It should be noted that this patient had 7 axillary lymph nodes with metastatic disease. Additionally, this patient's tumor had the highest stage (pT3N2, stage IIIA) of the 11 examined cases. One patient had a history of ipsilateral invasive ductal carcinoma 5 years prior to her basaloid breast cancer and then developed lung metastases 11 months after her diagnosis of basaloid breast cancer but has no evidence of disease at 124 months of follow-up after additional treatment for her metastatic disease. Material from the lung was not available for review but was reported to be consistent with metastases from a breast primary.
Discussion
We present a series of rare mammary carcinoma mimicking salivary basaloid adeno-carcinoma. All tumors displayed malignant histologic features of basal cells forming glands and solid nests of cells with microscopically infiltrative borders and tumor extension into surrounding adipose tissue. Within these common characteristics, a spectrum of histologic of features have been found to be useful in delineating differentiated from less differentiated tumors. Nonetheless, this morphologic spectrum does not appear to impact tumor behavior as in salivary gland-type tumors. Because these tumors also resemble other malignant breast tumors with basaloid features and poorer prognosis, their recognition and differentiation from other similar appearing tumors may be important for treatment and prognosis. To our knowledge, this is the largest case series reported of this type of tumor.
Other case series of basaloid salivary gland-type breast tumors have been reported. Shin and Rosen reported 9 cases of solid variants of mammary ACC with basaloid features [7-9]. With a mean follow-up of 32 months, 6 patients had no evidence of disease, 1 died of unknown causes, and 1 was lost to follow-up. Lamovec, et al. reported 9 cases of basaloid carcinoma of the breast with one death due to disease and another with metastatic invasive ductal carcinoma [10]. Three cases had nodal metastases.
Salivary gland-type tumors of the breast have been reported to have a relatively good prognosis compared to their salivary gland counterparts [5,6]. Experience with basaloid salivary gland-type tumors, however, is limited. Basal cell adenocarcinoma of the major salivary glands was first described in 1990 [11] and further described by Muller and Barnes in 1996 [12]. In 2012, Flynn, et al. [13] described 3 cases of basal cell adenocarcinoma arising in salivary gland metaplasia of the breast. All 3 patients were free of disease at 15, 23, and 25 months of follow-up. Although current evidence suggests that these tumors pursuea good outcome, the finding of axillary and distant metastases in some cases heightens the potentially aggressive nature of this entity [9,10,13]. Similarly, patient outcomes in our series were generally less aggressive, with no deaths in patients with sufficient follow-up information. However, two patients had lymph node metastases and one experienced distant metastasis. Two of these patients are alive at last follow-up. The common branching morphogenesis underlying the development of both mammary and salivary glands provides a unifying concept for the occurrence of salivary gland-type tumors in the breasts. However, important differences between the organs may explain the different behavior of salivary gland-type tumors in the two locations. Malignant tumors in the salivary glands, including ACC, generally follow a protracted course, but local/regional recurrence is, unfortuntely, common and results in poor long-term outcomes [3]. The limited spaces and complex anatomic structures of head and neck coupled with the propensity of the tumors to track along peripheral nerves results in difficulty achieving adequate negative margins and complete excision. In contrast, tumors involving the breast are amenable to complete excision and sparing of adjacent structures. Although salivary gland-type tumors of the breast certainly carry the possibility of regional lymph node and distant metastases, they are less frequently aggressive based on the limited numbers reported, although experience with these tumors is limited. Accordingly, standard management of less aggressive variants of breast carcinoma should be adequate for these tumors, whereas more aggressive treatment with concomitant adverse effects may not be helpful and can be avoided.
Interestingly, other salivary gland-type tumor of the breast may display overlapping features with basaloid adenocarcinoma, especially the solid-variant of ACC. This is identified by the presence of areas of cribri form morphology identical to those of ACC. Basal cell carcinoma, however, shows dense eosinophilic material in the cribri form spaces in contrast to light mucopolysaccharide material in ACC. Furthermore, all of the tumors in this series displayed significant peripheral palisading of basaloid cell nests. Nonetheless, a definitive distinction between these two categories may not be reached in the mammary gland context.
Basaloid salivary gland-type breast tumors may also show overlapping histologic features with other primary tumors of the breast. The differential diagnosis of a basaloid breast carcinoma may include a poorly differentiated ductal carcinoma, neuroendocrine carcinoma, and metaplastic carcinoma. Other diagnostic considerations are most important when the prognosis and behavior of the tumors are markedly different. Poorly differentiated ductal carcinoma, neuroendocrine carcinoma, and some metaplastic carcinomas of the breast have a relatively poor prognosis. None of our cases showed immunohistochemical evidence of neuroendocrine differentiation. A second issue is that of "triple negative" breast carcinoma, which is typically associated with poorly differentiated carcinomas that lack targeted therapy options and have a poor outcome [14]. In the breast, most salivary gland-type tumors are negative for hormone receptors and HER 2 overexpression [15]. It is important to distinguish these salivary gland-type tumors from other triple-negative and "basal-like" breast cancers with poorer prognoses and recognize that less aggressive treatment may be appropriate. Interestingly, not all of the tumors in our series were triple-negative. None of the tumors showed HER2 overexpression, but 4 tumors showed some combination of hormone receptor positivity. This suggests a level of heterogeneity at the molecular level within this group of tumors and supports routine testing for these prognostic/predictive markers in all breast cancers so that targeted therapy may be pursued when appropriate.
The results of our study along with those previously reported suggest that overall, salivary gland-type tumors of the breast, including basaloid tumors like those presented here, have a generally good prognosis. However, one of the (triple negative) cases progressed while the patient was receiving neoadjuvant chemotherapy. This progression suggests that neoadjuvant chemotherapy may not be helpful and that similar cases might best be treated by emphasizing complete excision, whether by partial or complete mastectomy, with adjuvant chemotherapy and radiation therapy and axillary exploration handled on a case by case basis, as would usually be done for a conventional breast carcinoma of similar stage. However, one must be cautious drawing conclusions based on a single case.
In summary, malignant basaloid salivary gland-analogue tumors of the breast are rare tumors that present with a spectrum of differentiation. Despite the morphologic spectrum, these tumors appear to behave in a manner similar to other salivary gland-type tumors of the breast and generally have a better outcome than their counterparts in the salivary gland. Although distinguishing these tumors from others, such as neuroendocrine carcinoma and other triple-negative invasive ductal carcinomas, is important owing to their differences in behavior and outcome, distinguishing these basaloid tumors from other salivary gland-type tumors such as ACC may not be strictly necessary because outcomes are uniformly good with conventional treatment.
References
-
Bennett AK, Mills SE, Wick MR (2003) Salivary-type neoplasms of the breast and lung. Semin Diagn Pathol 20: 279-304.
-
Pia-Foschini M, Reis-Filho JS, Eusebi V, Lakhani SR (2003) Salivary gland-like tumours of the breast: surgical and molecular pathology. J Clin Pathol 56: 497-506.
-
Ellis GL, Auclair PL (2008) Tumors of the Salivary Glands. AFIP Atlas of Tumor Pathology, Fourth Series, In: SG Silverberg, LH Sobin, 9, Silver Spring, Maryland: American Registry of Pathology.
-
Li N, Xu L, Zhao H, El-Naggar AK, Sturgis EM (2012) A comparison of the demographics, clinical features, and survival of patients with adenoid cystic carcinoma of major and minor salivary glands versus less common sites within the Surveillance, Epidemiology, and End Results registry. Cancer 118: 3945-3953.
-
Ghabach B, Anderson WF, Curtis RE, Huycke MM, Lavigne JA, et al. (2010) Adenoid cystic carcinoma of the breast in the United States (1977 to 2006): a population-based cohort study. Breast Cancer Res 12: R54.
-
Kulkarni N, Pezzi CM, Greif JM, Suzanne Klimberg V, Bailey L, et al. (2013) Rare breast cancer: 933 adenoid cystic carcinomas from the National Cancer Data Base. Ann Surg Oncol 20: 2236-2241.
-
Foschini MP, Krausz T (2010) Salivary gland-type tumors of the breast: a spectrum of benign and malignant tumors including "triple negative carcinomas" of low malignant potential. Semin Diagn Pathol 27: 77-90.
-
Cox KL, Korourian S, Klimberg VS (2009) Unusual tumors of the breast. Curr Probl Surg 46: 514-590.
-
Shin SJ, Rosen PP (2002) Solid variant of mammary adenoid cystic carcinoma with basaloid features: a study of nine cases. Am J Surg Pathol 26: 413-420.
-
Lamovec J, Falconieri G, Salviato T, Pizzolitto S (2008) Basaloid carcinoma of the breast: a review of 9 cases, with delineation of a possible clinicopathologic entity. Ann Diagn Pathol 12: 4-11.
-
Ellis GL, Wiscovitch JG (1990) Basal cell adenocarcinomas of the major salivary glands. Oral Surg Oral Med Oral Pathol 69: 461-469.
-
Muller S, Barnes L (1996) Basal cell adenocarcinoma of the salivary glands. Report of seven cases and review of the literature. Cancer 78: 2471-2477.
-
Flynn C, Kadivar M, Kwon MY, Bonzanini M, Haines GK, et al., (2012) Basal cell adenocarcinoma arising in salivary gland metaplasia of the breast: a novel salivary gland-type tumor developing in the breast. Hum Pathol 43: 1764-1771.
-
Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, et al. (2004) Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10: 5367-5374.
-
Reyes C, Jorda M, Gomez-Fernandez C (2013) Salivary gland-like tumors of the breast express basal-type immunohistochemical markers. Appl Immunohistochem Mol Morphol 21: 283-286.





