International Journal of Stem cell Research and Therapy
Enhancing the Potency of Mesenchymal Stem Cells for Tissue Regeneration
Sylvia Müller1, Kenny Dalgarno2, Anne Dickinson1, Xiao-nong Wang1* and Lindsay Nicholson1
1Institute of Cellular Medicine, Newcastle University, Newcastle upon Tyne, UK
2School of Mechanical and Systems Engineering, Newcastle University, Newcastle upon Tyne, UK
*Corresponding author: Xiao-nong Wang, Institute of Cellular Medicine, Newcastle University, Newcastle upon Tyne, UK, Tel: +44 191 2088792, Fax: +44 191 2226794, E-mail: X.N.Wang@ncl.ac.uk
Int J Stem Cell Res Ther, IJSCRT-2-013, (Volume 2, Issue 2), Short Review; ISSN: 2469-570X
Received: October 29, 2015 | Accepted: November 23, 2015 | Published: November 27, 2015
Citation: Müller S, Dalgarno K, Dickinson A, Wang XN, Nicholson L (2015) Enhancing the Potency of Mesenchymal Stem Cells for Tissue Regeneration. Int J Stem Cell Res Ther 2:013. 10.23937/2469-570X/1410013
Copyright: © 2015 Müller S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Mesenchymal stem cells (MSCs) are adult stromal cells with multi-lineage differentiation potential and immunomodulatory properties. They can be isolated relatively easily from several tissues, including bone marrow and adipose tissue and can be expanded ex vivo to yield the large numbers required for a therapeutic dose. As such, MSCs are considered a promising cell source that can be used in cellular therapy for a wide range of diseases, including bone and cartilage defects. However, despite the huge number of clinical trials utilising MSCs, their clinical application has yielded variable results, often attributable to different donor sources and expansion protocols. The objective of this concise review is to summarize recent developments regarding tissue source of MSCs, use of phenotypic markers for MSC selection, as well as potential in vitro modifications that may be stratified and applied to enhance the potency of MSCs for clinical use in regenerative medicine.
Keywords
Mesenchymal stem cell, CD271, Bone marrow, Clinical trials, Regenerative medicine
Introduction
Mesenchymal stem cells (MSCs) are adult stem cells with a fibroblast-like morphology. In the late 60's and 70's, transplantation studies showed that a small sub-population of cells present in the bone marrow could aid osteogenic regeneration [1]. Studies by Friedenstein, Chailakhjan and Lalykina [2] named this population 'colony forming unit fibroblasts', due to their colony forming ability. This was changed in 1987 to 'Osteogenic stem cell' [3]. The nomenclature used to describe this cell type is contentious. The term 'Mesenchymal stem cell' came into use in the 1990's, being coined by Caplan [4] and this remained as the term for these cells until an International Society of Cellular Therapy (ISCT) position paper by Horwitz et al., [5] proposed that the official term be changed to "multipotent mesenchymal stromal cells". The paper stated that as plastic adherent populations isolated from the bone marrow shared certain properties they should be classed together as one type of cell. However, the word 'stem' was removed as not all plastic adherent cells derived from the bone marrow exhibit the stem cell properties. The word stromal was used as its replacement owing to the stromal location MSCs are derived from in the original tissue.
The current classification of MSCs was set by the International Society of Cellular Therapy (ISCT) in the position paper by Dominici et al. [6]. This listed the three minimal criteria that must be met to define cells as MSCs: plastic adherence, tri-lineage differentiation potential (osteogenic, chondrogenic and adipogenic) and expression of a specific surface phenotype. In addition to the minimal criteria, MSCs have also been described as hypo-immunogenic [7] (though current research disputes this and have described them as immune-evasive [8]) and immunomodulatory [9-15] making an allogeneic "off-the-shelf" therapy an attractive possibility. Unsurprisingly, given MSC's capabilities they show great potential for a wide range of clinical uses, most notably in treating graft versus host disease (GvHD) [16-21] and defects in bone and cartilage [22-29]. Indeed, the number of indicated uses for these cells continues to gain pace; illustrated by the fact that as of October 2015 there were 554 clinical studies involving mesenchymal stem cells listed on clinicaltrials.gov. However, to date, MSCs used clinically have yielded heterogeneous results, though no adverse effects have ever been reported. This heterogeneity can be largely attributed to differences in donor source and ex vivo expansion methods. This review summarises the current MSC selection criteria, as well as potential in vitro modifications that may be utilised to enhance the potency of MSCs for clinical use, with an emphasis on regenerative medicine.
Donor Source
MSCs have been isolated from a number of tissues including the bone marrow (BM) [30], adipose tissue (AT) [31], umbilical cord blood (UCB) [32], Wharton's jelly [33] and dental pulp [34]. MSCs from different sources have been extensively compared and have shown to have a unified phenotype and similar morphology. The main variation in cells from different sources lies in their differentiation potential, proliferation rates and the potency of their immunosuppressive actions.
UCB-MSCs have faster growth rates than BM- and AT-MSCs [35,36] at the expense of a weaker differentiation potential [37]. UCB-MSCs can be problematic to isolate as the procedure requires a large volume of blood processed immediately after collection [38]. Bone marrow collection is an invasive and unpleasant process which may only yield a small number of stromal cells. Adipose tissue and lipoaspirate can be collected as biological waste from cosmetic procedures [39], and contains more cells per gram of tissue compared to bone marrow [40,41]. As well as their longevity in in vitro culture, AT-MSCs produce higher levels of cytokines and have stronger immunosuppressive potential [42]. However the differentiation potential of BM-MSCs has been seen to be stronger than AT-MSCs [43]. Clearly, depending upon the clinical need the donor source should be carefully considered.
Phenotype
A single marker that can identify all MSCs in a population has yet to be found, instead, a mixed panel of positive and negative markers has been put forward by the ISCT. The phenotype for a population of MSCs is 95% positive expression of CD73, CD90 and CD105 and less than 2% positivity for CD14, CD19, CD34, CD45 and HLA-DR [6]. The three positive markers are consistently found on MSCs, independent of passage or source and not found on other cell types in this combination. Non expression of the negative markers ensures the absence of contaminating cell types of haematopoietic lineage.
Though this panel is currently used as the standard for MSC classification, it is likely to change as more research is conducted into the properties of MSCs. The disadvantage of the current phenotypic classification of MSCs is that the CD73/CD90/CD105 population is highly heterogeneous in its functional capacity [13,44,45]. Clonal studies have found that during in vitro expansion, less than half of BM-MSCs clones possessed the tri-lineage differentiation potential [44]. This heterogeneity becomes more pronounced as with continual cell doubling and long term culture [45]. This suggests that those cells in the conventionally isolated MSCs have already been primed for specific lineage. To obtain a purer sample of MSCs, various single and combination markers have been examined as potential targets for positive selection, summarised in table 1, with the surface marker, CD271, either used alone or in combination with other markers leading the way as the frontrunner for MSC selection, as discussed below.
![]()
Table 1: Single and combination markers for MSC isolation and characterisation
View Table 1
CD271
CD271, also known as low-affinity nerve growth factor receptor (LNGFR) or p75 NTR (neurotrophin receptor), is one putative marker that potentially identifies a pure MSC population [46]. In the 1990s, studies using immunoelectron microscopy and immunohistochemistry found that fibroblast-like cells throughout the bone marrow expressed CD271 [47,48]. The majority of research using CD271+ MSCs has used bone marrow as the source tissue, including the pivotal work by Quirici et al., [49] which detailed the first successful isolation and characterisation of CD271+ cells. Work has also suggested that CD271+ MSCs are found in the greatest concentration in the trabecular bone surrounding the bone marrow [50,51]. Despite this however, some studies have successfully isolated CD271+ MSCs from adipose tissue [52] and dental pulp [53] though, to date, they have not been found in the umbilical cord blood or Wharton's jelly [54,55]. The characteristics of the CD271+ population of bone marrow-derived cells are in line with the standard requirements for MSC classification; however, they are coupled with more potent functional properties. This includes their osteochondral differentiation ability, colony forming potential and immunomodulatory characteristics.
The immunomodulatory properties of CD271+ MSCs were assessed by Kuçi et al. [56,57]. Even at low concentrations, the ability of CD271+ cells to inhibit mononuclear cell proliferation was greater than that of the CD271- MSC population. Additionally, Kuçi et al., [58] showed that co-transplantation of CD271+ MSCs and T cells can lead to an increased level of T cell repopulation. Jones et al. [59] proposed that the CD271+ cells are MSC precursor cells. Churchman et al. [60] published a transcriptional study of CD271+ MSCs and standard cultured MSCs found that the CD271+ population expressed greater levels of genes involved in bone production, suggestive that the CD271+ MSC cells are primed to undergo osteogenesis. In line with this, differentiation studies have found that human MSCs enriched for CD271+ cells have a greater osteogenic potential compared to the non-enriched fraction. Despite a possible bias for osteogenic expression, CD271+ MSCs have also been shown to undergo greater levels of chondrogenic differentiation in vivo and in vitro [61,62].
Conversely, work by Mikami et al. [53] showed that prolonged overexpression of CD271 in murine MSCs leads to the inhibition of lineage commitment. This disparity between commitment versus inhibition to lineage differentiation may be ascribed to species variation. Aomatsu et al. [63] found that signalling from the protein SCRG1 maintains expression of CD271 on MSCs in culture, with the production of SCRG1 decreasing during osteogenesis. This would suggest that CD271 is a MSC marker of stemness. The possible stemness properties of CD271+ cells have also been found in research on cancer cells. A population of CD271+ cells have been found in squamous cell carcinoma of the head and neck [64], melanoma [65], osteosarcoma [66], and oesophageal squamous cell carcinoma [67]. These studies have found that the CD271+ cells proliferate faster [68], are more likely to contain colony forming cells, have greater expression of stemness genes [66,67] and have a higher level of self-renewal [69]. Additionally, these CD271+ populations are more likely to metastasise [65] and are more chemo-resistant than CD271- cancer cells [67,70]. This is not to suggest that CD271 is a marker of tumorigenicity in MSCs, rather it exemplifies the fact that CD271 is a marker of greater growth potential and resilience.
It is important to note that MSCs lose the surface expression of CD271 in culture [71]. If CD271 is a marker of precursor MSCs with no lineage commitment, then the isolation of CD271+ cells could produce a population of cells with greater osteogenic potential as none (or very few) of the cells have committed to either the adipogenic or chondrogenic lineage. Taken together, this suggests that the CD271+ MSC populations are more potent in comparison to standard MSC populations and CD271 enrichment could yield a better product for clinical applications. With the advantages of CD271+ populations containing a higher concentration of colony forming cells [57] and having stronger differentiation and immunomodulatory potential, fewer cells would need to be isolated to be as efficacious as larger populations of standard MSCs. Furthermore, the requirement for a smaller cell number would reduce the total cost of production and decrease the risk of potential contamination during culture by using either uncultured cells or shorter expansion times. Indeed, a recent study by Cuthbert et al. [72] using clinical-grade immunomagnetic CD271+ positive selection to obtain an uncultured, enriched CD271+ MSC fraction from three different tissue sources found these cells to have greater osteogenic potential compared to the non-enriched fraction. This suggests that this cell population could be used without expansion, further improving the clinical applicability. However, the feasibility of using specific markers for MSC isolation is still unclear and more work would need to be conducted to clarify the potential for using these cells for therapeutic purposes.
in vitro Culture Modifications
As well as selecting specific cells from a defined tissue source, alterations to the standard method of cell expansion can have a positive impact on the potency of the resulting cell product. MSCs are highly responsive to chemical and physical signals from their surrounding environment and can be both negatively and positively impacted by alterations in their culture conditions [73]. Different modifications to the traditional culture system can have different impacts on the functional properties of MSCs, as discussed below.
Serum
Possibly one of the most common alterations to standard MSC culture conditions is the replacement of foetal calf serum (FCS), also known as foetal bovine serum. In standard culture conditions, MSCs are grown in media containing 10% FCS, providing the cells with the growth factors needed for expansion. Although the use of FCS for clinical-grade MSC expansion is currently permissive, it is associated with several disadvantages and implementation of a standard xeno-free culture condition is most likely required for future clinical production. For example, cells cultured in FCS can retain bovine proteins on their cell surface and these can cause an allergic reaction in the receiving patients [74-76]. Additionally, there is a risk of transmitting prion-based diseases from cows to humans. Though recent studies have shown that these proteins can be filtered out of contaminated media [77], sources are still restricted to certified prion-free countries such as Australia and New Zealand. These, coupled with the obvious ethical problems attached to the use of FCS in cell culture [78] make alternatives actively sought, whilst maintaining or enhancing, MSC function.
The current competitors to replace FCS in cell culture are human serum and platelet lysate. These are both derived from human sources, which would remove the use of xenogeneic material in culture [79]. Human serum for cell culture can be collected from either an autologous or allogeneic source. The use of autologous serum would reduce the chance of a negative reaction occurring from the serum-cultured cells; however the quality of the serum may vary from different patients. The use of allogeneic serum would eradicate this problem by pooling serums collected from numerous donors to produce a more standardised product. Cultures supplemented with 10% autologous serum were seen to have higher rates of proliferation and greater osteogenic potential compared to cultures with the standard 10% FCS [80,81]. Nimura et al. [82] suggested that the increased growth rate of cells in human autologous serum may be due to the increased level of platelet-derived growth factor (PDGF) compared to FCS. Studies have also seen that MSCs grown in autologous human serum are more genetically stable compared to those grown in FCS [83,84]. The use of allogeneic human serum for MSC expansion yielded comparable growth rates to FBS and MSCs of a similar quality and functionality [85,86]. In contrast, a study by Shahdadfar et al. [83] found that MSCs cultured with allogeneic serum were more likely to be affected by growth arrest.
Platelet lysate is obtained through repeated freeze-thaw cycles of platelets isolated from peripheral blood [87]. Human platelet lysate has been found to be applicable for the in vitro expansion of epithelial cells, fibroblasts, and MSCs from multiple sources [88-92]. Doucet et al., [93] demonstrated that platelet lysate is rich in growth factors which enhance the proliferation of MSCs. Further studies have found the phenotypic and morphological properties of MSCs grown in platelet lysate are strongly comparable to those grown in FCS. Expansion of MSCs has continuously been shown to be more efficient in platelet lysate compared to in FCS, as the platelet lysate-MSCs have faster growth rates [94-97]. Studies that have assessed the genetic stability of MSCs cultured in platelet lysate have reported a high level of chromosomal stability [97]. Lange et al. [98] suggested that culture of MSCs in platelet lysate could maintain their stemness properties due to an increased expression of cell cycle-related genes and a decrease in genes specific for lineage commitment. Additionally, studies have shown that in in vitro conditions the immunomodulatory properties of platelet lysate-expanded MSCs are either equal to, or stronger than, FCS-expanded MSCs [99]. However, a clinical trial by von Bonin et al., [100] using platelet lysate-expanded MSCs to treat acute GvHD, found that only 2 of the 13 patients responded positively to the initial treatment. Despite the low response rate in this trial, it did show that there was no negative reaction from using platelet lysate expanded MSCs, thus highlighting the clinical feasibility of using platelet lysate-expanded MSCs.
Some disadvantages to using platelet lysate for cell expansion have also been reported. Firstly, it has been suggested that MSCs cultured in platelet lysate are less plastic adherent due to a decreased expression of adhesion-related genes [101]. This could make the initial isolation of freshly harvested cells problematic as many isolation protocols rely on the plastic adherence of MSCs. Secondly; there is a level of lot-to-lot variability in different batches of platelet lysate which can result in problems with standardising for use in culture [102], however, the use of pooled platelet lysate batches can reduce this variability.
Growth Factor Supplements
The addition of recombinant growth factors in serum-supplemented media can enhance the growth potential of MSCs, making shorter culture times as effective as longer periods. Additionally, media containing recombinant growth factors could entirely replace serum supplementation in culture media. Matrin et al. [103] reported that the addition of 1 ng/ml fibroblast growth factor 2 (FGF-2, also known as basic fibroblast growth factor, bFGF) to media containing 10% FCS resulted in the formation of larger colonies and faster growth rates compared to the MSC cultured in control media. The addition of FGF-2 to standard osteogenic induction media also enhanced the level of matrix deposition from MSC samples. Other work has also found that the addition of FGF-2 can enhance the growth of MSCs in culture, as well as the eventual differentiation potential of the cells [104-106]. However, addition of FGF-2 can cause low level surface expression of HLA class I and II [107], which could potentially stimulate an immune response.
Platelet-derived growth factor (PDGF) is another growth factor often used to supplement MSC culture medium, sometimes with mixed results. Cassiede et al. [108] found that 5 ng/ml PDGF added to the culture media enhanced the proliferation of MSCs, though the osteochondral differentiation potential of the cells was not consistent in vitro and in vivo. The same paper showed addition of transforming growth factor beta (TGFβ) had a similar effect as PDGF on the growth of MSCs. Gharibi et al. [109] found that stimulation of the PDGF beta receptor (PDGFRβ) can induce the expression of cell cycle-associated proteins and increase the rate of proliferation, and Tokunaga et al. [110] suggest that PDGFRβ may also inhibit osteogenic differentiation.
Some work has found that a combination of different growth factors can be as effective as serum supplementation in culture media. Ng et al. [111] and Chase et al. [112] detail a combination of TGFβ, PDGF and FGF2 added to serum-free media being as effective as serum-containing media in stimulating proliferation without altering the gene expression profile of the cells.
Hypoxia
Certain culture modifications aim to replicate the internal environment that MSCs are subjected to within the body. Currently, the standard culture environment for cells is in an incubator at 37°C with 5% CO2 and 20% O2; however the oxygen content within our bodies varies from 1-11% [113,114]. Growth in hypoxic conditions (2-7% oxygen) has been known to benefit cell cultures since the mid-20th century [115] with multiple studies reporting that culture of MSC samples in hypoxia enhanced the rate of proliferation and maintained the lifespan of cultures [113,116-122], though when the oxygen concentration is too low (≤ 1%), the rate of proliferation can decrease [123].
Whether hypoxic conditions also enhance the differentiation potential of MSCs is still unclear with some reports showing greater differentiation from cells cultured in 2-5% O2 [122,123] and others suggesting that low oxygen inhibits differentiation [113,118,122]. The main consensus is that a hypoxic environment can maintain stemness in a cell population, with studies showing an increased or prolonged expression of genes such as Oct-4 and NANOG [116,120,124]. One such explanation for this improvement under low oxygen concentration is the maintenance of hypoxia-inducible factor 1-alpha (Hif-1α). This is a transcription factor that regulates the expression of hypoxia-dependant genes. Under normoxic conditions the protein is susceptible to degradation by the proteasome [125], therefore is only stable in low oxygen environments. Active Hif-1α can inhibit the activation of the E2A-p21 complex which leads to cellular senescence and apoptosis [126]. A second hypothesis is that the normoxic environment can lead to a production of reactive oxygen species (ROS) within the cell, causing oxidative stress [119] and compromising the stability of DNA [127].
Mechanical Stimulation
Similar to hypoxia, the addition of mechanical stimulation to culture is also based on physiological conditions. As the body moves and the blood pumps, cells are subject to a degree of shear stress. This can be replicated in vitro with the use of bioreactors in which cells can be cultured [128]. These can be 'spinner flasks' containing a propeller which continuously stir the cell-containing media in one direction or flow perfusion bioreactors, which pump media across the cell sample. A third option is to provide cells with mechanical stimulation by simulating mechanical loading.
The mechanical stimulation provided by the movement of fluid within these devices has been shown to lead to an increased activation of mitogen-activated protein kinase (MAPK)-linked signalling pathways [129-132]. As the MAPK signalling pathways are key regulators of differentiation and proliferation, the functional benefit of mechanical stimulation is an enhanced level of osteogenesis. Certain studies have sought to discover whether mechanical stimulation could induce dexamethasone-independent osteogenic differentiation in MSCs. Holtorf et al. [133] tested MSCs cultured on a titanium fiber scaffold with or without flow perfusion-derived mechanical stimulation in the absence or presence of dexamethasone. Analysis found high levels of alkaline phosphatase activity, calcium deposition and osteopontin secretion, key markers of osteogenesis, in samples subjected to flow perfusion only in those that were preconditioned with dexamethasone-treatment. Mechanical stimulation is not limited to osteogenesis; Mauck et al. [134] demonstrated that mechanical loading of MSCs in a hydrogel can improve chondrogenic differentiation in MSCs. This study showed an increased level of aggrecan gene transcription and glycosaminoglycan production, signifying the enhanced production of cartilage.
Osteogenesis can also be augmented through mechanical loading and strain. This is believed to be due to an increased activation of the ERK/MAPK signalling pathway which regulates differentiation through inhibiting adipogenesis in favour of osteogenesis [135-137]. Other studies have failed to show conclusive evidence that mechanical stimulation (whether by flow perfusion or mechanical loading) can provide osteogenic induction in the absolute absence of dexamethasone. However, it does appear that if cells are subjected to mechanical stimulation during osteogenic induction, they will produce higher levels of bone deposition compared to the cells differentiated without mechanical stimulation [138,139].
Scaffolds
Standard MSC culture utilises a two-dimensional plastic surface on to which the cells adhere. This may be of use to in vitro research; however, it does not replicate the natural environment in which cells have evolved in and grown. Though hypoxia and mechanical stimulation may provide some of the features of their in vivo environment, changing to a three-dimensional scaffold may also be required to obtain the best possible potency from a cell population. Scaffolds are often used for tissue regeneration and can be utilised to ensure direct application of cells into an area of damage. Depending on the specific requirements, scaffolds can be made from a range of different materials including ceramics, Bioglass®, synthetic polymers, hydroxyapatite, and biological materials such as hyaluronic acid or collagen. Shared characteristics of these different materials are that they are non- immunogenic and can support the growth of the surrounding cells [140].
The stiffness of a scaffold material can impact on the cellular functions by dictating the pathway of lineage commitment in the interacting cell. Fu et al. [141] highlighted that the stiffness of a material can affect MSC differentiation. The authors produced a scaffold made of elastic, 'polymer polydimethylsiloxane', the surface of which was covered in small protrusions. The rigidity of these was dependant on their length, with the shorter protrusions having the greater rigidity than the longer, more flexible protrusions. They found that the cells cultured on the rigid scaffold were more spread out (in their morphology) and displayed highly organised cytoskeletons. These cells were also seen to be more likely to undergo osteogenesis. In contrast, those cells grown on a more pliable structure were more rounded in shape with a disordered actin structure and predisposed to undergo adipogenic differentiation. Therefore, scaffolds made from stiff materials are more likely to be used for bone regenerative uses; this includes ceramics and bio-glasses, which can also bond quickly to bone [142]. Conversely, materials that are more pliable can be used for cartilage and soft tissue regeneration [143-145], as well as in the production of hydrogels for wound healing [146].
As well as directing the fate of cells through their basic structure and topography, scaffold materials can impact on the functions of cells through the presentation and release of proteins, molecules, and ions. The presentation of proteins and molecules will often be linked to the structure and function of the extracellular matrix (ECM) which provides structure and signals for cell growth and function. By studying the structural makeup of the ECM, appropriate additions can be made to scaffold materials. Schmitt, Murphy and Gopalan [147] isolated the functional epitope of fibronectin and added it to a polyethylene glycol (PEG) polymer sheet. This peptide sequence (Gly-Gly-Gly-Arg-Gly-Asp-Ser-Pro) had the same adhesive properties as the entire protein and by adding it to the polymer sheet they increased the rate of cell adhesion. Alternatively, some scaffolds can be used to cultivate osteoblasts to produce extracellular matrix (ECM) proteins and then be decellularised, leaving only the ECM behind [148,149]. In a similar vein, integration of signalling molecules into the scaffold material has also been used to improve cellular function. The addition of TGF-β3 into copolymer scaffolds was shown to enhance the regeneration of cartilage [150].
A final method by which scaffold materials can enhance the functional properties of MSCs is through ion dissolution. This was detailed by Xynos et al., [151] who show that the production of ions from a Bioglass® can have osteoinductive characteristics and this has been repeated by other groups showing that culture of cells in Bioglass®-conditioned media increases their expression of bone specific genes [152-154]. In further work, Xynos et al. [155] assessed how osteoblasts alter their gene expression after 48 hours of being cultured in Bioglass®-conditioned media. They found that genes controlling cell cycle, proliferation and adhesion were strongly upregulated in Bioglass®-conditioned cells. Similarly, Sun et al. [156] showed that culture in Bioglass®-conditioned media increases the rate of proliferation in osteoblasts.
Genetic Modification
Numerous studies in rodent and murine models have found that insertion of new genes into the genome of the MSC can prolong their survival, improve their functional characteristics, and enhance their production of therapeutic proteins (comprehensively reviewed in Park [157]). For example, the induced expression of CXCR4 in MSCs has been shown to enhance their homing potential and has led to improved repair of cardiac [158], liver [159], and bone tissues [160,161]. The overexpression of BCL-2 has been shown to protect cells from apoptosis and improve their differentiation potential [162]. Genetically modified MSCs can also be applied to cancer therapies, with studies showing that cells modified to express IFN-α [163], IFN-β [164], TNFSF10 [165], IL-12 [166] or CCL5 [167] can increase survival in murine models of haematological malignancies and solid tumours such as multiple myeloma [165] and pancreatic carcinoma [167], respectively.
Good Manufacturing Practice (GMP)
Irrespective of the donor source, the growth conditions or the status of genetic modification, the production of MSCs for any form of cellular therapy must be ensured to be compliant with Good Manufacturing Practices (GMPs), guaranteeing the safety, efficacy and reproducibility of MSC production. Any product designed for clinical application must follow strict GMP guidelines; thus any culture modifications used to enhance the potency of MSCs for clinical use must be GMP-transferrable. These are the rules and regulations that govern the production of medical products and therapies with the aim to safeguard the health of a patient by ensuring that all steps taken during production have been verified and that the product is regularly tested for contamination [168]. GMP compliance requires every process to be well documented so that any adverse event can be easily traced back to source, whilst also standardising the production process to guarantee a level of quality in each batch of product (Table 1 and Figure 1).
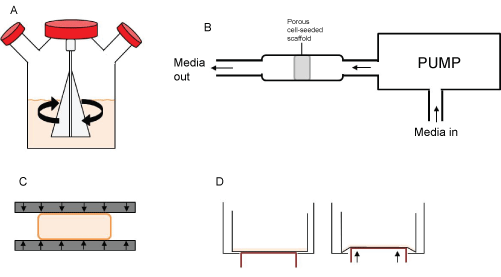
.
Figure 1: Mechanical stimulation for cell culture
Four schematic examples of mechanical stimulation that can be used in cell culture to enhance the properties of MSCs.
Image (A) shows a spinner flask bioreactor. These are flasks which contain a rotating propeller to continually stir the media and cells within. This provides homogeneous and dynamic culture conditions to the sample and can increase the level of osteogenic differentiation compared to static culture [140,186]. Flow perfusion (B) also creates a dynamic culture environment, but this is by pumping media through a porous scaffold. This has been shown to stimulate osteogenic differentiation in MSCs [133]. Mechanical compression (C) and mechanical strain (D) squeezes or stretches the cells on pliable substrates. Compression of MSCs can encourage chondrogenic differentiation [134,187] and strain can induce osteogenic differentiation [188].
View Figure 1
Conclusion
There is an increasing demand for MSCs in clinical applications, however, to date the research successes have yet to be fully translated from the "bench to the bedside". Only when the optimal donor source and selection criteria, in combination with the addition of bioactive materials and factors, are coupled with GMP-requirements can their full potential be exploited.
Acknowledgement
This work was supported by a grant from Arthritis Research UK Award 19429 and CellEurope project, FP7-people-2012-ITN, No. 315963.
References
-
Tavassoli M, Crosby WH (1968) Transplantation of marrow to extramedullary sites. Science 161: 54-56.
-
Friedenstein AJ, Chailakhjan RK, Lalykina KS (1970) The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Tissue Kinet 3: 393-403.
-
Friedenstein AJ, Chailakhyan RK, Gerasimov UV (1987) Bone marrow osteogenic stem cells: in vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet 20: 263-272.
-
Caplan AI (1991) Mesenchymal stem cells. J Orthop Res 9: 641-650.
-
Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, et al. (2005) Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 7: 393-395.
-
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, et al. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8: 315-317.
-
Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC (2003) Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 75: 389-397.
-
Ankrum JA, Ong JF, Karp JM (2014) Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol 32: 252-260.
-
Rasmusson I, Ringdén O, Sundberg B, Le Blanc K (2005) Mesenchymal stem cells inhibit lymphocyte proliferation by mitogens and alloantigens by different mechanisms. Exp Cell Res 305: 33-41.
-
Munn DH, Shafizadeh E, Attwood JT, Bondarev I, Pashine A, et al. (1999) Inhibition of T cell proliferation by macrophage tryptophan catabolism. J Exp Med 189: 1363-1372.
-
Glennie S, Soeiro I, Dyson PJ, Lam EW, Dazzi F (2005) Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 105: 2821-2827.
-
Yang SH, Park MJ, Yoon IH, Kim SY, Hong SH, et al. (2009) Soluble mediators from mesenchymal stem cells suppress T cell proliferation by inducing IL-10. Exp Mol Med 41: 315-324.
-
Liu WH, Liu JJ, Wu J, Zhang LL, Liu F, et al. (2013) Novel mechanism of inhibition of dendritic cells maturation by mesenchymal stem cells via interleukin-10 and the JAK1/STAT3 signaling pathway. PLoS One 8: e55487.
-
Yañez R, Oviedo A, Aldea M, Bueren JA, Lamana ML (2010) Prostaglandin E2 plays a key role in the immunosuppressive properties of adipose and bone marrow tissue-derived mesenchymal stromal cells. Exp Cell Res 316: 3109-3123.
-
Selmani Z, Naji A, Zidi I, Favier B, Gaiffe E, et al. (2008) Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+ regulatory T cells. Stem Cells 26: 212-222.
-
Le Blanc K, Rasmusson I, Sundberg B, Götherström C, Hassan M, et al. (2004) Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 363: 1439-1441.
-
Bartholomew A, Sturgeon C, Siatskas M, Ferrer K, McIntosh K, et al. (2002) Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 30: 42-48.
-
Müller I, Kordowich S, Holzwarth C, Isensee G, Lang P, et al. (2008) Application of multipotent mesenchymal stromal cells in pediatric patients following allogeneic stem cell transplantation. Blood Cells Mol Dis 40: 25-32.
-
Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, et al. (2008) Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 371: 1579-1586.
-
Arima N, Nakamura F, Fukunaga A, Hirata H, Machida H, et al. (2010) Single intra-arterial injection of mesenchymal stromal cells for treatment of steroid-refractory acute graft-versus-host disease: a pilot study. Cytotherapy 12: 265-268.
-
Zhou H, Guo M, Bian C, Sun Z, Yang Z, et al. (2010) Efficacy of bone marrow-derived mesenchymal stem cells in the treatment of sclerodermatous chronic graft-versus-host disease: clinical report. Biol Blood Marrow Transplant 16: 403-412.
-
Wakitani S, Mitsuoka T, Nakamura N, Toritsuka Y, Nakamura Y, et al. (2004) Autologous bone marrow stromal cell transplantation for repair of full-thickness articular cartilage defects in human patellae: two case reports. Cell Transplant 13: 595-600.
-
Centeno CJ, Busse D, Kisiday J, Keohan C, Freeman M, et al. (2008) Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells. Pain Physician 11: 343-353.
-
Giannini S, Buda R, Vannini F, Cavallo M, Grigolo B (2009) One-step bone marrow-derived cell transplantation in talar osteochondral lesions. Clin Orthop Relat Res 467: 3307-3320.
-
Giannini S, Buda R, Cavallo M, Ruffilli A, Cenacchi A, et al. (2010) Cartilage repair evolution in post-traumatic osteochondral lesions of the talus: from open field autologous chondrocyte to bone-marrow-derived cells transplantation. Injury 41: 1196-1203.
-
Buda R, Vannini F, Cavallo M, Grigolo B, Cenacchi A, et al. (2010) Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am 92 Suppl 2: 2-11.
-
Kasemkijwattana C, Hongeng S, Kesprayura S, Rungsinaporn V, Chaipinyo K, et al. (2011) Autologous bone marrow mesenchymal stem cells implantation for cartilage defects: two cases report. J Med Assoc Thai 94: 395-400.
-
Emadedin M, Aghdami N, Taghiyar L, Fazeli R, Moghadasali R, et al. (2012) Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med 15: 422-428.
-
Jo CH, Lee YG, Shin WH, Kim H, Chai JW, et al. (2014) Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells 32: 1254-1266.
-
Rebelatto CK, Aguiar AM, Moretão MP, Senegaglia AC, Hansen P, et al. (2008) Dissimilar differentiation of mesenchymal stem cells from bone marrow, umbilical cord blood, and adipose tissue. Exp Biol Med (Maywood) 233: 901-913.
-
Choudhery MS, Badowski M, Muise A, Harris DT (2013) Comparison of human mesenchymal stem cells derived from adipose and cord tissue. Cytotherapy 15: 330-343.
-
Qiao C, Xu W, Zhu W, Hu J, Qian H, et al. (2008) Human mesenchymal stem cells isolated from the umbilical cord. Cell Biol Int 32: 8-15.
-
Hass R, Kasper C, Böhm S, Jacobs R (2011) Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun Signal 9: 12.
-
Perry BC, Zhou D, Wu X, Yang FC, Byers MA, et al. (2008) Collection, cryopreservation, and characterization of human dental pulp-derived mesenchymal stem cells for banking and clinical use. Tissue Eng Part C Methods 14: 149-156.
-
Barlow S, Brooke G, Chatterjee K, Price G, Pelekanos R, et al. (2008) Comparison of human placenta- and bone marrow-derived multipotent mesenchymal stem cells. Stem Cells Dev 17: 1095-1107.
-
Jin HJ, Bae YK, Kim M, Kwon SJ, Jeon HB, et al. (2013) Comparative analysis of human mesenchymal stem cells from bone marrow, adipose tissue, and umbilical cord blood as sources of cell therapy. Int J Mol Sci 14: 17986-18001.
-
Kern S, Eichler H, Stoeve J, Klüter H, Bieback K (2006) Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 24: 1294-1301.
-
Zhang X, Hirai M, Cantero S, Ciubotariu R, Dobrila L, et al. (2011) Isolation and characterization of mesenchymal stem cells from human umbilical cord blood: reevaluation of critical factors for successful isolation and high ability to proliferate and differentiate to chondrocytes as compared to mesenchymal stem cells from bone marrow and adipose tissue. J Cell Biochem 112: 1206-1218.
-
Priya N, Sarcar S, Majumdar AS, SundarRaj S (2014) Explant culture: a simple, reproducible, efficient and economic technique for isolation of mesenchymal stromal cells from human adipose tissue and lipoaspirate. J Tissue Eng Regen Med 8: 706-716.
-
Fraser JK, Wulur I, Alfonso Z, Hedrick MH (2006) Fat tissue: an underappreciated source of stem cells for biotechnology. Trends Biotechnol 24: 150-154.
-
Semon JA, Maness C, Zhang X, Sharkey SA, Beuttler MM, et al. (2014) Comparison of human adult stem cells from adipose tissue and bone marrow in the treatment of experimental autoimmune encephalomyelitis. Stem Cell Res Ther 5: 2.
-
Melief SM, Zwaginga JJ, Fibbe WE, Roelofs H. (2013) Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med 2: 455-463.
-
Im GI, Shin YW, Lee KB (2005) Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthritis Cartilage 13: 845-853.
-
Muraglia A, Cancedda R, Quarto R (2000) Clonal mesenchymal progenitors from human bone marrow differentiate in vitro according to a hierarchical model. J Cell Sci 113: 1161-1166.
-
Whitfield MJ, Lee WC, Van Vliet KJ (2013) Onset of heterogeneity in culture-expanded bone marrow stromal cells. Stem Cell Res 11: 1365-1377.
-
Flores-Torales E, Orozco-Barocio A, Gonzalez-Ramella OR, Carrasco-Yalan A, Gazarian K, et al. (2010) The CD271 expression could be alone for establisher phenotypic marker in Bone Marrow derived mesenchymal stem cells. Folia Histochem Cytobiol 48: 682-686.
-
Cattoretti G, Schiró R, Orazi A, Soligo D, Colombo MP (1993) Bone marrow stroma in humans: anti-nerve growth factor receptor antibodies selectively stain reticular cells in vivo and in vitro. Blood 81: 1726-1738.
-
Caneva L, Soligo D, Cattoretti G, De Harven E, Deliliers GL (1995) Immuno-electron microscopy characterization of human bone marrow stromal cells with anti-NGFR antibodies. Blood Cells Mol Dis 21: 73-85.
-
Quirici N, Soligo D, Bossolasco P, Servida F, Lumini C, et al. (2002) Isolation of bone marrow mesenchymal stem cells by anti-nerve growth factor receptor antibodies. Exp Hematol 30: 783-791.
-
Jones E, English A, Churchman SM, Kouroupis D, Boxall SA, et al. (2010) Large-scale extraction and characterization of CD271+ multipotential stromal cells from trabecular bone in health and osteoarthritis: implications for bone regeneration strategies based on uncultured or minimally cultured multipotential stromal cells. Arthritis Rheum 62: 1944-1954.
-
Cox G, Boxall SA, Giannoudis PV, Buckley CT, Roshdy T, et al. (2012) High abundance of CD271(+) multipotential stromal cells (MSCs) in intramedullary cavities of long bones. Bone 50: 510-517.
-
Cuevas-Diaz, Duran R González-Garza MT, Cardenas-Lopez A, Chavez-Castilla L, Cruz-Vega DE, et al. (2013) Age-related yield of adipose-derived stem cells bearing the low-affinity nerve growth factor receptor. Stem Cells Int 2013: 372164.
-
Mikami Y, Ishii Y, Watanabe N, Shirakawa T, Suzuki S, et al. (2011) CD271/p75(NTR) inhibits the differentiation of mesenchymal stem cells into osteogenic, adipogenic, chondrogenic, and myogenic lineages. Stem Cells Dev 20: 901-913.
-
Álvarez-Viejo, M Menéndez-Menéndez Y, Otero-Hernández J (2015) CD271 as a marker to identify mesenchymal stem cells from diverse sources before culture. World J Stem Cells 7: 470-476.
-
Watson JT, Foo T, Wu J, Moed BR, Thorpe M, et al. (2013) CD271 as a marker for mesenchymal stem cells in bone marrow versus umbilical cord blood. Cells Tissues Organs 197: 496-504.
-
Kuçi Z, Kuçi S, Zircher S, Koller S, Schubert R, et al. (2011) Mesenchymal stromal cells derived from CD271(+) bone marrow mononuclear cells exert potent allosuppressive properties. Cytotherapy 13: 1193-1204.
-
Kuçi Z, Seiberth J, Latifi-Pupovci H, Wehner S, Stein S, et al. (2013) Clonal analysis of multipotent stromal cells derived from CD271+ bone marrow mononuclear cells: functional heterogeneity and different mechanisms of allosuppression. Haematologica 98: 1609-1616.
-
Kuçi S, Kuçi Z, Kreyenberg H, Deak E, Pütsch K, et al. (2010) CD271 antigen defines a subset of multipotent stromal cells with immunosuppressive and lymphohematopoietic engraftment-promoting properties. Haematologica 95: 651-659.
-
Jones EA, Kinsey SE, English A, Jones RA, Straszynski L, et al. (2002) Isolation and characterization of bone marrow multipotential mesenchymal progenitor cells. Arthritis Rheum 46: 3349-3360.
-
Churchman SM, Ponchel F, Boxall SA, Cuthbert R, Kouroupis D, et al. (2012) Transcriptional profile of native CD271+ multipotential stromal cells: evidence for multiple fates, with prominent osteogenic and Wnt pathway signaling activity. Arthritis Rheum 64: 2632-2643.
-
Hermida-Gómez T, Fuentes-Boquete I, Gimeno-Longas MJ, Muiños-López E, Díaz-Prado S, et al. (2011) Bone marrow cells immunomagnetically selected for CD271+ antigen promote in vitro the repair of articular cartilage defects. Tissue Eng Part A 17: 1169-1179.
-
Mifune Y, Matsumoto T, Murasawa S, Kawamoto A, Kuroda R, et al. (2013) Therapeutic superiority for cartilage repair by CD271-positive marrow stromal cell transplantation. Cell Transplant 22: 1201-1211.
-
Aomatsu E, Takahashi N, Sawada S, Okubo N, Hasegawa T, et al. (2014) Novel SCRG1/BST1 axis regulates self-renewal, migration, and osteogenic differentiation potential in mesenchymal stem cells. Sci Rep 4: 3652.
-
Murillo-Sauca O, Chung MK, Shin JH, Karamboulas C, Kwok S, et al. (2014) CD271 is a functional and targetable marker of tumor-initiating cells in head and neck squamous cell carcinoma. Oncotarget. 5: 6854-6866.
-
Civenni G, Walter A, Kobert N, Mihic-Probst D, Zipser M, et al. (2011) Human CD271 positive melanoma stem cells associated with metastasis establish tumor heterogeneity and long-term growth. Cancer Res 71: 3098-3109.
-
Tian J, Li X, Si M, Liu T, Li J (2014) CD271+ osteosarcoma cells display stem-like properties. PLoS One 9: e98549.
-
Li S, Yue D, Chen X, Wang L, Li J, et al. (2015) Epigenetic regulation of CD27 a potential cancer stem cell marker associated with chemoresistance and metastatic capacity. Oncol Rep 33: 425-432.
-
Redmer T, Welte Y, Behrens D, Fichtner I, Przybilla D, et al. (2014) The nerve growth factor receptor CD271 is crucial to maintain tumorigenicity and stem-like properties of melanoma cells. PLoS One 9: e92596.
-
Okumura T, Tsunoda S, Mori Y, Ito T, Kikuchi K, et al. (2006) The biological role of the low-affinity p75 neurotrophin receptor in esophageal squamous cell carcinoma. Clin Cancer Res 12: 5096-5103.
-
Huang SD, Yuan Y, Liu XH, Gong DJ, Bai CG, et al. (2009) Self-renewal and chemotherapy resistance of p75NTR positive cells in esophageal squamous cell carcinomas. BMC Cancer 9: 9.
-
Boxall SA, Jones E (2012) Markers for characterization of bone marrow multipotential stromal cells. Stem Cells Int 2012: 975871.
-
Cuthbert RJ, Giannoudis PV, Wang XN, Nicholson L, Pawson D, et al. (2015) Examining the feasibility of clinical grade CD271+ enrichment of mesenchymal stromal cells for bone regeneration. PLoS One 10: e0117855.
-
Murphy MB, Moncivais K, Caplan AI (2013) Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med 45: e54.
-
Spees JL, Gregory CA, Singh H, Tucker HA, Peister A, et al. (2004) Internalized antigens must be removed to prepare hypoimmunogenic mesenchymal stem cells for cell and gene therapy. Mol Ther 9: 747-756.
-
Sakamoto N, Tsuji K, Muul LM, Lawler AM, Petricoin EF, et al. (2007) Bovine apolipoprotein B-100 is a dominant immunogen in therapeutic cell populations cultured in fetal calf serum in mice and humans. Blood 110: 501-508.
-
Sundin M, Ringdén O, Sundberg B, Nava S, Götherström C, et al. (2007) No alloantibodies against mesenchymal stromal cells, but presence of anti-fetal calf serum antibodies, after transplantation in allogeneic hematopoietic stem cell recipients. Haematologica 92: 1208-1215.
-
Chou ML, Bailey A, Avory T, Tanimoto J, Burnouf T (2015) Removal of transmissible spongiform encephalopathy prion from large volumes of cell culture media supplemented with fetal bovine serum by using hollow fiber anion-exchange membrane chromatography. PLoS One 10: e0122300.
-
van der Valk J, Mellor D, Brands R, Fischer R, Gruber F, et al. (2004) The humane collection of fetal bovine serum and possibilities for serum-free cell and tissue culture. Toxicol In Vitro 18: 1-12.
-
Kocaoemer A, Kern S, Klüter H, Bieback K (2007) Human AB serum and thrombin-activated platelet-rich plasma are suitable alternatives to fetal calf serum for the expansion of mesenchymal stem cells from adipose tissue. Stem Cells 25: 1270-1278.
-
Stute N, Holtz K, Bubenheim M, Lange C, Blake F, et al. (2004) Autologous serum for isolation and expansion of human mesenchymal stem cells for clinical use. Exp Hematol 32: 1212-1225.
-
Kobayashi T, Watanabe H, Yanagawa T, Tsutsumi S, Kayakabe M, et al. (2005) Motility and growth of human bone-marrow mesenchymal stem cells during ex vivo expansion in autologous serum. J Bone Joint Surg Br 87: 1426-1433.
-
Nimura A, Muneta T, Koga H, Mochizuki T, Suzuki K, et al. (2008) Increased proliferation of human synovial mesenchymal stem cells with autologous human serum: comparisons with bone marrow mesenchymal stem cells and with fetal bovine serum. Arthritis Rheum 58: 501-510.
-
Shahdadfar A, Frønsdal K, Haug T, Reinholt FP, Brinchmann JE (2005) In vitro expansion of human mesenchymal stem cells: choice of serum is a determinant of cell proliferation, differentiation, gene expression, and transcriptome stability. Stem Cells. 23: 1357-1366.
-
Dahl JA, Duggal S, Coulston N, Millar D, Melki J, et al. (2008) Genetic and epigenetic instability of human bone marrow mesenchymal stem cells expanded in autologous serum or fetal bovine serum. Int J Dev Biol 52: 1033-1042.
-
Bieback K, Hecker A, Kocaömer A, Lannert H, Schallmoser K, et al. (2009) Human alternatives to fetal bovine serum for the expansion of mesenchymal stromal cells from bone marrow. Stem Cells 27: 2331-2341.
-
Bieback K, Hecker A, Schlechter T, Hofmann I, Brousos N, et al. (2012) Replicative aging and differentiation potential of human adipose tissue-derived mesenchymal stromal cells expanded in pooled human or fetal bovine serum. Cytotherapy 14: 570-583.
-
Warnke PH, Humpe A, Strunk D, Stephens S, Warnke F, et al. (2013) A clinically-feasible protocol for using human platelet lysate and mesenchymal stem cells in regenerative therapies. J Craniomaxillofac Surg 41: 153-161.
-
Kuznetsov SA, Mankani MH, Robey PG (2000) Effect of serum on human bone marrow stromal cells: ex vivo expansion and in vivo bone formation. Transplantation 70: 1780-1787.
-
Mirabet V, Solves P, Miñana MD, Encabo A, Carbonell-Uberos F, et al. (2008) Human platelet lysate enhances the proliferative activity of cultured human fibroblast-like cells from different tissues. Cell Tissue Bank 9: 1-10.
-
Castegnaro S, Chieregato K, Maddalena M, Albiero E, Visco C, et al. (2011) Effect of platelet lysate on the functional and molecular characteristics of mesenchymal stem cells isolated from adipose tissue. Curr Stem Cell Res Ther 6: 105-114.
-
Govindasamy V, Ronald VS, Abdullah AN, Ganesan Nathan KR, Aziz ZA, et al. (2011)Human platelet lysate permits scale-up of dental pulp stromal cells for clinical applications. Cytotherapy 13: 1221-1233.
-
Hofbauer P, Riedl S, Witzeneder K, Hildner F, Wolbank S, et al. (2014) Human platelet lysate is a feasible candidate to replace fetal calf serum as medium supplement for blood vascular and lymphatic endothelial cells. Cytotherapy 16: 1238-1244.
-
Doucet C, Ernou I, Zhang Y, Llense JR, Begot L, et al. (2005) Platelet lysates promote mesenchymal stem cell expansion: a safety substitute for animal serum in cell-based therapy applications. J Cell Physiol 205: 228-236.
-
Müller I, Kordowich S, Holzwarth C, Spano C, Isensee G, et al. (2006) Animal serum-free culture conditions for isolation and expansion of multipotent mesenchymal stromal cells from human BM. Cytotherapy 8: 437-444.
-
Schallmoser K, Bartmann C, Rohde E, Reinisch A, Kashofer K, et al. (2007) Human platelet lysate can replace fetal bovine serum for clinical-scale expansion of functional mesenchymal stromal cells. Transfusion 47: 1436-1446.
-
Avanzini MA, Bernardo ME, Cometa AM, Perotti C, Zaffaroni N, et al. (2009) Generation of mesenchymal stromal cells in the presence of platelet lysate: a phenotypic and functional comparison of umbilical cord blood- and bone marrow-derived progenitors. Haematologica 94: 1649-1660.
-
Crespo-Diaz R, Behfar A, Butler GW, Padley DJ, Sarr MG, et al. (2011) Platelet lysate consisting of a natural repair proteome supports human mesenchymal stem cell proliferation and chromosomal stability. Cell Transplant 20: 797-811.
-
Lange C, Cakiroglu F, Spiess AN, Cappallo-Obermann H, Dierlamm J, et al. (2007) Accelerated and safe expansion of human mesenchymal stromal cells in animal serum-free medium for transplantation and regenerative medicine. J Cell Physiol 213: 18-26.
-
Bernardo ME, Avanzini MA, Perotti C, Cometa AM, Moretta A, et al. (2007) Optimization of in vitro expansion of human multipotent mesenchymal stromal cells for cell-therapy approaches: further insights in the search for a fetal calf serum substitute. J Cell Physiol 211: 121-130.
-
von Bonin M, Stölzel F, Goedecke A, Richter K, Wuschek N, et al. (2009) Treatment of refractory acute GVHD with third-party MSC expanded in platelet lysate-containing medium. Bone Marrow Transplant 43: 245-251.
-
Bieback K, Ha VA, Hecker A, Grassl M, Kinzebach S, et al. (2010) Altered gene expression in human adipose stem cells cultured with fetal bovine serum compared to human supplements. Tissue Eng Part A 16: 3467-3484.
-
Horn P, Bokermann G, Cholewa D, Bork S, Walenda T, et al. (2010) Impact of individual platelet lysates on isolation and growth of human mesenchymal stromal cells. Cytotherapy 12: 888-898.
-
Martin I, Muraglia A, Campanile G, Cancedda R, Quarto R (1997) Fibroblast growth factor-2 supports ex vivo expansion and maintenance of osteogenic precursors from human bone marrow. Endocrinology 138: 4456-4462.
-
Solchaga LA, Penick K, Porter JD, Goldberg VM, Caplan AI, et al. (2005) FGF-2 enhances the mitotic and chondrogenic potentials of human adult bone marrow-derived mesenchymal stem cells. J Cell Physiol 203: 398-409.
-
Gharibi B, Hughes FJ (2012) Effects of medium supplements on proliferation, differentiation potential, and in vitro expansion of mesenchymal stem cells. Stem Cells Transl Med 1: 771-782.
-
Wu J, Huang GT, He W, Wang P, Tong Z, et al. (2012) Basic fibroblast growth factor enhances stemness of human stem cells from the apical papilla. J Endod 38: 614-622.
-
Sotiropoulou PA, Perez SA, Salagianni M, Baxevanis CN, Papamichail M (2006) Characterization of the optimal culture conditions for clinical scale production of human mesenchymal stem cells. Stem Cells 24: 462-471.
-
Cassiede P, Dennis JE, Ma F, Caplan AI (1996) Osteochondrogenic potential of marrow mesenchymal progenitor cells exposed to TGF-beta 1 or PDGF-BB as assayed in vivo and in vitro. J Bone Miner Res 11: 1264-1273.
-
Gharibi B, Ghuman MS, Hughes FJ (2012) Akt- and Erk-mediated regulation of proliferation and differentiation during PDGFRß-induced MSC self-renewal. J Cell Mol Med 16: 2789-2801.
-
Tokunaga A, Oya T, Ishii Y, Motomura H, Nakamura C, et al. (2008) PDGF receptor beta is a potent regulator of mesenchymal stromal cell function. J Bone Miner Res 23: 1519-1528.
-
Ng F, Boucher S, Koh S, Sastry KS, Chase L, et al. (2008) PDGF, TGF-beta, and FGF signaling is important for differentiation and growth of mesenchymal stem cells (MSCs): transcriptional profiling can identify markers and signaling pathways important in differentiation of MSCs into adipogenic, chondrogenic, and osteogenic lineages. Blood 112: 295-307.
-
Chase LG, Lakshmipathy U, Solchaga LA, Rao MS, Vemuri MC (2010) A novel serum-free medium for the expansion of human mesenchymal stem cells. Stem Cell Res Ther 1: 8.
-
Fehrer C, Brunauer R, Laschober G, Unterluggauer H, Reitinger S, et al. (2007) Reduced oxygen tension attenuates differentiation capacity of human mesenchymal stem cells and prolongs their lifespan. Aging Cell 6: 745-757.
-
Carreau AE, Hafny-Rahbi B, Matejuk A, Grillon C, Kieda C (2011) Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J Cell Mol Med 15: 1239-1253.
-
Cooper PD, Burt AM, Wilson JN (1958) Critical effect of oxygen tension on rate of growth of animal cells in continuous suspended culture. Nature 182: 1508-1509.
-
Grayson WL, Zhao F, Bunnell B, Ma T (2007) Hypoxia enhances proliferation and tissue formation of human mesenchymal stem cells. Biochem Biophys Res Commun 358: 948-953.
-
Dos Santos F, Andrade PZ, Boura JS, Abecasis MM, da Silva CL, et al. (2010) Ex vivo expansion of human mesenchymal stem cells: a more effective cell proliferation kinetics and metabolism under hypoxia. J Cell Physiol 223: 27-35.
-
Basciano L, Nemos C, Foliguet B, de Isla N, de Carvalho M, et al. (2011) Long term culture of mesenchymal stem cells in hypoxia promotes a genetic program maintaining their undifferentiated and multipotent status. BMC Cell Biol 12: 12.
-
Boregowda SV, Krishnappa V, Chambers JW, Lograsso PV, Lai WT, et al. (2012) Atmospheric oxygen inhibits growth and differentiation of marrow-derived mouse mesenchymal stem cells via a p53-dependent mechanism: implications for long-term culture expansion. Stem Cells 30: 975-987.
-
Hung SP, Ho JH, Shih YR, Lo T, Lee OK (2012) Hypoxia promotes proliferation and osteogenic differentiation potentials of human mesenchymal stem cells. J Orthop Res 30: 260-266.
-
Berniakovich I, Giorgio M (2013) Low oxygen tension maintains multipotency, whereas normoxia increases differentiation of mouse bone marrow stromal cells. Int J Mol Sci 14: 2119-2134.
-
Park IH, Kim KH, Choi HK, Shim JS, Whang SY, et al. (2013) Constitutive stabilization of hypoxia-inducible factor alpha selectively promotes the self-renewal of mesenchymal progenitors and maintains mesenchymal stromal cells in an undifferentiated state. Exp Mol Med 45: e44.
-
Holzwarth C, Vaegler M, Gieseke F, Pfister SM, Handgretinger R, et al. (2010) Low physiologic oxygen tensions reduce proliferation and differentiation of human multipotent mesenchymal stromal cells. BMC Cell Biol, 11:11.
-
Ren H, Cao Y, Zhao Q, Li J, Zhou C, et al. (2006) Proliferation and differentiation of bone marrow stromal cells under hypoxic conditions. Biochem Biophys Res Commun 347: 12-21.
-
Kallio PJ, Wilson WJ, O'Brien S, Makino Y, Poellinger L (1999) Regulation of the hypoxia-inducible transcription factor 1alpha by the ubiquitin-proteasome pathway. J Biol Chem 274: 6519-6525.
-
Tsai CC, Chen YJ, Yew TL, Chen LL, Wang JY, et al. (2011) Hypoxia inhibits senescence and maintains mesenchymal stem cell properties through down-regulation of E2A-p21 by HIF-TWIST. Blood 117: 459-469.
-
Estrada JC, Albo C, Benguría A, Dopazo A, López-Romero P, et al. (2012) Culture of human mesenchymal stem cells at low oxygen tension improves growth and genetic stability by activating glycolysis. Cell Death Differ 19: 743-755.
-
Yeatts AB, Choquette DT, Fisher JP (2013) Bioreactors to influence stem cell fate: augmentation of mesenchymal stem cell signaling pathways via dynamic culture systems. Biochim Biophys Acta 1830: 2470-2480.
-
Kapur S, Baylink DJ, Lau KH (2003) Fluid flow shear stress stimulates human osteoblast proliferation and differentiation through multiple interacting and competing signal transduction pathways. Bone 32: 241-251.
-
Kanno T, Takahashi T, Tsujisawa T, Ariyoshi W, Nishihara T (2007) Mechanical stress-mediated Runx2 activation is dependent on Ras/ERK1/2 MAPK signaling in osteoblasts. J Cell Biochem 101: 1266-1277.
-
Yan YX, Gong YW, Guo Y, Lv Q, Guo C, et al. (2012) Mechanical strain regulates osteoblast proliferation through integrin-mediated ERK activation. PLoS One 7: e35709.
-
Mu C, Lv T, Wang Z, Ma S, Ma J, et al. (2014) Mechanical stress stimulates the osteo/odontoblastic differentiation of human stem cells from apical papilla via erk 1/2 and JNK MAPK pathways. Biomed Res Int 2014: 494378.
-
Holtorf HL, Jansen JA, Mikos AG (2005) Flow perfusion culture induces the osteoblastic differentiation of marrow stroma cell-scaffold constructs in the absence of dexamethasone. J Biomed Mater Res A 72: 326-334.
-
Mauck RL, Byers BA, Yuan X, Tuan RS (2007) Regulation of cartilaginous ECM gene transcription by chondrocytes and MSCs in 3D culture in response to dynamic loading. Biomech Model Mechanobiol 6: 113-125.
-
Sen B, Styner M, Xie Z, Case N, Rubin CT, et al. (2009) Mechanical loading regulates NFATc1 and beta-catenin signaling through a GSK3beta control node. J Biol Chem 284: 34607-34617.
-
Case N, Xie Z, Sen B, Styner M, Zou M, et al. (2010) Mechanical activation of ß-catenin regulates phenotype in adult murine marrow-derived mesenchymal stem cells. J Orthop Res 28: 1531-1538.
-
Case N, Thomas J, Xie Z, Sen B, Styner M, et al. (2013) Mechanical input restrains PPAR?2 expression and action to preserve mesenchymal stem cell multipotentiality. Bone 52: 454-464.
-
Sikavitsas VI, Bancroft GN, Holtorf HL, Jansen JA, Mikos AG (2003) Mineralized matrix deposition by marrow stromal osteoblasts in 3D perfusion culture increases with increasing fluid shear forces. Proc Natl Acad Sci USA 100: 14683-14688.
-
Sittichokechaiwut A, Edwards JH, Scutt AM, Reilly GC (2010) Short bouts of mechanical loading are as effective as dexamethasone at inducing matrix production by human bone marrow mesenchymal stem cell. Eur Cell Mater 20: 45-57.
-
Frith JE, Thomson B, Genever PG (2010) Dynamic three-dimensional culture methods enhance mesenchymal stem cell properties and increase therapeutic potential. Tissue Eng Part C Methods 16: 735-749.
-
Fu J, Wang YK, Yang MT, Desai RA, Yu X, et al. (2010) Mechanical regulation of cell function with geometrically modulated elastomeric substrates. Nat Methods 7: 733-736.
-
Hench LL (2006) The story of Bioglass. J Mater Sci Mater Med 17: 967-978.
-
Kang SW, Bada LP, Kang CS, Lee JS, Kim CH, et al. (2008) Articular cartilage regeneration with microfracture and hyaluronic acid. Biotechnol Lett 30: 435-439.
-
Shah RN, Shah NA, Del Rosario Lim MM, Hsieh C, Nuber G, et al. (2010) Supramolecular design of self-assembling nanofibers for cartilage regeneration. Proc Natl Acad Sci USA 107: 3293-3298.
-
Li X, Ma X, Fan D, Zhu C (2012) New suitable for tissue reconstruction injectable chitosan/collagen-based hydrogels. Soft Matter 8: 3781-3790.
-
Rustad KC, Wong VW, Sorkin M, Glotzbach JP, Major MR, et al. (2012) Enhancement of mesenchymal stem cell angiogenic capacity and stemness by a biomimetic hydrogel scaffold. Biomaterials 33: 80-90.
-
Schmitt SK, Murphy WL, Gopalan P (2013) Crosslinked PEG mats for peptide immobilization and stem cell adhesion. Journal of Materials Chemistry B 1: 1349-1360.
-
Datta N, Holtorf HL, Sikavitsas VI, Jansen JA, Mikos AG (2005) Effect of bone extracellular matrix synthesized in vitro on the osteoblastic differentiation of marrow stromal cells. Biomaterials 26: 971-977.
-
Pati F, Song TH, Rijal G, Jang J, Kim SW, et al. (2015) Ornamenting 3D printed scaffolds with cell-laid extracellular matrix for bone tissue regeneration. Biomaterials 37: 230-241.
-
Park JS, Yang HJ, Woo DG, Yang HN, Na K, et al. (2010) Chondrogenic differentiation of mesenchymal stem cells embedded in a scaffold by long-term release of TGF-beta 3 complexed with chondroitin sulfate. J Biomed Mater Res A 92: 806-816.
-
Xynos ID, Hukkanen MV, Batten JJ, Buttery LD, Hench LL, et al. (2000) Bioglass 45S5 stimulates osteoblast turnover and enhances bone formation In vitro: implications and applications for bone tissue engineering. Calcif Tissue Int 67: 321-329.
-
Bielby RC, Boccaccini AR, Polak JM, Buttery LD (2004) In vitro differentiation and in vivo mineralization of osteogenic cells derived from human embryonic stem cells. Tissue Eng 10: 1518-1525.
-
Bielby RC, Pryce RS, Hench LL, Polak JM (2005) Enhanced derivation of osteogenic cells from murine embryonic stem cells after treatment with ionic dissolution products of 58S bioactive sol-gel glass. Tissue Eng 11: 479-488.
-
Jell G, Notingher I, Tsigkou O, Notingher P, Polak JM, et al. (2008) Bioactive glass-induced osteoblast differentiation: a noninvasive spectroscopic study. J Biomed Mater Res A 86: 31-40.
-
Xynos ID, Edgar AJ, Buttery LD, Hench LL, Polak JM (2001) Gene-expression profiling of human osteoblasts following treatment with the ionic products of Bioglass 45S5 dissolution. J Biomed Mater Res 55: 151-157.
-
Sun JY, Yang YS, Zhong J, Greenspan DC (2007) The effect of the ionic products of Bioglass dissolution on human osteoblasts growth cycle in vitro. J Tissue Eng Regen Med 1: 281-286.
-
Park JS, Suryaprakash S, Lao YH, Leong KW (2015) Engineering mesenchymal stem cells for regenerative medicine and drug delivery. Methods 84: 3-16.
-
Cheng Z, Ou L, Zhou X, Li F, Jia X, et al. (2008) Targeted migration of mesenchymal stem cells modified with CXCR4 gene to infarcted myocardium improves cardiac performance. Mol Ther 16: 571-579.
-
Du Z, Wei C, Yan J, Han B, Zhang M, et al. (2013) Mesenchymal stem cells overexpressing C-X-C chemokine receptor type 4 improve early liver regeneration of small-for-size liver grafts. Liver Transpl 19: 215-225.
-
Cho SW, Sun HJ, Yang JY, Jung JY, An JH, et al. (2009) Transplantation of mesenchymal stem cells overexpressing RANK-Fc or CXCR4 prevents bone loss in ovariectomized mice. Mol Ther 17: 1979-1987.
-
Lien CY, Chih-Yuan Ho K, Lee OK, Blunn GW, Su Y (2009) Restoration of bone mass and strength in glucocorticoid-treated mice by systemic transplantation of CXCR4 and cbfa-1 co-expressing mesenchymal stem cells. J Bone Miner Res 24: 837-848.
-
Li W, Ma N, Ong LL, Nesselmann C, Klopsch C, et al. (2007) Bcl-2 engineered MSCs inhibited apoptosis and improved heart function. Stem Cells 25: 2118-2127.
-
Ren C, Kumar S, Chanda D, Chen J, Mountz JD, et al. (2008) Therapeutic potential of mesenchymal stem cells producing interferon-alpha in a mouse melanoma lung metastasis model. Stem Cells 26: 2332-2338.
-
Ren C, Kumar S, Chanda D, Kallman L, Chen J, et al. (2008) Cancer gene therapy using mesenchymal stem cells expressing interferon-beta in a mouse prostate cancer lung metastasis model. Gene Ther 15: 1446-1453.
-
Ciavarella S, Grisendi G, Dominici M, Tucci M, Brunetti O, et al. (2012) In vitro anti-myeloma activity of TRAIL-expressing adipose-derived mesenchymal stem cells. Br J Haematol 157: 586-598.
-
Han J, Zhao J, Xu J, Wen Y (2014) Mesenchymal stem cells genetically modified by lentivirus-mediated interleukin-12 inhibit malignant ascites in mice. Exp Ther Med 8: 1330-1334.
-
Zischek C, Niess H, Ischenko I, Conrad C, Huss R, et al. (2009) Targeting tumor stroma using engineered mesenchymal stem cells reduces the growth of pancreatic carcinoma. Ann Surg 250: 747-753.
-
Unger C, Skottman H, Blomberg P, Dilber MS, Hovatta O (2008) Good manufacturing practice and clinical-grade human embryonic stem cell lines. Hum Mol Genet 17: R48-53.
-
Zhu H, Mitsuhashi N, Klein A, Barsky LW, Weinberg K, et al. (2006) The role of the hyaluronan receptor CD44 in mesenchymal stem cell migration in the extracellular matrix. Stem Cells 24: 928-935.
-
Ode A, Schoon J, Kurtz A, Gaetjen M, Ode JE, et al. (2013) CD73/5'-ecto-nucleotidase acts as a regulatory factor in osteo-/chondrogenic differentiation of mechanically stimulated mesenchymal stromal cells. Eur Cell Mater 25: 37-47.
-
Yang ZX, Han ZB, Ji YR, Wang YW, Liang L, et al. (2013) CD106 identifies a subpopulation of mesenchymal stem cells with unique immunomodulatory properties. PLoS One 8: e59354.
-
Covas DT, Panepucci RA, Fontes AM, Silva WA Jr, Orellana MD, et al. (2008) Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with CD146+ perivascular cells and fibroblasts. Exp Hematol 36: 642-654.
-
Tormin A, Li O, Brune JC, Walsh S, Schütz B, et al. (2011) CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood 117: 5067-5077.
-
Espagnolle N, Guilloton F, Deschaseaux F, Gadelorge M, Sensébé L, et al. (2014) CD146 expression on mesenchymal stem cells is associated with their vascular smooth muscle commitment. J Cell Mol Med 18: 104-114.
-
Simmons PJ, Torok-Storb B (1991) Identification of stromal cell precursors in human bone marrow by a novel monoclonal antibody, STRO-1. Blood 78: 55-62.
-
Gronthos S, Graves SE, Ohta S, Simmons PJ (1994) The STRO-1+ fraction of adult human bone marrow contains the osteogenic precursors. Blood 84: 4164-4173.
-
Oyajobi BO, Lomri A, Hott M, Marie PJ (1999) Isolation and characterization of human clonogenic osteoblast progenitors immunoselected from fetal bone marrow stroma using STRO-1 monoclonal antibody. J Bone Miner Res 14: 351-361.
-
Bensidhoum M, Chapel A, Francois S, Demarquay C, Mazurier C, et al. (2004) Homing of in vitro expanded Stro-1- or Stro-1+ human mesenchymal stem cells into the NOD/SCID mouse and their role in supporting human CD34 cell engraftment. Blood 103: 3313-3319.
-
Lin G, Liu G, Banie L, Wang G, Ning H, et al. (2011) Tissue distribution of mesenchymal stem cell marker Stro-1. Stem Cells Dev 20: 1747-1752.
-
Gang EJ, Bosnakovski D, Figueiredo CA, Visser JW, Perlingeiro RC (2007) SSEA-4 identifies mesenchymal stem cells from bone marrow. Blood 109: 1743-1751.
-
Mabuchi Y, Morikawa S, Harada S, Niibe K, Suzuki S, et al. (2013) LNGFR(+)THY-1(+)VCAM-1(hi+) cells reveal functionally distinct subpopulations in mesenchymal stem cells. Stem Cell Reports 1: 152-165.
-
Gronthos S, Zannettino AC, Hay SJ, Shi S, Graves SE, et al. (2003) Molecular and cellular characterisation of highly purified stromal stem cells derived from human bone marrow. J Cell Sci 116: 1827-1835.
-
Battula VL, Treml S, Bareiss PM, Gieseke F, Roelofs H, et al. (2009) Isolation of functionally distinct mesenchymal stem cell subsets using antibodies against CD56, CD27 and mesenchymal stem cell antigen-1. Haematologica 94: 173-184.
-
Bühring HJ, Battula VL, Treml S, Schewe B, Kanz L, et al. (2007) Novel markers for the prospective isolation of human MSC. Ann N Y Acad Sci 1106: 262-271.
-
Muñiz C, Teodosio C Mayado A et al. (2015) Ex vivo identification and characterization of a population of CD13(high) CD105(+) CD45(-) mesenchymal stem cells in human bone marrow. Stem Cell Res Ther 6: 169.
-
Teixeira GQ, Barrias CC, Lourenço AH, Gonçalves RM (2014) A multicompartment holder for spinner flasks improves expansion and osteogenic differentiation of mesenchymal stem cells in three-dimensional scaffolds. Tissue Eng Part C Methods 20: 984-993.
-
Kisiday JD, Frisbie DD, McIlwraith CW, Grodzinsky AJ (2009) Dynamic compression stimulates proteoglycan synthesis by mesenchymal stem cells in the absence of chondrogenic cytokines. Tissue Eng Part A 15: 2817-2824.
-
Zhang P, Wu Y, Jiang Z, Jiang L, Fang B (2012) Osteogenic response of mesenchymal stem cells to continuous mechanical strain is dependent on ERK1/2-Runx2 signaling. Int J Mol Med 29: 1083-1089.





