International Journal of Pediatric Research
Endogenic Intoxication and Kidney
Yurieva Eleonora Alexandrovna1, Vozdvizhenskaya Ekaterina Sergeevna1, Kushnareva Maria Vasilievna1* and Novikova Natalia Nicolaevna2
1Research Clinical Institute of Pediatrics, N.I.Pirogov Russian National Research Medical University, Ministry of Health of Russia, Moscow, Russia
2National research center "Kurchatov institute", Moscow, Russia
*Corresponding author: Kushnareva Maria Vasilievna, N.I.Pirogov Russian National Research Medical University, Research Clinical Institute of Pediatrics, Ministry of Health of Russia, Moscow, Russia, E-mail: mkuschnareva@mail.ru
Int J Pediatr Res, IJPR-2-022, (Volume 2, Issue 2), Review Article; ISSN: 2469-5769
Received: December 29, 2015 | Accepted: August 16, 2016 | Published: August 19, 2016
Citation: Alexandrovna YE, Sergeevna VE, Vasilievna KM, Nicolaevna NN (2016) Endogenic Intoxication and Kidney. Int J Pediatr Res 2:022. 10.23937/2469-5769/1510022
Copyright: © 2016 Alexandrovna YE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The following information about the endogenous intoxication in children (from 2 up to 17 y.o.) with nephrological diseases was presented. Endogenous intoxication as multisystemic pathological condition occurs as the result of action on the organism of toxic substances of endogenous or exogenous origin. It is characterized by the following: a) violation of the barrier function of the organism against toxins, b) violation of transport mechanisms of toxic substances to the organs of their transformation and/or excretion. Endogenous intoxication leads to disruption of the structure, function, and antigenic properties of protein molecules that can cause severe chronic renal disease.
Keywords
Endogenic intoxication, Kidney, Nephrological diseases, Children
Introduction
Endogenous intoxication is a polyethiological polipatogenny syndrome characterized by the accumulation in tissues and biological fluids of the endogenous toxic substances, which are excess products of normal or distressed metabolism. In this case the mechanism of detoxification of these substances and their biotransformation is suffered.
As a result of the action on the body of toxic substances of endogenous or exogenous origin appears pathological condition that is chronically stored under more or less constant contact of the organism with these substances and/or their accumulation in the body. It is a complex of multi-organ pathological condition. It includes the following: a) the violation of the barrier function of the body against toxins, b) the violation of the transport mechanisms of toxic substances to the organs of their transformation and/or excretion by the kidneys, c) the ability of biotransformation of toxins [1-5].
A kidney is one of the main organs that provide purification of the organism from endogenous and exogenous toxins and, at the same time, this organ is at higher risk to damage as being the target action of toxicants [2-8].
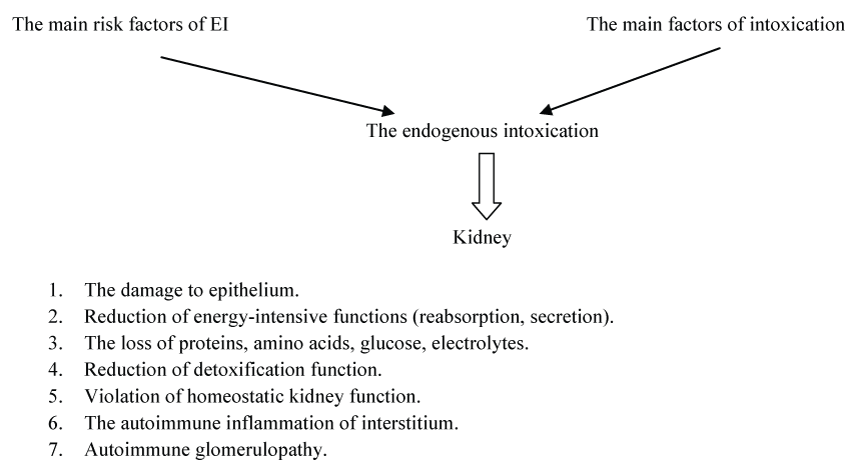
The table shows the risk factors of the endogenous intoxication's development and the main factors of intoxication in children. The figure is summarized the mechanism of the damaging effect of intoxication factors on the kidneys in children (Table 1 and Figure 1).
![]()
Table 1: The endogenous intoxication (EI) in infants.
View Table 1
It is evident from the table and figure that the endogenous intoxication is a multifactorial pathological condition.
The reasons of endogenous intoxication can be the following [2]:
1) The products of normal metabolism - lactate, pyruvate, creatinine, uric acid, bilirubin;
2) The products of disturbed metabolism - ketones, ammonia and others;
3) The breakdown products of cells and tissues or penetration of toxicants from gastrointestinal tract in case of the barrier dysfunction of membrane such as lipases, lysosomal enzymes, cationic proteins, myoglobin, phenol etc;
4) The immune xenogeneic cellular debris, antigens and immune complexes [2].
In addition, the intoxication matters due to the ingestion of exogenous toxicants (medicines, heavy metals, pesticides, a number of food products). Elimination of the various toxins occurs mainly through the gastrointestinal tract, kidney and macrophage-monocyte system (lung, perspiratory glands, saliva etc.). Therefore, the nature of the flow affects the endogenous intoxication in the pathological process involving different organs and systems [4,9].
Thus, according to the different authors, the nephropathy often combines with the diseases gastrointestinal tract, lungs, upper respiratory tract, cardiovascular system primary or it occurs because of the intoxication. In such cases, the pathology is caused by the accumulation or the massive intake of toxic substances in excess of the possibility detoxifying systems or their inferiority, particularly in the kidneys [2,5,10]. It is evident that it is important to consider the pathogenetic action of endogenous and exogenous toxins on the kidney function in nephropathies.
The leading process of toxins' excretion in the urine is filtration of xenobiotics (foreign substances) through biological barriers in the kidneys. Approximately 1 liter of blood clears through the kidneys for 1 hour, 40-120 ml of urine per hour is released from this liter, containing removed xenobiotics in the concentrated form and elimination metabolites. Such structures as the capillary endothelium, epithelium of tubules and glomeruli and glomerular basement membrane consisting of the polysaccharide glycocalyx of cell, endothelium and epithelium (2-4 nm diameter pores in the glomerular basement membrane) is involved in the kidney on the way of toxicants. As it is known, it is not penetrate normal macromolecular proteins in the Bowman's capsule, but all the other plasma (serum) components, xenobiotics and other low molecular weight compounds contain in the "primary" urine [2,4,11].
The concentration of low and medium weight compounds occurs many times over in the tubules due to the result of the reabsorption, secretion and the functioning of the specific systems (e.g., Geple loop).
Consequently, the concentrated substances material due to the high gradient between the tubules and plasma that are exposed reverse diffusion into the blood, although the barrier formed by the epithelium and the interstitial substance endothelium.
Reabsorption is exposed in the first place:
a) Liposoluble substances.
b) Non-ionized molecules of water-soluble substances.
c) Substances with the low molecular weight, including toxins.
Such recycling of toxicants leads to the slower process of their elimination. There is a transformation of liposoluble toxicants (substances that poorly excretes from the body) into water-soluble in the liver and its excretion facilitates. It has a value and charge of exteriorized substances through the kidneys: the ionized substances are worse reabsorbed and it is more easily excreted. Moreover, normally the active removal of individual compounds (specific secretion of antibiotics etc.) is performed in the tubules. The violation of these processes creates the risk of damage of the structures and functions of the kidneys with endogenous and exogenous intoxication [2,4,8].
These processes are created by the intensive work of the mitochondria-bioenergetic organelles of the cells. The particular importance has the impact of toxicants on the energy of kidney enzyme systems, which execute a catalytic and/or transport specific functions.
In this case may be the following:
1) Increase the catalytic activity (strengthen of synthesis, block of inhibitors protease, and peptidase during autolysis because of lysosomal enzymes excretion and increase of substrates for this enzyme).
2) Inhibit of the catalytic activity (inhibition of the synthesis, accelerating of proteases and peptidases degradation, substrate reduction).
3) The changing of the enzymes structure is also valued [2,4-6].
The inhibition of the catalytic activity of several enzymes may be the result of actions of the cofactors among which include a number of metals (Zn, Mg, Mo, Co) and vitamins (group B, E, K). Some toxicants are competitive or non-competitive inhibitors of enzymes: competitive inhibition occurs in the presence of competitive substrates, blockade of the enzyme's active center and/or non-competitive inhibition of the additional allosteric center [5,6,8,9].
One of the factors that affect the eliminated ability of the kidneys in relation to the toxicant is circadian frequency of the kidneys. The last one in the norm depends on the time of the day, the content of corticosteroids and other hormones in the blood, neurotransmitters in the nerve tissue, which causes the different enzyme activity during the day, but also the air temperature has a value.
Oxidative Stress
The most common endogenous toxicants (substances that lead to the damage of biological systems, disruption of its function and vitality) are products of oxidative stress, the effects of hypoxia [6,7].
These products can be accumulated because of the activation of free radical processes in the body as a result of the biotransformation of xenobiotics, metals such as Fe, Cu, cytostatics, phenylhydrazine and etc. (not only because of the endogenous causes), with the formation of the intermediate reactive products [1,11].
A selective damage of the one of the structures (the brush border of the renal epithelium, mitochondrial enzymes) occurs under the action of active oxygen as well as structurally functional changes of the several structures [11].
Having a sufficient stability the active forms of oxygen react with the different biomolecules, it is not only damaging them but also provoking the chain reactions of active radicals of further formation from lipids, amino acids, nucleic acids, etc [11].
The tissue damage is occurred by necrosis, apoptosis, and development of fibrosis in the damaged organs and even the appearance of tumors [9,11]. The result of the activation of free radical processes in the cells can be mutagenesis, carcinogenesis, photoallergy, hemolysis besides cell necrosis and fibrosis of the damaged organs [11].
The kidney for free-radical influences is one of the main organs of the targets due to the multiple increase in the area of lipid membranes by brush rims of the epithelium of the renal tubules. The antioxidant protection (enzymatic and non-enzymatic) processes is rather quickly get exhausted while strengthen the peroxidation especially in people with genetically determined decrease in activity of the antioxidant defense system (30% of the population) [11].
The increase of uric acid, middle molecules, peroxidation products and the decrease of antioxidant protection is detected in the blood with nephropathy [3].
Biochemical studies of daily urine' indicators for years have established the high frequency disorder of the antioxidant protection of urine in kids with dysmetabolic nephropathy, urolithiasis, and glomerulonephritis. The urine antioxidant capacity (the norm is more than 50%) is decreased 1.5-5 times in these kids [6].
At the same time, the significant amount of super acidulated of lipids and/or other peroxidic compounds (H2O2) is released with the urine, which are not detectable in normal urine. It is known that mitochondria cells under an oxidative stress (its basic energy organelles) which are either targets of reactive oxygen species, or the site of synthesis of reactive oxygen species as a result of hypoxia, ischemia tissue [6].
Not only the redox processes in the respiratory chain are broken during the hypoxic effect in mitochondria, but also the cell calcium metabolism with multiple effects including cell apoptosis system is disrupted earlier.
The violated intercellular isolation calcium phosphate "vessels" of the mitochondria with the release of these complexes from the cell. The indirect evidence of violations of the calcium-phosphorus homeostasis in the cells of kidney tissue under the oxidative stress is increasing the urinary excretion of calcium and phosphate. It is detectable with the help of Sulkovich' reaction (Ca) and the reaction with MgCl2 in an alkaline environment [6].
The lack of calcium phosphate and magnesium ammonium phosphate crystals in the urine test during transient disturbances of mitochondria has a value, but the presence of such crystals is typical for more serious violations of calcium-phosphate metabolism: hyperparathyreosis, systematic violation of bioenergy at risk of kidney stones developing. In such cases, calcium phosphate, magnesium phosphate crystals and also aggregates are identified (reducing of urine's anti crystal-forming ability) [6].
The typical expression of an oxidative stress (hypoxic syndrome) is the disturbance of the structure and activity of many enzymes, especially identified with an increase of a number of products in the blood and/or urine, depending on changes in their activity [1,10-13].
Uric Acid
The most famous indicators of dysfunction of enzymes in an oxidative stress are the uric acid and homocysteine, accumulated due to the violation of purine metabolism or metabolism of methionine, respectively [5].
Endogenous peroxide products and other toxicants cause the reduction in the activity of enzymes of purine metabolism and also cause their genetic mutations in the somatic cells, which are not inherited. So far, the most recognized marker of disorders of purine metabolism, along with hyperuricousuria is the reduction of the activity in the lymphocytes of one of the main enzymes' synthesis of purine nucleotides - hypoxanthine - guanine - phosphoribozyl - transferase (HGPRT), the genic locus of the enzyme is located in the long arm of the X-chromosome and easily susceptible to mutations in the somatic cells [5].
Changes in the activity of this enzyme are typical for endo- and ekzotoksikosis in the body. It is shown that the number of lymphocytes deficient on HGPRT among smoking women and their newborns is 3.08 ± 0.55 and 2.17 ± 0.24 × 10-6 cells, while the number of such cells among non-smoking women and their newborns was 1.07 ± 0.17 and 0.77 ± 0.13 × 10-6, respectively [2,6].
In subjects with defects of this enzyme increased biosynthesis of uric acid due to its accumulation in tissues precursors: hypoxanthine and guanine and xanthine oxidase increased activity (XA), for which the base is a substrate in the synthesis of uric acid. In turn, damage the XA activity causes a dramatic increase in the formation of reactive oxygen species (superoxide anion) and the activation of peroxide processes [5,14].
All the degrees of HGPRT deficiency are appeared by more or less significant hyperproduction and the excretion of uric acid. This may not be detected until the age of puberty due to the fact that the clearance of uric acid is much higher in younger children than it is in adults [5].
It is found a deficiency of one of the enzymes of the purine degradation metabolites - adenosine deaminase (ADA), beside HGPRT in children with nephropathy [2,15-17]. There is also often a decrease of ADA under the severe primary and secondary immunodeficiency. We found a close relationship with immunodeficiency failure of this enzyme. Maximum activity of adenosine deaminase found in the normal E-rosette-forming cells, the maximum occurs for S-phase of the cell division (mitosis) [2,5,17].
Furthermore, it was shown that these two enzymes of purine metabolism are regulators of nucleotide exchange, and its biochemistry is closely linked with lymphocyte physiology, both enzymes are targeted for chemical attack and determine the sensitivity to xenobiotics, including drugs that affect renal function [2].
Unfortunately, in such case the formed uric acid in excess became paradoxical factor of further activation of the peroxide processes. It happens despite its antioxidant action, which occurs under physiological concentration (0.25 mol/L) (i.e., the shuttle effect is observed). The increased amount of the uric acid contributes to an endothelial damage and the formation of atherosclerotic plaques in blood vessels [2,18].
Homocysteine
It has a similar action on the blood vessels. This amino acid is an intermediate in the metabolism of methionine. The increased of homocysteine in the blood is a recognized factor of the vascular endothelial damage and thrombus formation [18,19].
Homocysteine is one of highly pathogenic risk factors of the vascular endothelial damage, particularly in the kidneys [2,6]. Homocysteine and other endogenous toxicants are accumulated during prolonged and repeated situations with violation of the metabolic processes, they are created the risk of chronic-somatic diseases which are progressing with the age [19].
Proteins
It has been established that first of all the endogenous and exogenous toxicants damage the protein molecules. The proteins change their conformation with the emergence of new and extra ligand locus that bind metals and aggressively capture the trace elements that reinforce the altered conformation.
Such protein modification makes it enthetic, antigenic structure with auto-aggressive properties [12,13]. Modified proteins in microquantities are excreted with the urine and, obviously, poorly exposed to reabsorption by means of transport systems in the renal tubules due to structural changes [4].
Highly specific proteins - enzymes and transport proteins are affected particularly. Thus, the control system of blood coagulation activity with the change of proteins such as tissue plasminogen activator, plasminogen activator inhibitor type 1 is disrupted during the nephrotic syndrome. By changing the content of NO and natriuretic peptide type-C (SNP) in the blood can assume changes in the activity of the endothelial enzymes synthesis of regulators of vascular tone (eNOS synthase, SNP) [20]. Changing of the content of the vasoconstrictor endothelin-1 peptide occurs in violation of the functions of membrane proteases (endothelin-converting enzyme) in the endothelium [20].
Conclusion
Thus, the pathological condition occurs as a result of the action on the organism of toxic substances of the endogenous or exogenous origin. This condition chronically remains under constant contact the organism with such substances and/or their accumulation in the body.
This is a complex of multiple organ pathological condition, it includes:
a) The violation of the barrier function of the body against toxins,
b) The violation of the transport mechanisms of toxic substances to the organs of their transformation and/or excretion by the kidneys, and
c) The ability of the biotransformation of toxins.
It is especially necessary to emphasize that the endogenous intoxication is a risk factor pathological changes mainly protein molecules with the violation not only of their structure and function, but also changes of their antigenic properties. The result of these changes is the appearance and chronization of many disabling human diseases. An obvious need is not only to diagnose the endogenous intoxication, but, as far as it possible, to reduce its activity, particularly during nephropathy.
References
-
Santos EB, Koff WJ, Grezzana Filho Tde J, De Rossi SD, Treis L, et al. (2013) Oxidative stress evaluation of ischemia and reperfusion in kidneys under various degrees of hypothermia in rats. Acta Cir Bras 28: 568-573.
-
Kutsenko CA (2004) Basis of toxicology. S-Peterburg: Foliant: 750.
-
Yuryeva EA, Suchorukov VS, Murashova AN, Pimenov MC, Ilyushina IA, et al. (2012) Biochemical markers of aterogenecyti and protective action xidiphone in experemetal animals. Bull Expe Biol and Med 153: 445-448.
-
Yuryeva EA, Suchorukov VS, Tsaregorodtsev AD, Vozdvizhenskaya ES, Kharabadze MN, et al. (2013) Modification of protein molecules under endogenous intoxication as a risk factor of chronic metabolic diseases. J Molecular Medicine 3: 45-52.
-
Yuryeva EA, Yablonskaya MI, Raba GP (2013) Pathogenetic aspects of uric acid (purine) diathesis in children. Ros Vestn Perinatol I Pediat 58: 40-46.
-
Yuryeva EA, Dlin VV (2007) Handbook in children nephrology. Moskow, Med-Praktika 250.
-
Fine LG, Norman JT (2008) Cronic hypoxia as a mechanism of progresses of chronic kidney disease: from hypothesis to novel therapeutics. Kidney int 74: 867-872.
-
Novikova N, Kovalchuk M, Stepina N, Gaynutdinov R, Chukhrai E, et al. (2015) Distinct effect of xenobiotics on the metal-binding molecules. J Synchrotron Radiat 22: 1001-1007.
-
Patzer L (2008) Nephrotoxicity as a cause of acute kidney injury in children. Pediatr Nephrol 23: 2159-2173.
-
Schlondorff DO (2008) Overview of factors contributing to the pathophysiology of progressive renal disease. Kidney Int 74: 860-866.
-
Skulachev VP (2012) Mitochondria-targeted antioxidants as promising drugs for treatment of age-related brain diseases. J Alzheimers Dis 28: 283-289.
-
Novikova NN, Kovalchuk MV, Stepina ND, Konovalov OV, Yurieva EA, et al. (2011) Spectral-selective x-ray methods for structure diagnostics of ordered bioorganic nanosystems on the liquid surface. X-ray, synchrotron and neutron investigations 9: 6-11.
-
Novikova NN, Kovalchuk MV, Yurieva EA, Konovalov OV, Rogachev AV, et al. (2012) Total external reflection X ray fluorescence analysis of protein-metal Ion Interactions in biological systems. Crystallography Reports 57: 648-655.
-
Macásek J, Zeman M, Vecka M, Vávrová L, Kodydková J, et al. (2011) Reactive oxygen and nitrogen species in the clinical medicine. Cas Lek Cesk 150: 423-432.
-
Yamada Y, Nomura N, Yamada K, Kimura R, Fukushi D, et al. (2014) Hypoxanthine guanine phosphoribosyltransferase (HPRT) deficiencies: HPRT1 mutations in new Japanese families and PRPP concentration. Nucleosides Nucleotides Nucleic Acids 33: 218-222.
-
Yamada Y, Wakamatsu N, Taniguchi A, Kaneko K, Fujimori S (2011) Hypoxanthine guanine phosphoribosyltransferase (HPRT) mutations in the Asian population. Nucleosides Nucleotides Nucleic Acids 30: 1248-1255.
-
Booth C, Hershfield M, Notarangelo L, Buckley R, Hoenig M, et al. (2007) Management options for adenosine deaminase deficiency; proceedings of the EBMT satellite workshop. Clin Immunol 123: 139-147.
-
Hayden MR, Tyagi SC (2002) Uric acid: a new look at old risk marker for cardiovascular disease, metabolic syndrome, and type 2 diabetes mellitus: the urate redox shuttle. Card Diabetol 1: 3-15.
-
Ciaccio M, Bivona G, Bellia C (2008) Therapeutical approach to plasma homocysteine and cardiovascular risk reduction. Ther Clin Risk Manag 4: 219-224.
-
Sivceva EM, Dlin VV, Vozdvizhenskaya ES (2012) Endotelin-1 in blood plasma in children with nephropatic syndrome of glomerulonephritis. Ros Vestn Perinato Pediat 57: 29-35.