International Journal of Radiology and Imaging Technology
The Utility and Inter-Reader Agreement in the Evaluation of Pediatric Liver Masses with Hepatobiliary Contrast Enhanced MRI
Hansel J Otero1*, Asef B Khwaja2, Maria A Bedoya3, Nancy Chauvin2, Kassa Darge2 and Sudha A Anupindi2
1Department of Diagnostic Imaging and Radiology, University School of Medicine & Health Sciences, USA
2Department of Radiology, University of Pennsylvania Perelman School of Medicine, USA
3Department of Radiology, Hospital of the University of Pennsylvania, USA
*Corresponding author:
Hansel J Otero, MD, Department of Diagnostic Imaging and Radiology, Children's National Health System, George Washington University School of Medicine & Health Sciences, 111 Michigan Avenue, NW Washington, DC 20010, USA, Tel: 202-476-4252, Fax: 202-476-3644, E-mail: hotero@childrensnational.org
Int J Radiol Imaging Technol, IJRIT-3-020, (Volume 3, Issue 1), Original Research
Received: August 17, 2016 | Accepted: January 06, 2017 | Published: January 09, 2017
Citation: Otero HJ, Khwaja AB, Bedoya MA, Chauvin N, Darge K, et al. (2017) The Utility and Inter-Reader Agreement in the Evaluation of Pediatric Liver Masses with Hepatobiliary Contrast Enhanced MRI. Int J Radiol Imaging Technol 3:020.
Copyright: © 2017 Otero HJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To evaluate the utility of Gd-EOB-DTPA-enhanced MRI in pediatric liver lesions.
Methods and results: 22 patients with liver lesions evaluated on Gd-EOB-DTPA-enhanced MRI were selected. Two reviewers assigned a diagnosis and stratified the hepatobiliary images as: 1) High utility; 2) Potential utility; or 3) No utility.
95% of assigned diagnoses were correct with almost-perfect agreement (κ = 0.92). Hepatobiliary images were deemed useful by at least one reviewer in 82% cases with moderate agreement (k = 0.48).
Conclusion: Gd-EOB-DTPA-enhanced MRI had high or potential utility in the majority of cases with moderate inter-reader agreement and seemingly demonstrating a promising role in characterizing pediatric liver lesions.
Keywords
Hepatobiliary contrast, Gd-DTPA-EOB, Children, MRI, Liver masses, Inter-reader agreement, Utility
Introduction
Magnetic resonance imaging (MRI) is the modality of choice for imaging the liver and biliary tract in children, virtually replacing CT and ERCP [1]. Gadoxetate disodium (Gd-EOB-DTPA, Eovist, Bayer HealthCare Pharmaceuticals, Wayne, NJ) is a relatively new hepatocyte specific MR contrast agent useful for detecting and characterizing liver lesions. This agent highlights functioning hepatocyte-containing lesions and improves the depiction of lesions that lack functioning hepatocytes. Additionally, the same imaging techniques can be used for assessment of hepatocellular function and functional evaluation of the biliary tract [2]. The vast majority of clinical experience with this agent is in adults, however, increasing use among children is being reported [3,4]. In children, Gd-EOB-DTPA enhanced MRI has the same potential to improve characterization of liver lesions [5-7]. Only a few original publications have reported the initial clinical experience with Gd-EOB-DTPA in children [3,4,7]. A recent publication discusses initial experience with gadobenate dimeglumine (Gd-BOPTA)-enhanced MRI, a contrast agent with similar hepatobiliary affinity [8]. However, the hepatobiliary excretion of Gd-BOPTA is lower (3-5%) compared to Gd-EOB-DTPA (as much as 50%); allowing for 1) a lower dose of Gd-EOB-DTPA; 2) earlier imaging of the hepatobiliary phase (i.e.; 10-20 minutes with Gd-EOB-DTPA compared to 45-60-minute delay with Gd-BOPTA); and 3) shorter table time studies [9]. Additionally, the largest study to date evaluating Gd-EOB-DTPA in children was based on a single reviewer perceived diagnostic confidence [7]. Our objective was to investigate the inter-reader agreement and perceived utility of Gd-EOB-DTPA enhanced MRI in the evaluation of liver lesions in children.
Materials and Methods
Patient selection
This Health Insurance Portability and Accounting Act-compliant retrospective study was approved by our institutional review board. All patients who had undergone Gd-EOB-DTPA enhanced MRI examination at our institution between March 1, 2011 and July 31, 2013 were initially selected. MRI studies with a biliary indication (i.e.; for the evaluation of biliary obstruction, strictures or leaks) (n = 3) as well as the studies with no identifiable focal lesion (n = 9) were excluded. The studies that showed one or more focal liver lesions were selected for the final study group. If a single patient had more than one study, only the first one was included in the final study group. Patients' demographics, history and follow-up results were obtained from our electronic medical records.
MRI imaging protocol
All patients underwent abdominal MRI with either a 1.5-T MRI system (Avanto; Siemens Medical Systems, Erlangen, Germany) or a 3.0-T system (Trio; Siemens Medical Systems, Erlangen, Germany). Typical imaging protocol included axial and coronal T1 weighted images including axial T1 in and out of phase as well axial T1 weighted with fat saturation before the administration of intravenous Gd-EOB-DTPA. The contrast was administered using a power injector at a dose of 0.1ml/kg (0.025 m mol/kg) of body weight with a flow rate of 1ml/sec, followed by injection of 5-10mL of sterile saline solution. Dynamic axial contrast enhanced volumetric T1 weighted GRE images were obtained after Gd-EOB-DTPA administration in the axial plane in the arterial (10 seconds), portal-venous (45-55 seconds) and 3 minutes delayed venous phases. Axial and coronal fat saturated single shot fast spine echo (SSFSE) images, axial T2 weighted with fat saturation, axial diffusion weighted images and coronal balanced steady-state free precession without fat saturation images were obtained. Subsequently, axial and coronal T1 weighted GRE with fat saturation images during hepatobiliary phase (at least 20-minutes post-injection) were also performed. The typical imaging protocol is summarized in table 1.
![]()
Table 1: Typical pulse sequence protocol for liver tumor examination with Gadoxetate disodium-enhanced MRI.
View Table 1
Image analysis
Two pediatric radiologists (R1 and R2) with 15 years and 14 years of experience, respectively, independently reviewed anonymized images including dynamic T1-weighted post contrast images for each lesion under investigation. The two reviewers were blinded to the clinical data, but were disclosed gender and age of each subject. Each reviewer assigned a diagnosis after reviewing the initial images (T1, T2 and post contrast arterial, portal-venous and venous phase imaging). Subsequently, during the same session, additional images during the hepatobiliary phase were shown and the reviewers were asked to confirm their initial diagnosis and to stratify the hepatobiliary images as a score of: 1) High utility: allows for definitive diagnosis or change the differential diagnosis; 2) Potential utility: adds diagnostic certainty; 3) No utility: final diagnosis was reached before hepatobiliary phase images.
Statistical analysis
Descriptive analysis of patients' demographics, study indications, imaging findings and final clinical diagnosis was performed. The hepatobiliary phase utility scores were summarized and inter-reader variability was assessed using weighted kappa coefficient. The following value guidelines were followed in interpreting the kappa statistic: slight agreement: 0.00-0.20; fair agreement: 0.21-0.40; moderate agreement: 0.41-0.60; substantial agreement: 0.61-0.80; and almost perfect agreement: 0.80-1.00 [10]. All statistics were calculated using publicly available statistical software (Vassar Stats: Statistical Computation Website) [11].
Results
Twenty-two patients, meeting our inclusion criteria, (17 male, 5 female) ages 4 months -19 years (median: 9 years; 8 patients younger than 7 years and 14 patients 8 years of age and older) underwent Gd-EOB-DTPA-MRI for the evaluation of liver lesions during the study period. Fifteen patients (68%) underwent imaging in a 1.5 Tesla magnet while the remaining 7 (32%) underwent imaging in a 3 Tesla magnet. Of the 22 patients with liver lesions, 21 underwent Gd-EOB-DTPA-MRI for further evaluation of lesions identified by a different imaging study (11 from CT, 8 from ultrasound and 2 from non-dedicated MRI); while the remaining patients had a diagnosis of Alagille syndrome and underwent Gd-EOB-DTPA-MRI as screening for hepatocellular carcinoma because of the inherent increased risk. Seven of the 22 patients (31%) had a past medical history of cancer including neuroblastoma (n = 3), hepatoblastoma (n = 1), medulloblastoma (n = 1), leukemia (n = 1), and granulosa cell tumor (n = 1). The final diagnosis of the lesions was: focal nodular hyperplasia-FNH-, FNH-like, or adenoma (10), hepatoblastoma (3), Abscesses (2), regenerative nodules (3), hemangioma (2), focal fatty infiltration (1) and coagulative necrosis (1). Final imaging and clinical diagnoses as well as follow up data are summarized in table 2.
![]()
Table 2: Summary of study findings.
View Table 2
There was only one discrepancy between the reviewers' assigned diagnosis and the final clinical/pathological diagnosis. It was a case of a rim-enhancing lesion, which on biopsy proved to represent multiple discrete areas of coagulative necrosis with adjacent reactive changes of chronic inflammation and portal fibrosis. However, both reviewers characterized it as an abscess. In the final diagnosis of the focal liver lesions, there was inter-reader agreement in 20 of 22 cases with an almost perfect kappa coefficient (κ = 0.92). In the 2 cases in which the final diagnosis differed between the reviewers, one reviewer labeled the hepatic lesions as concerning and needing biopsy, while the second reviewer labeled the lesions as likely benign, regenerative nodule or FNH-like lesion, which would undergo imaging follow up. The former of the two patients underwent biopsy, which showed biliary cirrhosis with acute cholangitis (Figure 1); while the second patient underwent short term imaging follow up, which was reassuring for FNH-type lesion and was stable over the next 3 years.
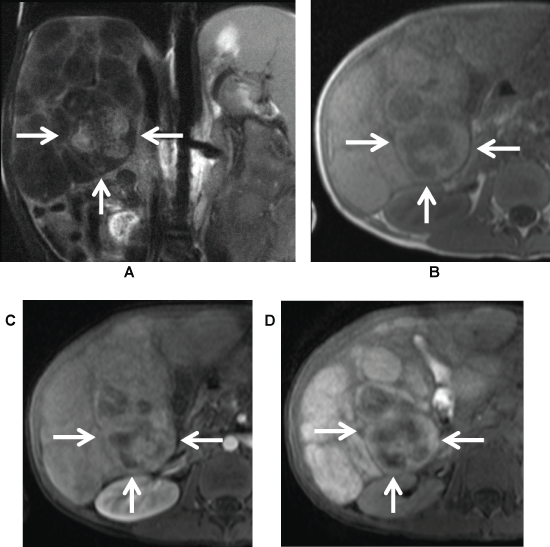
.
Figure 1: 9-year-old boy with history of cystic fibrosis and new focal lesion on CT. MRI shows A) Right hepatic lobe lesion (arrows) heterogeneously hyperintense in Coronal T2-weighted image; B) Hypointense in T1-weighted images; C) Shows heterogeneous enhancement in post contrast; D) Hepatobiliary phase images.
At biopsy this was found to represent focal irregular, severe fibrosis in the setting of multilobular biliary cirrhosis resulting from cystic fibrosis.
View Figure 1
In analysis of the added utility of Gd-EOB-DTPA, hepatobiliary phase images were given high utility scores in n = 10 cases (reviewer 1) and n = 6 cases (reviewer 2); while potential utility scores were given to n = 5 cases (reviewer 1) and n = 8 cases (reviewer 2) and no utility for n = 7 cases (reviewer 1) and n = 8 cases (reviewer 2). There was a moderate inter-reader agreement for the overall utility scores (weighted k = 0.48). However, there is high agreement regarding utility in cases of FNH/FNH-like lesions (Figure 2), regenerative nodules (Figure 3) and focal fatty infiltration as well as high agreement in the lack of utility for venous malformation ("adult hemangioma") and hepatoblastoma cases. Overall, the hepatobiliary images were deemed useful (scores of 1 or 2) by both reviewers in 50% (11/22) cases and by at least one reviewer in additional 32% (7/22) cases. Consequently, the hepatobilliary phase images during Gd-EOB-DTPA enhanced-MR are useful or at least potentially useful in a great majority (82%) of cases. In the remaining 18% (4/22) cases both reviewers agreed that hepatobiliary phase images were of no utility.
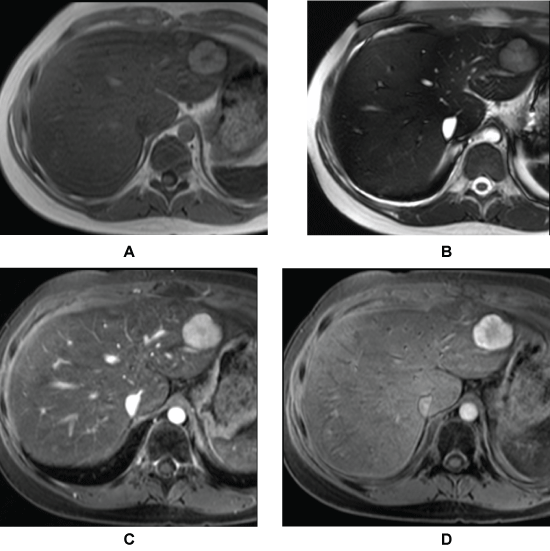
.
Figure 2: 14-year-old girl with history of leukemia and whole body radiation with left hepatic lobe lesion, which is A) Mildly hyperintense; B) T1; C) T2-weighted images and enhances avidly in post contrast image; D) Hepatobilliary phase image confirms the diagnosis of Focal Nodular Hyperplasia with avid hepatobiliary uptake.
View Figure 2
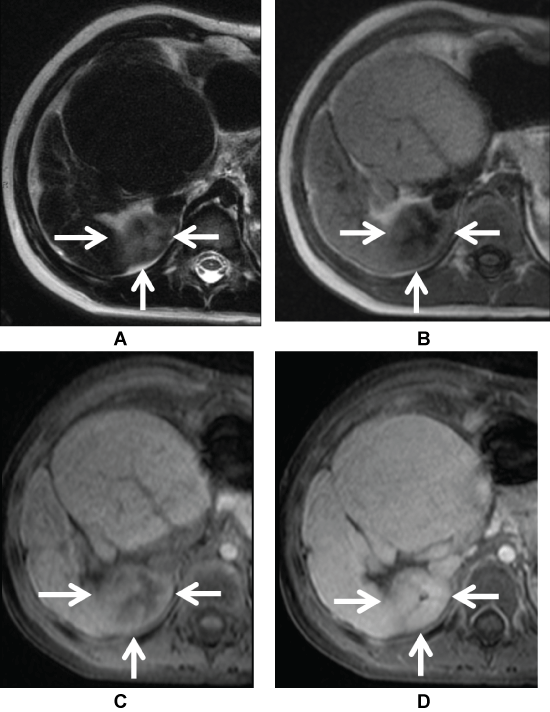
.
Figure 3: 4-year-old girl with Alagille syndrome related cirrhosis. Surveillance MR shows A) T2-weighted; B) Focal hyperintense lesion in the posterior segment of the right hepatic lobe with corresponding heterogeneous hypointensity on T1-weighted image; C) There is peripheral enhancement in early post-contrast image; D) Homogeneous enhancement, which is isointense to the rest of the liver on hepatobilliary phase image confirms the diagnosis of regenerative nodule.
View Figure 3
Discussion
The present study found almost perfect agreement between readers in diagnosing the liver lesions on liver MRI; which included Gd-EOB-DTPA-enhanced images. This highlights the excellent performance of contrast enhanced MR imaging in characterizing liver lesions, a well-established practice supported by literature in both adults [12-16] and children [1,17,18] before and after the advent of hepatobiliary contrast agents. Moreover, the results showed that hepatobiliary phase images are useful or at least potentially useful in a great majority (82%) of cases. These results are in accordance with previous studies that found significant improvement in the ability to characterize focal hepatic lesions with the addition of Gd-EOB-DTPA [7,19-21].
The results also showed moderate inter-reader agreement in terms of utility, which highlighted consensus regarding the type of lesions in which hepatobiliary phase images are most useful, namely FNH/FNH-like lesions and regenerative nodules. These results concur with previous reports in both the adult [22] and pediatric [7,8] literature.
High inter-reader agreement was seen regarding the type of lesions in which hepatobiliary contrast images are not useful, namely hepatoblastomas and hemangiomas. Regarding hepatoblastomas, the lack of perceived utility is most likely due to the limited differential diagnosis for large heterogeneous aggressive appearing enhancing hepatic masses seen in the 5 months- 3-year age group. Additionally, the study was not designed to identify additional lesions or stage malignancies. However, a prior publication highlighted the usefulness of Gd-EOB-DTPA in defining the relationship of the primary tumor with respect to hepatic vasculature and identifying intrahepatic lesions distant from the main tumor [4]. Hence, the previous publication does not contradict our findings but offers insight in other potential benefits of Gd-EOB-DTPA use beyond diagnosis [4].
Now, regarding hemangiomas, research has shown that some hemangiomas have a different enhancement pattern during Gd-DTPA-EOB than with purely extracellular gadolinium contrast, making it difficult to diagnose and in some cases this lesion may mimic hypervascular malignant tumors [23,24]. The added difficulty is due to the lack of hepatocyte phase contrast uptake that results in relative hypointensity of the lesion, which might be misinterpreted as "washout", particularly in small lesions that enhanced homogeneously in early images [25-27]. However, hemangiomas are usually characterized with confidence before hepatobiliary phase imaging. Hence, the variable pattern of enhancement during hepatobiliary phase is of little use and does not add to the radiologist's diagnostic confidence [7].
Differentiation between regenerating or dysplastic nodules and differentiated hepatocellular carcinoma (HCC) in the cirrhotic liver based on imaging alone can be challenging and at times impossible. However, lack of suspicious features on MRI might suffice to enroll the patient in long-term follow up to document stability over time. Hence, the additional information provided by Gd-EOB-DTPA has the potential to increase diagnostic confidence and in turn decrease the number of patients that require additional work-up, short term follow up and biopsies. In our sample, only few lesions required additional short term imaging (n = 4) or biopsy (n = 3); which creates a limitation for the study, but serves as further proof that MRI is clinically relevant in separating patients requiring additional work up and those that can be safely followed. The benefit might be even higher in a selected population with increased incidence of benign lesions; such as FNH-like lesions in cancer survivors or regenerative nodules in the setting of cirrhosis [28]. In these cases, the identification of FNH or regenerative nodules allows patients to return to their regular follow-up schedule.
Our study has several limitations mainly related to its retrospective nature, the small sample size and lack of pathologic correlate in a majority of the cases. Additionally, the utility scale is subjective and as such introduces a degree of "personal preference" into the analysis. However, the correlation among readers and the low number of patients that clinically required additional imaging follow-up or biopsy are encouraging findings that support the selective use of hepatobiliary contrast for the evaluation of focal liver lesions.
Our study showed excellent inter-reader agreement in characterizing liver lesions with Gd-EOB-DTPA, which is in keeping with a growing body of evidence suggesting an increased MR accuracy in the depiction and characterization of liver lesions using Gd-EOB-DTA [3,6,7,29,30]. The results also showed a perceived utility in a majority of cases with moderate inter-reader agreement, which means that there is potential for prospectively identifying whether hepatobiliary contrast is likely to be useful or not. Prospectively identifying the cases in which Gd-EOB-DTPA contrast is unlikely to be useful, is challenging, but attainable through careful scrutiny of clinical and laboratory data. Limiting the use of Gd-EOB-DTA to those studies with high pretest yield might prove a suitable approach to its incorporation in common clinical practice. Alternatively, Gd-EOB-DTPA could be used for characterization of focal liver lesions in all pediatric patients without sacrificing accuracy or diagnostic confidence.
Conclusion
Gd-EOB-DTPA enhanced MRI ranked as having high or potential utility in the majority of our cases with moderate inter-reader agreement and seems to demonstrate a promising role in improving the diagnosis and characterization of pediatric liver lesions.
References
-
Mitchell CL, Vasanawala SS (2011) An approach to pediatric liver MRI. AJR Am J Roentgenol 196: W519-W526.
-
Huppertz A, Haraida S, Kraus A, Zech CJ, Scheidler J, et al. (2005) Enhancement of focal liver lesions at gadoxetic acid-enhanced MR imaging: correlation with histopathologic findings and spiral CT--initial observations. Radiology 234: 468-478.
-
Tamrazi A, Vasanawala SS (2011) Functional hepatobiliary MR imaging in children. Pediatr Radiol 41: 1250-1258.
-
Meyers AB, Towbin AJ, Geller JI, Podberesky DJ (2012) Hepatoblastoma imaging with gadoxetate disodium-enhanced MRI--typical, atypical, pre- and post-treatment evaluation. Pediatr Radiol 42: 859-866.
-
Courtier JL, Perito ER, Rhee S, Tsai P, Heyman MB, et al. (2012) Targeted MRI Contrast Agents for Pediatric Hepatobiliary Disease. J Pediatric Gastroenterol Nutr 54: 454-462.
-
Meyers AB, Towbin AJ, Serai S, Geller JI, Podberesky DJ (2011) Characterization of pediatric liver lesions with gadoxetate disodium. Pediatr Radiol 41: 1183-1197.
-
Kolbe AB, Podberesky DJ, Zhang B, Towbin AJ (2015) The impact of hepatocyte phase imaging from infancy to young adulthood in patients with a known or suspected liver lesion. Pediatr Radiol 45: 354-365.
-
Chavhan GB, Mann E, Kamath BM, Babyn PS (2014) Gadobenate-dimeglumine-enhanced magnetic resonance imaging for hepatic lesions in children. Pediatr Radiol 44: 1266-1274.
-
Seale MK, Catalano OA, Saini S, Hahn PF, Sahani DV (2009) Hepatobiliary-specific MR contrast agents: role in imaging the liver and biliary tree. Radiographics 29: 1725-1748.
-
Viera AJ, Garrett JM (2005) Understanding interobserver agreement: the kappa statistic. Fam Med 37: 360-363.
-
Lowry R, VassarStats: Website for Statistical Computation.
-
Hamm B, Thoeni RF, Gould RG, Bernardino ME, Lüning M, et al. (1994) Focal liver lesions: characterization with nonenhanced and dynamic contrast material-enhanced MR imaging. Radiology 190: 417-423.
-
Semelka RC, Martin DR, Balci C, Lance T (2001) Focal liver lesions: comparison of dual-phase CT and multisequence multiplanar MR imaging including dynamic gadolinium enhancement. J Magn Reson Imaging 13: 397-401.
-
Elsayes KM, Narra VR, Yin Y, Mukundan G, Lammle M, et al. (2005) Focal hepatic lesions: diagnostic value of enhancement pattern approach with contrast-enhanced 3D gradient-echo MR imaging. Radiographics 25: 1299-1320.
-
Silva AC, Evans JM, McCullough AE, Jatoi MA, Vargas HE, et al. (2009) MR imaging of hypervascular liver masses: a review of current techniques. Radiographics 29: 385-402.
-
Chung YE, Kim MJ, Kim YE, Park MS, Choi JY, et al. (2013) Characterization of incidental liver lesions: comparison of multidetector CT versus Gd-EOB-DTPA-enhanced MR imaging. PLoS One 8: e66141.
-
Finn JP, Hall-Craggs MA, Dicks-Mireaux C, Spitz L, Howard ER, et al. (1990) Primary malignant liver tumors in childhood: assessment of resectability with high-field MR and comparison with CT. Pediatr Radiol 21: 34-38.
-
Weinreb JC, Cohen JM, Armstrong E, Smith T (1986) Imaging the pediatric liver: MRI and CT. AJR Am J Roentgenol 147: 785-790.
-
Raman SS, Leary C, Bluemke DA, Amendola M, Sahani D, et al. (2010) Improved characterization of focal liver lesions with liver-specific gadoxetic acid disodium-enhanced magnetic resonance imaging: a multicenter phase 3 clinical trial. J Comput Assist Tomogr 34: 163-172.
-
Halavaara J, Breuer J, Ayuso C, Balzer T, Bellin MF, et al. (2006) Liver tumor characterization: comparison between liver-specific gadoxetic acid disodium-enhanced MRI and biphasic CTVa multicenter trial. J Comput Assist Tomogr 30: 345-354.
-
Haimerl M, Wächtler M, Platzek I, Müller-Wille R, Niessen C, et al. (2013) Added value of Gd-EOB-DTPA-enhanced Hepatobiliary phase MR imaging in evaluation of focal solid hepatic lesions. BMC Med Imaging 13: 41.
-
Ichikawa T, Saito K, Yoshioka N, Tanimoto A, Gokan T, et al. (2010) Detection and characterization of focal liver lesions: a Japanese phase III, multicenter comparison between gadoxetic acid disodium-enhanced magnetic resonance imaging and contrast-enhanced computed tomography predominantly in patients with hepatocellular carcinoma and chronic liver disease. Invest Radiol 45: 133-141.
-
Pradella S, Lucarini S, Colagrande S (2012) Liver lesion characterization: the wrong choice of contrast agent can mislead the diagnosis of hemangioma. AJR Am J Roentgenol 199: W662.
-
Tateyama A, Fukukura Y, Takumi K, Shindo T, Kumagae Y, et al. (2012) Gd-EOB-DTPA-enhanced magnetic resonance imaging features of hepatic hemangioma compared with enhanced computed tomography. World J Gastroenterol 18: 6269-6276.
-
Goshima S, Kanematsu M, Watanabe H, Kondo H, Shiratori Y, et al. (2010) Hepatic hemangioma and metastasis: differentiation with gadoxetate disodium-enhanced 3-T MRI. AJR Am J Roentgenol 195: 941-946.
-
Tamada T, Ito K, Yamamoto A, Sone T, Kanki A, et al. (2011) Hepatic hemangiomas: evaluation of enhancement patterns at dynamic MRI with gadoxetate disodium. AJR Am J Roentgenol 196: 824-830.
-
Doo KW, Lee CH, Choi JW, Lee J, Kim KA, et al. (2009) "Pseudo washout" sign in high-flow hepatic hemangioma on gadoxetic acid contrast-enhanced MRI mimicking hypervascular tumor. AJR Am J Roentgenol 193: W490-496.
-
Cha DI, Yoo SY, Kim JH, Jeon TY, Eo H (2014) Clinical and imaging features of focal nodular hyperplasia in children. AJR Am J Roentgenol 202: 960-965.
-
Ye F, Liu J, Ouyang H (2015) Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid (Gd-EOB-DTPA)-Enhanced Magnetic Resonance Imaging and Multidetector-Row Computed Tomography for the Diagnosis of Hepatocellular Carcinoma: A Systematic Review and Meta-analysis. Medicine (Baltimore) 94: e1157.
-
Donati OF, Hunziker R, Fischer MA, Raptis DA, Breitenstein S, et al. (2014) MRI for characterization of primary tumors in the non-cirrhotic liver: added value of Gd-EOB-DTPA enhanced hepatospecific phase. Eur J Radiol 83:1074-1079.





