International Journal of Stem cell Research and Therapy
Enhancement of Human Adipose-Derived Stem Cell Expansion and Stability for Clinical use
Swenn Maxence Krähenbüh1, Anthony Grognuz, Murielle Michetti, Wassim Raffoul and Lee Ann Applegate*
Plastic, Reconstructive and Hand surgery, Regenerative Therapy Unit, University Hospital of Lausanne, Switzerland
*Corresponding author: Lee Ann Laurent-Applegate, University Hospital of Lausanne (CHUV), Department of Plastic and Hand Surgery, Regenerative Therapy Unit (UTR), EPCR/Croisettes 22, CH-1066 Epalinges, Switzerland, Tel: 41 21 314 3510, E-mail: Lee.Laurent-Applegate@chuv.ch
Int J Stem Cell Res Ther, IJSCRT-2-007, (Volume 2, Issue 1), Research Article; ISSN: 2469-570X
Received: March 28, 2015 | Accepted: April 04, 2015 | Published: April 06, 2015
Citation: Krähenbühl SM, Grognuz A, Michetti M, Raffoul W, Applegate LA (2015) Enhancement of Human Adipose-Derived Stem Cell Expansion and Stability for Clinical use. Int J Stem Cell Res Ther 2:007. 10.23937/2469-570X/1410007
Copyright: © 2015 Krähenbühl SM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Co-culture techniques associating both dermal fibroblasts and epidermal keratinocytes have shown to have better clinical outcome than keratinocyte culture alone for the treatment of severe burns. Since fat grafting has been shown to improve scar remodelling, new techniques such as cell-therapy-assisted surgical reconstruction with isolated and expanded autologous adipose-derived stem cells (ASCs) would be of benefit to increase graft acceptation. Therefore, integrating ASCs into standardized procedures for cultured skin grafting could be of benefit for the patient if cell quality and quantity could be maintained. The purpose of this study was to evaluate ASC processing from adult tissue with simple isolation (without enzymatic steps), expansion (low density of 325-3,000 cells/cm2) and storage conditions to assure methods to enhance the cellular resistance when transferred back to the patient. Co-culture with cell-banked skin progenitor cells (FE002-SK2) showed an increase of 40-50% ASCs yield at high passages alongside with a better preservation of morphology, proper adipogenic and osteogenic differentiation and efficient biocompatibility with 3D collagen scaffolds. ASCs can be considered as a valuable additional cell source to be delivered in biological bandages to the patient in a need of tissue reconstruction such as burn patients.
Keywords
Adipose-derived stem cells, Cell stability, Co-culture, Fat grafting, Lipofilling, Burn treatment, Biological bandages
Introduction
Multipotent stem cells may be beneficial in tissue engineering applications for reconstructive surgery, particularly for soft tissue enhancement and around implants for better integration [1,2] and potentially for burn patients [3-6].
Until recently, the predominant source of stem cells considered for clinical applications was bone marrow-derived mesenchymal stem cells (BM-MSCs). More recent studies using fresh stem cells derived from white adipose tissue, termed adipose-derived stem cells (ASCs) [7], have confirmed them as multipotent mesenchymal stem cells [8-11]. Advantages are high availability and easy accessibility from direct excision or from fresh lipoaspirate [12]. It was shown that their therapeutic potential is equal or even superior compared to BM-MSCs because of their higher number and viability upon transplantation [8,13].
In severely burned patients, it is now accepted that co-culture of both fibroblasts (dermal) and keratinocytes (epidermal) provides a better clinical outcome for burn applications [14]. With such patients, there is also a lack of soft tissue and adipose tissue injection has been proposed to help in scar tissue remodelling [3]. Therefore, with standardized methods for ASC culture using small quantities of tissue (that could be taken during initial debridement and biopsy for keratinocyte culture), these patient cells could be easily integrated into the co-cultured treatment for burns and perhaps provide a better overall treatment for skin and soft tissue reconstruction.
Isolation methods from lipoaspirate digested using collagenase, filtered and finally centrifuged have been used in research [2]. Recent techniques have shown that less aggressive enzymatic treatment using trypsin could be employed for better cellular stability but had lower overall cell yields. Reports have demonstrated that although mechanically isolated ASCs (mincing) revealed lower cell numbers, the viability was higher [15-17]. In addition, Zeng et al. [18] have shown isolation of ASCs without collagenase or trypsin although they treated the tissue for 3 consecutive days with 100% fetal bovine (FBS) serum to "digest" the tissue and allow cells to emerge from the tissue mass. Although many protocols have been tested and allowed to improve stability by decreasing enzymatic treatment, ASCs remain a cell population that loses growth potential already beginning in passage 4 (similar to that seen in BM-MSCs) and has difficult recovery following freezing [19]. Even if ASCs have been proposed as the best stem cell source in terms of quantity, large amounts of tissue are generally not available from burn patients, so we optimized a standard method for rapid ASC isolation revising latest results of protocols without enzymatic digestion and using only very small quantities of tissue. Important culture parameters for clinical use were controlled, including population growth and stability and differentiation potential. The growth of ASCs co-cultured with progenitor skin fibroblasts was evaluated for potential increase in cell number, quality, and overall robustness to better prepare cells for in vivo environments.
High expectations that these cells may help solve some of the reconstruction problems faced in clinics could be met if the cell source can be shown to be stable, reliable and routinely isolated, cultured and stocked under standardized systems allowing for safe clinical use.
Experimental Methods
Tissue processing, isolation and cell bank preparation
Adipose tissue from healthy patients (n = 15, between 27-61 yr - average age and standard deviation of 46.1 ± 2.0 years and mean BMI of 25.4 ± 0.8kg/m2) (Table 1) undergoing various elective reconstructive surgeries were obtained either by direct excision or manual aspiration (Protocol included in the Department of Musculoskeletal Biobank Regulation: 12/2012 "Conservation of Biological human material for research"). They were given a code (Table 1) and placed in 50ml sterile polypropylene tubes containing transport solution of phosphate buffered saline (PBS) (NaCl 6.8g/l, Na2HPO4 1.5g/l, KH2PO4 0.4g/l) and 1% penicillin-streptomycin (P/S). Samples were processed within 12 hours and were rinsed twice with 5% P/S in PBS for 10 min until connective tissue and blood were released. Tissue (~10g) was then minced into small pieces (diameter: < 5mm) and the washing step was again repeated in 2% P/S in PBS. Approximately 1g of tissue was then directly seeded onto 10cm petri dishes prepared with scalpel incisions to attach tissue fragments at < 20% density of the surface (Figure 1). Initial experiments using three patient tissues were accomplished to determine the influence of cell culture media, based on population doubling-time and morphological analysis. Culture media assessed were as follows: A. Dulbecco's modified Eagle medium (DMEM, Gibco)/FBS (Sigma)/l-glutamine (l-glut, Gibco) (89:10:1); B. DMEM / Ham-F12 - GlutaMAX®-I (Gibco)/FBS (45:45:10); C. DMEM/Iscove's Modified Dulbecco's Medium (IMDM, Gibco) - GlutaMAX®-I/FBS (45:45:10); D. DMEM/Minimum essential medium alpha (α-MEM, Gibco)/FBS/l-glut (59:30:10:1). Samples were cultured in an incubator (37°C, 95% humidity, 5% CO2) and culture media was renewed twice a week. Cells were then passed when reaching 80-90% confluency by removing cells from culture plates with 2 ml of TrypLE® Express (TrypLE: Gibco) for 10 min. Cells were then reseeded into 75 cm2 flasks for further expansion. Cell banks were established for each patient up to passages 3-4, by freezing a minimum of 20 vials of cells at 1 million cells in 1ml freezing media [50% culture media, 40% FBS and 10% dimethyl sulphoxide (DMSO)] and cryopreserved at -80°C until later use. Cells were cryopreserved for a minimum of 2 weeks to measure overall viability. They were then seeded into 6-well culture plates at 1,500 cells/cm2 density to assess for potential adverse impact on cell growth rate and recovery following freezing (hemocytometer total viable cell counts with trypan blue exclusion, n = 3). Cells that were cryopreserved were compared to cells that had not been cryopreserved in the same passage. Morphology of cells was captured in images at 8 days of culture and counting and doubling times were assessed at 14 days of culture.
![]()
Table 1: Adipose tissue from healthy patients undergoing elective reconstructive surgeries
View Table 1
Mechanical versus enzymatic isolation of adipose-derived stem cells
ASCs from two patients were isolated and harvested as described above, and the cells from two other patients were isolated with collagenase for either 30 min or 120 min according to the protocol from Buschmann et al. [20]. All four cell lines established were used at the same passage (P2) for experimentation. Cells from each cell line were seeded in triplicate into 96-well culture plates at 3,000 cells/cm2 in order to assess differences between growths of collagenase- and mechanically-isolated cells up to 19 days. Growth rates were assessed following a metabolic assay based on Cell Titer (Promega) and metabolite absorbance (Tecan Infinite F50 plate reader with 40 min incubation at 37°C and blank subtraction).
Impact of cell seeding density for ASC culture
Mechanically-isolated ASCs were seeded into 96-well cultures plates at various low densities from passages 2 to 5, in order to assess the lowest density for ASC culture and observe the evolution of population doubling time among the passages. Growth rates were assessed at day 12 according to a metabolic assay based on Cell Titer, as described above. Experiments were repeated three times with three different donors.
Differentiation culture assay and staining procedures
Differentiation assays were assessed to confirm that the differentiation potential is maintained with the retained protocols for mechanical mincing of small amounts of tissue, standard medium, low cell density seeding along with freezing of cell stocks. Cells from three patients were seeded at 3,000 cells/cm2 into 12-well plates and cultured with our standard culture medium (formulation A) for 3-5 days, before the beginning of the differentiation assay, in order to obtain around 50% confluence. Cells were then cultured in 2ml of the proper induction medium and changed twice a week over a period of 21 days from the beginning of the induction (three experiments conducted in triplicate).
Osteogenic potential was induced with alpha-MEM, 10% FBS, 1% l-glut, 50 μg/mL l-ascorbic acid, 5mM β-glycerophosphate and 100nM dexamethasone. Adipogenic potential was induced with DMEM, 1% l-glut, 1x ITS, 1μM dexamethasone, 100μM indomethacin and 100μM 3-isobutyl-1-methylxanthine (IBMX). A part of osteogenically-induced cells in monolayer were fixed in 4% paraformaldehyde for 10 min at room temperature and washed with deionized H2O and followed standard von Kossa (VK), and Alizarin red (AR) staining procedures in order to show visual mineralization. The other cells were fixed in a citrate/acetone solution for 30 sec and followed alkaline phosphatase (ALP) staining to highlight ALP activity. Similarly, adipogenic-induced cells in monolayer were fixed in 4% paraformaldehyde and washed as described above and stained using Oil Red O (ORO) standard procedure to reveal cytoplasmic lipid deposits.
Cell stability and yield over passages using co-culture (FE-002γ)
In order to assess potential growth stability and stimulation of ASC co-culture with progenitor fibroblasts, ASCs from 4 different patients were cultured between passages 4-6, with and without co-culture prepared with human progenitor skin cells (FE002-SK2) that had been gamma-irradiated with 200Gy (referred as FE002γ) (dose response was performed by using increment doses until no cell growth or colonies were formed). These off-the-shelf frozen cell banks are used for routine skin graft cultures for severely burned patients in our hospital to provide cytokines and growth factors for better quality cell growth (Ethics protocol #62/07 and organ donation registered under the Federal Transplantation Program complying with the laws and Biobank procedures of the University Hospital of Lausanne and SwissMedic. The cells are registered with the European Collection of Cell Cultures with the Health Protection Agency at Porton Down, ECACC-FE-002-SK2#12070301). These cells have been used in clinical trials as a viable fibroblast cell source for patients with burns and wounds [21,22]. Patient ASCs were seeded between 1,500-3,000 cells/cm2 into 75 cm2 culture flasks with FE002γ at a constant dose of 1,000 cells/cm2. Cell growth of ASCs was assessed by trypan blue exclusion with total viable cell counts using a Neubauer growth chamber (hemocytometer) at 3-6 days. Control flasks of FE002γ alone were prepared for each experiment and did not show any cell growth or viability (trypan blue) following the irradiation and freezing protocol for preparation of clinical batches.
Biocompatibility assessment of biodegradable scaffold
Aiming for further clinical use on patients and better localized cell delivery, we evaluated the biocompatibility of the ASCs when seeded into a collagen membrane (TissuFleece E®, Baxter). Briefly, cells were seeded at 10,000 cells/cm2 into the collagen matrix, and cellular colonisation of the matrix was evaluated by a Live/Dead metabolic assay at 3 different time points (24, 48, 72h in triplicate). The Live/Dead solution was prepared fresh [1ml PBS, 2 μl Ethidium homodimer-1 (EthD-1), 0.5 μl calcein acetsymethyl ester (calcein-AM)] as described by the manufacturer (Life Technologies, L-3244, Live/Dead Viability/Cytotoxicity). Membranes were placed in 6-well culture plates, rinsed with PBS and 50 μl of Live/Dead solution was pipetted onto each membrane. They were incubated at room temperature for 30 min and images were taken with an Olympus IX81 fluorescent microscope with an Andor iXon digital camera.
Results and Discussion
Standardization/simplification of isolation, expansion and stocking of adult ASCs
As regulations have been severely intensified for cell therapies, each step including tissue procurement, cell isolation, cell culture and stocking is very important. Within the technical scope of manipulating stem cells for clinical use, procedures need to be simplified to assure safety and respect manufacturing guidelines. Using simple materials and methods for isolation is of utmost importance in order to treat the tissue and the subsequent cells in the least aggressive manner possible. In this study, the dissociation of small quantities of tissue could be done rapidly and without any aggressive enzymes, centrifugation forces or high serum treatments. Adipose tissue harvested from 15 women patients was minced and placed into cell culture dishes (~1g) that had been scored to allow good attachment and early cell migration (Figure 1). Cells were seen to emerge as early as 3-4 days following this procedure and grown to confluence within 10-14 days (80-90% growth over culture surface) for all patients. By using this mechanical mincing and "direct attachment" procedure, only small amounts of tissue were necessary (Figure 1B,1D), which helped to avoid contamination since tissue could be dissected within small masses and not on the edges. Media optimization was then determined using three separate cell cultures and it was found that the most simple composition [DMEM / FBS / l-glut (89:10:1)] was the most stable between passages. Early passage ASCs showed similar population doubling times with all media used (~110 h) (Figure 2). At later passages, the population doubling time was seen to increase in a linear manner with the DMEM / FBS / l-glut (89:10:1), but the other media combinations portrayed high variability among passages. Once the culture conditions were determined, the use of collagenase treatment of various times (30 and 120 min) was compared to the mechanical isolation procedure we have defined for tissue isolation. Since digestion is currently used for cell separation, it was of interest to see if this step could be eliminated and still have rapid cell growth. We found that 30 min of collagenase treatment showed lower cell growth compared to the less aggressive treatment of tissue without collagenase treatment. If tissue in small quantities (~10g) was subjected to 120 min of collagenase, there was no cell growth observed indicating that this variable may be of interest to eliminate. Other previous studies have shown that cell growth is sensitive to trypsin enzyme treatments also [15]. Freezing the cell gently with temperature increment freezing, and keeping them stored at only -80°C instead of liquid nitrogen (which is normally more difficult to have in the clinical surgical site) allowed very high viability (~97-98%) after 2 weeks or more. More important is the actual recovery of cultured ASCs following freezing protocols to assure their growth stability and potential to stock patient cells until they are needed for therapeutic procedures. With our isolation method, we have seen a slight shift in recovery of ASCs when cryo-preserved for two weeks or more by approximately 3 days, as compared to cells at the same passage without freezing (Figure 3). Even with a slight decrease in cell recovery once ASCs had been cryopreserved, banked cells would however allow to obtain high potential number of cells for patient treatments.
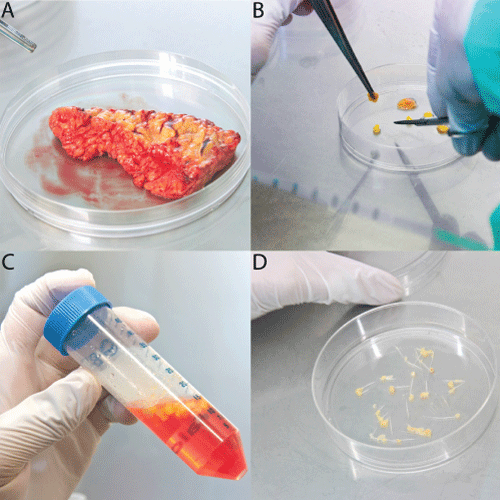
.
Figure 1: Processing of adipose tissue samples for primary culture
Adipose tissue was transported into the lab following tissue extraction (A) and following a first rinsing procedure in 5% P/S in PBS, adipose tissue was then minced into small pieces (< 5mm) (B), rinsed again in 2% P/S (C) and resulting tissue pieces were placed into cell culture dishes that had been deeply scored with a scalpel to attach the tissue into the culture plate for primary culture (D).
View Figure 1
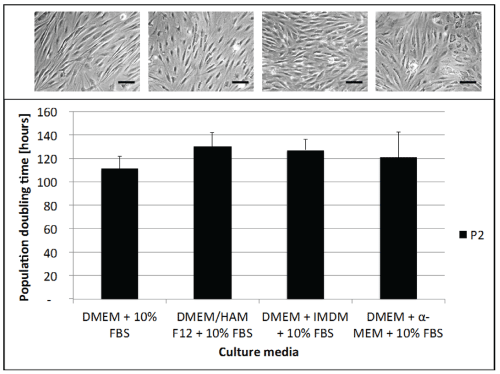
.
Figure 2: Media effect on ASC morphology and growth
Cell growth and morphology was compared using four different growth media. Resulting cell cultures plated at 3,000cells/cm2 were imaged at 8 days and cell growth for 14 days was assessed with doubling times. Experiments were done in triplicate (mean ± SEM). Scale bar on images represents 100 μm.
View Figure 2
Maintenance of differentiation potential
The ASC culture with our defined protocol assured that patient cells maintained differentiation potential after 21 days of stimulation in respective media for osteogenic or adipogenic cell culture conditions (Figure 4). Osteogenic induction was observed by the presence of mineral deposition and enzymatic activity with three different staining procedures including Alizarin red (Figure 4A,4E), von Kossa (Figure 4B,4F) and alkaline phosphatase (Figure 4C,4G). There was light staining of ALP already in non-induced ASCs and since it has been shown that ALP enzymatic activity varies with the age of the donor, the two other staining procedures were used to evaluate mineralization. Adipogenic potential was seen by the large lipid droplets stained with oil red O in the induced cells (Figure 4D,4H) and good overall differentiation potential was similar to that seen recently by Lin et al. [23] showing a uniform multipotent stem cell source.
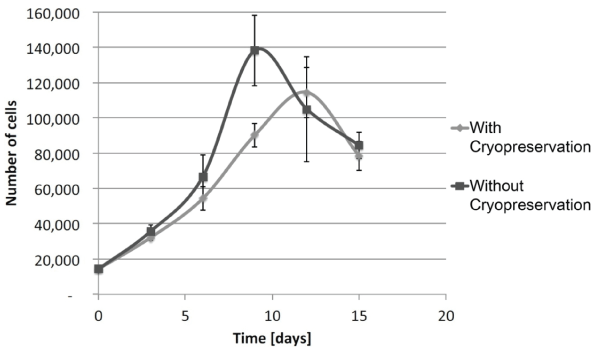
.
Figure 3: Recovery of ASCs following cryopreservation
Growth rate of cells after 2 weeks or more of cryopreservation (◊) were measured as a function of cell number over time in days. They were compared to cells at the same passages without cryopreservation ( ♦ ). Results are from experiments in triplicate (mean ± SEM).
View Figure 3
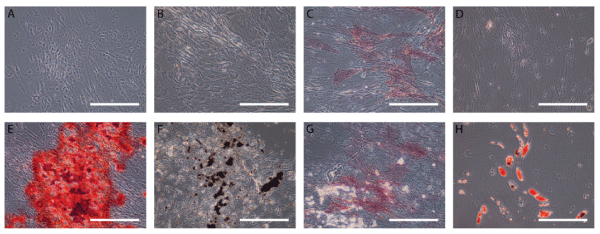
.
Figure 4: Osteogenic and Adipogenic differentiation of ASCs
Patient ASCs were cultured in an osteogenic-inducing medium and stained with Alizarin red (A and E) and von Kossa (B and F) showing mineral deposits and alkaline phosphatase (C and G) showing enzymatic activity. ASCs were cultured in adipogenic-inducing medium and stained with oil red O showing lipid deposits (D and H). Scale bar on images represents 250 μm.
View Figure 4
Optimization of cell growth by seeding density and co-culture
Low density seeding was accomplished over early passages (seeding densities of 325, 750, 1500 and 3000; passages 2-4) to observe full growth characteristics and all showed good cellular expansion capacity. The densities of 1500 to 3000 cells/cm2 would have a slight benefit to obtain enough cells rapidly. Of the 15 patients, only two had cell growth that extended to passages 6 and beyond, without the typical cell morphology changes (enlarged cells with wide cytoplasmic spread). This aspect of culturing adult stem cells has also been illustrated by others showing slowing of cell growth and large, spreading cell morphology already at passage 4-6 [19]. Primary cell cultures from patient tissues are known to show heterogeneous growth capacities with overall lower growth over time. Because of this, we evaluated the use of co-culture directly with skin progenitor cells (FE002γ) to culture or to help adult adipocytes to remain stable over longer periods and thus, even higher passages could be used to prepare protocols for clinical batches of cells if needed. As early cells can only be frozen after several amplifications, it is only at passage 2-3 that enough cells could be stocked. Therefore, placing the cells into culture with further expansion would bring them easily to passage 5-6. It would therefore be important, particularly for severely burned patients where multiple grafts are needed over time, to have the possibility to increase cell expansion potential for cells that may be relatively less stable over passages. This variability, when working with adult stem cells, has been difficult to integrate into standardized protocols. Cell growth of ASCs in the presence of FE002γ was found to be 40-50% higher in number and the morphology and stability were optimized (cells showing finer spindle-shaped morphology rather than large cytoplasm and large round cell morphology) (Figure 5). Thus, we could increase total cell number through at least two cell passages. Use of FE002γ could prepare a robust adult ASC population for clinical use, as has been shown for skin cells for burn patients [21,22], and could be used in co-culture association for burn therapy.
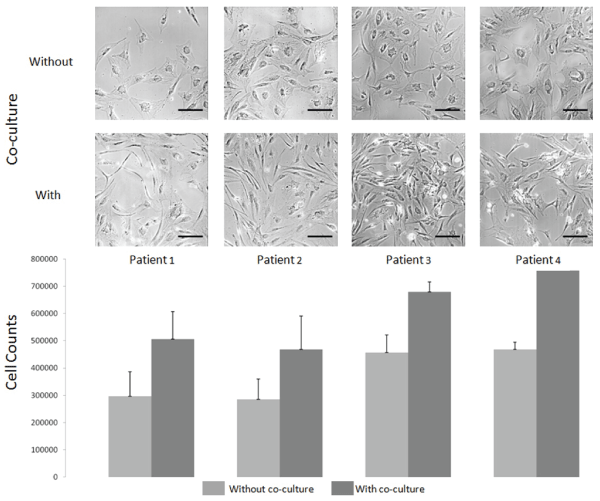
.
Figure 5: Cell stability and yield over passages using co-culture (FE-002γ)
Cells from passage 6 of four patients are illustrated with growth conditions with and without skin progenitor cells (FE002γ, progenitor skin fibroblasts pre-treated with gamma irradiation of 200Gy and kept as frozen stocks) portraying typical fibroblastic cultures of later passages with many cells larger with wide cytoplasm when under normal culture conditions (without co-culture). ASCs grown in combination with FE002γ showed cell populations more similar to those in passage 2-4 for most patients. Cell numbers were counted for each patient in triplicate without co-culture (dark grey) and with co-culture (light grey). Scale bar on images represents 100 μm.
View Figure 5
ASC association with delivery matrix for clinical application
By having more robust cells with the co-culture protocol, we have shown that these cells could be integrated easily with 3D scaffolds to provide a delivery system to the patient. Use of CE marked and clinical-grade hemostatic sponge collagens used routinely in surgery (TissuFleece®, Baxter) could be an elegant method to deliver ASCs now since they grow well in co-culture with fibroblasts. The ASCs showed integration into the matrix already at 24 h (Figure 6A) and following 48 h adhered strongly and migrated within the scaffolds (Figure 6B,6C). Patient ASCs could now be considered to be associated within biological bandages used for burn patients to help in overall skin repair and soft tissue reconstruction [21,22].
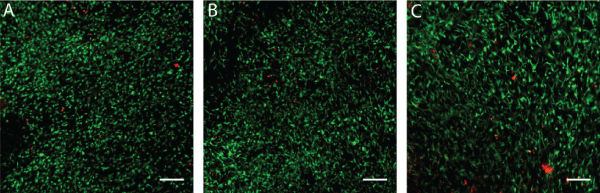
.
Figure 6: Biocompatibility and delivery potential of ASCs in collagen scaffold
Cells were seeded into collagen scaffold (TissuFleece®, Baxter) and stained with Live/Dead kit after 24h (A) 48h (B) and 72h with live cells stained in green and rare non-viable cells in red. Scale bar on images represents 250 μm.
View Figure 6
The recent study in the Lancet by K∅le and colleagues [1] showed that fat grafts supplemented with autologous culture-expanded ASCs were able to increase graft volume retention up to ~81% compared to only ~16% if no cultured cells were associated to the human fat grafts transplanted into the upper arm [1]. Therefore, the specific technique for cultured ASCs has shown to be of importance for cell selection and activity. This technique could prove to be a great advantage for the future of lipofilling and soft tissue reconstruction. More and more patients are in need of lipofilling with delays between operations due to their pathologies (i.e. scleroderma or damage from radiotherapy). There are also situations where multiple injections are needed such as for breast augmentation or scar corrections. There are particular problems around implant surfaces and matrix enriched with stem cells could provide a solution to help integration into the surrounding tissues with improved tolerance.
Importantly, ASCs could be added to grafting procedures routinely used for severely burned patients adding them with co-culture of keratinocytes and fibroblasts. Thus, having stocks of cells from individual patients would be of benefit for a large variety of interventions. The lower amounts of fat tissue from donor sites would increase safety and minimize donor site deformation. Furthermore, it would be possible to obtain sufficient amount of cultured ASCs also from patients with low fat deposit and for severely burned patients and their use could be a great advantage to be associated with fat grafts or skin grafts.
Methods and techniques for cellular therapies have to be performed under full Good Manufacturing Processes (cGMP compliance). As all the techniques for burn management use traditional methods employing basic media, fetal bovine serum and growth factors since 30 years in our hospital, it is of importance to include this new cell type with similar adapted culture conditions as has been positively illustrated in this study and adapt in a step-wise manner for potential improvements.
To this end, there has been much effort world-wide imagining the replacment of animal components in tissue and cell processing including patient serum [19], platelet derivatives [5,24,25] and recombinant growth factors [26,27]. These alternatives could now be analyzed in our long-term clinical setting to see if cellular growth and stability of cell stocks can be maintained whenever instigating changes in clinical protocols. All of these should be done step-by-step to assure the high quality necessary for patient use.
Conclusions
When working with tissue and stem cell isolation procedures that are destined for clinical use, it is important to have the simplest and the least aggressive treatments to allow robust cell populations. With mechanical isolation of ASCs instead of more common (but also more aggressive) enzymatic treatments, it is possible to obtain better cell growth alongside full maintenance of the differentiation potential. The cells could be frozen with a high survival rate (~98%) up to passages 4-6 and then co-cultured with FE002γ to improve the stability. Co-culture can significantly enhance primary ASC culture with higher numbers of patient cells obtained from a low quantity of tissue so that it would be possible to store the cells and thaw them when needed for transplantation. Finally, the fact that ASCs could be dispersed on collagen scaffolds with a good survival opens the possibility to use them in various manners, including filling of larger gaps and for skin grafting techniques routinely used to allow better soft tissue reconstruction with co-culture of keratinocytes and fibroblasts for burn patients (Figure 7). Cell culture assisted surgical techniques will need to be developed with Good Manufacturing Processing in mind since regulations have become more strict in the hospital environment, but taking care at integrating each cell type at each step of the tissue processing will increase benefits towards patient treatments and safety.
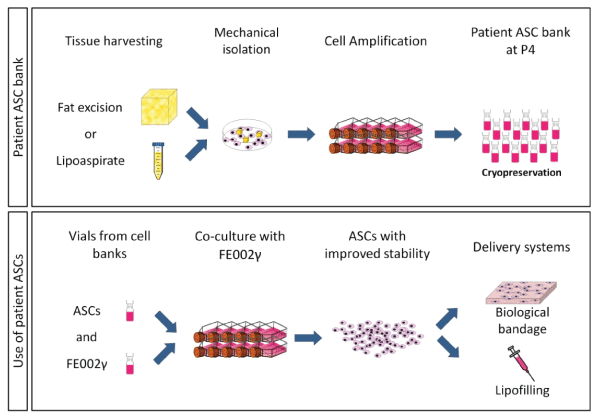
.
Figure 7: Development of ASC banks and protocol to enhance cell stability through co-culture
Patient adipose tissue is micro-dissected into culture plates to allow rapid cell outgrowth and amplification for cryopreservation. Cells can be either used directly if enough cells are acquired or continued for expansion. Enhancement of cell numbers and quality can be obtained by co-culture with progenitor cells before associating them with matrix materials.
View Figure 7
Ethical Statement
All of the work conducted in this study has been accept by the State Ethics Board with Progenitor skin cells under Ethics protocol #62/07 and the organ donation registration under the Federal Transplantation Program complying with the laws and regulations of the Biobank procedures of the University Hospital of Lausanne and SwissMedic. Adipose tissue collection is under Protocol #12/2012 "Department of Musculoskeletal Medicine Biobank conservation of biological human material for research and use" following all of the rules and regulations of this program approved by the State Ethics Board.
Acknowledgements
We thank Drs. Valerie Decrouy-Duruz, Pietro di Summa, Nathalie Koch, Giorgio Pietramaggiori, Laure Emilie Rey and Saja Sandra Scherer-Pietramaggiori of the surgical team of the Plastic, Reconstructive and Hand Surgery Service for providing the samples of adipose tissue, and Corinne Scaletta for valuable technical guidance and discussions. This study was supported in part by the Foundations Sandoz Family, S.A.N.T.E and Orthopaedic Hospital and the European Grant FP7-EPIONE.
References
-
KØlle SF, Fischer-Nielsen A, Mathiasen AB, Elberg JJ, Oliveri RS, et al. (2013) Enrichment of autologous fat grafts with ex-vivo expanded adipose tissue-derived stem cells for graft survival: a randomised placebo-controlled trial. Lancet 382: 1113-1120
-
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, et al. (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7: 211-228
-
Klinger M, Marazzi M, Vigo D, Torre M (2008) Fat injection for cases of severe burn outcomes: a new perspective of scar remodeling and reduction. Aesthetic Plast Surg 32: 465-469
-
Trottier V, Marceau-Fortier G, Germain L, Vincent C, Fradette J (2008) IFATS collection : Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells 26: 2713-2723
-
Kakudo N, Minakata T, Mitsui T, Kushida S, Notodihardjo FZ, et al. (2008) Proliferation-promoting effect of platelet-rich plasma on human adipose-derived stem cells and human dermal fibroblasts. Plast Reconstr Surg. 122: 1352-1360
-
Jeong JH (2010) Adipose stem cells and skin repair. Curr Stem Cell Res Ther 5: 137-140
-
Locke M, Feisst V, Dunbar PR (2011) Concise review: human adipose-derived stem cells: separating promise from clinical need. Stem Cells 29: 404-411
-
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, et al. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8: 315-317
-
di Summa PG, Kalbermatten DF, Raffoul W, Terenghi G, Kingham PJ (2013) Extracellular matrix molecules enhance the neurotrophic effect of Schwann cell-like differentiated adipose-derived stem cells and increase cell survival under stress conditions. Tissue Eng Part A 19: 368-379
-
Mitchell JB, McIntosh K, Zvonic S, Garrett S, Floyd ZE, et al. (2006) Immunophenotype of human adipose-derived cells: temporal changes in stromal-associated and stem cell-associated markers. Stem Cells 24: 376-385
-
Gronthos S, Franklin DM, Leddy HA, Robey PG, Storms RW, et al. (2001) Surface protein characterization of human adipose tissue-derived stromal cells. J Cell Physiol 189: 54-63
-
Dong Z, Luo L, Liao Y, Zhang Y, Gao J, et al. (2014) In vivo injectable human adipose tissue regeneration by adipose-derived stem cells isolated from the fluid portion of liposuction aspirates. Tissue Cell 46: 178-184
-
Strioga M, Viswanathan S, Darinskas A, Slaby O, Michalek J (2012) Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev 21: 2724-2752
-
Koch N, Erba P, Benathan M, Raffoul W (2010) Nouveautes dans la reconstruction cutanee : cultures de cellules et substituts dermiques et revue de la litterature. Ann Burns Fire Disasters 23: 131-136
-
Markarian CF, Frey GZ, Silveira MD, Chem EM, Milani AR, et al. (2014) Isolation of adipose-derived stem cells: a comparison among different methods. Biotechnol Lett 36: 693-702
-
Baptista LS, da Silva KR, Pedrosa C, Borojevic R (2011) Processing of lipoaspirate samples for optimal mesenchymal stem cells isolation. In: Advanced Techniques in Liposuction and Fat Transfer. (Ed. Serdev N) ISBN 978-953-307-668-3
-
Kim JB, Lee HM, Kim SY, Park KS, Bae J, et al. (2011) Cryopreservation of Human Adipose Tissues using Human Plasma. J Biotechnol Biomaterial 1: 4
-
Zeng G, Lai K, Li J, Zou Y, Huang H, et al. (2013) A rapid a efficient method for primary culture of human adipose-derived stem cells. Organogenesis 9: 287-295
-
Kocaoemer A, Kern S, Klüter H, Bieback K (2007) Human AB serum and thrombin-activated platelet-rich plasma are suitable alternatives to fetal calf serum for the expansion of mesenchymal stem cells from adipose tissue. Stem Cells 25: 1270-1278
-
Buschmann J, Gao S, Harter L, Hemmi S, Welti M, et al. (2013) Yield and proliferation rate of adipose-derived stromal cells as a function of age, body mass index and harvest site-increasing the yield by use of adherent and supernatant fractions? Cytotherapy 15: 1098-1105
-
Hohlfeld J, de Buys Roessingh AS, Hirt-Burri N, Chaubert P, Gerber S, et al. (2005) Tissue engineered foetal skin constructs to Avoid Autografting for Burns. Lancet 366: 840-842
-
de Buys Roessingh A, Scaletta C, Hirt-Burri N, Hohlfeld J, Gerber S, et al. (2006) Development and Characterization of a fetal skin bank for tissue engineering and its clinical application for acute and chronic wounds. Cell Transplantation 15: 823-834
-
Lin CY, Huang C-H, Wu Y-K, Cheng N-C, Yu J (2014) Maintenance of human adipose derived stem cell (hASC) differentiation capabilities using a 3D culture, Biotechnol Lett 36: 1529-1537
-
Doucet C, Ernou I, Zhang Y, Llense J-R, Begot L, et al. (2005) Platelet lysates promote mesenchemal stem cell expansion: a safety substitute for animal serum in cell-based therapy applications. J Cell Physiol 205: 228-236
-
Bieback K (2013) Platelet lysate as replacement for fetal bovine serum in mesenchymal stromal cell cultures. Transfus Med Hemother 40: 326-335
-
van der Valk J, Brunner D, De Smet K, Svenningsen AF, Honegger P, et al. (2010) Optimization of chemically defined cell culture media-replacing fetal bovine serum in mammalian in vitro methods. Toxicol in Vitro 24: 1053-1063
-
Lund P, Pilgaard L, Duroux M, Fink T, Zachar V (2009) Effect of growth media and serum replacements on the proliferation and differentiation of adipose-derived stem cells. Cytotherapy 11: 189-197





