International Journal of Stem cell Research and Therapy
CD271 Negative Human Dental Pulp Cells Yield Significantly More Adherent Colony Forming Cells than the Positive Phenotype
Matthew J Tomlinson1, Elena A Jones2, Peter V Giannoudis2,3, Xuebin B Yang1 and Jennifer Kirkham1*
1Division of Oral Biology, School of Dentistry, University of Leeds, Leeds, LS9 7TF, UK
2Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds, Leeds, LS9 7TF, UK
3Academic Department of Trauma and Orthopaedics, School of Medicine, University of Leeds, Leeds, LS1 3EX, UK
*Corresponding author: Jennifer Kirkham, Division of Oral Biology, School of Dentistry, University of Leeds, Leeds, LS9 7TF, UK, E-mail: j.kirkham@leeds.ac.uk
Int J Stem Cell Res Ther, IJSCRT-3-025, (Volume 3, Issue 1), Original Research; ISSN: 2469-570X
Received: November 27, 2015 | Accepted: February 05, 2016 | Published: February 08, 2016
Citation: Tomlinson MJ, Jones EA, Giannoudis PV, Yang XB, Kirkham J (2016) CD271 Negative Human Dental Pulp Cells Yield Significantly More Adherent Colony Forming Cells than the Positive Phenotype. Int J Stem Cell Res Ther 3:025. 10.23937/2469-570X/1410025
Copyright: © 2016 Tomlinson MJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Cell surface markers for isolating proliferative human dental pulp stromal cells are currently lacking. Other tissues containing mesenchymal stromal cells have been studied in greater depth and candidate markers for cell isolation identified, one such marker being CD271. Previous reports suggest CD271 as a marker for isolating dental pulp stromal cells from rat incisors. We aimed to study the utility of CD271 as a marker for isolating human dental pulp stromal cells. CD271 positive cells from both third molar dental pulp and bone marrow mononuclear cells were isolated by magnetic separation followed by in vitro expansion. Phenotypic analysis was performed by flow cytometry. Our data showed that although CD271 is present in the adult molar dental pulp, significantly greater numbers of colonies are produced from the CD271 negative fraction. The opposite was seen with bone marrow mononuclear cells; all colony forming cells were derived from the CD271 positive fraction. Phenotypic analysis of expanded CD271 negative cells showed that these cells are identical to dental pulp stromal cells isolated by non-selective plastic adherence. We conclude that, in contrast to bone marrow, CD271 is not a positive selection marker for the predominant colony forming cell types from human molar dental pulp.
Keywords
Dental pulp, CD271, Magnetic activated cell sorting, MACS, Mesenchymal stem cells, Flow cytometry
Introduction
Human dental pulp stromal cells (hDPSCs) are plastic adherent cells capable of in vitro colony formation, mineralisation and demonstrable mesenchymal differentiation potential [1,2]. They are of interest for tissue engineering and regenerative medicine applications, in particular dental and orthopaedic procedures, due to the potential for banking cells from the deciduous dentition [2,3]. Within the dental pulp (DP), hDPSCs are present in comparatively low numbers, but undergo expansion in vitro and are phenotypically similar to mesenchymal stromal cells (MSCs) from other tissues [4,5]. Ex vivo expansion is important because native hDPSC numbers are insufficient for regenerative therapies [6]. However, a potential problem with this is heterogeneity, a recognised issue with MSCs and one exacerbated by non-selective plastic adherence isolation [7]. Cell sorting can reduce heterogeneity by selecting specific cell populations but, to achieve this, cell surface markers for hDPSCs are required. MSCs from other tissues, such as bone marrow (BM), have been studied in greater depth and candidate markers for MSC enrichment have been identified such as CD146, CD105 and mesenchymal stem cell antigen 1 (MSCA-1) also known as tissue non-specific alkaline phosphatase (TNAP) [8-10]. One interesting marker is the neutrophin receptor CD271 which has shown specificity for colony forming cells from BM [11-13].
CD271 (also known as (L) NGFR, NTR and p75) is important for neuronal cell survival but is also found on MSCs from BM and both trabecular and cortical bone [1,14]. These studies selected multipotent MSCs expressing CD271 and showed standard MSC phenotype and trilineage differentiation [5]. Studies of developing human teeth have shown that prior to matrix production, CD271 expression maps to the inner enamel epithelial and the dental follicle [15]. Human exfoliated deciduous teeth show continued expression [16], as do mature DPs, with increased expression associated with dental caries [17,18]. CD271 has been identified within the human third molar apical papillae and studies with cultured hDPSCs described a subpopulation of cells with neurogenic potential that express CD271 [19,20]. Further studies with cultured hDPSCs have also shown that expression of CD271 may be induced if the cells are grown in non-adhesive culture vessels, allowing for the formation of spheroid aggregates [21]. This may be because the aggregates more accurately mimic the 3D environment from which the hDPSCs come compared to the 2D conditions which are used in standard cell culture.
Studies with rat DP have shown CD271 expression in epithelial, mesenchymal and nerve structures of developing and adult molar pulps, with expression in incisors mapping to polarising odontoblasts [22,23]. Further studies using rat incisor DP found that CD271 can be used to isolate proliferative DPSCs before expansion [24]. It is for these reasons that we have chosen to investigate CD271 as a marker for hDPSC isolation.
The aim of this study was to investigate CD271 expression by adult human DP and to subsequently determine its utility for hDPSC isolation. We hypothesised that hDPSCs would likely be isolated using CD271 given that it is an established marker for BMSCs, with evidence of utility in isolating rat DPSCs.
Materials and Methods
Cell isolation
Dental pulp: Healthy third molars (n = 14, 10 donors (3 male, 7 female), 18-33 years), obtained with written informed patient consent through the Leeds Dental Institute Research Tissue Bank (ethical approval for this study was granted by the Dental Research Ethics Committee of the University of Leeds, study number 07/H1306/93+5), were washed with 70% (v/v) ethanol before removal of attached tissue and fracturing. DPs were excised, mechanically disrupted and digested in 3 mg/mL collagenase I (Sigma-Aldrich, UK) and 4 mg/mL dispase (Roche, Germany) for 45 minutes at 37°C with agitation. Cell suspensions were filtered (70 μm) (BD Biosciences, NJ) and centrifuged at 200 × g for 5 minutes. Cells were cultured, sorted or analysed by flow cytometry. Cultured cells were resuspended in α-MEM (Life Technologies, UK) supplemented with 15% FCS (Biosera, UK), 2 mM L-glutamine (Sigma-Aldrich) and 100 units/mL penicillin/100 μg/mL streptomycin (Sigma-Aldrich) before incubation at 37°C in 5% CO2 for 10-14 days. Cells for sorting and flow cytometry were resuspended in 80 μL magnetic activated cell sorting (MACS) buffer (PBS, 2 mM EDTA (Alfa Aesar, UK) and 0.5% BSA (Sigma-Aldrich)).
Bone marrow: Primary posterior iliac crest BM (n = 4, 2 male, 2 female, 30-54 years), obtained with written informed consent following ethical approval for this study (NHS Health Research Authority, Leeds West Research Ethics Committee 06/Q1206/127) was diluted 1:1 with PBS, filtered at 70 μm and layered over 10 mL Biocoll Separating Solution (density 1.077 g/mL) (Biochrom, Germany). Cells were centrifuged at 445 × g for 35 minutes at 20°C. Mononuclear cells (MNCs) were aspirated and washed in MACS buffer before centrifugation at 200 × g for 10 minutes, twice. The resulting cell pellet was resuspended in 80 μL MACS buffer/1 mL of BM.
Cell culture
Subconfluent cells were passaged by treatment with 0.25% trypsin/0.02% EDTA (Sigma-Aldrich) and the resulting suspensions were transferred to T75 flasks, designated as passage 1 (p1). Passaged cells were subsequently sub-cultured in basal medium with 10% FCS until 90% confluency to p5.
CD271 sorting of dental pulp and bone marrow
Digested DP and isolated BM-MNCs, resuspended in 80 μL MACS buffer, were labelled with 10 μL anti-CD271-PE and 10 μL FcR blocking reagent (both Miltenyi Biotec, Germany) for 10 minutes at 4°C. Labelled cells were washed with 1 mL MACS buffer, centrifuged at 200 × g for 5 minutes and resuspended in 70 μL MACS buffer before incubation with 20 μL anti-PE microbeads (Miltenyi Biotec) and 10 μL FcR blocker at 4°C for 15 minutes. Cells were then washed, resuspended in 500 μL MACS buffer and added to a magnetised MS MACS column (1 column / DP or mL BM-MNCs), columns were then washed with 500 μL MACS buffer (x3). Eluted unlabelled cells were collected and designated the CD271- (negative) fraction. Labelled cells, retained on the column, were isolated by demagnetising the column and washing with 1 mL of MACS buffer, these cells were designated the CD271+ (positive) fraction. Separation efficiency was confirmed by the use of BM as a positive control. Depletion efficiency from DP was found to be in excess of 90%, i.e., more than 90% of CD271+ cells were removed from the CD271- fraction. All sorted cells were culture expanded in primary DP medium.
Colony forming unit fibroblast assay
Sorted cells from whole DP were plated into separate T25 flasks at the ratio of CD271+ and CD271- cells present within the DP (1 sorted DP or 1 mL sorted BM-MNC per flask) with medium changes every 4 days. After 14 days, flasks were fixed with 10% (v/v) neutral buffered formalin (Sigma-Aldrich) and stained with 1% (w/v) methyl violet (VWR, UK) for 30 minutes before washing and colony counting.
Flow Cytometry
Primary DP cells and cultured hDPSCs (unsorted and CD271-sorted) were resuspended in 80 μL MACS buffer and 10 μL FcR blocking buffer before antibody incubation (10 μL/1 × 106 cells) for 20 minutes at room temperature in the dark. MACS buffer (900 μL) was added before centrifugation and resuspension in 500 μL MACS buffer. Samples were analysed using a BD LSR Fortessa running BD Diva 7 software and data analysis was performed using Kaluza 1.3 software (Beckman Coulter, Pasadena, CA). Antibodies used were CD271-PE (mouse IgG1 κ monoclonal, clone ME20.4, WM245 melanoma cells as antigen), CD29-Alexa Fluor 488 (mouse IgG1 κ monoclonal, clone TS2/16), CD44-FITC (mouse IgG1 κ monoclonal, clone VI A034, normal human PBL as antigen), CD56-PE (mouse IgG2a κ monoclonal, clone VI NK26, KG-1 cell line as antigen), CD73-PE (mouse IgG1 κ monoclonal, clone AD2), CD90-APC (mouse IgG1 κ monoclonal, clone 5E10, HEL cells as antigen), CD105-FITC (mouse IgG1 κ monoclonal, clone 43A3, L-cells transfected with human CD105 as antigen), CD146-Alexa Fluor 488 (mouse IgG2a κ monoclonal, clone SHM-57, HEL cells as antigen) and CD166-PE (mouse IgG1 κ monoclonal, clone 3A6, cultured human thymic epithelial cells as antigen) (all Biolegend, San Diego, CA). Threshold values were obtained by measuring non-labelled cell autofluorescence and isotype matched control reactivity, colour compensation was performed to reduce channel crossover.
Statistical Analysis
All measurements were performed in at least triplicate and all graphs are displayed as the mean ± SEM. The data were subsequently analysed using a two sample t test. P values of less than 0.05 (*) were considered significant and 0.01 (**) very significant.
Results
CD271 expression in DP and hDPSCs
Analysis of uncultured DP by flow cytometry confirmed CD271+ cells were present at 1.24% ± 0.46 of total cells (Figure 1a). However, analysis of cultured hDPSCs, derived from plastic adherence, revealed that CD271 expression decreased to very low levels upon induction into 2 dimensional culture with an average expression of 0.2% across passages 2 to 4 (Figure 1b).
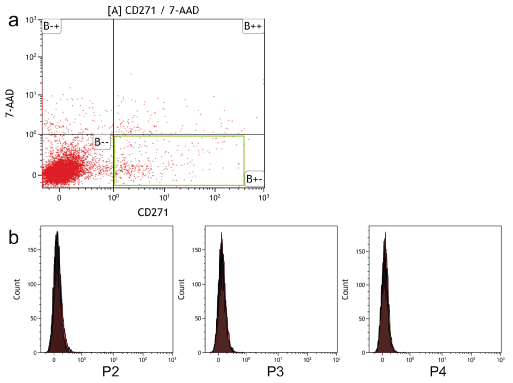
.
Figure 1: Expression of CD271 by primary DP cells. (a) Flow cytometry plot demonstrating low-level CD271 expression by a small population of cells from digested DP (quadrant B+- for viable CD271+ cells); (b) CD271 expression by adherent hDPSCs: Histograms of flow cytometric analyses of CD271 expression by cultured hDPSCs (red) and isotype controls (black) showing that this marker is not expressed by early (p2) to mid-passage (p4) cells.
View Figure 1
Sorting hDPSCs using the CD271 surface marker
Cells from whole digested DP were sorted for expression of CD271, isolated cells were used in colony formation unit fibroblast (CFU-F) assays, CD271+ and CD271- cells were plated at the ratio of cells found within the DP. The results showed that the mean number of colonies forming from the CD271+ fraction was 5 ± 2 per pulp compared to 55 ± 17 colonies per pulp from the CD271- fraction (Figure 2). These results show that significantly more colony forming cells were present in the CD271- fraction compared to the CD271+ cells (p = 0.027). Positive control experiments with BM showed the opposite relationship between CD271 expression and colony formation, with an average of 18 ± 5 colonies per mL of CD271+ sorted BM and 1 ± 1 colonies in the corresponding CD271- fraction, which was statistically significant (p = 0.039). These data are summarised in figure 2.
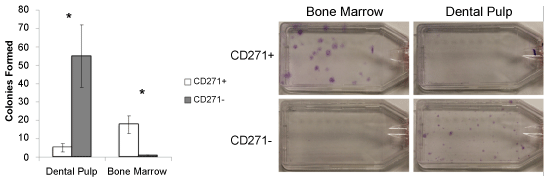
.
Figure 2: Averaged results of CFU-F assays performed with CD271+ and CD271- sorted digested DP compared with BM-MNCs isolated by density gradient centrifugation, white = CD271+ and dark grey = CD271-; photographs show representative colony counts for each condition, * = p < 0.05, n=10 for dental pulp and n = 4 for bone marrow, data shown ± SEM.
View Figure 2
Having shown the relative colony forming capacities of CD271+ and CD271- DP cells, we next determined whether cells derived from the CD271- fraction were phenotypically similar to cells expanded from unsorted DP. To achieve this hDPSCs from both unsorted and CD271- sorted DP were compared for the expression of various hDPSC and MSC markers [5,25]. The results of these analyses failed to show any significant differences in the expression patterns of the markers analysed, CD29 (p = 0.43), CD44 (p = 0.38), CD56 (p = 0.9), CD73 (p = 0.29), CD90 (p = 0.15), CD105 (p = 0.34), CD146 (p = 0.57) and CD166 (p = 0.21) (Table 1). These data showed that based upon the markers examined, the CD271- cells derived from primary DP are phenotypically identical to hDPSCs isolated by plastic adherence.
![]()
Table 1: Comparative flow cytometry data showing average percentage expression of hDPSC markers expressed by expanded hDPSCs from both unsorted and CD271- cultures. No statistically significant differences were observed in the marker expression profiles, n = 3.
View Table 1
Discussion
We have studied the presence of the human 'in vivo' BMSC marker CD271 by adult human DP and in vitro expanded hDPSCs. We showed that CD271 is weakly expressed in DP and cultured hDPSCs show no upregulation in expression. These results confirm previous studies whereby CD271 was found within adult human DP and also showed similar in vitro expression to BM [12,18].
Following confirmation of CD271 expression in adult DP, and given that CD271 has been used to isolate BMSCs [11,13,26], we attempted to use it to selectively isolate hDPSCs. These studies showed that while CD271 was expressed in DP, the CD271+ fraction contained significantly fewer colony forming cells than the CD271- fraction (p = 0.027). To validate these findings, we utilised the same experimental regimen to isolate BMSCs from BM-MNCs. The results of these experiments were in stark contrast to those for DP with significantly more colony forming cells found in the CD271+ fraction (p = 0.039) and are consistent with previously published BM data [11,13,14,26]. These previous studies also showed that collagenase digestion does not impair CD271 based selection [11,14].
Finally, we compared the expression of a range of common hDPSC markers by CD271- hDPSCs and unsorted hDPSCs, isolated by plastic adherence. These results showed near identical expression profiles, confirming that CD271- derived cells are the same as those isolated by plastic adherence and are in keeping with previously published data [27-29]. We can therefore conclude that cells from the CD271- fraction are indeed the cells commonly described as hDPSCs. This does not preclude the possibility that CD271+ cells also form colonies with similar profiles, but in smaller numbers. In this respect, it is important to mention that previous reports have shown that multiple proliferative cell populations exist within the human DP, including Oct-4 and Nanog expressing immature DP stem cells [24,30,31]. Recent reports have shown that cultured hDPSCs contain some cells of neural crest origin and it has been proposed in rats that CD271 is a marker for neural crest derived populations [24,32]. From our data, it is possible to say that CD271 identifies a small subpopulation of cells within dental pulp and these may be the neural crest derived cells. However, in our experimental conditions, selection based on its expression produces significantly fewer colonies than the CD271- fraction.
It is interesting to note that previous studies have identified CD271 in the apical papilla as well as the continuously erupting rat incisor [19,24]. It can therefore be postulated that CD271 may mark for early stage proliferating DP cells, and potentially prevents differentiation in deciduous teeth, but upon tissue maturation its expression is lost [16].
Our data also provide further evidence for differences in stromal cell biology between hDPSCs and BMSCs. These cell types are ostensibly similar with many commonalities [25,33]; however it is clear from our data that a key 'in vivo' marker of BMSCs -CD271- is lacking in the majority, if not all of colony forming hDPSCs. The rationale for this finding is likely related to the developmental origin of these tissues, with DP being a neural crest ectomesenchymal derivative and BM originating from, in the most part, the mesodermal germ layer [34-36]. It is therefore likely that despite the many phenotypic similarities following culture, the differences in embryonic origin mean that each cell type is likely to have fundamental differences in terms of 'in vivo' marker expression and inherent differentiation potentials [33]. Indeed this difference in differentiation potential is well known with hDPSCs producing globular dentin following in vivo transplantation whereas BMSCs produce lamellar bone [1]. We also note from our data that colonies formed from DP appear smaller in size than those from BM.
Conclusion
We conclude that CD271 is expressed within adult DP but that a significant majority of colony forming DP cells do not show CD271 expression. Following in vitro expansion these cells possess sufficiently similar expression profiles to conventionally isolated hDPSCs so as to be indistinguishable. CD271+ cells showed limited colony forming potential and may be a distinct neural crest subpopulation within the DP. Overall, we conclude that CD271 is not a specific marker for hDPSCs and that it should not be used to isolate these cells from mature DP.
Acknowledgement
This work was supported by WELMEC, a Centre of Excellence in Medical Engineering funded by the Wellcome Trust and EPSRC, under grant number WT 088908/Z/09/Z and NIHR through the Leeds Musculoskeletal Biomedical Research Unit.
References
-
Gronthos S, Brahim J, Li W, Fisher LW, Cherman N, et al. (2002) Stem cell properties of human dental pulp stem cells. J Dent Res 81: 531-535.
-
Zhang W, Walboomers XF, Shi S, Fan M, Jansen JA (2006) Multilineage differentiation potential of stem cells derived from human dental pulp after cryopreservation. Tissue Eng 12: 2813-2823.
-
Krebsbach PH, Robey PG (2002) Dental and skeletal stem cells: potential cellular therapeutics for craniofacial regeneration. J Dent Educ 66: 766-773.
-
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci USA 97: 13625-13630.
-
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini FC, et al. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8: 315-317.
-
Guillot PV, Cui W, Fisk NM, Polak DJ (2007) Stem cell differentiation and expansion for clinical applications of tissue engineering. J Cell Mol Med 11: 935-944.
-
Ho AD, Wagner W, Franke W (2008) Heterogeneity of mesenchymal stromal cell preparations. Cytotherapy 10: 320-330.
-
Battula VL, Treml S, Bareiss PM, Gieseke F, Roelofs H, et al. (2009) Isolation of functionally distinct mesenchymal stem cell subsets using antibodies against CD56, CD27, and mesenchymal stem cell antigen-1. Haematologica 94: 173-184.
-
Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, et al. (2008) A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3: 301-313.
-
Majumdar MK, Banks V, Peluso DP, Morris EA (2000) Isolation, characterization, and chondrogenic potential of human bone marrow-derived multipotential stromal cells. J Cell Physiol 185: 98-106.
-
Jones E, English A, Churchman SM, Kouroupis D, Boxall SA, et al. (2010) Large-scale extraction and characterization of CD271+ multipotential stromal cells from trabecular bone in health and osteoarthritis: implications for bone regeneration strategies based on uncultured or minimally cultured multipotential stromal cells. Arthritis Rheum 62: 1944-1954.
-
Jones EA, Kinsey SE, English A, Jones RA, Straszynski L, et al. (2002) Isolation and characterization of bone marrow multipotential mesenchymal progenitor cells. Arthritis Rheum 46: 3349-3360.
-
Quirici N, Soligo D, Bossolasco P, Servida F, Lumini C, et al. (2002) Isolation of bone marrow mesenchymal stem cells by anti-nerve growth factor receptor antibodies. Exp Hematol 30: 783-791.
-
Cox G, Boxall SA, Giannoudis PV, Buckley CT, Roshdy T, et al. (2012) High abundance of CD271(+) multipotential stromal cells (MSCs) in intramedullary cavities of long bones. Bone 50: 510-517.
-
Becktor KB, Hansen BF, Nolting D, Kjaer I (2002) Spatiotemporal expression of NGFR during pre-natal human tooth development. Orthod Craniofac Res 5: 85-89.
-
Mikami Y, Ishii Y, Watanabe N, Shirakawa T, Suzuki S, et al. (2011) CD271/p75(NTR) inhibits the differentiation of mesenchymal stem cells into osteogenic, adipogenic, chondrogenic, and myogenic lineages. Stem Cells Dev 20: 901-913.
-
Sakurai K, Okiji T, Suda H (1999) Co-increase of nerve fibers and HLA-DR- and/or factor-XIIIa-expressing dendritic cells in dentinal caries-affected regions of the human dental pulp: an immunohistochemical study. J Dent Res 78: 1596-1608.
-
Maeda T, Sato O, Iwanaga T, Takano Y (1992) Immunocytochemical localization of nerve growth factor receptor (NGFR) in human teeth. Proc Finn Dent Soc 88 Suppl 1: 557-562.
-
Abe S, Hamada K, Miura M, Yamaguchi S (2012) Neural crest stem cell property of apical pulp cells derived from human developing tooth. Cell Biol Int 36: 927-936.
-
Martens W, Wolfs E, Struys T, Politis C, Bronckaers A, et al. (2012) Expression pattern of basal markers in human dental pulp stem cells and tissue. Cells Tissues Organs 196: 490-500.
-
Xiao L, Tsutsui T (2013) Characterization of human dental pulp cells-derived spheroids in serum-free medium: stem cells in the core. J Cell Biochem 114: 2624-2636.
-
Byers MR, Schatteman GC, Bothwell M (1990) Multiple functions for NGF receptor in developing, aging and injured rat teeth are suggested by epithelial, mesenchymal and neural immunoreactivity. Development 109: 461-471.
-
Mitsiadis TA, Couble P, Dicou E, Rudkin BB, Magloire H (1993) Patterns of nerve growth factor (NGF), proNGF, and p75 NGF receptor expression in the rat incisor: comparison with expression in the molar. Differentiation 54: 161-175.
-
Waddington RJ, Youde SJ, Lee CP, Sloan AJ (2009) Isolation of distinct progenitor stem cell populations from dental pulp. Cells Tissues Organs 189: 268-274.
-
Huang AH, Chen YK, Chan AW, Shieh TY, Lin LM (2009) Isolation and characterization of human dental pulp stem/stromal cells from nonextracted crown-fractured teeth requiring root canal therapy. J Endod 35: 673-681.
-
Tormin A, Li O, Brune JC, Walsh S, Schütz B, et al. (2011) CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood 117: 5067-5077.
-
Kitraki E, Zakkas S, Synolaki E, Diamanti E, Tiniakos DG, et al. (2014) Dental pulp cells enhance bone healing in a rat osteotomy model. Ann Orthop Rheumatol 2: 1009.
-
Tomlinson MJ, Dennis C, Yang XB, Kirkham J (2015) Tissue non-specific alkaline phosphatase production by human dental pulp stromal cells is enhanced by high density cell culture. Cell Tissue Res 361: 529-540.
-
Alongi DJ, Yamaza T, Song Y, Fouad AF, Romberg EE, et al. (2010) Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential. Regenerative Medicine 5: 617-631.
-
Paino F, Ricci G, De Rosa A, D'Aquino R, Laino L, et al. (2010) Ecto-mesenchymal stem cells from dental pulp are committed to differentiate into active melanocytes. Eur Cell Mater 20: 295-305.
-
Kerkis I, Kerkis A, Dozortsev D, Stukart-Parsons GC, Gomes Massironi SM, et al. (2006) Isolation and characterization of a population of immature dental pulp stem cells expressing OCT-4 and other embryonic stem cell markers. Cells Tissues Organs 184: 105-16.
-
Pisciotta A, Carnevale G, Meloni S, Riccio M, De Biasi S, et al. (2015) Human dental pulp stem cells (hDPSCs): isolation, enrichment and comparative differentiation of two sub-populations. BMC Dev Biol 15: 14.
-
Giannoudis P, Jones E, Yang X, Mcgonagle D (2013) Mesenchymal Stem Cells and Skeletal Regeneration. Waltham, MA: Academic Press.
-
Blentic A, Tandon P, Payton S, Walshe J, Carney T, et al. (2008) The emergence of ectomesenchyme. Dev Dyn 237: 592-601.
-
Chai Y, Jiang X, Ito Y, Bringas P Jr, Han J, et al. (2000) Fate of the mammalian cranial neural crest during tooth and mandibular morphogenesis. Development 127: 1671-1679.
-
Komada Y, Yamane T, Kadota D, Isono K, Takakura N, et al. (2012) Origins and properties of dental, thymic, and bone marrow mesenchymal cells and their stem cells. PLoS One 7: e46436.





