Impact of Donor Cytomegalovirus Serology and duration of Prophylaxis on Follow-Up Strategy in Lung Transplant Recipients
Jos� M. Borro1*, Mar�a Delgado1, Teresa Rey2 and Pablo Rama2
1Thoracic Surgery Department, Hospital Universitario de A Coru�a, Spain
2Anesthesia and Reanimation Department, Hospital Universitario de A Coru�a, Spain
*Corresponding author:
Jos� M. Borro, Hospital Universitario de A Coru�a, A Coru�a, Spain, Tel: (+34) 981178187, E-mail: Jose.MA.Borro.Mate@sergas.es
Int J Transplant Res Med,
IJTRM-1-007, (Volume 1, Issue 2),
Original Article
Received: May 25, 2015: Accepted: July 2, 2015: Published: July 4, 2015
Citation: Borro JM, Delgado M, Rey T, Rama P (2015) Impact of Donor Cytomegalovirus Serology and duration of Prophylaxis on Follow-Up Strategy in Lung Transplant Recipients. Int J Transplant Res Med 1:007
Copyright: © 2015 Borro JM. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We investigated the effect of the duration of cytomegalovirus prophylaxis on cytomegalovirus infection rates, drug side effects, and patient survival in a retrospective study of 139 patients who received a lung transplant and oral valganciclovir between 2003 and 2010. Multivariate analysis was performed to explore the impact of cytomegalovirus, bacterial and fungal infections, and acute and chronic rejection on survival. Cox regression methods were used to analyze the effects of prophylaxis and its duration. Mean patient age was 49.8 � 12.6 years; 60% were male. Cytomegalovirus infection occurred in 45.3% patients during the first year post-transplantation, with recurrent infection in 57%. Mean treatment duration was 117 � 40 days. No relationship was found between the duration of prophylaxis and cytomegalovirus infection rate, number of relapses or development of disease, irrespective of donor-recipient cytomegalovirus serology. Overall survival rate was 54% at 5 years. There was no relationship between cytomegalovirus infection and death (p=0.90). Our results suggest that valganciclovir prophylaxis protects against cytomegalovirus infection only during treatment. Treatment for more than 3-6 months (depending on donor-receptor serology) did not seem to modify the probability of infection, and was associated with a higher risk of drug side effects, higher healthcare costs, and no improvement in survival.
Keywords
Cytomegalovirus disease, Cytomegalovirus infection, Lung transplant, Prophylaxis, Valganciclovir
Introduction
Cytomegalovirus (CMV) infection is the most prevalent opportunistic infection after lung transplantation. It is reported in between 20% and 50% of cases after discontinuation of prophylaxis, depending on the series [1]. CMV usually remains dormant in the lymphatic system, and its reactivation, due to immunosuppressive therapy, may have considerable adverse consequences in the immunocompromised host [2]. Its many and varied effects include inflammation, increased morbidity, and decreased graft and patient survival.
Most viral reactivation occurs during the first year after transplant surgery. Effects on survival are twofold: direct effects, caused by the development of invasive disease; and indirect effects, due to overgrowth of other opportunistic infections, malignancy or development of bronchiolitis obliterans [3]. Established scientific evidence indicates that prophylaxis during periods of high-dose immunosuppressive therapy is beneficial in the first months after transplantation, as well as during acute rejection episodes. Without prophylactic treatment, the incidence of infection is very high and the risk of disease increases in the first months after surgery [4]. Recent studies provide some reliable data providing guidance for both general long-term prophylaxis and more tailored strategies based on pre-emptive therapy. A randomized placebo-controlled multicenter trial showed that 12 continuous months of valganciclovir substantially reduced CMV rates during the first 18 months after lung transplant (CMV occurred in 4% of patients), as compared to 3 months of valganciclovir (CMV occurred in 32% of patients), the standard of care at the start of the trial [5]. The authors concluded that extended prophylaxis reduced CMV infection, disease, and disease severity without increased ganciclovir resistance or toxicity. Finlen Copeland et al. also showed a sustained benefit with twelve months of valganciclovir, as compared to 3, over the course of 3.9 years of follow-up in a single-center study [6]. However, there is still no consensus on the protocol to be followed or the duration of treatment [7], and some authors advocate shorter treatments (3 months) followed by a pre-emptive strategy to protect against CMV infections, prevent resistance and avoid valganciclovir toxicity. Some studies and meta-analyses focused on solid-organ transplant recipients found no differences in efficacy between these two approaches [8,9], while adverse effects such as leukopenia have been reported in patients on prophylaxis [10]. For these reasons, the 2011 consensus document of the Spanish Transplantation Infection Study Group (GESITRA) of the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC) recommends short courses of prophylactic treatment, followed by pre-emptive therapy [11]. This approach has been the standard of care in our group since 2003.
Our goal in this study was to establish the effects of a 3-month course of valganciclovir prophylaxis followed by pre-emptive therapy on the CMV infection rate in our series, and to determine the number of recurrences, and patient and graft survival, taking into account patient/donor serologies. Another aim was to describe the side effects associated with treatment and the emergence of resistance reported in our center.
Patients and Methods
Patients
The protocol was approved by the Research Ethics Committee of Hospital Universitario de A Coru�a. A retrospective study was conducted of patients undergoing lung transplantation between 2003 and 2010 in Hospital Universitario de A Coru�a, who received oral valganciclovir for CMV prophylaxis. Patients who died during the first 3 months after transplantation were excluded.
Definitions
Our cases were defined in accordance with the GESITRA-SEIMC consensus document [12]. Thus, "active infection" (CMV infection) was considered when the viral genome, viral proteins or total virus was detected in any tissue or body fluid. Infection was "primary" when virus was detected in a previously seronegative patient. "Recurrent infection" was the renewed detection of CMV at least 4 weeks after the infection had been controlled, due to either reactivation of the same endogenous latent strain or reinfection with a new CMV strain. "CMV disease" was considered when the infected patient presented symptoms or signs of viral syndrome or organ involvement. The term "viremia" was reserved for isolation of the virus in blood cultures, and "antigenemia" was determined when the viral antigen pp65 was directly detected in leukocytes. Finally, "viral syndrome" was characterized by fever (≥ 38 �C) of at least 2 days' duration within a 4-day period, with neutropenia, thrombocytopenia, elevated transaminases and CMV detection.
General care protocol for lung transplant recipients
The patients were selected for inclusion in the transplantation waiting list according to the International Society for Heart and Lung Transplantation criteria [13]. According to this protocol, patients perform physical and respiratory exercises during the waiting period. In addition, candidates for single-lung transplantation receive antifungal prophylaxis with weekly amphotericin B lipid complex via aerosol. Subjects with a history of repeated infections also receive tobramycin before surgery [14].
The surgical technique used by our group has not undergone substantial changes since the beginning of our program in 1999, and is similar to the procedure recently described by the Toronto group [15]. Triple therapy immunosuppressive treatment was administered, including basiliximab for induction, oral or intravenous cyclosporine for maintenance initiation, and azathioprine and decreasing doses of corticosteroids in all cases. Cyclosporine and/or azathioprine were switched to tacrolimus and/or mycophenolate after repeated acute rejection or persistent rejection [14,16], and trimethoprim with sulfamethoxazole was given on alternate days for 9 months to prevent Pneumocystis carinii infection [17,18].
Episodes of acute rejection (ISHLT grade ≥ A2) were treated with 1gr prednisone boluses for 3 days combined with increased immunosuppressive treatment if necessary and the appropriate prophylactic therapy was resumed if it had been previously suspended.
Post-transplant prophylaxis for CMV was ganciclovir (10mg/kg/day, IV) during the first 15 days after surgery for all patients, followed by oral administration of valganciclovir 900 mg/day in a single dose. We never use anti-CMV immunoglobulin as prophylaxis in these patients. The treatment regimen was adjusted between month 3 and 6 of the treatment period, according to risk group. CMV viremia was detected by shell vial assay (between 2003 and June 2007) or whole-blood quantitative polymerase chain reaction (PCR, 2007-2009). CMV detection was routinely performed during oral prophylaxis with valganciclovir, on a weekly basis during the 2-3 months after surgery and at all scheduled follow-up visits thereafter. CMV testing was also performed if infection was suspected, regardless of donor/receptor (D/R) serology. PCR was considered positive when >700 copies/mL was determined at the beginning of the study. Neither positive PCR nor antigenemia occurred during prophylaxis in this 7-year experience and these tests are no longer performed routinely in our department, unless CMV infection is suspected due to disease manifestations.
In case of asymptomatic infection or CMV syndrome, valganciclovir 900mg was administered twice a day [6]. If the patient developed disease, ganciclovir (10mg/kg/day, IV) was prescribed. In case of resistance, foscarnet was used as second-line treatment, with or without immunoglobulins. Hyperinflation during the long-term postoperative period was treated with surgical or bronchoscopic volume reduction [19].
Variables and statistical analysi
All data were collected from the electronic clinical records of the patients included. CMV events included infection, CMV syndrome, and disease. Death was attributed to CMV if the patient died of CMV disease during the course of treatment. Patients with a CMV event who presented increases in viral load and/or clinical worsening of their disease during appropriate treatment with ganciclovir were defined as clinically resistant.
Continuous variables are reported as mean � standard deviation (SD). Dichotomous variables were compared using the χ2 test or Fisher's exact test when the sample size did not allow the former. Survival analysis was performed using the Kaplan-Meier method and curves were compared using the log-rank test. The impact of CMV, bacterial and fungal infections, and acute and chronic rejection on survival were calculated using multivariate analyses. The effect of prophylaxis and its duration (Cox regression) were also analyzed. P-value was considered significant at <0.05. The PASW Statistics 18 program was used for statistical analysis.
Results
A total of 139 eligible patients with lung transplant were identified during the inclusion period, of whom 60% were men and 40% were women. The most frequent indications for transplantation were pulmonary fibrosis (42.4%), chronic obstructive pulmonary disease (25.2%) and cystic fibrosis (10%). General demographic and clinical data are shown in table 1.
![]()
Table 1: Demographic Characteristics and Clinical Data
View Table 1
Donor/recipient CMV serological status in our cohort covered all combinations (Table 1), the most frequent being D+/R+ (n=83; 57%) and the least frequent D+/R- (n=16; 10.7%). The mean duration of follow-up was 31.45 � 21.0 months after transplantation. During the first year after surgery, 45.3% of patients presented a CMV-related event, and the mean disease-free period after transplant was 6 � 3.3 months, with 57% subsequently presenting recurrence.
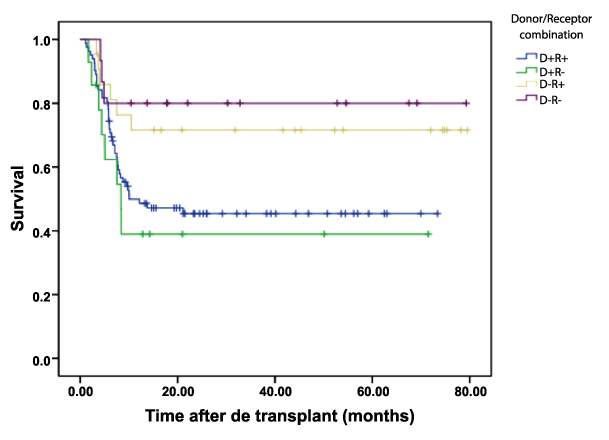
We analyzed serological D/R status as a risk factor for CMV infection. Univariate analysis with Kaplan-Meier curves (Figure 1) showed a lower cumulative probability of survival in patients with serology matches D+/R- and D+/R+. Their respective probabilities tended to be grouped together and fell to an estimated probability of survival of 40-50% 10 months after transplant, compared to the other 2 matches (statistically significant difference: P=0.035). In the multivariate analysis adjusted for age, only age reached significance as a risk factor (OR=1.032; 95% CI=1.007 1.058). The Wald Test indicated that D/R serology should be included in the model (P=0.044) and Cox regression indicated, as expected after Kaplan-Meier analysis, that D+/R+ and D+/R- would be risk factors (B=0.802 and 1.293 respectively) if significantly associated, but none of the different matches analyzed independently reached significance (Table 2,3).
![]()
Table 2: Multivariate analysis of serological D/R match and survival, adjusted for age (Cox regression)
View Table 2
![]()
Table 3: Multivariate analysis of infections, rejection and survival, adjusted for age (Cox regression)
View Table 3
The mean duration of prophylaxis with valganciclovir was 117 � 40 days (range 60-210 days). There was no relationship between the duration of prophylaxis and the incidence of CMV events (CMV infection, the number of recurrences, or the development of disease), irrespective of CMV donor-recipient serology (P=0.98).
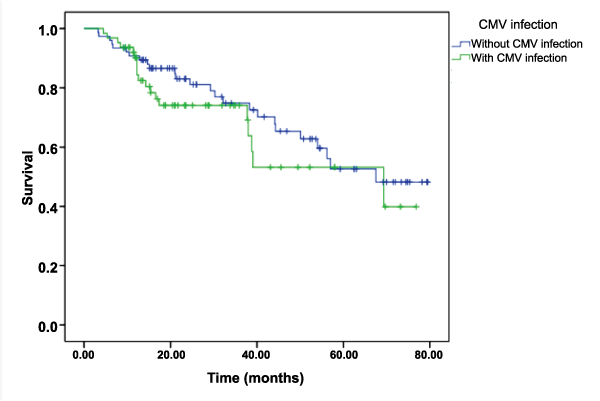
The most frequent adverse effects of prophylactic treatment were increased creatinine and leukopenia. A total of 26% of transplant recipients developed these events, which lead to the discontinuation of prophylaxis in 5 patients. The dose had to be adjusted in 25 patients and 6 needed concomitant treatments, such as colony-stimulating factors (granulocyte colony-stimulating factor), for the continuation of prophylaxis. The incidence of CMV infection was not associated with mortality (P = 0.900) after adjustment for the presence of bacterial and fungal infections, acute rejection, and chronic rejection. Overall estimated probability of survival was 54% at 5 years (Figure 2). Only 9.6% (n = 6) of patients who experienced a CMV event developed disease. Four of them developed CMV pneumonitis and 2 had gastrointestinal tract involvement. None of the deaths were clinically attributed to the CMV event. Two patients who developed clinical disease were resistant to ganciclovir and second-line treatments had to be initiated to achieve resolution.
Discussion
The frequency of CMV infection and disease in lung transplant recipients varies from series to series; incidences of between 54% and 92% in the absence of prophylaxis have been reported [2,20], and even in cohorts receiving prophylactic treatment a 26% incidence has been reported [1]. In our study, viremia occurred in 45% of patients, which is similar to the rate reported by Humar et al. [21], but lower than in the series of Schr�eder et al. who reported a CMV infection incidence of 68.3% in the first year after surgery in patients treated with ganciclovir [22]. According to Singh et al. the period of greatest risk is the first 3 months after transplantation [23], but some recent studies indicate that longer treatments may be desirable to prevent CMV disease [5,6]. There is still a lack of consensus on the optimal duration of prophylaxis and the treatment protocol to be followed in lung transplantation. Zuk et al. recently conducted an international survey to explore procedures in 59 centers in 5 continents. The disparity in strategies was confirmed, as the duration of prophylaxis in the different centers was found to range from 3 months to indefinitely. Most centers reported protocols with prophylaxis durations of between 3 and 6 months; 35.6% did not follow any strategy for D-/R- patients, and only 3.4% used pre-emptive therapy [7]. A recent multicenter study demonstrated the benefits of prophylactic treatment with valganciclovir for longer than 3 months [5], with no increase in valganciclovir toxicity or resistance, and this may convince some practitioners of the value of this approach.
Pre-emptive therapy, a strategy focused on viral monitoring and early treatment instead of universal prophylaxis, has been suggested as an appropriate alternative. Pre-emptive therapy allows controlled low-level replication, thus inducing immune response in the host and preventing late-onset CMV infection [11,23,24]. However, meta-analyses reviewing this strategy have failed to demonstrate its superiority [3,25]. Moreover, a recent pilot study found that valganciclovir prophylaxis did not prevent the development of CMV-specific T cell responses [26], suggesting that the host would not need viral replication in the absence of antiviral drugs to develop an immune response. Paraskeva et al. also reported that sub-clinical viral replication, such as pre-emptive therapy would allow, may be related with pneumonitis and the development of bronchiolitis obliterans syndrome (BOS) [27]. Extended prophylaxis has been shown to prevent CMV infection and disease with effects lasting 6 months after treatment completion, yet the long-term consequences of extended prophylaxis on CMV prevention and BOS were not explored by Palmer et al. [5]. Finlen Copeland et al. did address this issue in their single-center study, reporting a long-term increase in survival during a follow-up of almost 4 years. However, it should be noted that both studies compared 3-month and 12-month prophylaxis treatments with valganciclovir, without the use of pre-emptive therapy, so further studies are needed to shed more light on this question.
In the absence of any clear consensus, practical, empirical and economic reasons have led to the implementation of mixed strategies in clinical environments. This situation has arisen because the costs of each strategy are still under discussion [28,29] and the approach that should be used in different patients with different risk levels is equally unclear [30,31]. The general CMV prevention policy of our group is universal prophylaxis with valganciclovir for 3 months, extended to 6 months in the case of D+/R- patients or when the need for immunosuppressive therapy (acute rejection episodes) creates a high-risk situation. This strategy seems to have good results as regards patient outcome, as only 9.6% of our patients between 2003 and 2010 developed CMV disease. As mentioned above, routine PCR determinations during prophylaxis were discontinued, because infection is rare in this period and, indeed, never occurred in our series. We felt that testing should only be performed when infection is suspected after the suspension of prophylaxis, and this has resulted in an improved management of our resources that has not negatively affected patient outcome. As to when pre-emptive therapy should be indicated after the prophylaxis period, Gerna et al. [32] have discussed the importance of selecting an appropriate cutoff level for viral DNA copies in blood. Although some studies have suggested that DNA replication is not needed to elicit specific immune response and prophylaxis does not seem to prevent T cell response [26], Gerna et al. indicate that a high cutoff in PCR determinations (300,000 copies/mL in their practice) would be more efficient in inducing host immune response. In any case, pre-emptive therapy is not currently recommended in clinical practice in Spain, due to the lack of strong evidence [33].
With regard to donor/receptor serology as a risk factor for CMV infection, Caspar da Cunha et al. reported on a series of 242 solid organ transplant patients, 89 of which were lung transplants. They found that donor seropositivity was a major risk factor for CMV infection, irrespective of whether the recipient was seronegative or positive [34]. These findings are reflected in our series, in which D+ patients have a clear tendency towards a lower estimated probability of survival (i.e. probability of not presenting CMV infection) than those receiving organs from seronegative donors. Our results, however, did not reach significance. This is possibly due to the small population and the loss of statistical power occurring when the analyses were performed after classification of the sample by serology. Both groups of patients, D+/R+ and D+/R-, tend to have a higher probability of CMV infection, which could be explained by a primary infection with donor CMV, rather than a reactivation of the latent virus in R+ patients. It is common for R- receptors of CMV-positive organs to develop CMV infection despite prolonged prophylaxis: of the 15 cases in our series, only 6 did not develop infection. Nevertheless, closer examination of these 6 cases revealed that 1 died 1 month after discontinuing prophylaxis and another 2 developed the infection 2 and 4 months after data collection was complete. The remaining 3 patients became seropositive but did not develop clinical infection during postoperative follow-up.
As for the length of prophylaxis, Valentine et al. defend the need for indefinite treatment, reporting a low incidence of CMV pneumonia in patients who do not discontinue [35]. However, the authors do not specify if pre-emptive therapy was used in case of suspected CMV infection in patients who stopped their prophylactic treatment. Absence of pre-emptive treatment could contribute to the high rates of CMV pneumonitis reported in these patients. On the other hand, Mitsani et al. studied 170 lung transplant patients followed between 2003 and 2008 who received valganciclovir prophylaxis. No relationship was found between duration and dosage of prophylaxis treatment and the development of CMV disease or viremia. The authors stressed the need to define genetic risk markers that promote disease development, in order to guide both treatment and prophylaxis strategies [36].
The long-term use of antiviral drugs in prophylaxis is associated with adverse effects that sometimes lead to treatment discontinuation [25], the development of viral resistance, most frequently in the form of UL97 and UL54 mutations [37,38], and increased healthcare costs [31]. Significant adverse effects, namely leukopenia and increased creatinine, were observed in 26% of our patients. These figures are consistent with the literature. Authors who defend prolonged prophylaxis for longer than 6 months in high-risk D+/R- patients admit that the development of resistance and toxicity associated with prophylaxis are the main factors limiting their protocols [39]. The incidence of viral resistance to ganciclovir is around 5-10% and this proportion rises in D+/R- combinations and lung transplant recipients [40]. The development of resistance is also associated with a higher degree of tissue invasion and a poorer prognosis [41].
In this study, resistance to treatment was suspected when there was an increase in viremia or clinical progression in patients receiving valganciclovir treatment. CMV disease development in our series was infrequent (9.6%), but 33% of these cases were clinically resistant to ganciclovir. This suggests, as reported elsewhere [39], that resistance to antiviral treatment is a risk factor for the development of disease. Our second-line treatment for resistant strains was foscarnet, also used by Reddy et al. [42] to good effect. Several authors report that if CMV pneumonia is correctly treated, BOS can be avoided with no negative consequences on long-term survival [39,43].
Our results support the hypothesis that extended prophylaxis (6 months in our case) can delay CMV events, but does not prevent subsequent infections. This theory has been repeatedly supported by results, for example, from Gavalda et al. [33] Prophylactic treatment during the first 3 to 6 months after transplant combined with antiviral treatment during rejection episodes could be a very useful strategy for improving the survival of recipients in the early months after lung transplant or during periods when high-dose immunosuppression increasing the risk of disease is needed. This protocol reduces the side effects and costs associated with prolonged prophylaxis. Our findings also support the importance of taking high-risk serologies, such as D+, into consideration when establishing the prophylaxis protocol. Consequently, our D- patients received shorter courses, while high-risk patients received longer prophylactic treatments of 6 months. This coincides with the findings of Witzke et al. in kidney transplant recipients [44], who reported significant benefits with a 100-day prophylactic therapy with valganciclovir, especially in D+ patients. Clinical follow-up with PCR tests when infection is suspected allows early diagnosis and prevents disease development in most cases. The most reliable data are those calculated using the actual time of administration of prophylaxis, which is not the case in some trials. In a randomized prospective trial, Palmer et al. found that extended prophylaxis (12 months) significantly reduced CMV disease in lung transplant recipients, compared to prophylaxis periods of 3 months [5]. However, 11 different centers participated in this study, and differences in long-term clinical management methods after discontinuing prophylaxis in each center could have influenced the results. Reduced incidence of CMV disease was also reported in lung recipients in the INCA study [45]. Both studies used a fixed treatment length protocol, but Monforte et al. did not report on possible discontinuations during the prophylaxis period. This would have been interesting, as patient compliance might be affected by adverse effects, resulting in fewer days on prophylaxis [45]. If this was the case, the short treatment period would in fact be less than the 100 or 120 days of treatment specified in the original protocol.
Our results suggest that a follow-up protocol designed to avoid the need for long-term prophylaxis may have benefits in terms of fewer adverse effects for the patients and lower costs, with no negative impact on patient outcome. The detection of patients at risk of developing CMV disease or viral resistance to treatment is a clinical goal that would allow effective and tailored prophylaxis for lung recipients, while reducing side effects.
Acknowledgements
The authors thank Dr. Blanca Piedrafita of Medical Statistics Consulting S.L. for her editorial assistance, Dr Salvador Pita for his advice and collaboration in the study design and statistical analyses and Ms Covadonga Lemos, who collaborated in data management and contact with the patients.
Authorship
All authors contributed to clinical decisions and patient care during the study, including patient selection and data acquisition. J.M. Borro and M. Delgado managed the study database and performed the statistical analysis. J.M. Borro wrote the first manuscript draft, and all the authors revised the subsequent versions and approved the final version.
References
-
Kelly J, Hurley D, Raghu G (2000) Comparison of the efficacy and cost effectiveness of pre-emptive therapy as directed by CMV antigenemia and prophylaxis with ganciclovir in lung transplant recipients. J Heart Lung Transplant 19: 355-359
-
Ettinger NA, Bailey TC, Trulock EP, et al. (1993) Cytomegalovirus infection and pneumonitis. Impact after isolated lung transplantation. Washington University Lung Transplant Group. Am Rev Respir Dis 147: 1017-1023.
-
Kalil AC, Levitsky J, Lyden E, Stoner J, Freifeld AG (2005) Meta-analysis: the efficacy of strategies to prevent organ disease by cytomegalovirus in solid organ transplant recipients. Ann Intern Med 143: 870-880.
-
Hodson EM, Craig JC, Strippoli GF, Webster AC (2008) Antiviral medications for preventing cytomegalovirus disease in solid organ transplant recipients. Cochrane Database Syst Rev 16: CD003774.
-
Palmer SM, Limaye AP, Banks M, et al (2010) Extended valganciclovir prophylaxis to prevent cytomegalovirus after lung transplantation: a randomized, controlled trial. Ann Intern Med 152: 761-169.
-
Finlen Copeland CA, Davis WA, Snyder LD, et al. (2011) Long-term efficacy and safety of 12 months of valganciclovir prophylaxis compared with 3 months after lung transplantation: a single-center, long-term follow-up analysis from a randomized, controlled cytomegalovirus prevention trial. J Heart Lung Transplant 30: 990-996.
-
Zuk DM, Humar A, Weinkauf JG, Lien DC, Nador RG et al. (2010) An international survey of cytomegalovirus management practices in lung transplantation. Transplantation 90: 672-676.
-
Small LN, Lau J, Snydman DR (2006) Preventing post-organ transplantation cytomegalovirus disease with ganciclovir: a meta-analysis comparing prophylactic and preemptive therapies. Clin Infect Dis 43: 869-880.
-
Khoury JA, Storch GA, Bohl DL, et al. (2006) Prophylactic versus preemptive oral valganciclovir for the management of cytomegalovirus infection in adult renal transplant recipients. Am J Transplant 6: 2134-2143.
-
Strippoli GF, Hodson EM, Jones C, Craig JC (2006) Preemptive treatment for cytomegalovirus viremia to prevent cytomegalovirus disease in solid organ transplant recipients. Transplantation 81: 139-145.
-
de la Torre-Cisneros J, Farinas MC, Caston JJ, et al. (2011) GESITRA-SEIMC/REIPI recommendations for the management of cytomegalovirus infection in solid-organ transplant patients. Enferm Infecc Microbiol Clin 29: 735-758.
-
Torre-Cisneros J, Fortun J, Aguado JM, et al. (2005) [Consensus document from GESITRA-SEIMC on the prevention and treatment of cytomegalovirus infection in transplanted patients]. Enferm Infecc Microbiol Clin 23: 424-437.
-
(1998) International guidelines for the selection of lung transplant candidates. The American Society for Transplant Physicians (ASTP)/American Thoracic Society(ATS)/European Respiratory Society(ERS)/International Society for Heart and Lung Transplantation(ISHLT). Am J Respir Crit Care Med 158: 335-339.
-
Borro JM, Sole A, de la Torre M, et al. (2008) Efficiency and safety of inhaled amphotericin B lipid complex (Abelcet) in the prophylaxis of invasive fungal infections following lung transplantation. Transplant Proc 40: 3090-3093.
-
Boasquevisque CH, Yildirim E, Waddel TK, Keshavjee S (2009) Surgical techniques: lung transplant and lung volume reduction. Proc Am Thorac Soc 6: 66-78.
-
Borro JM (2013) Advances in immunosuppression after lung transplantation. Medicina Intensiva 37: 44-9.
-
Gadea I, Cuenca-Estrella M (2004) Guidelines for fungal diagnoses and antifungal sensitivity studies. Enferm Infecc Microbiol Clin 22: 32-39.
-
Gavalda J, Ruiz I (2003) [Guidelines for the treatment of invasive fungal infection. Invasive fungal infection by Candida spp. Invasive Fungal Infection Study Group (MICOMED) and Infection in Transplantation Study Group (GESITRA) of the Spanish Society for Infectious Diseases and Clinical Microbiology (SEIMC)]. Enferm Infecc Microbiol Clin 21: 498-508.
-
Pato O, Rama P, Allegue M, Fernandez R, Gonzalez D,et al. (2010) Bronchoscopic lung volume reduction in a single-lung transplant recipient with natal lung hyperinflation: a case report. Transplant Proc 42: 1979-1981.
-
Duncan AJ, Dummer JS, Paradis IL, et al. (1991) Cytomegalovirus infection and survival in lung transplant recipients. J Heart Lung Transplant 10: 638-646.
-
Humar A, Kumar D, Preiksaitis J, et al. (2005) A trial of valganciclovir prophylaxis for cytomegalovirus prevention in lung transplant recipients. Am J Transplant 5: 1462-148.
-
Schroeder R, Michelon T, Wurdig J, et al. (2007) The incidence of cytomegalovirus infection in lung transplant recipients under universal prophylaxis with intravenous ganciclovir. Braz J Infect Dis 11: 212-214.
-
Singh N (2006) Antiviral drugs for cytomegalovirus in transplant recipients: advantages of preemptive therapy. Rev Med Virol 16: 281-287.
-
Singh N (2001) Preemptive therapy versus universal prophylaxis with ganciclovir for cytomegalovirus in solid organ transplant recipients. Clin Infect Dis 32: 742-751.
-
Puius YA, Snydman DR (2007) Prophylaxis and treatment of cytomegalovirus disease in recipients of solid organ transplants: current approach and future challenges. Curr Opin Infect Dis 20: 419-424.
-
Snyder LD, Medinas R, Chan C, et al. (2011) Polyfunctional cytomegalovirus-specific immunity in lung transplant recipients receiving valganciclovir prophylaxis. Am J Transplant 11: 553-560.
-
Paraskeva M, Bailey M, Levvey BJ, et al. (2011) Cytomegalovirus replication within the lung allograft is associated with bronchiolitis obliterans syndrome. Am J Transplant 11: 2190-2196.
-
McGillicuddy JW, Weimert NA, Taber DJ, et al. (2010) Can preemptive cytomegalovirus monitoring be as effective as universal prophylaxis when implemented as the standard of care in patients at moderate risk? Transplantation 89: 1218-1223.
-
Pescovitz MD (2007) A cost too high to bear? Prophylaxis versus preemptive therapy to prevent post-transplantation cytomegalovirus. Kidney Int 72: 912-913.
-
Baillie GM (2006) Prevention of cytomegalovirus disease in solid organ transplant patients: prophylactic versus preemptive therapy. Am J Health Syst Pharm 63: S10-16.
-
Patel N, Snyder LD, Finlen-Copeland A, Palmer SM (2012) Is prevention the best treatment? CMV after lung transplantation. Am J Transplant 12: 539-544.
-
Gerna G, Baldanti F, Middeldorp J, Lilleri D (2000) Use of CMV transcripts for monitoring of CMV infections in transplant recipients. Int J Antimicrob Agents 16: 455-460.
-
Gavalda J, Monforte V, Len O (2011) [Prevention of cytomegalovirus disease in lung transplantation]. Enferm Infecc Microbiol Clin 29: 46-51.
-
da Cunha-Bang C, Sorensen SS, Iversen M, et al. (2011) Factors associated with the development of cytomegalovirus infection following solid organ transplantation. Scand J Infect Dis 43: 360-365.
-
Valentine VG, Weill D, Gupta MR, et al. (2008) Ganciclovir for cytomegalovirus: a call for indefinite prophylaxis in lung transplantation. J Heart Lung Transplant 27: 875-881.
-
Mitsani D, Nguyen MH, Girnita DM, et al. (2011) A polymorphism linked to elevated levels of interferon-gamma is associated with an increased risk of cytomegalovirus disease among Caucasian lung transplant recipients at a single center. J Heart Lung Transplant 30: 523-529.
-
Drew WL (2000) Ganciclovir resistance: a matter of time and titre. Lancet 356: 609-610.
-
Iwasenko JM, Scott GM, Naing Z, Glanville AR, Rawlinson WD (2011) Diversity of antiviral-resistant human cytomegalovirus in heart and lung transplant recipients. Transpl Infect Dis 13: 145-153.
-
Mitsani D, Nguyen MH, Kwak EJ, et al. (2010) Cytomegalovirus disease among donor-positive/recipient-negative lung transplant recipients in the era of valganciclovir prophylaxis. J Heart Lung Transplant 29: 1014-1020.
-
Boivin G, Goyette N, Gilbert C, Humar A, Covington E (2005) Clinical impact of ganciclovir-resistant cytomegalovirus infections in solid organ transplant patients. Transpl Infect Dis 7: 166-170.
-
Emery VC, Griffiths PD (2000) Prediction of cytomegalovirus load and resistance patterns after antiviral chemotherapy. Proc Natl Acad Sci U S A 97: 8039-8044.
-
Reddy AJ, Zaas AK, Hanson KE, Palmer SM (2007) A single-center experience with ganciclovir-resistant cytomegalovirus in lung transplant recipients: treatment and outcome. J Heart Lung Transplant 26: 1286-1292.
-
Tamm M, Aboyoun CL, Chhajed PN, Rainer S, Malouf MA, et al. (2004) Treated cytomegalovirus pneumonia is not associated with bronchiolitis obliterans syndrome. Am J Respir Crit Care Med 170: 1120-1123.
-
Witzke O, Hauser IA, Bartels M, Wolf G, Wolters H, et al. (2012) Valganciclovir prophylaxis versus preemptive therapy in cytomegalovirus-positive renal allograft recipients: 1-year results of a randomized clinical trial. Transplantation 93: 61-68.
-
Monforte V, Lopez C, Santos F, et al. (2009) A multicenter study of valganciclovir prophylaxis up to day 120 in CMV-seropositive lung transplant recipients. Am J Transplant 9: 1134-1141.