International Journal of Pathology and Clinical Research
Aortopathy in Bicuspid Aortic Valve and Marfan Syndrome is Characterized by a Lack of Activation Potential of the Epicardium in the Ascending Aorta
Nimrat Grewal1,2, Marie-José Goumans3, Marco C DeRuiter2, Robert JM Klautz1, Robert E Poelmann4, Barbara JM Mulder5, Ad JJC Bogers6, Adriana C Gittenberger-de Groot2,7*
1Department of Cardiothoracic Surgery, Leiden University Medical Center, Leiden, the Netherlands
2Department of Anatomy and Embryology, Leiden University Medical Center, Leiden, the Netherlands
3Department of Molecular Cell Biology, Leiden University Medical Center, Leiden, the Netherlands
4Department of Biology, Integrative Zoology, Leiden University, Leiden, the Netherlands
5Department of Cardiology, Academic Medical Center, Amsterdam, the Netherlands
6Department of Cardiothoracic Surgery and Heart Valve Bank, Erasmus University Medical Center, Rotterdam, the Netherlands
7Department of Cardiology, Leiden University Medical Center, Leiden, the Netherlands
*Corresponding author:
Adriana Gittenberger de Groot, Department of Cardiology, Leiden University Medical Center, Postal zone: S-5-24, P.O. Box 9600, 2300 RC Leiden, Netherlands, Tel: +31-71-526-3704/9306, Fax: +31-71-526-6809, E-mail: acgitten@lumc.nl
Int J Pathol Clin Res, IJPCR-3-051, (Volume 3, Issue 1), Original Article; ISSN: 2469-5807
Received: August 26, 2016 | Accepted: February 17, 2017 | Published: February 20, 2017
Citation: Grewal N, Marie-José G, DeRuiter MC, Klautz RJM, Poelmann RE, et al. (2017) Aortopathy in Bicuspid Aortic Valve and Marfan Syndrome is Characterized by a Lack of Activation Potential of the Epicardium in the Ascending Aorta. Int J Pathol Clin Res 3:051. 10.23937/2469-5807/1510051
Copyright: © 2017 Grewal N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Patients with a bicuspid aortic valve (BAV) and Marfan syndrome (MFS) have increased susceptibility for development of aortopathy. In the heart, epicardial cells expressing Wilms tumor suppressor protein (Wt1) are known to become activated after myocardial infarction. We hypothesize that epicardium covering the aorta might show a similar response in BAV and MFS in pathologic conditions.
Methods: Non- and dilated ascending aorta specimen of BAV (n = 36), tricuspid aortic valve (TAV) (n = 23), and MFS (8) were investigated. The aorta was studied by immunohistochemistry and immunofluorescence, using Wt1 and endothelial nitric oxide, which regulates Wt1 expression. Functionality and therefore activity of Wt1 was confirmed by co-expression with retinaldehyde dehydrogenase-II enzyme in the same cells.
Results: The endothelium did not show a difference in the expression of the markers between the groups. In the aortic media Wt1 and eNOS expression were significantly higher in the TAVs as compared to the BAVs and MFS. All groups exhibited Wt1 positive cells in a quiescent stage in the epicardial cells, whereas only the dilated TAVs showed a significant higher Wt1 expression (p < 0.05) in the activated cuboid epicardial cells.
Conclusion: In all non-dilated patients, a baseline expression of Wt1 is present in epicardial and vascular smooth muscle cells. A significant increase in Wt1 activity is solely seen in the dilated TAV. Immature vascular smooth muscle cells and absence of pathologic features of cardiovascular ageing in BAV and MFS might explain the observed lack of activation potential in these patients.
Keywords
Bicuspid aortic valve, Marfan syndrome, Aorta, Aneurysm, Basic science
Introduction
Patients with a bicuspid aortic valve (BAV) and patients with Marfan syndrome (MFS) carry an increased risk for aortic dilation as compared to patients with a tricuspid aortic valve (TAV). Earlier research has shown that both syndromes have similarities in the aortic media architecture, characterized by an increased MMP activity, decreased fibrillin-1 expression, immature vascular smooth muscle cells (VSMCs) and lack of features of cardiovascular ageing in the dilated aorta [1-4]. This is in contrast to the aortic wall in TAV, which harbours differentiated VSMCs and shows a significant increase in expression of progerin [4], a marker of cardiovascular ageing in the dilated aorta [5,6]. A different pathobiological mechanism thus seems to underlie aortic wall pathology in BAV and MFS as compared to the TAV, probably superimposed on the immature state of the aortic media.
Research on aortic dilation in BAV, MFS and TAV has till date mainly approached the media or adventitia of the vessel wall [7,8]. In this study we sought to investigate the outermost covering of the ascending aorta, the epicardium, which has not received much attention so far. It has been shown that epicardial cells fulfil many important functions in the heart, during embryogenesis and adult life [9-11]. During the development of the heart, the epicardium undergoes a process called epithelial-to-mesenchymal transition. During epithelial-to-mesenchymal transition the morphologic appearance of the epicardial cells alter from squamous to cuboidal and the basement membrane dissolves. The epicardial cell delaminates, becoming an epicardium derived cell which invades the subepicardial layer and subsequently migrates into the myocardium [9,12]. Epicardium derived cells are mandatory for the development of the VSMCs of the coronary vasculature and contribute to the atrioventricular valves (reviewed: [9,13]). A large proportion of epicardium derived cells differentiate into the cardiac interstitial fibroblasts of the heart, contributing to the fibrous skeleton. In adult life the epicardium and epicardium derived cells are in a quiescent stage, but it has been shown that these inactive cells can become active in response to pathological processes such as myocardial ischemia [14].
As BAV and MFS patients are highly susceptible for aortic complications, we postulate that a similar activation reaction of the arterial epicardium is plausible, whereby aortic wall pathology drives the activation of these cells.The activation of the epicardial cells might therefore serve as an early marker for future aortopathy, which could be assessed clinically to identify patients at risk of aortopathy.
To address this hypothesis, we investigated the ascending aortic wall on the expression of Wt1, as it has been shown that in the developing heart, WT1 is required for proliferation of vascular progenitors arising from the epicardium [15-17]. Also, that WT1 is required for maintenance of the undifferentiated state in these progenitors [16]. We also studied a retinoic acid (RALDH2) marker, as RALDH2 is under direct transcriptional control of WT-1 and thereby an indicator for functional activity of Wt1 [18]. RALDH2 is further identified as a specific marker for the epicardium derived from the second heart field [19]. It has also been shown that endothelial nitric oxide (eNOS) regulates the expression of Wt1, and a deficiency of eNOS inhibits epicardium derived cell migration [20]. Hence, we also investigated the expression of eNOS in the aortic wall layers and its relation to the Wt1 expression.
Methods
Ascending aortic wall samples were obtained from individuals with BAV and TAV, both with and without dilation and individuals with MFS, without dilation. Dilation was clinically defined by reaching an ascending aortic wall diameter of > 45 mm [1,21]. Sample collection and handling was carried out according to the official guidelines of the Medical Ethical Committee of the Leiden University Medical Centre, Leiden, the Netherlands and the code of conduct of the Dutch federation of Biomedical Scientific Societies. The Heart Valve Bank, Thoraxcentre, Erasmus Medical Centre, Rotterdam, provided 6 BAV samples without aortic dilation as these were not suitable for transplantation, as approved by their Scientific Advisory Board. Furthermore the Academic Medical Centre provided us with 8 MFS biopsy specimen, with approval of the Medical Ethical Committee. The study population was divided in 5 groups: 1) TAV without ascending aorta dilation; 2) TAV with ascending aorta dilation; 3) BAV without ascending aorta dilation; 4) BAV with ascending aorta dilation; 5) MFS without ascending aorta dilation, termed MFS (Table 1).
![]()
Table 1: Patient characteristics are shown in table 1. TA: Tricuspid Valve, without Dilation; TAD: Tricuspid Valve, with Dilation; BA: Bicuspid Valve, without Dilation; BAD: Bicuspid Valve, with Dilation; BAV: Bicuspid Aortic Valve; MFS: Marfan Syndrome, without Dilation; LUMC: Leiden University Medical Center, EMC: Erasmus Medical Center, AMC: Academic Medical Center.
*data unavailable, clinically defined as non-dilated by pathologist, **data unavailable for 5 patients, clinically defined as non-dilated by pathologist, ***aortic root diameters unavailable.
View Table 1
Following excision, all specimen were fixed in 4% formalin for 24 hours, decalcified in Kristensen's solution (a formic acid buffer) for 120 hrs and subsequently embedded in paraffin. Transverse sections (5 µm) were mounted on pre-coated Starfrost slides (Klinipath B.V., 3057-1, Duiven, The Netherlands) to allow comparing different expression profiles on sequential sections.
Immunohistochemical staining was performed following the protocol for deparaffinization, antigen retrieval and staining as described in our previous studies [1,21]. Primary antibodies applied to the slides were Wt1 1/300 (Product number CA1026, Calbiochem, Billerica, USA), RALDH2 1/100 (Product number ab75674, Abcam, Cambridge, UK) and eNOS 1/100 (Product number, PA1037, Thermo scientific, Rockford, USA). Secondary antibodies applied were: 1/200 goat-anti-rabbit-biotin, (Product number BA-100, Vector Laboratories, Burlingame, USA) and 1/66 normal goat serum, (Product number S1000, Vector Laboratories, Burlingame, USA).
To detect co-expression, we performed double immunofluorescent stainings. Sections were deparaffinated, rehydrated and subjected to antigen retrieval as described [1,21]. Tissue sections were incubated with primary antibodies Wt1 1/100 (Product number MAB4234, Chemicon, Billerica, USA), RALDH2 1/100 and eNOS 1/100 overnight (4 °C), followed by incubation with secondary antibody Cy3 donkey anti mouse IgG (Product number 715-165-150 Jackson Immunoresearch, 715-165-150) for Wt1 and Alexa Fluor 647 donkey anti-rabbit IgG (Invitrogen, A-31573) for eNOS (1 hr, 20 °C). Cy3 and Alexa Fluor 647 were preferred secondary antibodies, because of green autofluorescence of the elastic lamellae. Nuclei were visualized with DAPI (4',6-diamidino-2-phenylindole, Sigma-Aldrich). Finally, slides were mounted with ProlonGold (Invitrogen, P36930).
Sections were studied with a Leica BM5000 microscope equipped with plan achromatic objectives (Leica Microsystems, Wetzlar, Germany).
As handling during excision of aortic tissue can easily damage epicardial cells, we first identified Wt1 stained specimen in which the adventitia showed an intact monolayer of epicardial cells: non-dilated TAV N = 4 (total N = 11), dilated TAV N = 4 (total N = 12), non-dilated BAV N = 4 (total N = 17), dilated BAV N = 8 (total N = 19) and MFS N = 2 (total N = 8). The number of MFS patients was too small to allow an adequate statistical analysis of the epicardial cells. Analysis of epicardial Wt1, RALDH2 and eNOS expression was therefore solely performed on the non- and dilated BAV and TAV groups. In the MFS group, only the morphologic appearance of the epicardial cells could be described.
To determine the level of expression of the Wt1, eNOS and RALDH2 markers in the aortic media of BAV, TAV and MFS patients, the number of positively stained nuclei (for Wt1 and eNOS) and cytoplasmic positive cells (for RALHD2) was counted using ImageJ on 3 predetermined locations (left, middle and right) of every section. These we refer to as 'microscopic fields' and were maintained in evaluation of all sections. A threshold was applied to filter background noise. The total number of cells (positively stained and negative nuclei and cytoplasm) was not different in all specimens. In each microscopic field the number of cells positive for Wt1, RALDH2 and eNOS was therefore normalized to the total number of cells per 105 μm2. Finally, the number of normalized positive cells for each staining was averaged between the 3 microscopic fields. All specimens were re-evaluated by an independent, experienced histopathologist who was blinded to the clinical data.
All numerical data are presented as mean ± SD of 3 fixed microscopic fields on each stained slide. Statistical differences were evaluated with the Mann-Whitney U-test for comparison between the groups. Significance was assumed when p < 0.05 using SPSS 20.0 software program (SPSS Inc. Chicago, USA). We have performed a 1, 2 and 3 way ANCOVA test to correct for age and gender. Graphpad software was used to create graphics of statistical analysis.
Results
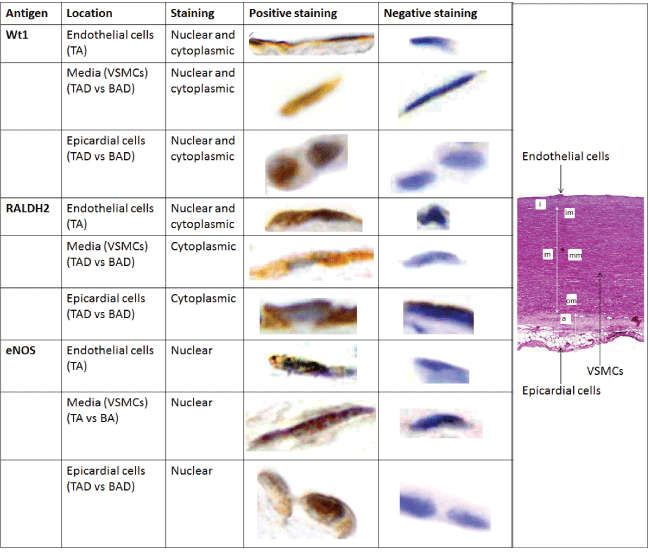
The aortic wall consists of 3 layers: the tunica intima lined by endothelium, media and adventitia lined by arterial epicardium. Expression of Wt1, RALDH2 and eNOS was seen in all 3 layers, as will be presented in the following paragraphs. Figure 1 gives an overview of the markers was expressed the endothelial cells, VSMCs and the epicardial cells, positive and negative staining is indicated, either cytoplasmic or nuclear. Figure 1 also shows a complete overview of the aortic wall to indicate the location of the stained cells.
.
Figure 1: An overview is given of the stained markers: Wilms tumor suppressor protein (Wt1), Retinaldehyde dehydrogenase-II (RALDH2) and endothelial nitric oxide (eNOS), in the endothelial cells, vascular smooth muscle cells (VSMCs) and the epicardial cells. Positive and negative staining is indicated, either cytoplasmic or nuclear.
In the last column a transverse histologic section (5μ) of the aortic wall of a non-dilated tricuspid aortic valve, stained for hematoxylin eosin is shown to indicate the location of the stained cells. The figure indicates the intimal layer (i) lined with endothelial cells; the medial layer (m) with vascular smooth muscle cells (VSMCs), subdivided in the inner media (im), middle media (mm) and outer media (om); and the adventitial layer (a) where epicardial cells are located.
Magnification: 100x.
View Figure 1
Endothelial cells lining the luminal surface of the tunica intima stained predominantly positive for Wt1 in the nucleus. Morphologic appearance of all Wt1 positive cells was similar, being squamous. We analysed the total Wt1 positive population in each sample of the 5 groups and calculated the ratio of Wt1 positive compared to Wt1 negative cells. No significant difference was seen between the 5 groups (Figure 1 and Table 2).
![]()
Table 2: An overview of the results and the statistical significance in the endothelial cells. Wilms Tumor suppressor protein: Wt1, Retinaldehyde Dehydrogenase-II: RALDH2, endothelial Nitric Oxide: eNOS. TA: Tricuspid Valve, without Dilation; TAD: Tricuspid Valve, with Dilation; BA: Bicuspid Valve, without dilation; BAD: Bicuspid Valve, with Dilation; MFS: Marfan Syndrome, without dilation. *mean ratio positive/negative cells.
View Table 2
All Wt1 positive endothelial cells also showed eNOS and RALDH2 expression, with no difference in the ratio of positive compared to negative cells between the 5 groups (Figure 1 and Table 2).
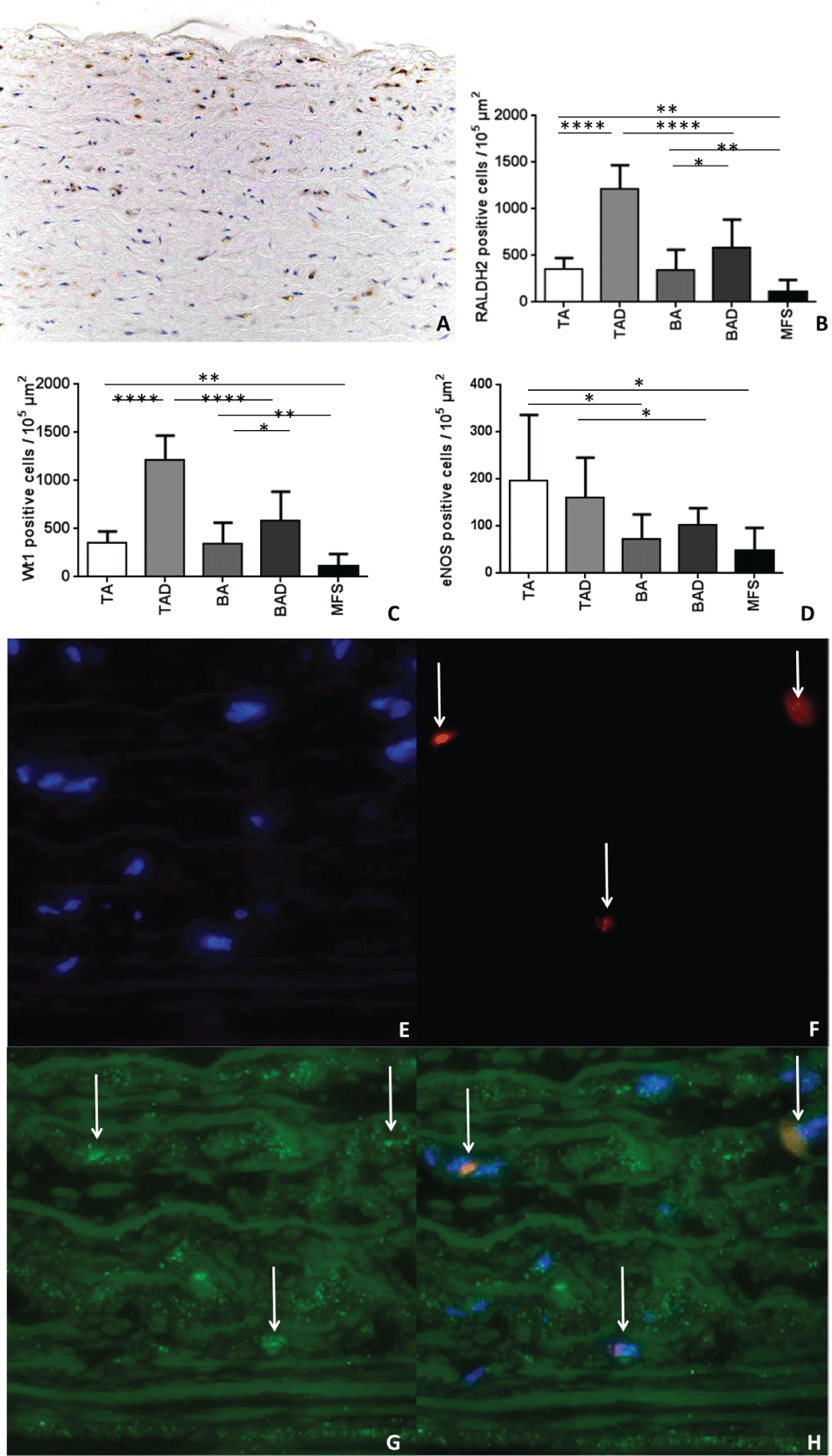
In the aortic media, the total number of VSMCs was not different between all investigated specimens [4,21]. Figure 2 gives an overview of the expression of Wt1, RALDH2 and eNOS in the aortic media (2A-H). In the aorta a subset of VSMCs variably stained positive for Wt1 in the nucleus and/or cytoplasm. For analysis only the nuclear Wt1 level of expression was considered.
.
Figure 2: Figure 2A is a transverse histologic section (5μ) of a non-dilated tricuspid aortic valve (TA). DAB stained for Retinaldehyde dehydrogenase-II (RALDH2), the vascular smooth muscle cells stained positive for RALDH2 are brown and the negative stained vascular smooth muscle cells remain blue. Expression of RALDH2, Wilms tumor suppressor protein (Wt1) and endothelial nitric oxide (eNOS) in the VSMCs of the aortic media are shown in graph 2B, 2C and 2D respectively.
Figure 2E-2H shows transverse histologic sections of the aortic wall in the non-dilated tricuspid aortic wall (5μ). The white arrows indicate staining with immunofluorescent staining for DAPI (blue, 2E), Wt1 (red, 2F), eNOS (green, 2G) and the overlay is shown in 2H for the co-expression of Wt1 and eNOS.
TA: Tricuspid Valve, without Dilation; TAD: Tricuspid Valve, with Dilation; BA: Bicuspid Valve, without Dilation; BAD: Bicuspid Valve, with Dilation, MFS: Marfan Syndrome, without Dilatation. * = p < 0.05, ** = p < 0.01, *** = p < 0.001, **** = p < 0.0001.
Magnification: 2A: 20×; 2E-2H: 40×.
View Figure 2
The number of positively stained VSMCs in the aortic media was significantly the highest in the dilated TAV group. Although in the dilated BAV the VSMCs stained significantly less for Wt1 as compared to the dilated TAV (p < 0.0001), the expression was higher than in the non-dilated BAV group (p = 0.015). No significant difference was found in medial expression of Wt1 between the non-dilated BAV and TAV group, whereas the MFS showed significantly less expression as compared to both these groups (p = 0.0022 and p = 0.0018 respectively) (Figure1 and Figure 2C).
All Wt1 positive VSMCs also stained cytoplasmic for RALDH2 (Figure 1, Figure 2A, Figure 2B and Figure 2C). Wt1 and RALDH2 positive VSMCs also stained positive in the nucleus for eNOS in the aortic media (Figure 2D, Figure2E, Figure2F, Figure2G and Figure2H). Expression was however not completely similar to the Wt1 and RALDH2, as some VSMCs only stained positive for eNOS (Figure 2E, Figure 2F, Figure 2G and Figure 2H). The number of eNOS positive cells was significantly higher in all TAVs (non- and dilated), as compared to all BAVs (non- and dilated) (p = 0.0234 and p = 0.0353 respectively) (Figure 2D). In the MFS group, the number of eNOS positive cells was comparable to the non-dilated BAV group (p = 0.1529) (Figure 2D).
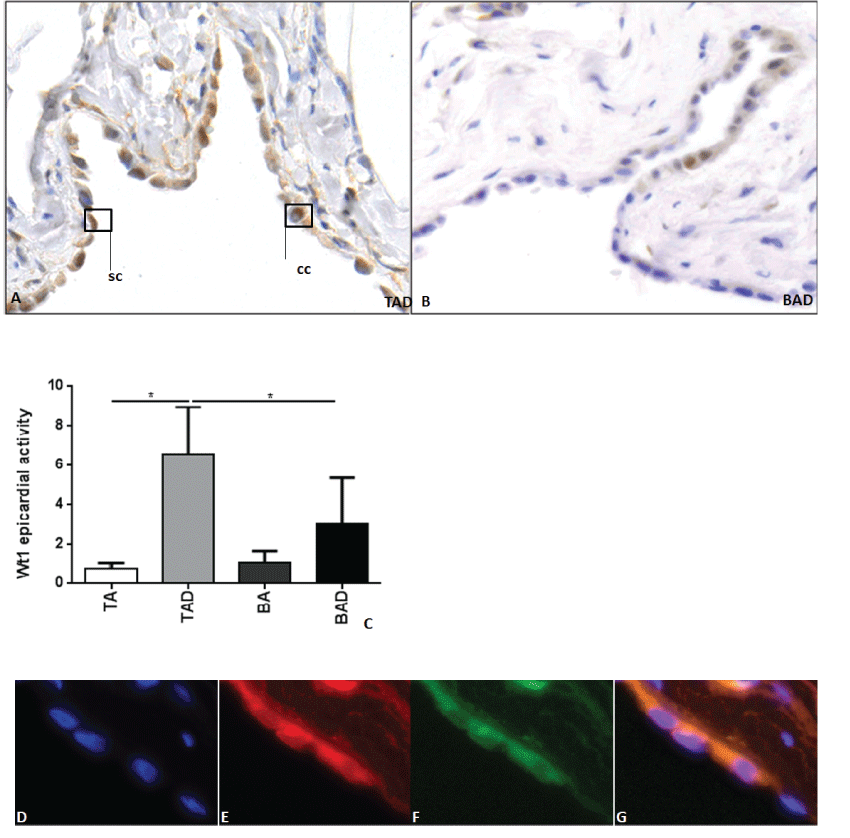
The adventitial side of the vessel wall, bordering the pericardial cavity, is lined by arterial epicardial cells. Wt1 expression was variably present in the nucleus and/or cytoplasm. An epicardial cell was identified as positive for Wt1 if either the nucleus or cytoplasm stained positive. The epicardial cells could morphologically be distinguished in 3 types: Wt1 negative squamous, Wt1 positive squamous and Wt1 positive cuboidal. The Wt1 positive cells were often found in epicardial invaginations or inpocketings (Figure 3A and Figure 3B). As handling during excision of aortic tissue can easily damage epicardial cells, we first identified Wt1 stained specimen in which the adventitia showed an intact monolayer of epicardial cells, which was in the TA, TAD, BA and BAD groups. The number of MFS patients with an epicardial outer covering was too small to allow an adequate statistical analysis of the epicardial cells. Analysis of epicardial Wt1, RALDH2 and eNOS expression was therefore solely performed on the non- and dilated BAV and TAV groups. In the MFS group, only the morphologic appearance of the epicardial cells could be described.
.
Figure 3: In figure 3A and 3B transverse histologic sections (5μ) are shown of the dilated tricuspid aortic valve (3F) and bicuspid aortic valve (BAD) group stained for Wt1 with DAB staining. Epicardial cells positive for Wt1 stain brown and can either have a squamous (sc) or cuboidal (cc) phenotype. The graph shown in figure 3C indicates the epicardial activity, defined as the ratio of Wt1 positive squamous (sc) and cuboidal (cc) epicardial cells.
Figure 3D-3G shows transverse histologic sections (5μ) of the epicardial cells in the dilated tricuspid aortic valve group (TAD). The epicardial cells shown are stained with immunofluorescence for DAPI (blue, 3D), Wilms tumor suppressor protein (Wt1) (red, 3E) and endothelial nitric oxide (eNOS) (green, 3F) and the overlay is shown in 3G for the co-expression of Wt1 and eNOS.
TA: Tricuspid Valve, without Dilation; TAD: Tricuspid Valve, with Dilation; BA: Tricuspid Valve, with Dilation; BAD: Bicuspid Valve, with Dilation. * = p < 0.05.
Magnification 3A, 3B: 40×; 3D-3G: 100×
View Figure 3
We first analysed the total Wt1 positive population (both squamous and cuboidal) in each sample and calculated the ratio of Wt1 positive compared to Wt1 negative cells. No significant difference was seen between the 4 groups (mean ratio 0.44). The Wt1 positive squamous epicardial cells represent an initial phase of epicardial activity, whereas cuboidal cells are prone for epithelial-to-mesenchymal transition. Therefore, we assessed the epicardial activity, defined as the transition from an initial quiescent phase to active epicardium, by the ratio of Wt1 positive squamous and cuboidal epicardial cells. The ratio was 0.73 in TA, 6.54 in TAD, 1.05 in BA and 2.99 in BAD. A significant increase in epicardial activity based on number of cuboid cells was seen in the dilated TAV as compared to the non-dilated TAV (p = 0.0286), and the epicardial activity was significantly greater in dilated TAV compared to dilated BAV (p < 0.0485) (Figure1 and Figure 3C). The epicardial cells of the ascending aorta of the MFS group were mostly squamous in appearance and showed minimal Wt1 expression and when present only in the cytoplasm. In dilated specimen of TAV invasion of epicardial cells into the subepicardial layer was also seen (not quantified), demonstrating epithelial-to-mesenchymal transition resulting in epicardium derived cells entering the adventitia, which was not prominent in BAV and MFS patients. Wt1 positive epicardial cells also stained positive cytoplasmatic for its downstream mediator RALDH2 in all groups (Figure 1). Moreover expression of eNOS was also observed in the same epicardial cells (Figure 1, Figure 3D, Figure 3E, Figure 3F and Figure 3G).
Discussion
Thoracic aortic dilation is a relatively common medical problem. The fact that the first presentation can be life threatening in the form of dissection or rupture of the aortic wall makes it even more crucial to understand the underlying pathobiology. The clinical course in BAV and MFS is more often complicated with thoracic aortic dilation as compared to TAV. In recent years research on differences between the aortic wall of both valve types and MFS has shown that the pathobiology is different in both syndromes with an intrinsically different, less mature aortic wall, as compared to the TAV [4]. However, the question remains what causes the progressive weakening of the aortic wall in bicuspidy and MFS. In this paper we focused on the activation of the arterial epicardium and VSMCs in both BAV and MFS aortic wall tissue as a possible marker for future aortopathy. As in the healthy adult heart the epicardium covering the myocardium is in a quiescent stage and also becomes active under pathological influences, such as myocardial infarction. The activated epicardial cells regulate and then take part in the repair processes that follows [14].
Although Wt1 has been shown to be a marker for the epicardium, recent literature confirms its expression in the endothelium as well [22,23]. Besides during normal development, an enhanced cardiac endothelial Wt1 expression has also been described in coronary vascular pathology in a rat model [20]. In our studied aortic specimen no difference in Wt1 and RALDH2 expression in the arterial endothelium was found between the dilated and the non-dilated aortic wall of BAV, TAV and MFS. Expression of eNOS in the endothelial cells was also similar in the 5 patient groups. Although co-expression of Wt1 and eNOS has been linked in epicardial cells [20] before, the observed co-expression in the endothelium has not been described earlier.
In all samples of the 5 groups the epicardial layer exhibited patches of quiescent epicardial cells alternating with cuboid epicardial cells that expressed Wt1, accordingly all specimens showed epithelial-to-mesenchymal transitionof the epicardial layer. An increase in Wt1 activity was most striking for the dilated TAV. No increase was seen for the dilated BAV patients. Additional statistical analysis showed that age and gender were not confounding in this study. As the absolute diameters of the ascending aorta were somewhat similar in dilated BAV and TAV, Wt1 expression in the epicardium is not likely to be associated with the degree of dilation. We have earlier shown that the aortic wall in TAV is structurally different from the BAV and MFS, with pathological differences related to inflammation and pathological ageing correlating with increased progerin expression. In the literature too, comparable characteristics of cardiovascular ageing leading to myocardial ischemia and subsequent activation of the epicardial Wt1 have been described in the coronary vasculature [14]. Epicardial activation might therefore not per se be associated with aortic dilation as an end point of aortopathy, rather with the pathologic cardiovascular ageing features leading to it. Besides Wt1 and RALDH2, expression of eNOS was also noted in the epicardial cells. Although in eNOS deficient mice an inhibition of epicardium derived cell migration has been noted [20], a prominent role of eNOS in the arterial epicardium of BAV cannot be concluded from this study. Future research should focus on arterial epicardial cells in BAV mouse models, to elucidate a possible role of eNOS related to the observed decreased Wt1 activity. Comparable to the epicardium we further see a difference in the VSMCs Wt1 activity between the BAV, MFS and TAV. We postulate that that this difference, like in the epicardial cells, is based on the immature state of the vessel wall and absence of features of cardiovascular ageing in the BAV and MFS as compared to the TAV.
Wt1 activity in epicardial and VSMCs is thus seen in ageing ascending aortic walls. Although this marker is not suitable in identifying BAV and MFS patients at risk for future aortopathy, more research is warranted to find out whether the activation state of epicardial cells could be of therapeutic value in TAV patients identified with an early stage of ascending aortic wall dilation.
Study Limitations
We designed our study by comparing the expression of the markers Wt1, eNOS and RALDH2 in cases of non- and dilated aorta of BAV, TAV and MFS patients. A limitation of our qualitative and semi-quantitative study is that we did not have frozen tissue samples of all the aortic wall specimens. Therefore quantitative evaluation by e.g. PCR or Western blot to correlate our findings to immunohistochemistry was not performed.
Conflict of Interest
None.
Acknowledgements
We thank the Heart Valve Bank Rotterdam, The Netherlands, for providing the described cryopreserved valves.
References
-
Grewal N, Franken R, Mulder BJ, Goumans MJ, Lindeman JH, et al. (2016) Histopathology of aortic complications in bicuspid aortic valve versus Marfan syndrome: relevance for therapy? Heart Vessels 31: 795-806.
-
Nataatmadja M, West M, West J, Summers K, Walker P, et al. (2003) Abnormal extracellular matrix protein transport associated with increased apoptosis of vascular smooth muscle cells in marfan syndrome and bicuspid aortic valve thoracic aortic aneurysm. Circulation 108: II329-II334.
-
Fedak PW, de Sa MP, Verma S, Nili N, Kazemian P, et al. (2003) Vascular matrix remodeling in patients with bicuspid aortic valve malformations: implications for aortic dilatation. J thorac cardiovasc surg 126: 797-806.
-
Grewal N, Gittenberger-de Groot AC, Poelmann RE, Klautz RJ, Lindeman JH, et al. (2014) Ascending aorta dilation in association with bicuspid aortic valve: a maturation defect of the aortic wall. J Thorac Cardiovasc Surg 148: 1583-1590.
-
Olive M, Harten I, Mitchell R, Beers JK, Djabali K, et al. (2010) Cardiovascular pathology in Hutchinson-Gilford progeria: correlation with the vascular pathology of aging. Arterioscler Thromb Vasc Biol 30: 2301-2309.
-
Bokenkamp R, Raz V, Venema A, DeRuiter MC, van Munsteren C, et al. (2011) Differential temporal and spatial progerin expression during closure of the ductus arteriosus in neonates. PLoS One 6: e23975.
-
Ikonomidis JS, Jones JA, Barbour JR, Stroud RE, Clark LL, et al. (2007) Expression of matrix metalloproteinases and endogenous inhibitors within ascending aortic aneurysms of patients with bicuspid or tricuspid aortic valves. J Thorac Cardiovasc Surg 133: 1028-1036.
-
Phillippi JA, Green BR, Eskay MA, Kotlarczyk MP, Hill MR, et al. (2014) Mechanism of aortic medial matrix remodeling is distinct in patients with bicuspid aortic valve. J Thorac Cardiovasc Surg 147: 1056-1064.
-
Gittenberger-de Groot AC, Winter EM, Bartelings MM, Goumans MJ, DeRuiter MC, et al. (2012) The arterial and cardiac epicardium in development, disease and repair. Differentiation 84: 41-53.
-
Lie-Venema H, van den Akker NM, Bax NA, Winter EM, Maas S, et al. (2007) Origin, fate, and function of epicardium-derived cells (EPDCs) in normal and abnormal cardiac development. ScientificWorldJournal 7: 1777-1798.
-
Smart N, Dube KN, Riley PR (2013) Epicardial progenitor cells in cardiac regeneration and neovascularisation. Vascul Pharmacol 58: 164-173.
-
Gittenberger-de Groot AC, Vrancken Peeters MP, Mentink MM, Gourdie RG, Poelmann RE (1998) Epicardium-derived cells contribute a novel population to the myocardial wall and the atrioventricular cushions. Circ Res 82: 1043-1052.
-
Germani A, Foglio E, Capogrossi MC, Russo MA, Limana F (2015) Generation of cardiac progenitor cells through epicardial to mesenchymal transition. J Mol Med (Berl) 93: 735-748.
-
Gittenberger-de Groot AC, Winter EM, Poelmann RE (2010) Epicardium-derived cells (EPDCs) in development, cardiac disease and repair of ischemia. J Cell Mol Med 14: 1056-1060.
-
Moore AW, Schedl A, McInnes L, Doyle M, Hecksher-Sorensen J, et al. (1998) YAC transgenic analysis reveals Wilms' tumour 1 gene activity in the proliferating coelomic epithelium, developing diaphragm and limb. Mech Dev 79: 169-184.
-
Hohenstein P, Hastie ND (2006) The many facets of the Wilms' tumour gene, WT1. Hum Mol Genet 15: R196-R201.
-
Perez-Pomares JM, de la Pompa JL (2011) Signaling during epicardium and coronary vessel development. Circ Res 109: 1429-1442.
-
Guadix JA, Ruiz-Villalba A, Lettice L, Velecela V, Munoz-Chapuli R, et al. (2011) Wt1 controls retinoic acid signalling in embryonic epicardium through transcriptional activation of Raldh2. Development 138: 1093-1097.
-
Xavier-Neto J, Shapiro MD, Houghton L, Rosenthal N (2000) Sequential programs of retinoic acid synthesis in the myocardial and epicardial layers of the developing avian heart. Dev Biol 219: 129-141.
-
Liu Y, Lu X, Xiang FL, Poelmann RE, Gittenberger-de Groot AC, et al. (2014) Nitric oxide synthase-3 deficiency results in hypoplastic coronary arteries and postnatal myocardial infarction. Eur Heart J 35: 920-931.
-
Grewal N, Gittenberger-de Groot AC, DeRuiter MC, Klautz RJ, Poelmann RE, et al. (2014) Bicuspid aortic valve: phosphorylation of c-Kit and downstream targets are prognostic for future aortopathy. Eur J Cardiothorac Surg 46: 831-839.
-
Duim SN, Kurakula K, Goumans MJ, Kruithof BP (2015) Cardiac endothelial cells express Wilms' tumor-1: Wt1 expression in the developing, adult and infarcted heart. J Mol Cell Cardiol 81: 127-135.
-
Rudat C, Kispert A (2012) Wt1 and epicardial fate mapping. Circ Res 111: 165-169.





