International Journal of Radiology and Imaging Technology
Assessment of Cervical Cancer Using Blood Oxygen-Level Dependent and Diffusion Weighted Magnetic Resonance Imaging
Jessica B Robbins1*, Emily F Dunn2, Kristin A Bradley2, James J Brittin1, Alejandro Munoz Del Rio1 and Elizabeth A Sadowski1,3
1Department of Radiology, School of Medicine and Public Health, University of Wisconsin, USA
2Department of Human Oncology, School of Medicine and Public Health, University of Wisconsin, USA
3Department of Obstetrics and Gynecology, University of Wisconsin Madison, USA
*Corresponding author:
Jessica B Robbins, MD, Department of Radiology, University of Wisconsin School of Medicine and Public Health, Clinical Science Center, MC 3252, 600 Highland Avenue, Madison, WI 53792-3252 USA, Tel: 608-263-9028, Fax: 608-263-0140, E-mail: jrobbins@uwhealth.org
Int J Radiol Imaging Technol,
IJRIT-1-006, (Volume 1, Issue 1),
Original Article
Received: October 7, 2015: Accepted: November 30, 2015: Published: December 02, 2015
Citation: Robbins JB, Dunn EF, Bradley KA, Brittin JJ, Rio AMD, et al. (2015) Assessment of Cervical Cancer Using Blood Oxygen-Level Dependent and Diffusion Weighted Magnetic Resonance Imaging. Int J Radiol Imaging Technol 1:006.
Copyright: © 2015 Robbins JB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose:To determine if blood oxygen level-dependent (BOLD) MRI can distinguish between normal cervix and cervical cancer and if there is a correlation between R2* and apparent diffusion coefficient (ADC).
Materials and methods:The picture archiving and communication system was used to retrospectively identify patients with MRI who had either cervical cancer (study group) or endometrial cancer and a pathologically proven normal cervix (control group). BOLD and diffusion weighted (DW) MRI were performed in all patients. R2* and ADC in cervical tumors and normal cervix were measured, compared, and their correlation determined.
Results:There were 7 patients each in the study and control groups. R2* of cervical cancer, 18.99 s-1, is significantly less than R2* of normal cervix, 31.10 s-1, (p = 0.012). ADC of cervical cancer, 0.96 × 10-3 mm2/s, is significantly less than ADC of normal cervix, 1.35 × 10-3 mm2/s, (p = 0.002). There is moderate correlation between R2* and ADC, (r = 0.78, p = 0.001).
Conclusion:R2* and ADC are significantly lower in cervical cancer compared to normal cervix and there is only a modest correlation between these parameters. In cervical cancer, BOLD MRI may provide information regarding oxygen bioavailability which is additive to DW MRI.
Keywords
BOLD, DWI, Cervical cancer
Introduction
Cervical cancer is the most common gynecological malignancy in the world, with more than 500,000 cases diagnosed per year [1]. There is a marked disparity in disease incidence and mortality between developed and underdeveloped regions of the world with nearly seventy percent of cases occurring in underdeveloped regions. The annual incidence of cervical cancer in sub-Saharan Africa is 35/100,000 women with an annual mortality rate of 23/100,000 as compared to an annual incidence of 6.6/100,000 women and an annual mortality rate of 2.5/100,000 in developed countries. The discrepancy in incidence is primarily ascribed to differences in screening with the Papanicolaou test (Pap smear) and availability and utilization of the human papilloma virus vaccine [2], whereas the differences in mortality are due to access to contemporary imaging and treatment regimens [1,2].
Even in developed regions of the world, 38% (2.5/6.6) women die of cervical cancer each year and cervical cancer is the leading cause of cancer death in women less than 35 year of age [3,4]. Survival in advanced stages of disease is disappointing despite recent advances in chemo-radiation therapy; in Stage II the survival ranges between 58-63% [5]. Investigators are targeting this population for advanced image-guided therapies. Functional magnetic resonance imaging (MRI) can be used to determine characteristics of the residual tumor early in the course of therapy; depending on the residual tumor characteristics, the course of therapy can be adapted [6]. Functional MRI includes perfusion, diffusion weighted (DW) imaging and blood oxygen level-dependent (BOLD) imaging. These techniques examine the physiological properties of tissue, not just the signal intensity of the tissue as seen on standard T1- and T2-weighted MRI sequences [7-10]. The ability to measure blood flow, water diffusibility and oxygen bioavailability has enabled investigators to observe changes in the physiology of cervical cancer before changes in tumor size and signal characteristics are detectable [11-18]. Changes in size and signal of the tumor can take 3-6 months at a minimum to develop [7] whereas alteration in blood flow, water diffusion, and oxygen level of the tumor can develop more quickly, in just days to weeks [11,13,18]. This allows for more rapid reassessment and a potential to alter treatment to the regions that may not be responding.
There is a paucity of studies investigating BOLD MRI in cervical cancer; yet, alteration in tumor oxygenation is one of the earliest physiologic changes, potentially even before changes in blood flow [19-21]. Additionally, BOLD MRI could be used during the course of treatment to provide a roadmap identifying hypoxic regions which could benefit from a higher radiation dose during treatment [22,23]. Since little has been published in the literature on BOLD MRI of the cervix, our goals were to determine if BOLD MRI could be used to distinguish between normal cervical tissue and cervical cancer and to determine if there is a correlation between R2* and ADC values.
Materials and Methods
Study population
This retrospective study is compliant with the Health Insurance Portability and Accountability Act and was approved by our institutional review board. The need for informed consent was waived.
A search of the picture archiving and communication system (PACS) identified consecutive patients with cervical cancer who were treated at our center with chemo-sensitized radiation therapy and had their pre-treatment diagnostic MRI between August 2, 2012 and March 4, 2013. Women with MRI examinations of the pelvis for the indication of endometrial cancer and a pathologically proven normal cervix were selected from the same time interval to serve as controls. All women underwent routine clinical MRI of the pelvis, which included BOLD and DW MRI. Patients in the study group had biopsy proven cervical cancer prior to the pre-treatment diagnostic MRI. Patients in the control group had a pre-treatment diagnostic MRI, subsequently underwent hysterectomy for endometrial cancer or endometrial hyperplasia, and the cervical tissue was pathologically proven to be benign. Exclusion criteria for the study group included tumor excluded from the BOLD field of view and artifact obscuring the cervical tumor. Patients were excluded from the control group if they had neoplastic involvement of the cervix.
MRI parameters
In all patients, MRI of the pelvis was performed according to clinical protocols with an 8 channel cardiac coil on a 1.5 Tesla magnet (GE Optima 450W, Waukesha, WI), similar to what is published in the literature [24] and displayed in table 1. The BOLD imaging plane was oblique axial oriented to the cervix. The BOLD imaging parameters were: 8 channel cardiac coil, TR = 87 ms, TE = 6.8-56.2 ms (16 echoes with ΔTE = 2.3 ms), flip angle = 40°, matrix = 256 × 256, FOV = 32-34 cm, slice thickness = 4.0 mm (skip 0.5 mm). DW imaging plane was axial through the pelvis, including cervix. The DW imaging parameters were: 8 channel cardiac coil, TR = 12,000 ms, TE = 81 ms, b factors = 0 and 500, matrix = 128 × 128 FOV = cm, slice thickness = 4.0 mm (skip 0.5 mm). All patients were imaged while breathing room air.
![]()
Table 1: Cervical cancer MRI parameters.
View Table 1
Image analysis
BOLD image processing was performed on a stand-alone workstation by one author (JBR) with 6 years of experience in women's imaging. Using GE Functool® R2* map analysis software, T1 weighted gray scale anatomic images and color R2* maps were generated. R2* values were recorded in units of 1/seconds (s-1) and were obtained by manually placing a region of interest (ROI) over the cervical tumor (Figure 1) or normal cervix (Figure 2) in the controls. In the setting of a cervical tumor, the ROI was placed to best encompass the tumor. Regions of susceptibility artifact from adjacent rectal gas were avoided. Both the T1-weighted gray scale and color maps were used to appropriately place the ROI. The mean area of the ROI for the cervical tumor was 124 mm (+/- 48 mm, range 49-168 mm). In the control group, 3 small ROI's were placed within normal cervical stroma at approximately 12 o'clock, 4 o'clock, and 7 o'clock positions and the mean value was determined; three small ROI's were chosen over a single ROI in order to maximize the volume of cervical stroma measured while excluding the endocervical canal. The mean area of the ROI for normal cervix was 63 mm (+/- 25 mm, range 21-122 mm).
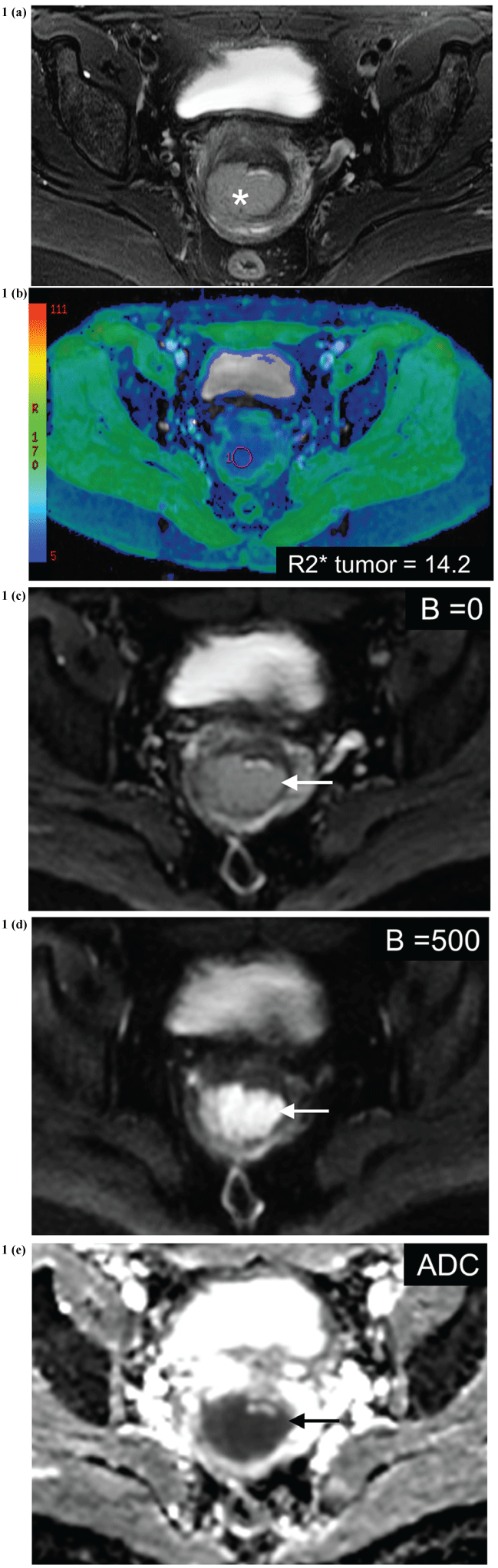
.
Figure 1(a-e): Patient with stage IIB cervical cancer at baseline before chemo-sensitized radiation treatment.
(a) Axial T2 weighted image with fat saturation demonstrates a large cervical tumor (white asterisk), predominantly located within the posterior lip of the cervix.
(b) R2* color map through the cervical tumor, with blue color representing lower R2* values and green representing higher R2* values. R2* of the tumor measures 14.2 s-1 (magenta ROI).
(c-e) DW images, B = 0, B = 500 and ADC map demonstrate the cervical cancer as having restricted diffusion (white arrow). The ADC of the tumor measures 0.78 × 10-3 mm2/s.
View Figure 1
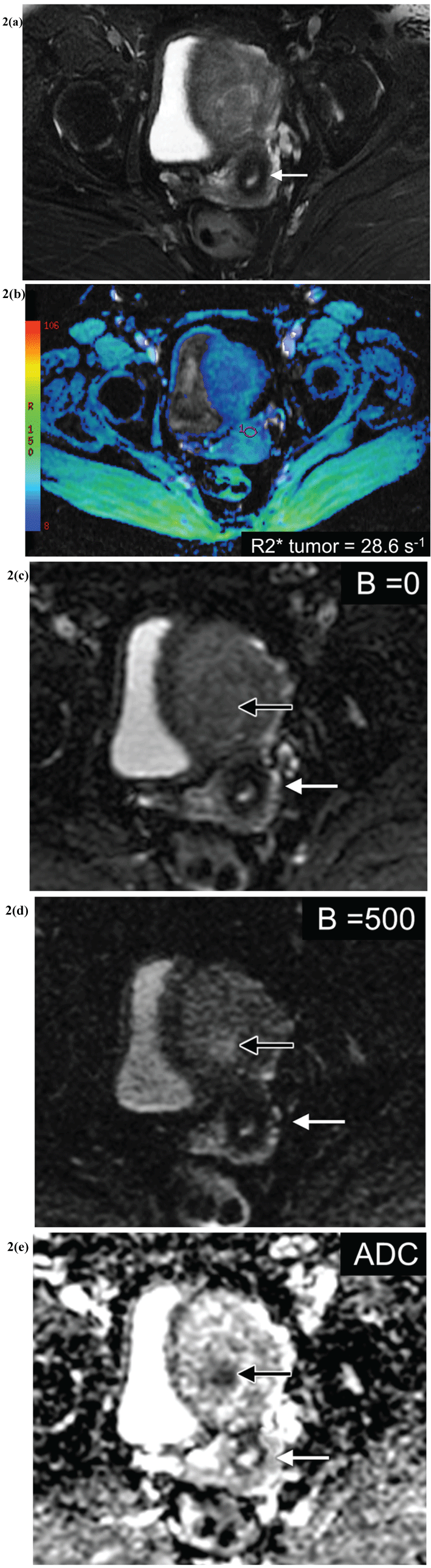
.
Figure 2 (a-e): Patient with pathologically proven normal cervix, who underwent hysterectomy for endometrial cancer.
(a) Axial T2 weighted image with fat saturation demonstrates a normal appearing cervix (white arrow).
(b) R2* color map through the cervical tumor, with blue color representing lower R2* values and green representing higher R2* values. R2* of the tumor measures 28.6 s-1 (magenta ROI).
(c-e) DW images, B = 0 (C), B = 500 (D) and ADC map (E) demonstrate the normal cervix (white arrow) and the patient know grade 1 endometrial cancer in the mid body of the uterus (black arrow). The ADC of the normal cervix measures 1.32 × 10-3 mm2/s.
View Figure 2
Apparent diffusion coefficient (ADC) values were measured in both normal cervical stroma and cervical tumors directly on the PACS work station. The ROI's were placed on the ADC maps, similar to the ROI's placed on R2* maps, as described in the methods described above; ADC values were recorded in the units of 10-3 mm2/s.
Statistics
For R2* and ADC measurement, mean and standard deviation (SD) for each group were calculated. Their distribution within controls and cervical cancer subjects was compared with a Kruskal-Wallis test. Linear regression was used to estimate the relationship between mean ADC as a linear function of R2*. The coefficient of determination R2 was used to assess the percentage of variation in the response (mean ADC) explained by the model; its square root, r, corresponds to the Pearson product moment correlation coefficient. Models were fitted to each group separately, as well as to combined groups. P < 0.05 (two-sided) was used as the criterion for statistical significance. All statistical graphics and computations were generated in R 3.0.1 (R Core Team 2013).
Results
10 patients with cervical cancer presenting for chemo-sensitized radiation treatment were identified. Three patients were excluded; BOLD sequence was not acquired in one, the tumor was excluded from the BOLD field of view in the second patient, and susceptibility artifact from rectal gas obscured the tumor in the third. Therefore, a total of 7 patients were included in the study group. All 7 patients had squamous cell carcinoma pathology. The stage of disease was IB1 in 3, IB2 in 2 and IIB in 2 with a mean tumor diameter of 4.4 cm +/- 1.3 cm (range 2.6-6.6 cm). Average age was 49.7 years +/- 10 years (range 35-64). Table 2 presents the data on a per patient basis.
![]()
Table 2: Demographic, mean R2* and ADC values of cervical tumor in patients with cervical cancer.
View Table 2
The control group was composed of 7 patients 55.2 years old +/- 10 (range 42-72). There was no significant difference in age versus the study group (p = 0.17). DW imaging was not obtained for one of the patients in the control group; therefore, DW imaging was available for 6 patients in this group. Table 3 presents the data on a per patient basis.
![]()
Table 3: Demographic, mean R2* and ADC values of normal cervix in controls.
View Table 3
The mean R2* of the cervical cancer is 18.99 s-1 (+/- 7.26, range 11.60-31.70). The mean R2* of the normal cervical stroma in the control group is 31.10 s-1 (+/- 5.5, range 23.49-35.55). R2* of cervical tumor is significantly less than the R2* of normal cervical stroma (p = 0.012). The data is presented in table 4.
![]()
Table 4: Summary of R2* and ADC values in cervical cancer and normal cervical stroma.
View Table 4
The mean ADC of cervical tumors was 0.96 × 10-3 mm2/s (+/- 0.95, range 0.80-1.10) while the mean ADC of normal cervical stroma was 1.35 × 10-3 mm2/s (+/- 0.16, range 1.20-1.60). The ADC of the cervical tumors is significantly lower than that of normal cervical stroma (p = 0.002). The data is presented in table 4.
There was moderate correlation between R2* values and ADC values, with r = 0.78, p = 0.001. The correlation plot is displayed in figure 3.
Discussion
The management of cervical cancer varies by region and outcomes are based on availability of advanced imaging techniques and treatments. Even in developed countries, nearly 40% of women diagnosed with cervical cancer will die from their disease. Although the Federation of Gynecology and Obstetrics (FIGO) classification system continues to be based upon the clinical staging of cervical cancer, they do advocate the use of MRI, when available, to plan the management of patients with more locally advanced and advanced disease (> 1B2). In addition to the basic T1- and T2-weighted MR imaging sequences, functional imaging with DW and perfusion MRI are also used in the evaluation of cervical cancer. Furthermore, there is much interest in investigating the role of functional MRI at baseline and early during therapy to predict tumor response and to focus interval modulated therapy [7]. Our aim was to determine if BOLD MRI could be used as a functional imaging technique to detect differences in oxygen bioavailability between normal cervical stroma and cervical cancer, and to determine if there is a correlation between R2* and ADC values.
The results of our study reveal that there is a significant increase in oxygenation of cervical cancer compared to normal cervical stroma as demonstrated by the significant decrease in the R2* values of cervical cancer compared to normal cervical stroma. The mean R2* value for cervical cancer was 18.99 s-1 +/- 7.26 compared with a mean R2* value of 31.10 s-1 +/- 5.49 for normal cervix (p = 0.012). Similar differences are seen when ADC values are compared. The mean ADC value of cervical cancer was 0.96 × 10-3 mm2/s +/- 0.95, while the mean ADC of normal cervical stroma was 1.35 × 10-3 mm2/s +/- 0.16 (p = 0.002).
To our knowledge, there have only been two other studies investigating the use of BOLD MRI in the setting of cervical cancer. The first study evaluated the response of normal cervical stroma and cervical cancer to a hyperoxic gas challenge (inhalation of 100% oxygen) in a small number of patients [20]; they did not report on static R2* measurements in the cervical tumor. Our results indicate that there is a significant difference in the static R2* values between cervical tumor and normal cervical stroma without a hyperoxic gas challenge. The second study performed by Kim et al. [19] evaluated BOLD MRI at 3T in 30 patients with cervical cancer before and after treatment. These investigators reported a mean R2* value in cervical tumor of 21.1 s-1, which is similar to our reported mean R2* in cervical tumors of 18.99 s-1. While it is important to investigate the utility of BOLD MRI at 3T, our study demonstrates the feasibility at 1.5T, a magnet strength that may be more widely available around the world. Given the high incidence of cervical cancer worldwide, the ability to utilize BOLD with both field strengths potentially increases its utility. Interestingly, Kim et al demonstrated a significant increase in R2* values following treatment, with post-treatment R2* values of 39.4 s-1 at 3T [19]. In light of the difference in field strength affecting the R2* values, their results are similar to the R2* values measured at 1.5 T in our patients with normal cervical tissue (R2* = 31.10 s-1), indicating that treatment may normalize the R2* values in the cervix.
The assessment of cervical cancer with DW MRI has been more extensively studied in the literature and it is well known that there are significant differences in the ADC values of cervical tumor as compared to normal cervical stroma [11-18]. The mean ADC value of the cervical tumors in our study was significantly lower than in normal cervix: 0.96 × 10-3 mm2/s versus 1.35 × 10-3 mm2/s respectively (P = 0.002). In addition to baseline difference between normal cervical tissue and cervical cancer, DW imaging can be used as a biomarker for disease prognosis, treatment response assessment and adaptive treatment of residual cervical cancer [6]. Assessment of the prognosis and response with functional MRI in our patients is ongoing.
We demonstrated only a moderate correlation between R2* and ADC values (Figure 3). Both R2* and ADC values are significantly abnormal in cervical cancer when compared with normal tissue; this in itself may contribute to the underlying correlation between the two functional parameters. However, this moderate degree of correlation may signify that BOLD and DW MRI each provides functional information about different aspects of the underlying tumor physiology. Edema within the tumor affects the intrinsic ADC value and may impact oxygen bioavailability (R2*). There are likely other factors which impact R2*, independent of ADC, such as the permeability of the tumor neovascularity. Certainly, further investigation into how these parameters potentially differ and complement each other in the evaluation of cervical cancer and its response to treatment are needed.
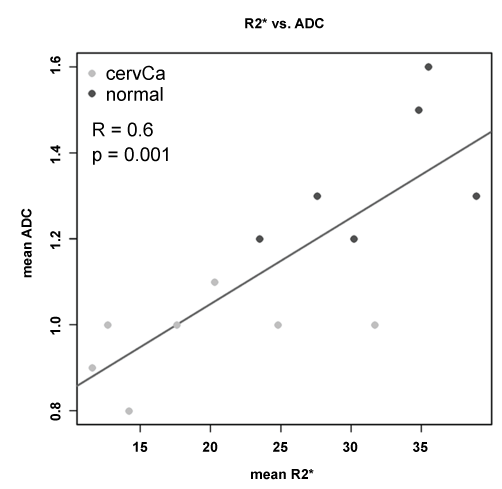
.
Figure 3: Correlation plot of mean ADC as a function of mean R2* in cervical cancer and normal cervix. There is a moderate correlation (R = 0.6; p = 0.001).
View Figure 3
The main limitation of our study includes small sample size. Despite the small population, we were able to demonstrate that there are significant differences in both the R2* and ADC values between cervical cancer and normal cervical stroma. We also cannot currently confirm if the R2* values of the cervix normalize after treatment or if BOLD imaging can predict response to treatment; however this assessment in our population is ongoing. We are using BOLD as a surrogate measure of tumor oxygen content and do not have invasive measurements to confirm in situ measurements of tumor oxygen content; however, this has been performed in other organs and BOLD MRI is an accurate measure of tissue oxygen content [25]. Finally, R2* values can be affected by blood hemoglobin concentration [26]; unfortunately, we were unable to obtain serum hemoglobin values concurrent with the diagnostic imaging for all patients in this study cohort.
In conclusion, there is a significant difference in the oxygen bioavailability and water diffusion within cervical tumors as compared to normal cervical stroma and there is a moderate correlation between R2* and ADC values in the cervix. Since it is well known in the radiotherapy literature that hypoxic tumors have a worse prognosis and well oxygenated tumors are more radiosensitive than those which are poorly oxygenated, more work is needed to determine if the pretreatment R2* value of a cervical tumor can be correlated to prognosis, risk of recurrence, or to early response to therapy. Since BOLD imaging can be performed on any strength magnet and can be performed in a noninvasive fashion, it can be widely disseminated. BOLD MRI has the potential to be a powerful functional tool for the evaluation of cervical tumors and may be able to shape treatment plans and predict outcomes.
References
-
WHO Latest World Cancer Statistics (2013) International Agency for Research on Cancer Press.
-
Waggoner SE (2003) Cervical cancer. Lancet 361: 2217-2225.
-
http://info.cancerresearchuk.org/cancerstats/types/cervix/incidence/
-
Green JA, Kirwan JM, Tierney JF, Symonds P, Fresco L, et al. (2001) Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix: a systematic review and meta-analysis. Lancet 358: 781-786.
-
Survival Rates for Cervical Cancer by Stage (2014) American Cancer Society.
-
Tanderup K, Georg D, Pötter R, Kirisits C, Grau C, et al. (2010) Adaptive management of cervical cancer radiotherapy. Semin Radiat Oncol 20: 121-129.
-
Barwick TD, Taylor A, Rockall A (2013) Functional imaging to predict tumor response in locally advanced cervical cancer. Curr Oncol Rep 15: 549-558.
-
Kido A, Fujimoto K, Okada T, Togashi K (2013) Advanced MRI in malignant neoplasms of the uterus. J Magn Reson Imaging 37: 249-264.
-
Sala E, Rockall A, Rangarajan D, Kubik-Huch RA (2010) The role of dynamic contrast-enhanced and diffusion weighted magnetic resonance imaging in the female pelvis. Eur J Radiol 76: 367-385.
-
Zand KR, Reinhold C, Abe H, Maheshwari S, Mohamed A, et al. (2007) Magnetic resonance imaging of the cervix. Cancer Imaging 7: 69-76.
-
Fu C, Bian D, Liu F, Feng X, Du W, et al. (2012) The value of diffusion-weighted magnetic resonance imaging in assessing the response of locally advanced cervical cancer to neoadjuvant chemotherapy. Int J Gynecol Cancer 22: 1037-1043.
-
Kim HS, Kim CK, Park BK, Huh SJ, Kim B (2013) Evaluation of therapeutic response to concurrent chemoradiotherapy in patients with cervical cancer using diffusion-weighted MR imaging. J Magn Reson Imaging 37: 187-193.
-
Kuang F, Yan Z, Wang J, Rao Z (2014) The value of diffusion-weighted MRI to evaluate the response to radiochemotherapy for cervical cancer. Magn Reson Imaging 32: 342-349.
-
Levy A, Caramella C, Chargari C, Medjhoul A, Rey A, et al. (2011) Accuracy of diffusion-weighted echo-planar MR imaging and ADC mapping in the evaluation of residual cervical carcinoma after radiation therapy. Gynecol Oncol 123: 110-115.
-
Levy A, Medjhoul A, Caramella C, Zareski E, Berges O, et al. (2011) Interest of diffusion-weighted echo-planar MR imaging and apparent diffusion coefficient mapping in gynecological malignancies: a review. J Magn Reson Imaging 33: 1020-1027.
-
Nakamura K, Kajitani S, Joja I, Haruma T, Fukushima C, et al. (2013) The posttreatment mean apparent diffusion coefficient of primary tumor is superior to pretreatment ADCmean of primary tumor as a predictor of prognosis with cervical cancer. Cancer medicine 2: 519-525.
-
Park JJ, Kim CK, Park SY, Park BK, Kim B (2014) Value of diffusion-weighted imaging in predicting parametrial invasion in stage IA2-IIA cervical cancer. Eur Radiol 24: 1081-1088.
-
Somoye G, Harry V, Semple S, Plataniotis G, Scott N, et al. (2012) Early diffusion weighted magnetic resonance imaging can predict survival in women with locally advanced cancer of the cervix treated with combined chemo-radiation. Eur Radiol 22: 2319-2327.
-
Kim CK, Park SY, Park BK, Park W, Huh SJ (2014) Blood oxygenation level-dependent MR imaging as a predictor of therapeutic response to concurrent chemoradiotherapy in cervical cancer: a preliminary experience. Eur Radiol 24: 1514-1520.
-
Hallac RR, Ding Y, Yuan Q, McColl RW, Lea J, et al. (2012) Oxygenation in cervical cancer and normal uterine cervix assessed using blood oxygenation level-dependent (BOLD) MRI at 3T. NMR in biomedicine 25: 1321-1330.
-
Jiang L, Weatherall PT, McColl RW, Tripathy D, Mason RP (2013) Blood oxygenation level-dependent (BOLD) contrast magnetic resonance imaging (MRI) for prediction of breast cancer chemotherapy response: a pilot study. J Magn Reson Imaging 37: 1083-1092.
-
Hockel M, Schlenger K, Aral B, Mitze M, Schaffer U, et al. (1996) Association between tumor hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res 56: 4509-4515.
-
Lim K, Chan P, Dinniwell R, Fyles A, Haider M, et al. (2008) Cervical cancer regression measured using weekly magnetic resonance imaging during fractionated radiotherapy: radiobiologic modeling and correlation with tumor hypoxia. Int J Radiat Oncol Biol Phys 70: 126-133.
-
Sala E, Rockall AG, Freeman SJ, Mitchell DG, Reinhold C (2013) The added role of MR imaging in treatment stratification of patients with gynecologic malignancies: what the radiologist needs to know. Radiology 266: 717-740.
-
Zhang JL, Morrell G, Rusinek H, Warner L, Vivier PH, et al. (2014) Measurement of renal tissue oxygenation with blood oxygen level-dependent MRI and oxygen transit modeling. Am J Physiol Renal Physiol 306: F579-587.
-
Zhao JM, Clingman CS, Närväinen MJ, Kauppinen RA, van Zijl PC (2007) Oxygenation and hematocrit dependence of transverse relaxation rates of blood at 3T. Magn Reson Med 58: 592-597.





