International Journal of Stem cell Research and Therapy
Generation of Antigen-Presenting Cells from Human Vascular Adventitia-Resident Progenitors
Mohamad Almoussa1,2, Volha Stoll1, Diana Klein3 and Süleyman Ergün1*
1Institute of Anatomy and Cell Biology, Julius-Maximilians-University Würzburg, Würzburg, Germany
2Institute of Anatomy, University Hospital Essen, University of Duisburg-Essen, Essen, Germany
3Institute of Cell Biology (Cancer Research), University Hospital Essen, University of Duisburg-Essen, Essen, Germany
*Corresponding author: Prof. Dr. Süleyman Ergün, Institute of Anatomy and Cell Biology, Julius-Maximilians-University Würzburg, Koellikerstrasse 6, 97070 Würzburg, Germany, Fax: 0931-31 82712, Tel: 0931-31 82707, E-mail: sueleyman.erguen@uni-wuerzburg.de
Int J Stem Cell Res Ther, IJSCRT-3-040, (Volume 3, Issue 2), Original Article; ISSN: 2469-570X
Received: June 05, 2016 | Accepted: August 09, 2016 | Published: August 13, 2016
Citation: Almoussa M, Stoll V, Klein D, Ergün S (2016) Generation of Antigen-Presenting Cells from Human Vascular Adventitia-Resident Progenitors. Int J Stem Cell Res Ther 3:040. 10.23937/2469-570X/1410040
Copyright: © 2016 Almoussa M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Macrophages and dendritic cells play a central role in the immune system. Here, we identify CD44(+) vascular wall-resident multipotent stem cells (VW-MPSCs) as progenitors for macrophages and dendritic cell-like cells. Using human internal thoracic artery (hITA) tissue specimens we performed ex vivo sprouting assays followed by immunohistochemistry. hITA rings and hITA wall-derived cells were treated with GM-CSF+IL-4. The cells were then characterized by immunostaining and flow cytometry for CD31, CD34, CD44, CD45, CD68 and CD80. While neither CD68(+) nor CD80(+) cells were found in freshly isolated hITA sections, they were present after sprouting assay. Double immunofluorescence analysis after sprouting assay revealed the presence of both CD45(+)CD68(+) and CD44(+)CD68(+) macrophages as well as CD44(+)CD80(+) dendritic cell-like cells within the adventitia and among the sprouting cells. Cultivation of total hITA wall-derived cells for 7-10 days resulted in enhanced accumulation of CD44(+) cells while CD34 and CD45 cells were not detectable regardless of the media used such as ECGM, RPMI or IMDM, respectively. Remarkably, a similar immunophenotype was maintained when the cells were cultured up to eight weeks. Only cultivation of the total hITA wall-derived cells in a high confluent state resulted in the in vitro generation of CD68(+) macrophages and CD80(+) dendritic cell-like cells as detected by FACS. The stimulation of total hITA wall-derived cells with GM-CSF+IL-4 did not change the number of CD68(+) and CD80(+) cells significantly. Taken together, our results show the differentiation of CD44(+) VW-MPSCs into CD44(+)CD68(+) macrophages and CD44(+)CD80(+) dendritic cell-like phenotypes. Thus, vascular adventitia might serve as a local source for the generation of antigen-presenting cells.
Keywords
Macrophages, Dendritic cells, Vascular wall, Stem/Progenitor cells
Introduction
Identification and characterization of endogenous sources of stem and progenitor cells is of essential relevance for regenerative medicine. Enormous progress in this field was made in the past 15 years. Several tissue niches containing different types of adult stem and progenitor cells were identified and the mechanisms regulating their differentiation into mature cells were partially characterized [1]. The wall of adult veins and arteries, basically the adventitial layer and the sub-endothelial zone have been identified to harbor stem cells [2,3]. Vascular adventitial cells were reported to contribute to vascular stenosis [4]. Within the vascular adventitia progenitor and stem cells have been shown to be predominently localized within the so-called "vasculogenic zone" which has been described as a circular zone close to the smooth muscle layer of the human adult vascular wall [5]. This niche was confirmed by further very recent studies [6-9] showing the presence of progenitors for endothelial and hematopoietic cells, smooth muscle cells and pericytes as well as inflammatory cells [9-12] in this zone. The two major cell populations residing within the adventitial vasculogenic zone are CD34(+) and CD44(+) cells. Until now, CD34(+) cells were characterized to differentiate into endothelial cells [5] and pericytes [13] and CD44(+) cells were shown to differentiate into smooth muscle cells and pericytes [7,10]. Furthermore, Sca1(+) cells differentiating into smooth muscle cells [9] and c-kit(+) cells were also found in vascular adventitia [9,14]. CD44 is a cell-surface glycoprotein that functions as a receptor for hyaluronic acid (HA) and interacts with other ligands such as osteopontin, collagens, and matrix metalloproteinases (MMPs). Thereby it plays an essential role in cell adhesion and migration and it is highly expressed on mesenchymal stem cells (MSCs). Moreover, CD44 is upregulated on effector T cells, facilitates their extravasation from the blood and their mobilization at sites of infection and inflammation [15]. CD44 is also expressed by different cancer cells and it enhances tumor aggressiveness and metastasis [16]. However, the CD44(+) stem cell type identified in the adventitia of human internal thoracic artery (hITA) was characterized by the additional expression of CD90(+) CD73(+) while being negative for the hematopoietic lineage markers CD34 and CD45 [17]. These cells were shown to be multipotent, they exhibited typical mesenchymal character and were named as vascular wall-resident multipotent stem cells (VW-MPSCs). VW-MPSCs contribute to new vessel maturation by differentiating into pericytes and smooth muscle cells which were shown to cover the endothelial layer of the newly formed capillary structures in vitro and in vivo [10,17].
Remarkably, macrophages, a specific mature cell type of the innate immune system, was found to be generated from adventitial progenitors [5], which still have not been exactly identified yet. While it was widely believed that tissue macrophages are derived from circulating monocytes, data obtained from mouse models with experimental bone marrow (BM) depletion or chimeras as well as using genetic approaches showed undoubtedly that mature macrophages can be generated from progenitors resident in the vascular adventitia independently from BM [5,12,18,19]. More recently, yolk-sac-derived erythro-myeloid progenitors rather than BM-derived cells were identified to deliver tissue-resident macrophages [20]. These findings together strongly indicate that only a small fraction of tissue-resident macrophages is of BM origin. The majority of them is likely dependent on local stem and/or progenitor cells, e.g. vascular adventitia-resident stem cells. Until now, it is unclear which vascular stem and/or progenitor cell type(s) might give rise to vascular adventitia-derived antigen-presenting cells.
We suspected that the CD44(+) VW-MPSCs might be a candidate for generating vascular wall-derived macrophages and for other antigen-presenting cells like dendritic cells. Our current data show that while neither CD68(+) nor CD80(+) cells were found in the adventitia of freshly isolated hITA, they were generated in a high number after performing sprouting assay. Interestingly, both CD45(+)CD68(+) and CD44(+)CD68(+) cells could be generated from the hITA adventitia after sprouting assay. The majority of CD80(+) dendritic cell-like cells generated from the hITA adventitia after sprouting assay was double positive for CD44(+)CD80(+). Such cells were found within the activated adventitia as well as within the collagen gel outside the hITA rings. Conformingly, hITA wall cells extracted after dispersion and cultured for 7-10 days differentiated into CD68(+) and CD80(+) cells when they were cultured in a highly confluent status. A considerable part of these cells was also positive for CD44. Stimulation of hITA-rings and hITA wall-derived cells with GM-CSF+IL-4 showed no significant effect on the generation of CD44(+)CD68(+) macrophages and CD44(+)CD80(+) dendritic cell-like cells. Our results suggest that CD44(+)VW-MPSCs serve as a source for both adult vessel wall-derived macrophages and dendritic cell-like cells.
Materials and Methods
Reagents and antibodies
Mouse anti-human CD45 (clone MEM-28, final dilution 1:50) was purchased from Immuno Tools (Germany) and CD80 (clone EP1155Y, final dilution1:100) from Abcam.
CD31 (cloneJC70A, final dilution 1:25), CD34 (Clone QBEnd-10, final dilution 1:50) and CD68 (clone KP1, final dilution 1:50) antibodies were purchased from Dako Denmark A/S (Hamburg, Germany), CD90 antibody (clone Thy-1, 0.5 μg per test in FACS) was provided by eBioscience (CA, USA) and the rat anti-human/mouse CD44 antibody (clone A020, final dilution 1:100) was purchased from Merck Chemicals Ltd. (United Kingdom). Human rGM-CSF and human rIL-4 were obtained from Immuno Tools (Germany). Endothelial cell growth medium MV (ready-to-use) and endothelial basal medium were purchased from Promocell (Heidelberg, Germany), monocyte-dendritic cell differentiation medium (ready-to-use) and MACS system were provided by Miltenyi Biotec GmBH (Bergisch Gladbach, Germany). RPMI, PBS, penicillin, streptomycin, fetal calf serum and DMEM (10x) were purchased from Gibco BRL (Karlsruhe, Germany). Paraformaldehyde was obtained from Sigma (Taufkirchen Germany). Purecol for hITA ring assay was obtained from Biomatrix (CA, USA). All peroxidase- and fluorescently-labelled secondary antibodies were provided by Dako Cytomation Denmark A/S (Denmark) and Dianova (Hamburg, Germany).
Tissue material
Human internal thoracic artery (hITA) samples were obtained during surgery (sparse material) according to local ethical and biohazard regulations and provided from the Clinics of Thoracic and Cardiovascular Surgery, University Hospital Essen for our institute. All experiments were approved by the local ethics committee. Written informed consent (Nr.10-4363) was obtained from the Ethikkommission of the University Medical Faculty, Essen, Germany.
Dispersion cell culture from hITA
The hITA samples were excised under a dissection microscope to remove fat and muscle tissue surrounding the hITA wall. After several washes in PBS, vessels were mechanically minced and enzymatically dissociated into a single cell suspension with 0.2% type II-collagenase (Worthington, Lakewood, USA) and 5 U/ml elastase (Sigma) at 37 °C for 30 minutes. After two washes in PBS containing 5% fetal calf serum (FCS), cellular suspensions were passed through 70 μm pore size filters. After centrifugation at 1000 rpm for 10 min, cells were cultured in endothelial cell growth medium MV (ECGM MV) supplemented with 5% FCS, 100 U/ml penicillin and 100 mg/ml streptomycin at 37°C supplied with 5% CO2. Medium was changed after 24 h of initial plating and then every second day.
Purification of CD44 (+) cells via MACS technology
The MACS system (Miltenyi Biotec) was used according to the manufacturer's instructions and as already described [21]. Briefly, a single cell suspension of hITA cells was prepared as described above. The cell number was determined. After centrifugation at 300 g for 10 min, cells were washed and resuspended in binding buffer containing phosphate-buffered saline (PBS) (pH 7.2), 0.5% BSA, and 2 mM ethylene diamine tetraacetic acid (EDTA) and incubated with mouse anti-human CD44 immunoglobulin G microbeads at 4°C for 15-20 min. The cell suspension was loaded onto a MACS column and placed into a magnetic field. The column was rinsed with buffer and negative cells passed through. Labeled CD44 positive cells were eluted after removing the column from the magnet. To increase purity, the CD44 positive fraction was reloaded onto a new column. The column was rinsed with buffer; the positive fraction was eluted as described above.
Ex vivo ring assay using hITA
The arterial ring or sprouting assay is an ex vivo model used basically to study angiogenesis. In this model the small organ- in our case hITA-specimens will be cultured within matrigel or collagen gel as also used in our studies. The benefit of this assay is that the intact vascular wall is cultured and thus the vascular wall cells can be studied within their own in situ microenvironment. It allows the analysis of mobilization, migration and proliferation of vascular wall cells including VW-derived stem cells as well as their morphogenesis within the collagen and differentiation into different cell types. After finishing the assay it allows to prepare and use sections from the arterial rings or tissue pieces for immunohistochemistry in order to characterize the immunophenotypic differentiation of the VW-SCs. Furthermore, the effect of selective cytokines and growth factors on VW-SC differentiation can be studied by adding them directly to the rings as we did for GMCF and IL-4 [22].
Fresh hITA specimens were dissected and transferred directly into sterile PBS on ice. Then, fatty tissue surrounding the vessel wall from the outside was removed carefully under a dissection microscope with fine forceps and scissors with paying special attention not to damage the vascular wall, particularly the vascular adventitia. The cleaned hITAs were cut into 1 mm thick rings using a surgical scalpel. Meanwhile, the collagen gel was prepared by mixing sodium hydroxide (PH 7.4-7.5), glutamine, sodium bicarbonate, sodium pyruvate, vitrogen-gel (Purecol), MEM (10x), antibiotics (penicillin/streptomycin) and sterile water. All these materials were kept on ice during the preparation of the collagen gel. A first layer of collagen gel (150 μl) was plated per well, using 48-well cluser tissue culture plates, and incubated at 37°C in a humidified atmosphere with 5% CO2 for 30 min. Afterwards the rings were placed into the first layer of collagen gel and reincubated in a 37°C, 5% CO2 humidified incubator for 10 min. After addition of the second layer of collagen gel (150 μL), the rings were reincubated for one hour, before they were seeded with 300 mL EPCs media containing only 5% FCS. Embedded rings were incubated at 37°C, 5% CO2. The medium was changed every third day. Rings were evaluated every day using a phase contrast microscope (Zeiss, Jena, Germany) equipped with a digital camera (Zeiss, Jena Germany). After nine days in culture, rings were fixed in 4% paraformaldehyde (PFA), dehydrated and embedded in paraffin wax for immunohistochemistry or immunofluorescence.
Immunohistochemistry and immunofluorescence
Immunohistochemical staining for CD31, CD34, CD44, CD45, CD68 and CD80 (final dilutions mentioned above) was performed on paraffin sections obtained from five different freshly isolated hITA samples and five independent hITA ring assay cultures. Paraffin embedded tissue sections were hydrated using a descending alcohol series, incubated for 10-20 min in target retrieval solution (Dako) and then treated with blocking solution. After permeabilization, sections were incubated with primary antibodies at 4°C overnight. Antigen was detected by exposing the sections to the corresponding biotinylated secondary antibody at appropriate dilutions (1:250) followed by development of DAB staining (Dako). Specimens were analyzed by phase contrast microscopy. For immunofluorescence double staining on paraffin sections, secondary antibodies corresponding to the host of the primary antibodies were used at 1:400 dilution. DAPI was used to visualize the nuclei.
Immunocytochemistry
Cells were placed on chamber slides (BD Bioscience) prior to fixation with 4% PFA, washed in PBS for 10 min and incubated with blocking solution (2% NGS/PBS) for 30 min. After permeabilization cells were incubated with mouse anti-CD31, -CD34, -CD45, -CD90, -CD68 or rat anti-CD44, respectively at 4°C overnight. Subsequently, fluorescently-labelled secondary antibodies were added. Specimens were analyzed by confocal microscopy.
Culture of hITA rings for sprouting assay with and without stimulation by GM-SCF +IL-4
The hITA rings were cultured in endothelial basal medium with and without adding GM-CSF (50 ng/mL) + IL-4 (1000 IU/mL) at 37°C and 5% CO2. The media was changed every third day and the culture of hITA rings was stopped after nine days. Controls were performed without addition of cytokines. After stopping the sprouting on culture day nine, the rings were fixed in 4% PFA, dehydrated and embedded in paraffin wax for immunohistochemistry and immunofluorescence analysis.
Treatment of hITA wall-derived cells with GM-SCF +IL-4 in vitro
After dispersion of hITA pieces cells were isolated and cultured either in endothelial growth medium as it was used in hITA sprouting assays above or in RPMI-1640 media. After reaching a sufficient cell number to perform FACS analysis usually after 7-10 days in culture, the cells were cultured in monocyte-dendritic cell differentiation medium (Miltenyi BiotecInc, USA) and in RPMI-1640 media without cytokines as a negative control for 10 days. The medium was replaced every second day. The cells were characterized by FACS before and after stimulation.
Flow cytometry
For FACS analysis cells were detached from the plates by incubation at 37°C with 0.05% EDTA-Trypsin, washed and fixed (4% PFA) on ice for 30 min, then washed and re-suspended in 100 μl of FACS buffer (PBS with 3% FCS) containing diluted primary antibodies or corresponding isotype controls on ice for one hour. The cells were then washed three times with FACS buffer and incubated for 30 min with 100 μl of secondary fluorescein isothiocynate (FITC) conjugated antibody diluted 1:50 in FACS buffer. Afterwards, cells were washed and re-suspended in 200 μl of PBS containing 200 μg/mL propidium iodide to exclude the apoptotic cells. For the detection of intracellular antigens the cells were fixed and permeabilized using BD Cytofix/Cytoperm™ Fixation/Permeabilization Kit (BD biosciences, USA) for 20 min on ice as instructed by the manufacturer. After washing twice with 1×BD Perm/Wash™ buffer, cells were blocked on ice for 30 min with normal serum to avoid non-specific staining. Finally, stained cells were measured with a FACS Calibur instrument (Becton Dickinson, USA) and data were analyzed by using Cell Quest software (Becton Dickinson, USA).
Results
Histological evaluation of freshly isolated adult hITA prior to ring assay
First, we evaluated the presence of macrophages and dendritic cells within the freshly prepared hITA wall. Immunohistochemistry for CD68 as macrophage marker and for CD80 as a maker for dendritic cells resulted in completely negative staining. Only occasionally one to two single cells marked for CD68 were found (Figure 1, first column). Mature endothelial cells lining the hITA lumen were marked by immunostaining for CD31. To visualize the adventitial vasculogenic zone we performed immunostaining for CD34 and CD44. Finally, hITA sections were also stained for CD45 as a marker for pan-hematopoietic cells. As a control, hITA sections were exposed to corresponding IgG isotype only (not shown). Confirming our previously published data [5,17] immunostaining showed CD31 expression only in the endothelial layer (tunica intima) and CD34(+) cells were identified in both, tunica intima and adventitia with a particular accumulation at the border between the media and adventitia, within the so called "vasculogenic zone" (Figure 1, first column). To a lesser extent, also CD44(+) and single CD45(+) cells were detected within the adventitia (Figure 1, first column).
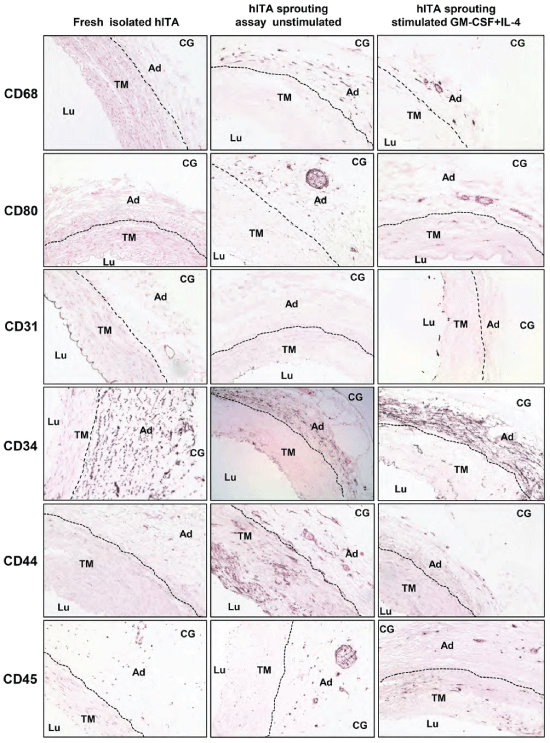
.
Figure 1: Immunohistochemical characterization of adult hITA.
Sections from all hITA groups (first column: fresh isolated hITA, second column: unstimulated hITA ring assay, third column: stimulated hITA ring assay with GM-CSF+IL-4) were stained with DAB (3,3′-Diaminobenzidine) for indicated markers as described in Materials and Methods. Lu: lumen, TM: tunica media, Ad: adventitia, CG: collagen gel. Original magnifications, ×20.
View Figure 1
Generation of macrophages and dendritic cell-like cells using hITA sprouting assay
To determine which one of the aforementioned cell types has the capacity to differentiate into antigen-presenting immune cells like macrophages and dendritic cells we performed arterial sprouting assay. We used collagen type I gel as a matrix in which small pieces of hITA rings of 1 mm thickness were embedded and cultured up to nine days. Then, the rings and the collagen gel were removed, embedded in paraffin and used for production of tissue sections. Immunostaining on such sections revealed a high number of CD68(+) cells and a considerable number of CD80(+) cells (Figure 1, second column) while no staining for these markers was detectable in hITA sections prior to the sprouting assay (Figure 1, first column). No significant change of the CD31 staining intensity was observed, but there were many intimal gaps lacking CD31 immunostaining indicating endothelial cell detachment from the intima due to their activation during the sprouting assay (Figure 1, second column). In comparison to freshly isolated hITA the number of CD34(+) cells within the hITA adventitia was reduced after sprouting assay. Particularly in the outer adventitial zone (Figure 1, second column) either only few CD34(+) cells were present or they disappeared completely (Figure 1, second column). This is probably due to both a) their migration into the collagen gel and b) down-regulation of CD34 after leaving the adventitial niche. In contrast, a remarkable increase of CD44(+) cells within the hITA adventitia was reproducibly detected after applying the sprouting assay (Figure 1, second column). These findings are in line with the previously published findings [17]. Furthermore, a little but not significant increase of CD45(+) cells in the hITA adventitia was also observed (Figure 1, second column) as it was reported previously [5]. Interestingly, besides a high number of CD68(+) cells also a considerable number of CD80(+) cells was seen while no staining for these markers was detectable in hITA sections prior to the ring assay (Figure 1, second column).
Influence of GM-CSF+IL-4 on hITA wall-derived macrophages and dendritic cell-like cells
Next, we wanted to test whether the number of CD68(+) macrophages and CD80(+) dendritic cell-like cells can be increased via treatment of the hITA rings with GM-CSF+IL-4. It has been reported that stimulation of blood monocytes with GM-CSF or GM-CSF+IL-4 in vitro leads to their differentiation into macrophages and dendritic cells, respectively [23,24].
To this end, the hITA rings were divided into two groups and cultured in the presence or absence of GM-CSF+IL-4 as described in Material and Methods. Surprisingly, immunohistochemical analysis on hITA ring sections after nine days in culture demonstrated a decrease in the number of CD68(+) cells within the adventitia of hITA rings stimulated with GM-CSF+IL-4 (Figure 1, third column) in comparison to unstimulated sprouting assays. The effect of GM-CSF+IL-4 stimulation on CD80(+) cells was more prominent as these cells either disappeared completely or were present only as few single cells (Figure 1, third column). The number of CD44(+) cells was decreased in the intima and media with no considerable change of their number in the adventitia (Figure 1, third column). No significant change was detected in staining pattern and intensity of CD31, CD34 and CD45 (Figure 1, third column) in comparison with untreated hITA sprouting assays (Figure 1, second column).
CD44(+)CD68(+) and CD45(+)CD68(+) hITA-derived cells after sprouting assay
Recently, the multipotent CD44(+)hITA wall-resident cells named as VW-MPSCs were shown to differentiate into smooth muscle cells and pericytes after in vitro expansion [10,17]. In line with these findings our data presented above showed a strong expansion of CD44(+) cells after sprouting assay. Thus, we hypothesized that CD44(+) VW-MPSCs can be one of the potential local progenitors giving rise to the generation of CD68(+) macrophages and CD80(+) dendritic cell-like cells from the hITA wall. Double immunofluorescence analyses performed on paraffin sections of hITA sprouting assays revealed a clear co-localization of CD68 and CD45 staining (Figure 2, first column). In-depth analyses showed that CD45(+)CD68(+) cells are also present in the adventitial vasa vasorum and within the collagen gel (Figure 2, first column). Similar expression patterns were found for CD68(+)CD44(+) cells (Figure 2, second column). Remarkably, cells exhibiting CD80 staining were found to be also positive for CD44 after hITA sprouting assay (Figure 2, third column). Both CD44(+)CD68(+) and CD44(+)CD80(+) cells have been mostly identified within the adventitia and the wall of vasa vasorum as well as within the collagen gel outside the rings (Figure 2). Moreover, the cells expressing the co-stimulatory molecule CD80 were found to be more roundly shaped and to be of bigger size than CD68(+) cells which were smaller and exhibited a spindle shape (not shown).
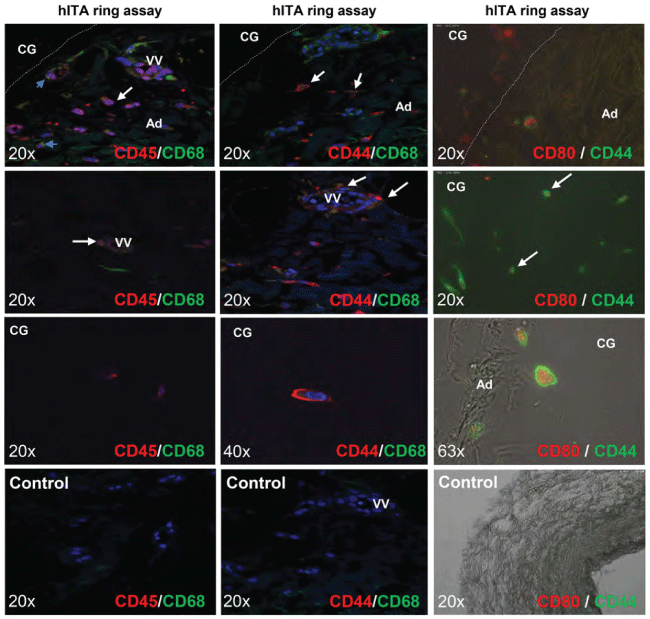
.
Figure 2: Co-localisation of CD45+ CD68+ cells, CD44+ CD68 + cells, and CD44 + CD80+ cells within the vessel wall of hITA after ring assay (9 days, ECGM, no stimulation).
The double immunofluorescent staining for CD44CD68, CD45CD68 and CD44CD80 on sections of embedded hITA rings demonstrate the presence of positive CD45 (red) CD68 (green) (first column) and CD44 (red) CD68 (green) cells (second column), and CD44 (green) CD80 (red) within the adventitia, vasa vasorum and outside of hITA rings in collagen gel. Third column was taken by Nikon Eclipse. Ti: inverted microscope. Lu: lumen, TM: tunica media, Ad: adventitia, CG: collagen gel, VV: vasa vasorum.
View Figure 2
Long term cultivation of total hITA-derived cells and sorted CD44(+) MSC-like VW-MPSCs
Based on the in situ findings above we wanted to further characterize the immunophenotype of total hITA wall-derived cells as well as of MACS-sorted CD44(+) cells in vitro. To this end, we cultured both sorted CD44(+) and total hITA wall-derived cells for 10 days and in long-term cultivation for up to eight weeks (data not shown). FACS analysis revealed that the major part of the total hITA wall-derived cells and as expected also the sorted CD44(+) cells maintained the expression of mesenchymal stem cell markers such as CD44, CD73 (not shown) and CD90 but they remained negative for CD80 (Figure 3A). Furthermore, they were weakly positive for the hematopoietic cell marker CD45 and for CD68 but almost completely negative for CD34 (Figure 3A). These findings were confirmed by immunostaining on hITA wall-derived cells which were fixed on glass slides by cytospin preparation showing CD44(+) and CD90(+) cells but no staining for CD34 (not shown), CD68 (not shown) and CD45 (Figure 3B). The presented data indicate that hITA wall-derived cells could keep mesenchymal characteristics even after culturing them for long-term periods, e.g. over eight weeks.
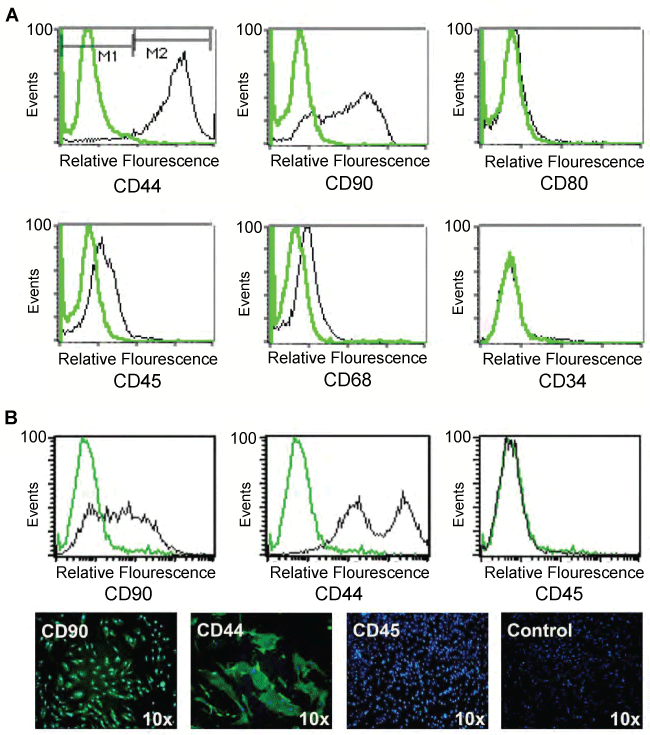
.
Figure 3: Characterization of cultured total cells from the hITA without purification.
FACS analysis of cultured total hITA cells at 10 days in culture shows that the cells are positive for mesenchymal stem cells markers CD44 and CD90 with a week expression of pan-hematopoetic marker CD45, and no expression of macrophages marker CD68 and dendritic cells marker CD80 (A). The expression of mesenchymal stem cells markers CD44 and CD90 is still dominant, whereas no expression of CD45 was detectable in hITA cells at 4 weeks in culture in FACS and in immunocytochemistry (B).
View Figure 3
Differentiation of hITA wall-derived cells into macrophages and dendritic cell-like cells in vitro under high-confluent culture conditions
As shown above, the application of GM-CSF and IL-4 to hITA sprouting assays had no impact on the generating of CD68(+) macrophages and CD80(+) dendritic cell-like cells. To prove this finding at the cellular level hITA wall-derived VW-MPSCs were sorted in CD44(+) and CD44(-) fractions using MACS technology as described in Material and Methods. Both cell populations were cultured with or without GM-CSF+IL-4. Cells were characterized via FACS before and after each stimulation experiment. Confirming our results from the hITA ring assay analyses the stimulation of neither total nor sorted CD44(+) hITA wall-derived cells with GM-CSF+IL-4 resulted in a significant change of the expression of the analyzed cell surface markers such as CD44, CD45, and CD80 except a weak increase in CD68 expression (Figure 4,first and second column). Next, we hypothesized that paracrine effects created by cell-cell-interactions during hITA sprouting assays might be the driving force for the generation of macrophages and dendritic cell-like cells from the hITA wall. To test this, we cultured the total hITA wall-derived cells in endothelial cell growth medium (ECGM) and RPMI at a high confluency. Surprisingly, FACS analysis revealed a significantly increased number of cells expressing CD68 and CD80 even in the absence of GM-CSF+IL-4 in both ECGM and RPMI (Figure 4, third and fourth columns). Noteworthy, a high number of CD44(+) cells was clearly detectable in all cultures (Figure 4).
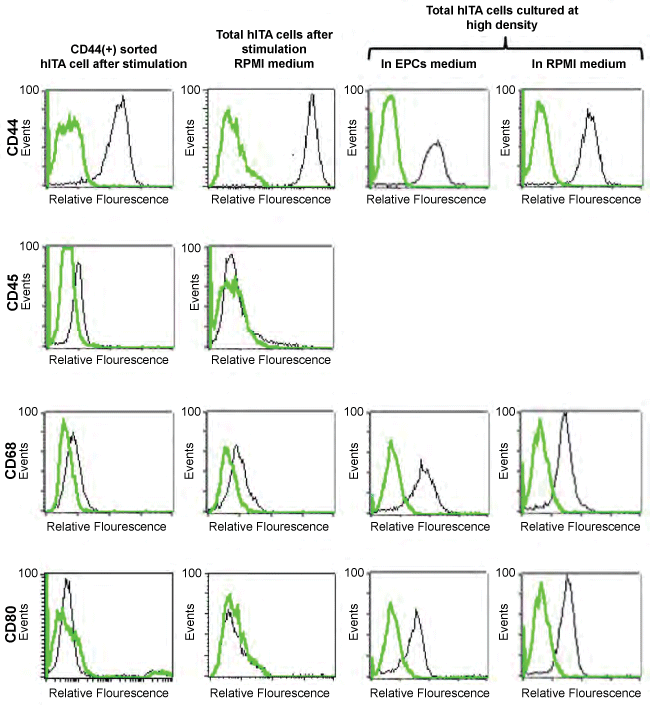
.
Figure 4: Effect of GM-CSF+IL-4 stimulation and culture at high density of total hITA cells on differentiation into macrophages and dendritic cell like cells.
FACS characterization of CD 44(+) sorted cells from the hITA after stimulation with GM-CSF + IL-4 (first column) and total hITA cells after stimulation with GM-CSF+IL-4 (second column). Unstimulated total hITA cells cultured at high density showed the surface phenotype of macrophage and dendritic cells (third column) regardless of media type used for culture.
View Figure 4
Discussion
Here, we show for the first time that MSC-like VW-MPSCs from the vasculogenic zone of the vascular adventitia give rise to tissue-resident CD68(+) macrophages and CD80(+) dendritic cell-like cells. Several studies performed during the last decade already demonstrated that the adventitia of postnatal murine and rat aorta as well as adult hITA contains tissue macrophage precursor cells. In particular, a significant increase in the number of CD68(+) cells within the hITA wall has been identified after sprouting assay using the hITA rings embedded in collagen gel [5]. Accumulation of macrophages after rat femoral artery ligation and depletion of BM cells with cyclophosphamide treatment has been observed within the adventitia of collateral artery walls grown after arterial occlusion in vivo [5]. Along these lines, it has been shown that tissue-resident macrophages rather than circulating and BM-derived macrophages contribute to collateral vessel growth after ligation of the femoral artery [18]. Another study confirmed extensive periaortic outgrowth of CD45(+) CD68(+) mononuclear cells with ultrastructural features of macrophages and dendritic cells after treatment of rat aortic cultures with M-CSF [24]. This is in line with the data demonstrating that murine adventitial Sca-1(+) CD45(+) precursors are a source for tissue macrophages [12,19]. Recently, it has been shown that follicular dendritic cells can be generated from PDGFRβ+ perivascular precursors using a mouse model with PDGFRβ-Cre-driven reporter gene recombination [25].
However, macrophages are a heterogeneous cell population. Moreover, they acquire different phenotypes depending on the microenvironment that surrounds them. Furthermore, most of these studies could only identify the general macrophage markers, e.g. CD68, which to some extent is also expressed by several non-hematopoietic cell types [26]. The major part of the published work was dealing with hematopoietic progenitor cells differentiating into macrophages. Up to date little is known about vascular adventitial cell types differentiating into macrophages, but recently published data undoubtedly identified Sca-1(+)CD45(+) cells as progenitors for vascular adventitia-derived macrophages, which remained present in the aortic adventitia even when circulating monocytes were depleted by liposomal clodronate treatment [12]. In contrast, almost nothing is known about whether there are further adventitial stem cell types with the capacity to deliver macrophages and serve as dendritic cell progenitors. To our knowledge our data are the first showing that CD44(+) MSC-like VW-MPSCs give rise to both macrophages and dendritic cell-like cells using the hITA model. We recently showed that CD44(+) MSC-like cells from the adventitial vasculogenic zone of hITA are multipotent having the capacity to differentiate into different mature cell types such as adipocytes, chondrocytes, smooth muscle cells and pericytes [7].
The present data together with our recently published findings that identified the vascular adventitial CD44(+) cells as multipotent MSC-like cells [7,10] let us postulate that CD44(+) VW-MPSCs might also serve as progenitors for generating antigen-presenting cells such as macrophages and dendritic cells from the vessel wall. To check this hypothesis, we cultured adult hITA rings in the presence or absence of GM-CSF+IL-4. We found that different culture conditions such as endothelial cell growth medium (ECGM MV) plus FCS vs. ECGM without FCS + GM-CSF+IL-4 did not affect the generation and distribution of CD68(+) and CD80(+) in hITA rings. Double immunofluorescence analyses revealed the presence of two immunophenotyptic subsets: CD44(+)CD68(+), CD45(+)CD68(+) and CD44(+)CD80(+) cells within the adventitia of unstimulated hITA wall. Such cell immunophenotypes were also found within the collagen gel after hITA sprouting assay. There are several explanations for the observed phenomenon. First, one could speculate that unpredictable factors present in our ECGM MV culture media may trigger and enhance the differentiation of CD44(+) MSC-like VW-MPSCs into CD68(+) macrophages and CD80(+) dendritic cells. Another explanation could be that vascular adventitia-derived CD44(+) cells need cell-cell interaction with other types of VW-SCs in order to switch to an inflammatory phenotype, e.g. Sca-1(+)CD45(+) adventitial cells.
In line with our findings, follicular dendritic cells could be generated from the PDGFRβ+ perivascular progenitors [25] suggesting that these local progenitors might be involved in the de novo generation of organized lymphoid tissue. We cannot exclude that part of CD45(+)CD68(+) macrophages changes their phenotype into CD44(+)CD68(+) cells by local secretion of different cytokines and growth factors from CD44(+) MSC-like VW-MPSCs, e.g. via paracrine mechanisms or direct cell-cell-interaction within the adventitial stem cell niche. Moreover, double immunofluorescence analysis on sections after aortic sprouting assay revealed the presence of both CD44(+)CD68(+) and CD45(+)CD68(+) cells not only within the vessel wall but also in collagen gel, suggesting that these cells may support the aortic sprouting activity and play a role in angiogenesis.
Opposite to immunostaining studies on native hITA tissue sections and sections of hITA ring assay, CD31(+), CD34(+) and CD45(+) cells were not detectable in FACS and immunofluorescence analysis of cells isolated from dispersed hITA pieces and cultured for different time periods, even when we used different kinds of media including ECGM, RPMI or IMDM. In contrast, the CD44(+) MSC-like VW-MPSC population was always predominant in total hITA cell cultures at all studied time points. This is in line with already published data showing that cells isolated from human saphenous vein and human thoracic aortae expressed MSC markers to a great majority upon culturing [11,13]. Performing FACS analysis on hITA cells after their extraction from hITA pieces was not successful to detect cells expressing hematopoietic markers probably due to the lesser number of adventitial progenitor cells expressing these markers. Alternatively, some of these markers might be down regulated because the progenitors have left the adventitial stem cell niche. This observation in turn may highlight the importance of the vascular niche for the functional activity and communication between the cells. Dispersed cells of the vascular wall without this microenvironment and communication may enter two ways: either they differentiate very fast into other cell types or completely lose some of their surface markers for yet unknown reasons. This is very different from the hITA ring sprouting model where the cells are not exposed to an enzymatic digestion and stay in their niche as well as in sprouting cell cords within the collagen matrix in close contact to each other. To mimic this in situ situation we cultured hITA-wall derived cells in a high vs. low confluency with and without stimulation with IL-4+GM-CSF. Confirming the importance of the cell-cell interaction within the vascular niche the generation of CD68(+) and CD80(+) cells from hITA wall-derived cells was only achieved when they were cultured at a high density. These data particularly underline our interpretation that cell-cell contact between activated VW-SCs and probably factors released following such cell-cell contacts may trigger and direct the differentiation of VW-SC subtypes as already described for BM-MSCs [27,28].
Our findings are of clinical relevance for two reasons: 1) Increasing evidence in the literature already demonstrated that MSCs have not only potent regenerative but also immunomodulatory properties [29-31]. 2) The cell surface marker CD44 functions as hyaluronan receptor and hyaluronan fragments were shown to be increasingly released in inflammatory processes, e.g. after a myocardial infarction. Hyaluronan fragments in turn were reported to act as potent activators of inflammation and their clearance by CD44(+) MSC-like cells was reported to essentially influence the intensity and duration of the inflammatory reaction [32]. In line with these data, absence of CD44 was shown to enhance inflammatory activity [33-36]. Independent from these basic properties of CD44(+) MSCs, their capacity to differentiate into multiple cellular lineages and their trophic effects on other progenitor cells has initiated interest in the use of these cells for regenerative therapy [33-36]. Furthermore, MSCs were shown to shift macrophages from the inflammatory M1 into the anti-inflammatory M2 phenotype [35,37,38] and locally administered MSC could turn the attracted macrophages into a regulatory phenotype [39,40]. More recently, it was shown that MSCs derived from placenta or BM, play an important role in angiogenesis, promote differentiation of human monocytes into macrophages and influence macrophage function [35,37,41,42]. Stagg et al. showed that BM-MSCs acquire antigen-presenting function upon stimulation with interferon-gamma (IFNγ) and thus become able to activate antigen-specific immune responses and express CD80 under syngenic conditions [43]. Furthermore, mouse BM-MSCs expressing CD80 could inhibit the activation of T cells by their cognate peptides in vitro and have thus been suggested for use in BM transplantation in order to control host-versus-graft (HvG) and GVHD reaction [44]. Our data suggest that such immunomodulatory effects can also be attributed to vascular adventitia-derived CD44(+) multipotent cells with MSC properties. Since these cells are omnipresent in the adventitia of blood vessels, including the organs' own vessels, they might play a critical role in organ transplantation, e.g. of kidney and liver including living liver transplantation. Taking together, we postulate that CD44(+) VW-MSCs may also play a role in local immune modulation and be involved in the regulation of macrophage function. However, further functional analysis and studies of these cells are needed before a possible clinical application can be achieved.
Conclusion
In conclusion, the data presented here demonstrate that human VW-SCs derived from the hITA wall have the capacity to differentiate into CD80(+) dendritic cell-like cells which is depending on a specific local microenvironment created by a tight cell-cell interaction between different VW-SC subtypes and factors released by these cells. We further demonstrate that the MSC-like VW-MPSC marker CD44 is also stably expressed on long-term cultured VW-SCs isolated from the vasculogenic zone of human vascular adventitia. A part of these stem cells gives rise to both CD44(+)CD68(+) macrophage-like inflammatory and CD44(+)CD80(+) dendritic cell-like cells. In summary, these data show the generation of antigen-presenting cell-like cells outside the BM, namely from the vascular adventitia-resident VW-SCs. This knowledge is of clinical and physiological relevance for a better understanding of local immune modulatory processes in health and disease.
Acknowledgement
The authors are grateful to Stefanie Kürten for language editing. We thank Erna Kleinschroth for the excellent technical assistance.
Ethical Statement
All experiments were approved by the local ethic committee. Informed consent (written form, Nr.10-4363) was obtained from Ethik-Kommission, University Medical Faculty, Essen, Germany.
References
-
Lander AD, Kimble J, Clevers H, Fuchs E, Montarras D, et al. (2012) What does the concept of the stem cell niche really mean today? BMC Biol 10: 19.
-
Ergün S, Tilki D, Klein D (2011) Vascular wall as a reservoir for different types of stem and progenitor cells. Antioxid Redox Signal 15: 981-995.
-
Tilki D, Hohn HP, Ergün B, Rafii S, Ergün S (2009) Emerging biology of vascular wall progenitor cells in health and disease. Trends Mol Med 15: 501-509.
-
Hu Y, Zhang Z, Torsney E, Afzal AR, Davison F, et al. (2004) Abundant progenitor cells in the adventitia contribute to atherosclerosis of vein grafts in ApoE-deficient mice. J Clin Invest 113: 1258-1265.
-
Zengin E, Chalajour F, Gehling UM, Ito WD, Treede H, et al. (2006) Vascular wall resident progenitor cells: a source for postnatal vasculogenesis. Development 133: 1543-1551.
-
Campagnolo P, Cesselli D, Al Haj Zen A, Beltrami AP, Kränkel N, et al. (2010) Human adult vena saphena contains perivascular progenitor cells endowed with clonogenic and proangiogenic potential. Circulation 121: 1735-1745.
-
Klein D, Hohn HP, Kleff V, Tilki D, Ergün S (2010) Vascular wall-resident stem cells. Histol Histopathol 25: 681-689.
-
Pacilli A, Pasquinelli G (2009) Vascular wall resident progenitor cells: a review. Exp Cell Res 315: 901-914.
-
Passman JN, Dong XR, Wu SP, Maguire CT, Hogan KA, et al. (2008) A sonic hedgehog signaling domain in the arterial adventitia supports resident Sca1+ smooth muscle progenitor cells. Proc Natl Acad Sci USA 105: 9349-9354.
-
Klein D, Benchellal M, Kleff V, Jakob HG, Ergün S (2013) Hox genes are involved in vascular wall-resident multipotent stem cell differentiation into smooth muscle cells. Sci Rep 3: 2178.
-
Pasquinelli G, Tazzari PL, Vaselli C, Foroni L, Buzzi M, et al. (2007) Thoracic aortas from multiorgan donors are suitable for obtaining resident angiogenic mesenchymal stromal cells. Stem Cells 25: 1627-1634.
-
Psaltis PJ, Puranik AS, Spoon DB, Chue CD, Hoffman SJ, et al. (2014) Characterization of a resident population of adventitial macrophage progenitor cells in postnatal vasculature. Circ Res 115: 364-375.
-
Campagnolo P, Cesselli D, Al Haj Zen A, Beltrami AP, Kränkel N, et al. (2010) Human adult vena saphena contains perivascular progenitor cells endowed with clonogenic and proangiogenic potential. Circulation 121: 1735-1745.
-
Bearzi C, Leri A, Lo Monaco F, Rota M, Gonzalez A, et al. (2009) Identification of a coronary vascular progenitor cell in the human heart. Proc Natl Acad Sci U S A 106: 15885-15890.
-
Baaten BJ, Li CR, Bradley LM (2010) Multifaceted regulation of T cells by CD44. Commun Integr Biol 3: 508-512.
-
Thapa R, Wilson GD (2016) The Importance of CD44 as a Stem Cell Biomarker and Therapeutic Target in Cancer. Stem Cells Int 2016: 2087204.
-
Klein D, Weisshardt P, Kleff V, Jastrow H, Jakob HG, et al. (2011) Vascular wall-resident CD44+ multipotent stem cells give rise to pericytes and smooth muscle cells and contribute to new vessel maturation. PLoS One 6: e20540.
-
Khmelewski E, Becker A, Meinertz T, Ito WD (2004) Tissue resident cells play a dominant role in arteriogenesis and concomitant macrophage accumulation. Circ Res 95: E56-64.
-
Psaltis PJ, Harbuzariu A, Delacroix S, Witt TA, Holroyd EW, et al. (2012) Identification of a monocyte-predisposed hierarchy of hematopoietic progenitor cells in the adventitia of postnatal murine aorta. Circulation 125: 592-603.
-
Perdiguero EG, Geissmann F (2016) The development and maintenance of resident macrophages. Nat Immunol 17: 2-8.
-
Klein D, Demory A, Peyre F, Kroll J, Augustin HG, et al. (2008) Wnt2 acts as a cell type-specific, autocrine growth factor in rat hepatic sinusoidal endothelial cells cross-stimulating the VEGF pathway. Hepatology 47: 1018-1031.
-
Bellacen K, Lewis EC (2009) Aortic ring assay. J Vis Exp 33: 1564.
-
Lehtonen A, Ahlfors H, Veckman V, Miettinen M, Lahesmaa R, et al. (2007) Gene expression profiling during differentiation of human monocytes to macrophages or dendritic cells. J Leukoc Biol 82: 710-720.
-
Zorzi P, Aplin AC, Smith KD, Nicosia RF (2010) Technical Advance: The rat aorta contains resident mononuclear phagocytes with proliferative capacity and proangiogenic properties. J Leukoc Biol 88: 1051-1059.
-
Krautler NJ, Kana V, Kranich J, Tian Y, Perera D, et al. (2012) Follicular dendritic cells emerge from ubiquitous perivascular precursors. Cell 150: 194-206.
-
Gottfried E, Kreutz M, Haffner S, Holler E, Iacobelli M, et al. (2007) Differentiation of human tumour-associated dendritic cells into endothelial-like cells: an alternative pathway of tumour angiogenesis. Scand J Immunol 65: 329-335.
-
Oswald J, Boxberger S, Jørgensen B, Feldmann S, Ehninger G, et al. (2004) Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells 22: 377-384.
-
Reyes M, Lund T, Lenvik T, Aguiar D, Koodie L, et al. (2001) Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood 98: 2615-2625.
-
Eggenhofer E, Hoogduijn MJ (2012) Mesenchymal stem cell-educated macrophages. Transplant Res 1: 12.
-
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, et al. (2002) Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99: 3838-3843.
-
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, et al. (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284: 143-147.
-
Frangogiannis NG (2012) Regulation of the inflammatory response in cardiac repair. Circ Res 110: 159-173.
-
Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, et al. (2005) Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 102: 11474-11479.
-
Horwitz EM, Prockop DJ, Fitzpatrick LA, Koo WW, Gordon PL, et al. (1999) Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med 5: 309-313.
-
Nakajima H, Uchida K, Guerrero AR, Watanabe S, Sugita D, et al. (2012) Transplantation of mesenchymal stem cells promotes an alternative pathway of macrophage activation and functional recovery after spinal cord injury. J Neurotrauma 29: 1614-1625.
-
Williams AR, Trachtenberg B, Velazquez DL, McNiece I, Altman P, et al. (2011) Intramyocardial stem cell injection in patients with ischemic cardiomyopathy: functional recovery and reverse remodeling. Circ Res 108: 792-796.
-
Abumaree MH, Al Jumah MA, Kalionis B, Jawdat D, Al Khaldi A, et al. (2013) Human placental mesenchymal stem cells (pMSCs) play a role as immune suppressive cells by shifting macrophage differentiation from inflammatory M1 to anti-inflammatory M2 macrophages. Stem Cell Rev 9: 620-641.
-
English K (2013) Mechanisms of mesenchymal stromal cell immunomodulation. Immunol Cell Biol 91: 19-26.
-
Maggini J, Mirkin G, Bognanni I, Holmberg J, Piazzón IM, et al. (2010) Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile. PLoS One 5: e9252.
-
Németh K, Leelahavanichkul A, Yuen PST, Mayer B, Parmelee A, et al. (2009) Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med 15: 42-49.
-
Boomsma RA, Geenen DL (2012) Mesenchymal stem cells secrete multiple cytokines that promote angiogenesis and have contrasting effects on chemotaxis and apoptosis. PLoS One 7:e35685.
-
van Royen N, Voskuil M, Hoefer I, Jost M, de Graaf S, et al. (2004) CD44 regulates arteriogenesis in mice and is differentially expressed in patients with poor and good collateralization. Circulation 109: 1647-1652.
-
Stagg J, Pommey S, Eliopoulos N, Galipeau J (2006) Interferon-gamma-stimulated marrow stromal cells: a new type of nonhematopoietic antigen-presenting cell. Blood 107: 2570-2577.
-
Krampera M, Glennie S, Dyson J, Scott D, Laylor R, et al. (2003) Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101: 3722-3729.





