International Journal of Radiology and Imaging Technology
A Retrospective Comparison of Pre-Operative Computed Tomographic Angiography (CTA) Performed at 100 versus 120 kVp Tube Energies and Surgical Findings at Laparoscopic Donor Nephrectomy
Pardeep Mittal1*, Courtney C Moreno1, Jianhai Li1, Kenneth Newell2, William E Torres1 and William Small1
1Departments of Radiology and Imaging Sciences, Emory University School of Medicine, USA
2Department of Surgery, Division of Transplantation, Emory University School of Medicine, USA
*Corresponding author:
Pardeep K Mittal, MD, Department of Radiology & Imaging Sciences, Emory University School of Medicine, 1365 Clifton Road NE, Building A, Suite AT-627, Atlanta, Georgia 30322, USA, Tel: 404-778-3800, Fax: 404-778-3080, E-mail: pmittal@emory.edu
Int J Radiol Imaging Technol, IJRIT-2-017, (Volume 2, Issue 2), Retrospective Review
Received: May 04, 2016; Accepted: July 16, 2016; Published: July 19, 2016
Citation: Mittal P, Moreno CC, Li J, Newell K, Torres WE, et al. (2016) A Retrospective Comparison of Pre-Operative Computed Tomographic Angiography (CTA) Performed at 100 versus 120 kVp Tube Energies and Surgical Findings at Laparoscopic Donor Nephrectomy. Int J Radiol Imaging Technol 2:017.
Copyright: © 2016 Mittal P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction:A retrospective review of patients who underwent preoperative Computed Tomographic Angiography (CTA) prior to living laparoscopic renal donation was performed. Image quality and accuracy were compared at 120 and 100 kVp tube energies with respect to quantitative image parameters and prediction of surgical findings.
Material and methods:A total of 193 patients were reviewed, all of whom underwent laparoscopic donor nephrectomy with preceding CTA evaluation over a 5.5 year period. Eighty-four patients had CTA performed at 120 kVp and 109 at 100 kVp. Images were evaluated using several quantitative parameters, including mean aortic contrast attenuation, noise level, and calculated signal-to-noise (SNR) and contrast-to-noise (CNR) ratios. Comparison was made between accuracy in predicting surgical findings between the two groups. Dosimetry was also compared using the volume CT dose index (CTDIvol).
Results:All CTA evaluations were diagnostically acceptable regardless of tube energy. Mean post-contrast attenuation values were greater in both the aorta and the renal arteries at 100 versus 120 kVp (357.4 and 327.2 versus 270.8 and 239.6 HU), respectively. Correspondingly increased mean noise was also noted: 34.2 versus 24.3 HU, respectively. Both SNR and CNR were slightly increased at lower tube energy but without statistical significance. Per-patient accuracy in predicting surgical vascular anatomy was similar at both energies, 96%. There was a mean CTDIvol reduction of 44.5% at lower tube energy.
Conclusion:Decreasing tube energy from 120 to 100 kVp is a consistently viable method of accurately predicting surgical anatomy at donor nephrectomy with improved vascular enhancement and significant dose reduction.
Keywords
Renal donor, CTA
Introduction
The shortage of organ donors is a well known chronic issue for patients in need of transplant therapy to address organ failure. One source of assistance in addressing a shortage of transplant organs is unique to the issue of renal failure in that, in the normal potential donor (cadaveric or living), there are duplicate organs. In the case of a cadaveric donor, two potential recipients benefit; in the case of a living donor there is the obvious case of only a single recipient. However, there is a significant benefit of a more direct transfer of organ from donor to recipient and a shorter period of cold-ischemia.
In the setting of a living donation, a vigorous program of evaluation is required to ensure the suitability of the donor in general and the best possible match for the designated recipient. From an imaging perspective, such evaluation of a potential donor involves several issues, including verification of the presence of two healthy kidneys, the absence of unexpected extra-renal abnormalities that might compromise the candidate's suitability for the rigors of donation, and determination of the anatomic issues that come into play should the donation go forward. This anatomic checklist includes such issues as the number of arteries and veins, any early arterial branching or venous anomalies that would require vascular reconstruction or at least recognition at the time of surgery, and any issues involving duplication of the collecting systems and ureters. These anatomic elements are that much more crucial given the limited exposure provided by the preferred laparoscopic method of organ harvest [1].
The majority of donor evaluation is done with CT angiography (CTA), which has shown to be effective in this preoperative evaluation process. CTA techniques in general have undergone a significant evolution with the benefit of multidetector arrays, various dose-modulation methods, and the migration to lower energy acquisitions which provide the dual benefit of increased attenuation effects derived from intravenous contrast and inherent decrease in radiation dose.
The current study represents a retrospective review of our experience in CTA prediction of vascular anatomy for patients, all of whom underwent laparoscopic renal donation. We compared the results of two different groups of patients: those imaged under a CTA protocol at 120 kVp and a second group at 100 kVp. Our study had two primary goals: to compare (1) the performance of the two protocols both by the use of several quantitative parameters of vascular enhancement and image quality as well as the ability to prospectively predict surgical findings and (2) the dose-consequence of the change from a 120 kVp to a 100 kVp protocol. Due to quite small size of some accessory renal arteries a decrease of kVp below 100 was not studied in an attempt to preserve diagnostic accuracy while still saving prospective donor radiation exposure.
Materials and Methods
This retrospective study was approved by our Institutional Review Committee Research Board, and the requirement for informed consent was waived. The study protocol was in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
Patients
By searching the radiological and transplant surgery database of electronic medical records, patients who underwent pre-operative CTA as part of a preoperative evaluation prior to renal donation between July 2008 and January 2013 were retrieved. The study cohort included 193 subjects. The mean age was 41.8 ± 11.3 years; ranging from 21 years to 67 years. There were 67 male patients with a mean age of 41.8 ± 10.7 years (range: 21-64 years) and 126 female patients with a mean age of 44.5 ± 11.5 years (range: 23-67 years).
CT technical protocols
Eighty-four patients were scanned under a 120 kVp protocol and 109 patients under a 100 kVp tube energy protocol. The initial 84 patients were scanned consecutively using 120 kVp technique. Subsequently, due to a desire to decrease dose, the next 109 patients were scanned consecutively using a lower, 100 kVp tube potential. Tube energy was fixed at either 120 or 100 kVp during evaluation of the two respective patient populations, no kVp adjustment was made as a consequence of patient size.
Of the patients scanned at 120 kVp, 50 were scanned using a GE 64-slice MDCT unit (GE Medical Systems, Milwaukee, Wisconsin, USA; Light Speed VCT) with a noise index of 21, minimum mA of 100 mAs, maximum mA of 400 mAs, and contrast volume of 100 cc using either Omnipaque 350 or Isovue 370 triggered using bolus tracking of contrast arrival to the aorta with a threshold value of 150 HU. A scan delay of approximately 6 seconds after the aortic enhancement threshold was reached allowed breathing the patient and table repositioning with subsequent arterial phase acquisition at 1.25 mm. A second post-contrast dedicated venous phase was obtained with 2.5 mm collimation after an additional 25 second delay.
The remaining 34 patients scanned at 120 kVp were scanned using a Siemens Definition MDCT scanner (Siemens Medical Solutions, Malvern, PA, USA, Siemens Definition) with effective mA of 270, using either 100 cc of Omnipaque 350 or Isovue 370 and once again bolus triggering of arterial-phase scanning at 1 mm collimation using contrast arrival in the aorta with a threshold value of 150 HU. Venous imaging at 2 mm collimation was performed after a 25 second delay at 2 mm collimation. In all 84 cases, both arterial and venous scan ranges extended from the diaphragm to the mid-pelvis.
The 109 donors scanned at 100 kVp were all evaluated on the same Siemens Definition with identical scan parameters with the exception of the change in tube potential from 120 to 100 kVp.
Image evaluation
As part of pre-operative workup all scans were reviewed by one of three dedicated abdominal imaging-trained radiologists with 15, 20, and 30 years of experience. Images were reviewed to determine: (1) the number of arteries supplying each kidney, (2) the number of veins draining each kidney, (3) any arterial abnormalities, including early branching (defined as branching occurring within 1 cm of the arterial origin from the aorta), (4) any venous abnormalities/variants, including duplication and retroaortic and/or cirmcumaortic variations, and (5) any ureteral duplications (Figure 1).
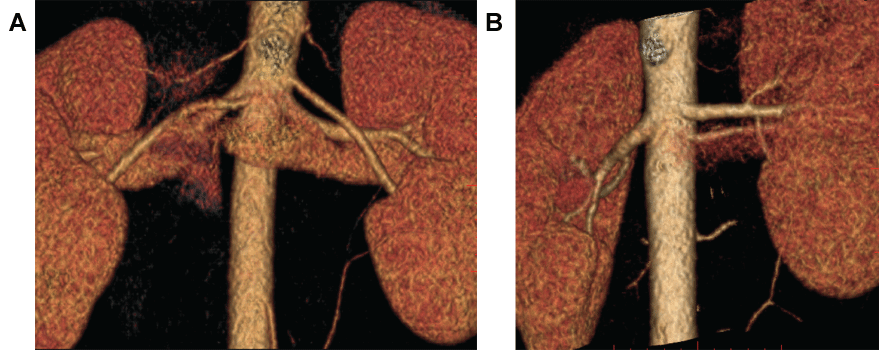
.
Figure 1: Volume-rendered CTA images of two living renal donor patients with vascular anatomy confirmed at laparoscopic left nephrectomy. A) Donor with 3 left renal arteries with imaging performed at 120 kVp; B) Donor with 3 left renal arteries with imaging performed at 100 kVp.
View Figure 1
All CTA interpretations were compared to findings at laparoscopic resection with respect to vascular anatomy (number of arteries, number of veins, vascular anomalies) and number of ureters. The transplant surgeons provided a separate evaluation of the vascular anatomy of the transplant kidney prior to implantation, allowing the ability to accurate compare predicted findings based on CTA to actual surgical findings. The predictive accuracy of surgical findings via CTA studies performed at 120 kVp was compared to those performed at 100 kVp.
Quantitative comparison was made between imaging studies performed at 100 and 120 kVp through several measurements performed by a single investigator with eight years of experience in CTA interpretation. Measurements were confined to scans obtained using the Siemens Definition in order to minimize differences in technique as scan parameters when using this device differed only in tube energy. In all cases, region of interest (ROI) density measurements were obtained of both the aorta (at the level of the main renal artery origins) and the lumen of both dominant renal arteries. Image noise was estimated via region of interests (20-30 mm2) drawn over the two psoas muscles with determination of the mean standard deviation (SD). The signal-to-noise ratio (SNR) was calculated as the measured mean vascular density in the two dominant renal arteries divided by the noise as measured in the psoas muscle. Contrast-to-noise was calculated via a determination of the quantity:
in a manner similar to that performed previously [2].
Data collection
For each patient, the body weight was obtained from the electronic medical record (EMR) (Power Chart, Cerner, North Kansas City, MO, USA) by one of the co-authors. Dosimetry (CTDI) was obtained from a dose report on a picture archiving and communication system (PACS) workstation (GE centricity 2.0) by the same co-author. Dosimetry was compared by evaluating the CTDIvol of the arterial phase of each study. Ranges of values were recorded and the mean arterial phase CTDIvol was calculated for all CTA studies performed at 120 and 100 kVp, respectively. However, only studies performed on the Siemens Definition were compared in order to minimize differences in technique as the only difference in technique for this comparison was in the tube voltage. The majority of the qualitative and quantitative parameters for image quality were derived from the arterial phase portion of the study, as such, the dosimetry from these respective arterial phases were utilized as a surrogate of total dose to evaluate dose savings.
Statistical analysis
Statistical analysis was performed with Microsoft Excel (Microsoft Excel, Microsoft Corporation, Redmond, WA). All quantitative results were expressed in the form mean ± SD. Comparison of patient ages, weights, quantitative vascular densities, SNR and CNR, and mean arterial-phase CTDI was performed using the independent samples Student's t-test with statistical significance defined as p < 0.05.
Results
A total of 193 patients who underwent successful laparoscopic renal harvesting for donation were included in this retrospective study. There was no significant difference in the mean age or weight of men or women between those who underwent CTA at 120 versus 100 kVp (Table 1). All cases involved resection of the left kidney. All patients underwent pre-surgical CTA evaluation with no adverse events related to the CTA procedure or contrast exposure. A total of 109 patients underwent CTA using a 100 kVp protocol and 84 using a 120 kVp protocol. All studies were of diagnostic quality regardless of tube potential. Quantitative results of both vascular attenuation and calculated SNR and CNR values were compared between studies performed at 100 versus 120 kVp, as summarized in table 1. Mean attenuation of both the aorta and the mean of the two dominant renal arteries were statistically higher at 100 kVp. Similarly, noise was found to be higher with lower tube energy. Though there was a trend toward an increase in both SNR and CNR mean values, statistical significance was not reached. Finally, a significant dose reduction benefit was achieved under the 100 kVp protocol, with an overall decrease in mean dose of 44.5%, as estimated by CTDIvol.
![]()
Table 1: Vital statistics and quantitative imaging parameters of different living donor groups who underwent pre-operative CTA imaging at 100 or 120 kVp. Values are listed in the format: mean ± standard deviation, (range).
View Table 1
Table 2 summarizes the degree of variability of vascular supply to the harvested left kidneys, including the number of vessels and patterns of variable early arterial branching and venous anomalies determined by CTA evaluation. In the 84 renal donors who underwent CTA at 120 kVp there were two misinterpretations of arterial anatomy relative to surgical findings: in one donor the left kidney at surgery was found to have three arteries but only two had been recognized at CTA, and in a second donor two supplying left renal arteries were found at surgery but the smaller accessory second vessel was missed at CTA. In both cases, a retrospective review of the case allowed visualization of the originally missed vessel. In each case, the missed vessel size was 1.5 mm or less in size and was ligated at the time of surgery. In addition, a duplication of the IVC was not prospectively appreciated in a third case. Consequently, a correct assessment of vascular anatomy was achieved in 81 of 84 cases, for an overall per-patient accuracy of 96.4%.
![]()
Table 2: Summary of vascular anatomy found at laparoscopic resection of living renal donors with pre-operative CTA at either 100 or 120 kVp.
View Table 2
In the 109 renal donors who underwent CTA at 100 KVp, there were also two misinterpretations of arterial anatomy: one in a donor with three arteries supplying the left kidney and a second in a donor with two arteries. In each case, one of the accessory vessels was missed prospectively by CTA interpretation, although it was seen in retrospect. In a third patient, two small branch arterial aneurysms were missed. Finally, a case of IVC duplication was also missed prospectively in this group. Therefore, a correct assessment of vascular anatomy was achieved in 105 of 109 cases, for an overall per-patient accuracy of 96.3%.
A single ureter was appreciated at imaging in all cases, and this was confirmed at the time of resection in each case.
Discussion
Vascular evaluation using CTA techniques has benefitted from a variety of technological innovations, including multidetector arrays and various dose reduction techniques. Such advancements have improved the speed of imaging with better use of tighter boluses of higher density contrast while still providing high resolution imaging with thin collimation. Lower kVp tube settings provide a higher level of enhancement relative to higher energies due to the closer approximation of the mean energy of the CT beam to the k-edge of the iodine within the contrast agent [3]. Through a lower kVp setting, this increase in contrast is particularly of benefit when the primary goal is centered on vascular imaging (i.e., CTA versus routine CT), and has been used with benefit in a variety of vascular applications in different regions of the body [4,5] and in prior evaluation of lower kVp studies in CT renal donor angiography [2,6]. A corresponding decrease in patient dose is an additional benefit, particularly in CTA applications as there is a geometric decrease in dose with this same kVp decrease that provides increased iodine contrast effect. This decrease in dose is usually maintained even though there is often some degree of mA increase at lower kVp to partially compensate for increased noise at lower kVp. The increase in mA with linear effect on dose is more than compensated by the decrease in dose proportional to square of tube energy provided by kVp decrease.
Using MDCT renal angiography in preparation for laparoscopic donation has been shown to be effective in predicting vascular anatomy and variants, with accuracy ranging from 93-100% [2,7-12]. These studies have largely been done with higher tube energy values ranging from 120-140 kVp but success has also been noted in studies performed at lower energies [2,6]. Our findings in a somewhat larger group of donors - all of whom have surgical correlation - confirms this accuracy level with essentially identical 96% accuracy at both 100 and 120 kVp. Furthermore, re-evaluation of imaging following the appreciation of differences in predicted and actual findings at surgery revealed each of these discrepancies to be errors of perception rather than errors due to non-diagnostic image quality.
The recorded trends within the quantitative imaging parameters were correctly predicted, both in the increase in vascular attenuation of the aorta and renal arteries and the increased noise when tube energy was dropped from 120 to 100 kVp. Overall values of vascular attenuation are somewhat lower when compared to earlier studies [2,6], but this is likely due to a smaller contrast volume. Ratios that incorporate both vascular attenuation and image noise were largely similar with a slight, non-significant increase in mean SNR and CNR. Such values have been found to be dependent on a number of variables, including variances in allowed peak mA, gantry rotation time, and pitch [2]. Preserving a relatively similar SNR and CNR allows for both quantitative and qualitative consistency in image quality when the tube energy is decreased during CTA, even with small target vessels as in renal artery CTA.
There was a significant 44.5% decrease in dose as estimated by mean CTDIvol in studies performed at 100 kVp. While this dose reduction is similar in magnitude to that seen with studies performed at 100 and 80 kVp, there is some variability due to the inconstancy of the scanner parameters described above. This study has a large number of surgical confirmations, adding to the evidence that the reduced contrast exposure afforded at lower energy still preserves vascular characterization with a high level of accuracy. In our institution there is a weekly conference where imaging as well as non-imaging findings are reviewed for prospective renal donors by the surgeons and transplant nephrologists. Our surgical and nephrology colleagues were completely satisfied with the image quality at the lower 100 kVp.
Our study has several limitations. While this study captures a larger group of donors than previous studies, the study size in each group was still relatively small and the comparisons of imaging findings and their concordance with surgical anatomy was retrospective. In addition, the sample size for dose comparison was smaller still because evaluations done on the GE 64-slice were excluded in order to eliminate further confounding variables. Surgical correlation was also only possible for the side of resection (in all cases the left kidney), so appreciation of potential errors in determining the vascular anatomy of the right kidney was not possible as no right kidney was harvested during this series of patients. Also, we did not attempt to evaluate the effect of other features such as body habitus. Finally, our findings were largely centered on that of vascular anatomy. Small lesions, typically cysts or occasional small angiomyolipomas, typically less than 5 mm seen during imaging could not be consistently verified at the time of surgery due to their small size and as such any decreased ability to detect or characterize small lesions by decreasing kVp from 120 to 100 could not be evaluated.
Our results provide additional support for the migration of CTA vascular imaging to lower tube potential protocols in the application of surgical planning for laparoscopic resection. Even in such scenarios where consistent small vessel detection is needed, the overall image quality and improved vascular enhancement at lower kVp preserves accurate vessel detection along with a significant reduction in radiation exposure.
References
-
Ratner LE, Ciseck LJ, Moore RG, Cigarroa FG, Kaufman HS, et al. (1995) Laparoscopic live donor nephrectomy. Transplantation 60: 1047-1049.
-
Davarpanah AH, Pahade JK, Cornfeld D, Ghita M, Kulkarni S, et al. (2013) CT Angiography in Potential Living Kidney Donors: 80 kVp Versus 120 kVp. AJR Am J Roentgenol 201: w753-760.
-
Curry TS, Dowdey JE, Murry RC (1990) Basic interactions between X-rays and matter. In: Curry TS, Dowdey JE, Murry RC, Christensen's physics of diagnostic radiology. (4th edn), Lippincott Williams & Wilkins, Philadelphia, PA, 61-69.
-
Ertl-Wagner BB, Hoffmann RT, Bruning R, Herrmann K, Snyder B, et al. (2004) Multidetector row CT angiography of the brain at various kilovoltage settings. Radiology 231: 528-535.
-
Sigal-Cinqualbre AB, Hennequin R, Abada HT, Chen X, Paul JF (2004) Low-kilovoltage multidetector row chest CT in adults: feasibility and effect on image quality and iodine dose. Radiology 231: 169-174.
-
Sahani DV, Kalva SP, Hahn PF, Saini S (2007) 16-MDCT Angiography in Living Kidney Donors at Various Tube Potentials: Impact on Image Quality and Radiation Dose. AJR 188: 115-120.
-
Chu LC, Sheth S, Segev DL, Montgomery RA, Fishman EK (2012) Role of MDCT Angiography in Selection and Presurgical Planning of Potential Renal Donors. AJR Am J Roentgenol 199: 1035-1041.
-
Chai JW, Lee W, Yin YH, Jae HJ, Chung JW, et al. (2008) CT angiography for living kidney donors: accuracy, cause of misinterpretation and prevalence of variation. Korean J Radiol 9: 333-339.
-
Laugharne M, Haslam E, Archer L, Jones L, Mitchell D, et al. (2007) Multidetector CT angiography in live donor renal transplantation: experience from 156 consecutive cases at a single centre. Transpl Int 20: 156-166.
-
Namasivayam S, Small WC, Kalra MK, Torres WE, Newell KA, et al. (2006) Multidetector-row CT angiography for preoperative evaluation of potential laparoscopic renal donors: how accurate are we? Clin Imaging 30: 120-126.
-
Sahani DV, Rostogi N, Greenfield AC, Kalva SP, Ko D, et al. (2005) Multi-detector row CT in evaluation of 94 living renal donors by readers with varied experience. Radiology 235: 905-910.
-
Raman SS, Pojchamarnwiputh S, Muangsomboon K, Schulam PG, Gritsch HA, et al. (2006) Utility of 16-MDCT angiography for comprehensive preoperative vascular evaluation of laparoscopic renal donors. AJR Am J Roentgenol 186: 1630-1638.





